Abstract
The therapeutic benefits of bone marrow mononuclear cells (BM-MNCs) in many diseases have been well established. To advance BM-MNC-based cell therapy into the clinic for peripheral nerve repair, in this study we developed a new design of tissue-engineered nerve grafts (TENGs), which consist of a chitosan/fibroin-based nerve scaffold and BM-MNCs serving as support cells. These TENGs were used for interpositional nerve grafting to bridge a 10-mm-long sciatic nerve defect in rats. Histological and functional assessments after nerve grafting showed that regenerative outcomes achieved by our developed TENGs were better than those achieved by chitosan/silk fibroin scaffolds and were close to those achieved by autologous nerve grafts. In addition, we used green fluorescent protein-labeled BM-MNCs to track the cell location within the chitosan/fibroin-based nerve scaffold and trace the cell fate at an early stage of sciatic nerve regeneration. The result suggested that BM-MNCs could survive at least 2 weeks after nerve grafting, thus helping to gain a preliminary mechanistic insight into the favorable effects of BM-MNCs on axonal regrowth.
Keywords
Introduction
For severe peripheral nerve injuries, interpositional nerve grafting is required to bridge the formed nerve defect. Tissue-engineered nerve grafts (TENGs) have been extensively developed and are a promising alternative to autografts, the commonly accepted gold standard for nerve grafting.
A TENG is typically composed of a nerve scaffold with added biochemical components (3,10,28). Nerve scaffolds can be prepared with different biomaterials and in various configurations. In our previous research efforts (4,5,32,35), several natural biomaterials, such as chitosan and silk fibroin (SF), and synthetic biomaterials, such as polyglycolic acid (PGA) and poly(L-latic-co-glycolic acid) (PLGA), were employed to engineer nerve scaffolds, whereas a composite scaffold configuration was created, which consisted of a nerve guidance conduit (NGC) and fiber- or filament-shaped lumen fillers. In addition, there have been several published reports concerning the use of chitosan-based biomaterials for the fabrication of neural scaffolds (9,12,21,29). Biochemical cues, furnished by support cells and/or growth factors, are introduced to a nerve scaffold in order to enhance the regenerative effects of TENGs on peripheral nerve regeneration. Schwann cells (SCs) and many types of stem cells have been tried as cellular components of TENGs (16,25). Among them, bone marrow-derived mesenchymal stem cells (BM-MSCs) are incorporated into a chitosan/PLGA composite scaffold or a SF-based scaffold, leading to satisfactory nerve regeneration in rat, dog, and monkey models of sciatic nerve transection (4,14,33,35). The safety of BM-MSC transplantation has been validated in primates, which is important in terms of translation of this research (14).
Mounting evidence shows that BM-MSCs can function as well as SCs in peripheral nerve repair and overcome the limitations associated with clinical use of SCs (13,20). The use of BM-MSCs, however, is still hindered by some drawbacks, including the limited availability and the varied differentiation potential (15,22). Accordingly, more support cells deserve to be examined for their application in the construction of TENGs.
Bone marrow mononuclear cells (BM-MNCs) consist of multiple progenitor/stem cells, including BM-MSCs, hematopoietic stem cells (HSCs), and endothelial progenitor cells (EPCs). Due to better availability and fewer ethical concerns related to use of BM-MNCs compared to other cell types and sources of those cells, an increasing research interest has focused on BM-MNC-based therapy for various diseases (31).
Based on our previous work, this study aimed to engineer a BM-MNC-containing, chitosan/SF-based TENG and evaluate the outcomes of repairing rat sciatic nerve defects by this new TENG. Histological and functional assessments were performed after nerve grafting, and the results, for the first time, suggested that BM-MNC-based transplantation for peripheral nerve repair was feasible.
Materials and Methods
All animal experiments were performed in accordance with the institutional guidelines of the Animal Care and Use Committee at Nantong University, and protocols were approved by the Administration Committee of Experimental Animals (Jiangsu Province, China).
