Abstract
We recently demonstrated that the local transplantation of human peripheral blood (PB) CD34+ cells, an endothelial/hematopoietic progenitor cell-rich population, contributes to fracture repair via vasculogenesis/angiogenesis and osteogenesis. Human PB mononuclear cells (MNCs) are also considered a potential cell fraction for neovascularization. We have previously shown the feasibility of human PB MNCs to enhance fracture healing. However, there is no report directly comparing the efficacy for fracture repair between CD34+ cells and MNCs. In addition, an unhealing fracture model, which does not accurately resemble a clinical setting, was used in our previous studies. To overcome these issues, we compared the capacity of human granulocyte colony-stimulating factor-mobilized PB (GM-PB) CD34+ cells and human GM-PB MNCs in a nonunion model, which more closely resembles a clinical setting. First, the effect of local transplantation of 1 × 105 GM-PB CD34+ cells (CD34+ group), 1 × 107 GM-PB MNCs (containing approximately 1 × 105 GM-PB CD34+ cells) (MNC group), and phosphate-buffered saline (PBS) (PBS group) on nonunion healing was compared. Similar augmentation of blood flow recovery at perinonunion sites was observed in the CD34+ and MNC groups. Meanwhile, a superior effect on nonunion repair was revealed by radiological, histological, and functional assessment in the CD34+ group compared with the other groups. Moreover, through in vivo and in vitro experiments, excessive inflammation induced by GM-PB MNCs was confirmed and believed to be one of the mechanisms underlying this potency difference. These results strongly suggest that local transplantation of GM-PB CD34+ cells is a practical and effective strategy for treatment of nonunion after fracture.
Introduction
In the field of regenerative medicine, adult stem/progenitor cells are currently considered one of the most important elements for the repair of various tissues. Many reports have given evidence that stem/progenitor cells derived from a variety of adult tissues are capable of maintaining, regenerating, and repairing other tissues as well as the original tissue (6, 16, 38, 50, 59, 69).
Endothelial progenitor cells (EPCs) were isolated as CD34+ mononuclear cells (MNCs) from adult peripheral blood (PB) (3, 4, 56). Recent studies have demonstrated that adult EPCs are mobilized from bone marrow (BM) into PB under tissue ischemia, incorporate into ischemic lesions, differentiate into mature endothelial cells, and contribute to neovascularization (3, 4, 61). The fundamental feasibility of EPCs for therapeutic neovascularization has commanded considerable attention from a number of researchers. The therapeutic potential of EPCs on hindlimb ischemia and myocardial infarction (MI) was reported in experimental animal models (25, 26, 28, 30, 33, 36, 67). More recent preclinical investigations have revealed favorable findings post-EPC transplantation in broader fields of regenerative medicine, including cerebral diseases, full-thickness skin wounds, or ligament injuries (27, 58, 60, 63). Several subsequent clinical trials have been performed to evaluate the efficacy of EPC transplantation and have revealed favorable outcomes in ischemic and nonischemic diseases (5, 7, 32, 44, 53). The results of these clinical trials suggest that CD34+ cell transplantation is safe enough to be applied to clinical treatment and has the potential to become a new therapeutic strategy for various diseases.
Recent progress in stem cell biology has clarified that human CD34+ cells or CD133+ cells, which were formerly considered as hematopoietic or EPCs, have a phenotypical aspect for osteogenesis. In vitro, CD34+ cells and CD133+ cells are capable of differentiating into osteoblasts (8, 64). The presence of osteoblastic lineage cells positive for osteocalcin (OC) and alkaline phosphatase (ALP) in PB was confirmed by flow cytometry. These cells formed mineralized nodules in vitro and regenerated bone in vivo (11). Moreover, it was noticed that approximately 40% of OC-positive and 50% of ALP-positive cells in human PB coexpressed CD34 (12). These findings strongly suggest that therapeutic application of BM-derived circulating CD34+ cells may be beneficial not only for vasculogenesis, but also for osteogenesis.
A significant proportion (5–10%) of bone fractures fail to heal and result in delayed union or persistent nonunion mainly due to inappropriate neoangiogenesis (2, 10, 19, 45). Based on the vasculogenic and osteogenic properties of CD34+ cells, our group performed a series of studies regarding CD34+ cell transplantation for fracture repair (47, 48). We reported previously that human PB CD34+ cells systemically infused into immunocompromised rats with unhealing fracture and contributed to functional fracture repair via vasculogenesis/angiogenesis and osteogenesis (47). Subsequently, it was revealed that local administration of human granulocyte colony-stimulating factor-mobilized PB (GM-PB) CD34+ cells also contributed to fracture healing via vasculogenesis and osteogenesis in a dose-dependent manner (48). On the other hand, the potential of PB or BM total MNCs containing both CD34+ and CD34- cells for therapeutic neovascularization has been reported in hindlimb and myocardial ischemia (15, 20, 29, 39, 57, 62). We previously reported that human GM-PB MNCs also have the potency to enhance fracture repair via vasculogenesis/osteogenesis (14). However, there have been no studies directly comparing the efficacy of human GM-PB CD34+ cells on bone healing with that of human GM-PB MNCs. In the current study, we investigated the potency of human GM-PB CD34+ cells compared to MNCs for bone repair in nonunion animal models, in which the degree of damage is more severe than that of unhealing fracture models.
Materials and Methods
Ethics Statement
All experimental procedures were conducted in accordance with the Japanese Physiological Society Guidelines for the Care and Use of Laboratory Animals, and the study protocol was approved by the Ethical Committee at the Institute of Biomedical Research and Innovation.
Preparation of Human Cells
We purchased human GM-PB CD34+ cells obtained from three healthy males (20-year-old African-American; 19-year-old Caucasian; 21-year-old Caucasian) and human GM-PB MNCs obtained from three healthy males (25-year-old African-American; 25-year-old African-American; 22-year-old Caucasian) from Lonza (Valais, Switzerland) and AllCells (Berkeley, CA, USA). These cells were used at random for the in vivo experiments.
Human GM-PB CD34+ cells and human GM-PB MNCs obtained from the same three healthy male donors (29-year-old Caucasian; 22-year-old Mexican and Caucasian; 22-year-old Caucasian) were obtained from AllCells and used at random for the in vitro real-time RT-PCR and enzyme-linked immunosorbent assay (ELISA) experiments.
Human GM-PB CD34+ cells and human GM-PB CD34-obtained from three subjects (42-year-old Caucasian male; 38-year-old Caucasian female; 28-year-old Caucasian male), who took part in the clinical trial that we previously reported (41), were used for in the vitro coculture experiments. The GM-PB CD34- cells were isolated by removal of the GM-PB CD34+ cells from the GM-PB MNCs using the CliniMACS system consisting of a CliniMACS Instrument, CD34 reagent, PBS/EDTA buffer, and a tubing set (Miltenyi Biotec, Bergisch Gladbach, Germany).
Surgical Procedure
Female athymic nude rats (F344/N Jclrnu/rnu; CLEA Japan, Inc., Tokyo, Japan) aged 8-12 weeks and weighing 150-170 g were used in this study. Anesthesia was induced by intraperitoneal administration of a ketamine hydrochloride (60 mg/kg; Daiichi Sankyo, Tokyo, Japan) and xylazine hydrochloride (10 mg/kg; Bayer, Tokyo, Japan) mixture. Unhealing fracture was induced in the right femur with cauterizing periosteum according to the methods of Einhorn (13) and Kokubu et al. (35). Eight weeks after the creation of the unhealing fracture, we radiographically confirmed nonunion with no bridging callus and defined these animals as the “nonunion models.” These rats with nonunion received local administration of the following materials with atelocollagen (KOKEN, Tokyo, Japan), a bovine-derived bio-absorbable scaffold (23, 24): (a) 1 × 105 human GM-PB CD34+ cells in 100 μl of PBS (Gibco, Grand Island, NY, USA) (CD34+ group), (b) 1 × 107 human GM-PB MNCs (containing approximately 1 × 105 human GM-PB CD34+ cells) in 100 μl of PBS (MNC group), and (c) 100 μl of PBS alone (PBS group) (n = 23 in each group). The cell suspension was mixed with atelocollagen in a microtube with a 22-gauge needle. The left intact femur of each animal served as a control. Rats were euthanized with an overdose of ketamine (Daiichi-Sankyo, Tokyo, Japan) and xylazine (Bayer Yakuhin, Osaka, Japan) for biomechanical and histological analysis in the indicated time course described below. The femurs were directly embedded in OCT compound (Sakura Finetek, Tokyo, Japan), snap frozen in liquid nitrogen, and stored at −80°C as fresh frozen samples.
Physiological Assessment of Tissue Blood Flow by Laser Doppler Perfusion Imaging (LDPI)
A LDPI system (Moor Instrument, Wilmington, DE, USA) (43, 66) was used to measure serial blood flow in both hindlimbs at 0, 1, 2, and 3 weeks post-cell transplantation. The blood flow recovery following cell transplantation was evaluated as the ratio of mean flux within the fracture region to the corresponding region in the intact (contralateral) limb.
Radiographic Assessment of the Nonunion Healing
Rats were fixed in a supine position under anesthesia, and radiographs of the injured hindlimbs were serially taken at 0, 4, 8, and 12 weeks following cell transplantation. Bone union was identified by the presence of a bridging callus on the two cortices. All radiographs were examined by three observers blinded to the study groups.
Histological Assessment of the Nonunion Healing
Samples were sectioned at 6 μm thickness on cryofilm (Leica Microsystems, Tokyo, Japan) (34) and attached to slides followed by fixation with 4% paraformaldehyde (Wako, Osaka, Japan) at 4°C for 5 min. Toluidine blue (Sigma-Aldrich, St. Louis, MO, USA) staining was performed to histologically evaluate the process of endochondral ossification at 4, 8, and 12 weeks after cell transplantation. The degree of fracture healing was evaluated at 12 weeks after cell transplantation using a 5-point scale (grades 0-4) proposed by Allen et al. (2).