Preparation of BM-MNCs and Green Fluorescent Protein-Labeled BM-MNCs
The BM-MNCs and green fluorescent protein (GFP)-labeled BM-MNCs (GFP-BM-MNCs) were harvested from adult female Sprague–Dawley (SD) rats and transgenic SD rats, respectively, as described previously (26). After animals were euthanized by intraperitoneal (IP) injection of an overdose of sodium pentobarbital solution (Sigma-Aldrich, St. Louis, MO, USA), the femur and tibia bones were dissected aseptically, stripped of connective tissues, and placed in phosphate-buffered saline (PBS, pH 7.4; Corning, Tewskbury, MA, USA) over ice. Both ends of the bone were cut away from the diaphyses, and the bone marrow cavity was exposed. The bone marrow was aspirated with 10-ml syringes that had been rinsed with heparin (Sigma-Aldrich). BM-MNCs were isolated from the marrow aspirate by density gradient centrifugation on Histopaque® 1077 (Sigma-Aldrich) at 400 × g for exactly 30 min at room temperature. After centrifugation, the collected BM-MNCs were washed twice with PBS before cell counting using a hemocytometer. The cell viability was assessed by trypan blue (Sigma-Aldrich) exclusion assay. Finally, the BM-MNCs were suspended in PBS and adjusted to the cell density of 1 × 108 cells/ml prior to use. GFP-BM-MNCs were harvested by the same method as mentioned above. For characterization of BM-MNCs, cells were incubated in several labeled monoclonal antibodies, such as CD3-PE, CD25-FITC, CD29-FITC, and CD44-FITC (Sigma-Aldrich). BM-MNCs that were incubated in PBS served as a control. Phenotype analysis was carried out using flow cytometry on a Becton Dickinson (BD) FACS caliber analyzer (San Jose, CA, USA).
Surgical Procedure
Animal surgery was conducted as previously described (35). Adult female SD rats were anesthetized by IP injection of 3% sodium pentobarbital solution (40 mg/kg body weight; Sigma-Aldrich). The sciatic nerve was exposed by making a skin incision and splitting the underlying muscles in the left lateral thigh. A segment of sciatic nerve was transected and removed, leaving a 10-mm-long defect after retraction of the nerve ends. A total of 63 rats were then randomly divided into three groups (n = 21 each group), referred to as Scaffold, TENG, and Autograft groups. In these three groups, the sciatic nerve defect was, respectively, bridged by three different nerve grafts: 1) a chitosan/SF-based scaffold, which consisted of a chitosan NGC (i.d. 2.0 mm) prepared as described previously (please refer to Chinese patent ZL 0110820.9 for technical details), and 120 of SF fibrous fillers (1.4 cm long, diameter 8 μm) prepared as described previously (34); 2) a BM-MNC-containing, chitosan/SF-based TENG, which was prepared on site by injecting BM-MNCs (30 μl of cell suspension) into a chitosan/SF-based scaffold; 3) an autologous nerve graft, the transected nerve segment.
In addition, another cohort of rats (n = 12) were subjected to nerve grafting to bridge the 10-mm-long sciatic nerve defect by using GFP-BM-MNC-containing, chitosan/SF-based TENGs, which were prepared through the same procedure as that for BM-MNCs-containing, chitosan/SF-based TENGs, but unlabeled BM-MNCs were replaced with GFP-BM-MNCs. At 1, 7, 10, and 14 days after nerve grafting, the regenerating nerves, together with TENGs, were removed from rats (n = 3 each time point) and then subjected to GFP fluorescence monitoring through microscopic observation and immunostaining with anti-NF200 according to the procedure described below.
Behavioral Analysis
The CatWalk XT 9.0 gait analysis system (Noldus, Wageningen, The Netherlands) was used to assess motor functional recovery. At 12 weeks after nerve grafting, the animals (n = 7) in three groups were placed on the right side of a runway consisting of a glass surface and black plastic walls. The animals were motivated to traverse the runway toward the left end where food pellet rewards were located, and the dynamic process of rats was recorded. The scale categorizes a combination of posture, hindlimb movements, hindlimb force, and joint motion. The sciatic function index (SFI) value was calculated by the formula used by Bain et al. (2): SFI = 109.5(ETS – NTS)/NTS – 38.3(EPL – NPL)/NPL + 13.3(EIT – NIT)/NIT – 8.8, where TS is the total toe spread, IT is the intermediate toe spread, PL is the footprint length, N refers to the contralateral uninjured (normal) side, and E refers to the injured side.