Biomechanical Analysis of Bone Union
Four rats in each treatment group were used for biomechanical evaluation at week 12 post-cell transplantation at the Kureha Special Laboratory (Tokyo, Japan). Injured femurs and the contralateral noninjured femurs were prepared, and intramedullary fixation pins were removed before the bending test. The standardized three-point bending test was performed using load torsion and a “MZ-500S” bending tester (Maruto Instruments, Tokyo, Japan). The bending force was applied with a cross-head at a speed of 2 mm/min until rupture occurred. Ultimate stress (N), extrinsic stiffness (N/mm), and failure energy (N/mm) were recorded. The relative ratio of the injured (right) femur to the noninjured (left) femur was calculated for each parameter.
Quantitative Real-Time RT-PCR Analysis for Inflammatory Cytokines In Vitro
We seeded 3.5 × 105 human GM-PB CD34+ cells or 3.5 × 107 human GM-PB MNCs in a 60-mm dish (Becton Dickinson, Franklin Lakes, NJ, USA) with 5 ml Dulbecco's modified Eagle's medium low glucose (Invitrogen, Gaithersburg, MD, USA) supplemented with 10% fetal bovine serum (FBS) (Vitromex, Vilshofen, Germany). After 6 h of culture, we collected the respective cells, and the total RNA was extracted with an RNeasy Mini Kit (Qiagen, Hilden, Germany) following the manufacturer's protocol. First-strand cDNA was synthesized using a PrimeScript RT Reagent Kit (Takara, Otsu, Japan) according to the standard protocol, and the converted cDNA samples (2 μl) were amplified in triplicate by real-time PCR (ABI PRISM 7700; Applied Biosystems, Foster City, CA, USA) in a final volume of 20 μl using SYBR Green Master Mix reagent (Applied Biosystems) with gene-specific primers of inflammatory cytokines, including tumor necrosis factor (TNF
Specific Primers for Real-Time RT-PCR Amplifications
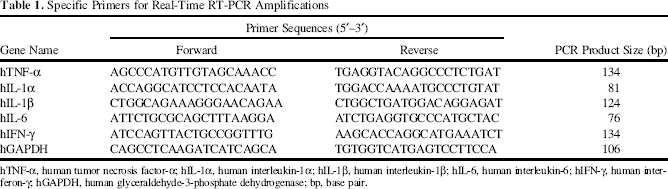
hTNF-α, human tumor necrosis factor-α; hIL-1α, human interleukin-1α; hIL-1β, human interleukin-1β; hIL-6, human interleukin-6; hIFN-γ, human interferon-γ hGAPDH, human glyceraldehyde-3-phosphate dehydrogenase; bp, base pair.
ELISA for Measuring the Concentration of Inflammatory Cytokines in the Cell Culture Supernatants
Human GM-PB CD34+ cells (6 × 105 cells/ml) or human GM-PB MNCs (6 × 107 cells/ml) were resuspended in RPMI-1640 (Sigma-Aldrich) with 10% FBS and phytohe-magglutinin (20 μg/ml; Sigma-Aldrich) and incubated at 37°C for 48 h. The cell-free supernatants were harvested after centrifugation at 300 × g for 10 min and stored at −20°C until used. The concentrations of human inflammatory cytokines, including TNF-α, IL-1α, IL-1β, IL-6, and IFN-γ, were measured by a commercially available ELISA kit (R&D Systems, Minneapolis, MN, USA).
Immunohistochemical Assessment of Inflammation at the Perinonunion Sites After Cell Transplantation
To detect the inflammatory cells in the rat perinonunion sites, immunohistochemistry was performed using fresh frozen tissue samples obtained 7 days after cell transplantation with the following antibodies: polyclonal rabbit antibody against CD68 of humans and rats (1:50; Santa Cruz Biotechnology, Santa Cruz, CA, USA) to detect both human and rat macrophages and mouse antibody against rat-specific granulocytes (1:100; BMA Biomedicals, Augst, Switzerland). The secondary antibodies for each immunostaining are as follows: Alexa Fluor 488-conjugated goat anti-rabbit IgG (1:500; Molecular Probes, Invitrogen Japan K.K., Tokyo, Japan) for human–rat CD68 staining, and Alexa Fluor 594-conjugated goat anti-mouse IgG1 (1:1,000; Molecular Probes) for rat granulocyte staining. 4′,6-Diamidino-2-phenylindole (DAPI) (Invitrogen) solution was applied for 5 min for nuclear staining.
Inflammatory cell density was morphometrically evaluated as the average value in four randomly selected fields of soft tissue at the perinonunion site. All morphometric studies were performed by two examiners blinded to the study groups.
Analysis of Cell Apoptosis around Nonunion Sites
Fresh frozen sections were obtained 7 days after cell transplantation, and the terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) assay was performed using an In Situ Cell Death Detection Fluorescein Kit (Roche, Penzberg, Germany) in accordance with the manufacturer's instructions. Briefly, sections were incubated with 4% paraformaldehyde dissolved in PBS for 10 min at 4°C and then permeabilized by treatment with 0.1% Triton X-100 (ICN Biomedicals, Costa Mesa, CA, USA) in 0.1% sodium citrate (Wako) at 4°C for 2 min. Slides were washed with PBS, and TUNEL reaction reagent was placed on the sections for 1 h at room temperature. After washing in PBS, DAPI solution was applied for 5 min for nuclear staining.
TUNEL-positive cell density was morphometrically evaluated as the average value in six randomly selected fields of soft tissue at the perinonunion site. All morphometric studies were performed by two examiners blinded to the study groups.
Coculture of GM-PB CD34+ Cells with or Without GM-PB CD34- Cells
To investigate the influence of GM-PB CD34- cells on GM-PB CD34+ cells, a coculture experiment was performed. CD34+ cells, 4 × 104, were incubated in the lower chamber of a 24-well plate separated by a cell culture insert (0.4 μm pore size) (Becton Dickinson) with or without 4 × 106 CD34- cells in the upper chamber in StemSpan-SFEM (StemCell Technologies, Vancouver, Canada) with lipopolysaccharide (10 ng/ml) (Sigma-Aldrich) conditioning. To correspond the inflammatory conditioning in the acute ischemic tissues, lipopolysaccharide was used for stimulation. Both upper and lower chambers were incubated at 37°C for 24 h with five growth factors; human vascular endothelial growth factor (VEGF; 50 μg/ml; Wako), human stem cell factor (SCF; 100 μg/ml; Wako), human IL-6 (20 μg/ml; Wako), human Flt3-ligand (100 μg/ml; Wako), and human thrombopoietin (20 μg/ml; Wako). After coculture, the GM-PB CD34+ cells were evaluated.
Evaluation of Cell Viability and Apoptosis
Cell viability was assessed by trypan blue exclusion assay. After the coculture, GM-PB CD34+ cells were resuspended in PBS, and 20 μl of 0.4% trypan blue (Gibco) was added to the cell suspension. After approximately 5 min of equilibration, the cells were counted under a microscope.
GM-PB CD34+ cell apoptosis was evaluated by using TUNEL with an In Situ Cell Death Detection Fluorescein Kit (Roche) according to the manufacturer's instructions. The quantification of apoptosis was determined by counting the TUNEL-positive cell nuclei from three random fields per section and was expressed as percentage of total cell nuclei.
EPC Colony-Forming Assay
An EPC colony-forming assay established in our laboratory was performed as reported previously (46). The number of EPC colonies was assessed at day 19 in methyl cellulose-containing medium H4236 (StemCell Technologies) with 100 ng/ml SCF (Wako), 50 ng/ml VEGF (Wako), 20 ng/ml IL-3 (Wako), 50 ng/ml basic fibroblastic growth factor (bFGF; Wako), 50 ng/ml epidermal growth factor (EGF; Wako), 50 ng/ml insulin-like growth factor-1 (IGF-1; Wako), and 2 U/ml heparin (Ajinomoto, Tokyo, Japan).
Statistical Analysis
The results were statistically analyzed using a software package (GraphPad Prism™; MDF Software, Inc., San Diego, CA, USA). All values were expressed as mean ± SEM. Comparisons between multiple groups or two groups were tested via one-way ANOVA followed by post hoc testing with Tukey's procedure or unpaired t-test, respectively. The comparison of radiological results was performed with a chi-square test. A probability value less than 0.05 was considered to be significant.
Results
Serial Improvement of Blood Flow at Nonunion Sites
To evaluate blood flow recovery at nonunion sites by a physiological approach, LDPI was serially performed at 0, 1, 2, and 3 weeks in each group (Fig. 1A). There was no significant difference in the blood flow ratio (intact site/contralateral site) 1 h after cell transplantation (week 0) between each group. While the ratio at weeks 1 and 2 was significantly higher in the CD34+ and MNC groups compared with the PBS group, there was no significant difference between the CD34+ and MNC groups (week 1: CD34+, 1.563 ± 0.066; MNC, 1.508 ± 0.158; PBS, 1.303 ± 0.093, respectively, p < 0.01 for CD34+ or MNC vs. PBS group, week 2: CD34+, 1.435 ± 0.037; MNC, 1.352 ± 0.093; PBS, 1.283 ± 0.052, respectively, p < 0.05 for CD34+ or MNC vs. PBS group). At week 3, there was no significant difference between the three groups (CD34+, 1.323 ± 0.031; MNC, 1.088 ± 0.141; PBS, 1.235 ± 0.057, respectively, p = NS) (Fig. 1B).
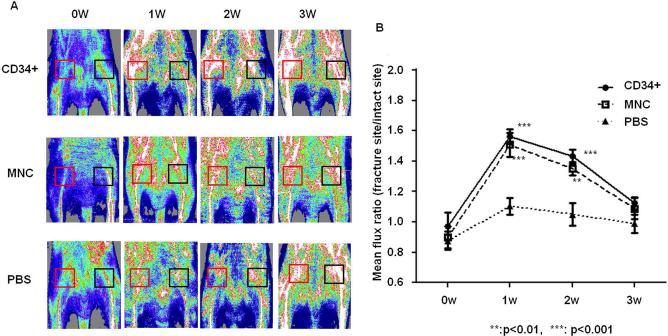
Serial improvement of blood flow at nonunion sites. (A) Representative laser Doppler perfusion imaging at weeks 0, 1, 2, and 3 in each group. (B) Quantitative analyses of local blood perfusion. The mean flux ratios at weeks 1 and 2 were significantly higher in the CD34+ group and the MNC group than the PBS group.
These results indicate that local transplantation of 1 × 105 human GM-PB CD34+ cells or 1 × 107 human GM-PB MNCs similarly contributes to the rapid improvement of tissue perfusion at the nonunion site.
Radiographic and Histological Evidence of Bone Healing
To evaluate bone repair morphologically, radiographic and histological examinations were performed. In 20% of the rats at week 4 and 90% of the rats at weeks 8 and 12 in the CD34+ group, nonunion healing with bridging callus formation was observed radiographically, while nonunion healing was observed in only 10% of the rats at week 4 and 30% of rats at weeks 8 and 12 in the MNC group. No animals in the PBS group showed bridging callus formation during the 12 weeks of observation. The frequency of bone union was significantly greater in the CD34+ group than in the other groups at weeks 8 and 12 (p < 0.001 for CD34+ vs. PBS group and p < 0.01 for CD34+ vs. MNC group) (Fig. 2A, B).

Radiographical evidences of bone healing following the cell transplantation. (A) Representative radiographs of nonunion sites at weeks 0, 4, 8, and 12. (B) The union rate in all groups. Frequency of the bone union was significantly greater in the CD34+ group than the other groups at weeks 8 and 12.
To evaluate bone healing histologically, toluidine blue staining was performed. At week 4, nonunion sites of the CD34+ group and the MNC group demonstrated enhanced endochondral ossification consisting of numerous chondrocytes and newly formed trabecular bone. In the CD34+ group, bridging callus formation at week 8 and complete union at week 12 was observed. Meanwhile, a thick but nonbridging callus formation at week 8 and complete bone union at week 12 was observed in the MNC group. In the PBS group, fracture gaps were filled with fibrous tissue, and no chondrogenesis or new bone formation was observed during the 12-week period (Fig. 3A). The degree of bone healing assessed by Allen's classification (2) was significantly greater in the CD34+ group than in the MNC and PBS groups. The degree of bone healing was also greater in the MNC group compared to the PBS group at week 12 (CD34+, 3.25 ± 0.70; MNC, 1.75 ± 1.07; PBS, 0.0 ± 0.00; respectively, p < 0.001 for CD34+ or MNC vs. PBS group, p < 0.05 for CD34+ vs. MNC group) (Fig. 3A, B).
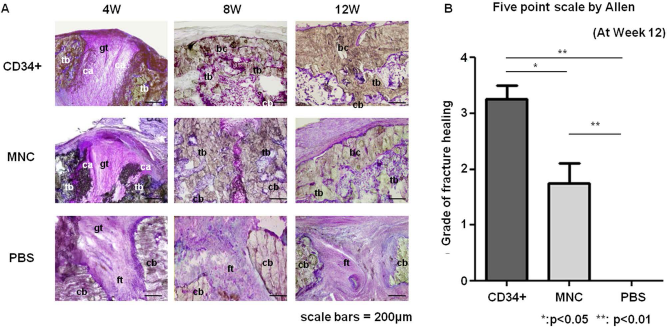
Histological evidence of bone healing following the cell transplantation. (A) Histological evaluation of endochondral ossification by toluidine blue staining. The abbreviations in the figures indicate as follows: bc, bridging callus; cb, cortical bone; ca, cartilage; ft, fibrous tissue; gt, granulation tissue; tb, trabecullar bone. (B) The extent of bone healing assessed by Allen's classification was significantly higher in the CD34+ group than the other groups at week 12, as well as in the MNC group compared to the PBS group.
These findings indicate that the transplantation of human GM-PB CD34+ cells induces nonunion repair more effectively than the injection of human GM-PB MNCs and PBS.
Functional Bone Recovery by Three-Point Bending Test
To confirm the functional recovery from nonunion, biomechanical evaluation using a three-point bending test was performed at week 12 in all groups. Specimen length was similar in the CD34+ group (15.8 ± 2.3 mm), MNC group (15.9 ± 4.1 mm), and the PBS group (15.6 ± 3.3 mm). In the CD34+ group, the ratios between the injured/contralateral femur of all parameters except extrinsic stiffness were significantly superior to those in the MNC and PBS groups (Percent failure energy: CD34+, 141.0 ± 5.8; MNC, 77.2 ± 18.4; PBS, 50.3 ± 9.0%; respectively, p < 0.01 for CD34+ vs. PBS group and p < 0.05 for CD34+ vs. MNC group. Percent ultimate stress: CD34+, 115.2 ± 2.5; MNC, 49.4 ± 23.8; PBS, 21.4 ± 3.8%; respectively, p < 0.01 for CD34+ vs. PBS group and p < 0.05 for CD34+ vs. MNC group). The ratio for extrinsic stiffness in the CD34+ group was significantly superior to that in the PBS group and tended to be greater than that in the MNC group (Percent extrinsic stiffness: CD34+, 27.7 ± 5.7; MNC, 12.4 ± 3.4; PBS, 8.6 ± 1.9%; respectively, p < 0.05 for CD34+ vs. PBS group, p = NS for CD34+ vs. MNC group) (Fig. 4).

Functional bone recovery; three-point bending test. Functional recovery after nonunion is assessed in a biomechanical three-point bending test at week 12. Percentage of each parameter (failure energy, ultimate stress, and extrinsic stiffness) indicates the ratio of each value between the injured sites and the contralateral intact sites.
These results indicate that functional repair from nonunion is more prominent after local transplantation of human GM-PB CD34+ cells compared with human GM-PB MNCs and PBS injection.
mRNA Expression of Inflammatory Cytokines in CD34+ Cells and MNCs
We performed real-time RT-PCR to quantify the mRNA expression of inflammatory cytokines (TNF-α, IL-1α, IL-1β, IL-6, and IFN-γ) in human GM-PB CD34+ cells and human GM-PB MNCs in vitro. The RNA expression levels of all inflammatory cytokines were significantly greater in human GM-PB MNCs compared with human GM-PB CD34+ cells (TNF-α: CD34+, 20.9 ± 5.0; MNC, 986.3 ± 424.3, p < 0.05. IL-1α: CD34+, 116.8 ± 22.32; MNC, 500.1 ± 94.09, p < 0.001. IL-1β: CD34+, 618.5 ± 60.75; MNC, 5318.1 ± 629.8, p < 0.001. IL-6: CD34+, 37.1 ± 11.2; MNC, 316.9 ± 54.7, p < 0.001. IFN-γ: CD34+, 124.6 ± 32.1; MNC, 715.3 ± 268.7, p < 0.05.) (Fig. 5).

Real-time RT-PCR analysis for inflammatory cytokines expressed in CD34+ cells and MNCs. Relative mRNA expressions of human TNF-α, IL-1α, IL-1β, IL-6, and IFN-γ were significantly greater in the MNC group than the CD34+ group.
Protein Expression of Inflammatory Cytokines in CD34+ Cells and MNCs
We cultured human GM-PB CD34+ cells (6 × 105 cells/ml) or human GM-PB MNCs (6 × 107 cells/ml) with PHA at 37°C for 48 h. The protein levels of all inflammatory cytokines (TNF-α, IL-1α, IL-1β, IL-6, and IFN-γ) in the culture supernatants tended to be greater in human GM-PB MNCs than in GM-PB CD34+ cells, although the differences were not statistically significant (TNF-α: CD34+, 6.5 ± 4.8; MNC, 1,036.3 ± 477.8 pg/ml; IL-1α: CD34+, 0.3 ± 0.2; MNC, 1,140.9 ± 1,095 pg/ml; IL-1β: CD34+, 1.9 ± 0.8; MNC, 19,650.6 ± 18,444 pg/ml; IL-6: CD34+, 25.1 ± 12.6; MNC, 62,565.2 ± 31,752 pg/ml; IFN-γ: CD34+, 7.6 ± 0.7; MNC, 33,290.4 ± 15,288 pg/ml).
Immunohistochemical Assessment of Inflammatory Cell Invasion After Cell Transplantation
To investigate the distribution of inflammatory cells around nonunion sites, immunohistochemistry for polyclonal CD68, known as a marker for both human and rat macrophages, was performed in all groups. The density of CD68+ cells within perinonunion sites was significantly greater in the MNC group than the CD34+ or PBS group (CD34+, 173.0 ± 18.6; MNC, 775.6 ± 34.4; PBS, 106.8 ± 6.0 cells/mm2; p < 0.001 for MNC vs. CD34+ and PBS groups) (Fig. 6A, B).

Immunohistochemistry for CD68+ cells at the perinonunion sites. (A) Representative images of immunohistochemical staining with polyclonal CD68, a marker for both human and rat macrophages, on tissue samples of perinonunion sites 7 days after cell transplantation. (B) The number of CD68+ cells was significantly greater in the MNC group than the other groups.
Furthermore, immunohistochemical staining for rat-specific granulocytes revealed that the density of rat granulocytes around nonunion sites was significantly greater in the MNC group than in the other groups (CD34+, 171.0 ± 15.0; MNC, 275.6 ± 35.2; PBS, 111.4 ± 7.8 cells/mm2; p < 0.001 for MNC vs. PBS group and p < 0.05 MNC vs. CD34+ group) (Fig. 7A, B).

Immunohistochemistry for granulocytes at the perinonunion sites. (A) Representative images of immunohistochemical staining with rat granulocyte antibody on tissue samples of perinonunion sites 7 days after cell transplantation. (B) The number of rat granulocytes was significantly greater in the MNC group than in the other groups.
These findings suggest that the transplantation of human GM-PB MNCs induces more excessive inflammation at the nonunion sites than that of human GM-PB CD34+ cells.
Apoptosis Analyses at the Cell Transplantation Site
To analyze apoptosis histologically, TUNEL staining was performed using tissue samples obtained 7 days after cell transplantation. Cell apoptosis was more abundant at the cell transplant site of the MNC group compared with the other groups (Fig. 8A). Quantification of TUNEL staining showed that the MNC group had significantly more apoptotic cells compared with the other groups, and there was no significant difference between the CD34+ and PBS groups (CD34+, 22.86 ± 1.534; MNC, 53.29 ± 6.494; PBS, 13.71 ± 1.614, respectively, p < 0.001 for MNC vs. CD34+ and PBS groups, p = NS for CD34+ vs. PBS group) (Fig. 8B).

Apoptosis analyses at the cell transplanted sites. (A) Representative images of TUNEL staining on tissue samples of perinonunion sites 7 days after cell transplantation. (B) The number of TUNEL-positive cells was significantly greater in the MNC group than in the other groups.
These findings suggest that the transplantation of human GM-PB MNCs induced more apoptosis at the nonunion sites than human GM-PB CD34+ cells.
Influence of GM-PB CD34- Cells on GM-PB CD34+ Cell Viability and Apoptosis
We evaluated the influence of GM-PB CD34- cells on the function of GM-PB CD34+ cells. The ratios of trypan blue-positive CD34+ cells with and without coculture with CD34- cells were nearly the same (CD34+, 89.33 ± 0.713; CD34+ with CD34-, 86.43 ± 0.028, p = NS) (Fig. 9A). This finding suggests that GM-PB CD34- cells have no significant influence on the viability of GM-PB CD34+ cells.

Influences of GM-PB CD34- cells on GM-PB CD34+ cells. (A) There was no significant difference between the viability of GM-PB CD34+ cells with coculture with GM-PB CD34- cells and that without coculture with GM-PB CD34- cells. (B) The ratio of TUNEL-positive cells in GM-PB CD34+ cells with coculture with GM-PB CD34- cells was significantly lower than that without coculture with GM-PB CD34- cells. (C) The numbers of EPC colonies from GM-PB CD34+ cells without coculture with GM-PB CD34- cells was significantly greater than that with coculture with GM-PB CD34- cells.
In contrast, the ratio of TUNEL-positive cells in the coculture group was approximately four times higher than that of the noncoculture group (CD34+, 3.63 ± 2.23%; CD34+ with CD34-, 14.37 ± 2.54%, p < 0.05) (Fig. 9B). This finding suggests that GM-PB CD34- cells significantly induce the apoptosis of GM-PB CD34+ cells.
EPC Colony-Forming Units From GM-PB CD34+ Cells
To evaluate the influence of GM-PB CD34- cells on the ability of GM-PB CD34+ cells to function as EPCs in vitro, GM-PB CD34+ cells with or without coculture with GM-PB CD34- cells were analyzed by EPC colony-forming assay (46). The average number of EPC colonies per plate in the noncoculture group was significantly greater than that of the coculture group (CD34+, 24.33 ± 0.67%; CD34+ with CD34-, 18.67 ± 0.33%, p<0.01) (Fig. 9C). This finding suggests that GM-PB CD34- cells decrease the ability of GM-PB CD34+ cells to function as EPCs in vitro.
Discussion
We have previously demonstrated the efficacy of the systemic transplantation of human PB CD34+ cells and the local transplantation of human GM-PB CD34+ cells in nude rats with unhealing fracture (47, 48). Our group recently started a phase I/II clinical trial regarding local transplantation of GM-PB CD34+ cells in patients with nonunion. In the first patient, fracture healing was observed radiographically and clinically by 12 weeks post-cell therapy (40). We also reported the usefulness of the local transplantation of human GM-PB MNCs, which can be prepared in a shorter time and at a lower cost than human GM-PB CD34+ cells, in an unhealing fracture model (14). A case study on BM MNC transplantation for delayed union after tibia fracture has been reported, but there are no studies regarding PB MNC transplantation (65). In both of our previous studies focusing on the local transplantation of human GM-PB CD34+ cells and human GM-PB MNCs immediately after the fracture, immunohistochemistry and real-time RT-PCR for human-specific markers around the fracture sites indicated that one of the mechanisms underlying the angiogenic and osteogenic effects of these cells may be direct differentiation into endothelial and osteoblastic lineages. Furthermore, paracrine effects of the transplanted cells on resident cells were also proved by quantitative real-time RT-PCR for rat-specific primers for angiogenic and osteogenic growth factors or cytokines (14, 48). Regarding the therapeutic potential of GM-PB MNCs and CD34+ cells for fracture healing, the union rate of fractures following human GM-PB MNC local transplantation in our previous study (33% at week 8) (14) was obviously lower compared with the union rate after human GM-PB CD34+ cell transplantation demonstrated in another study (100% at week 8) (48). However, efficacy has yet to be directly compared between the two strategies, and the mechanism of the potency difference has yet to be investigated. Another issue in the previous studies concerns the animal model used for cell transplantation. Because a bone fracture heals in most patients after conventional surgery, candidates for cell-based therapy would be a handful of patients with persistent nonunion. Considering this clinical demand, the ideal setting for cell transplantation would be a chronic nonunion state, not immediately after the fracture as in preclinical studies. Therefore, the state of chronic nonunion was a key factor in this study.
The most striking finding of this study is that human GM-PB CD34+ cell transplantation demonstrated significantly superior potency for bone healing radiographically, histologically, and physiologically compared with human GM-PB MNC transplantation, although blood flow recovery was similar in the two groups. Although the number of transplanted CD34+ cells was approximately equal between the CD34+ and the MNC groups, GM-PB MNCs contained a number of CD34- cells that were depleted in the CD34+ group. The presence of CD34- cells in GM-PB MNCs may be a key factor in the lower therapeutic potency in the MNC group compared to the CD34+ group. As a possible mechanism underlying the difference in therapeutic efficacy between human GM-PB CD34+ cells and MNCs, we focused on inflammation around the fracture site after cell therapy. Human GM-PB MNCs comprising EPCs and osteoblasts (29) contributed to neovascularization and osteogenesis; meanwhile, the high number of inflammatory lymphocytes and monocytes/macrophages present in the CD34- MNC population could induce excessive inflammation at the transplanted site (54). Our group previously exhibited the superior efficacy of human GM-PB CD34+ cells for preserving myocardial integrity and function after MI compared to human GM-PB MNCs, suggesting that inflammatory cells derived from transplanted human GM-PB MNCs may play a key role in accelerating myocardial damage (31). Grounded on the assumption that excessive inflammation induced by total MNCs might have a negative effect on bone healing, we measured the mRNA expression of inflammatory cytokines and the concentration of produced proteins of inflammatory cytokines by real-time RT-PCR and ELISA, respectively, in vitro. All examined inflammatory cytokines showed significantly greater mRNA expression levels in human GM-PB MNCs compared with human GM-PB CD34+ cells from the same donor. Similarly, in ELISA, the expression levels of all inflammatory cytokines produced by human GM-PB MNCs were greater than that of human GM-PB CD34+ cells from the same donor. To evaluate the severity of inflammation induced by cell transplantation in vivo, we conducted immunofluorescent staining for representative inflammatory cells, macrophages and granulocytes. Abundant distribution of the inflammatory cells indicating serious inflammation was observed after transplantation of human GM-PB MNCs compared with GM-PB CD34+ cells. Furthermore, to assess the influence of inflammation induced by CD34- cells on CD34+ cells, we performed a coculture study of GM-PB CD34+ cells with or without GM-PB CD34- cells. This result showed the negative influence of GM-PB CD34-cells on the function of GM-PB CD34+ cells as EPC, which is believed to be efficacious for fracture repair (47, 48). The discrepancy between the results of cell viability assay using trypan blue staining and TUNEL assay is believed to come from the difference in sensitivity for living cells between the two assays. In the current study, the influences of cell–cell interaction arising from the difference in transplanted cell numbers between the two groups was not considered, which should be regarded as a limitation.
It is well known that inflammatory signals play an important role in regulating fracture healing (42, 49). In the early stages of fracture healing, the mRNA expression levels of inflammatory cytokines, including TNF-α, IL-1, and IL-6, are significantly elevated within the first few days after injuries at the perifracture sites (9, 19, 37, 52). These signals induce the recruitment of inflammatory cells and angiogenesis (19, 55). In studies using fracture models in TNF receptor 1 and 2 knockout mice, TNF-α signaling is reported to be necessary for intramembranous ossification and endochondral repair of fracture healing (17, 18). IL-1 and IL-6 also stimulate angiogenesis and promote cartilaginous callus formation that stabilizes the fracture sites (55, 68). A previous study reported that IFN-γ stimulates ALP to activate in human osteoblasts (22).
However, inflammatory cytokines have a positive effect for bone healing only in cases in which their signal is highly regulated and brief (49). Inflammatory molecules are well known for their destructive effects on bone and cartilage in patients with rheumatic diseases. High levels of circulating TNF-α and IL-1 are directly related to bone destruction (51). In a previous study using a mouse arthritis model, the absence of IL-1 was associated with the prevention of bone and joint disease (21). Moreover, in a previous study using a fracture model in diabetic mice, it was suggested that diabetes may interfere with fracture repair because high levels of TNF-α increase the expression of resorptive factors in chondrocytes and lead to accelerated loss of cartilage (1). In the present study, we transplanted 1 × 107 of human GM-PB MNCs containing a large amount of inflammatory cells. In GM-PB MNCs, the mRNA expression levels are several score times, and the protein levels of inflammatory cytokines a few hundred to approximately a million times, greater than those in GM-PB CD34+ cells. It is strongly suggested that the excessive inflammation induced by GM-PB MNCs infiltrating the nonunion sites not only suppresses the favorable efficacy of GM-PB CD34+ cells, such as EPCs in fracture repair, but also inhibits bone repair by impeding endochondral ossification. However, direct evidence of the negative influence on endochondral ossification by GM-PB MNC transplantation was not shown in the present study, which should be regarded as a limitation.
In conclusion, the transplantation of human GM-PB CD34+ cells contributes to functional bone repair even in nonunion models and is more effective than the transplantation of human GM-PB MNCs. These results indicate that cell therapy with human GM-PB CD34+ cells can be a practical and potent strategy for therapeutic vasculo-osteogenesis in the healing of nonunion.
Footnotes
Acknowledgments
The authors would like to thank the animal facility of RIKEN Center for Developmental Biology for providing the space to perform animal surgery and Mr. Garret R. Cooper for his editing assistance in preparing this manuscript. The authors declare no conflicts of interest.