Immunohistochemical and Electrophysiological Assessment
At 1 or 2 weeks after surgery, seven rats in each group were sacrificed, and then the nerve tissue in the place of the original nerve defect were excised and cut into 12-mm-thick longitudinal or transverse sections on a freezing microtome (HM400E; Microm, Walldorf, Germany). The sections were allowed to incubate with mouse anti-neurofilament 200 (NF200) (1:400; Sigma-Aldrich) at 4°C for 24 h, followed by a reaction with anti-mouse IgG-Alex-488 (1:500; Sigma-Aldrich) at 4°C overnight and observed under a fluorescent microscope (DMR; Leica, Wetzlar, Germany). Morphometric analysis was performed to measure the length of regenerated nerve fibers.
At 12 weeks after surgery, the remaining rats (n = 7) in each group, on which gait analysis had just finished, were subjected to electrophysiological tests as previously described (34). Briefly, the sciatic nerve on the injured side was reexposed under anesthesia with sodium phenobarbital. Electrical stimuli were applied to the proximal and distal nerve stumps, the compound muscle action potentials (CMAPs) were recorded on the belly of target gastrocnemius muscle, as described previously (32). The conduction velocity of motor nerves was calculated by dividing the CMAP amplitude by the distance between two stimulation sites. The CMAPs recorded on the contralateral uninjured side was used as the normal control.
At the end of electrophysiological tests, immunohistochemistry was carried out to the transverse sections of the regenerated nerve through the same processes as described above. To calculate the number of regenerated nerve fibers, a Q550 IW image analysis system and a Leica QWin software package were used as described previously (32).
Histological Observation and Morphometric Analysis of Target Muscles
At the end of electrophysiological tests, gastrocnemius and anterior tibialis muscles on the injured side and contralateral, uninjured side were harvested and weighed immediately for determining the wet weight ratio of muscles (the wet weight of muscle at the injured side/the wet weight of muscle at the uninjured side). The muscle samples were harvested from the medial belly of gastrocnemius muscles, fixed in 4% paraformaldehyde (Xilong Chemical Co., Ltd., Guangzhou, China), and cut into sections, in which motor endplates were stained with α-bungarotoxin (Sigma-Aldrich) for observation under light microscopy. The contralateral uninjured muscle samples were used as normal controls.
Transmission Electron Microscopy
The sections of regenerated nerves were fixed in precooled 2.5% glutaraldehyde (SCRC, Beijing, China) for 3 h, postfixed with 1% osmium tetraoxide solution (Sigma-Aldrich) for 1 h, dehydrated, embedded in Epon 812 epoxy resin (Fluke, Beijing, China), and cut into 60-nm-thick, ultrathin sections for staining with lead citrate and uranyl acetate (both from Sigma-Aldrich). The stained sections were observed under a transmission electron microscope (JEOL Ltd., Tokyo, Japan), and images were taken from 10 random fields of each section to determine the number of myelin sheath layers, the thickness of myelin sheaths, and the diameter of myelinated nerve fibers using Image Pro Plus software (Media Cybernetics, Rockville, MD, USA).
Statistical Analysis
The data are presented as means ± standard deviation (SD). Statistical comparisons were made using one-way ANOVAs followed by Scheffé's post hoc test. Statistical significance was accepted at a value of p < 0.05.
Results
Characterization of Rat BM-MNCs
The BM-MNCs isolated from rat bone marrow were heterogeneous cells. Phenotype analysis by flow cytometry was conducted for BM-MNC characterization. We noted that about 95% of total cells were CD29 positive, and about 30% of total cells were positive for CD3, CD25, or CD44 (data not shown).
The Fate of Transplanted BM-MNCs
At 1, 7, 10, or 14 days after nerve grafting, the regenerating sciatic nerve, together with the GFP-BM-MNC-containing TENG, was taken from rats, and immunohistochemical analysis with anti-NF200 and fluorescence microscopy was carried out to observe axonal regrowth and track the GFP-BM-MNCs.
NF200 immunostaining showed that axonal regrowth gradually increased with time during the 1- to 14-day period after nerve grafting. Meanwhile, the number of GFP-BM-MNCs inside the chitosan/SF-based scaffold gradually decreased with time at the same early stage of sciatic nerve regeneration, but GFP-BM-MNCs were still present until 14 days after nerve grafting (Fig. 1).
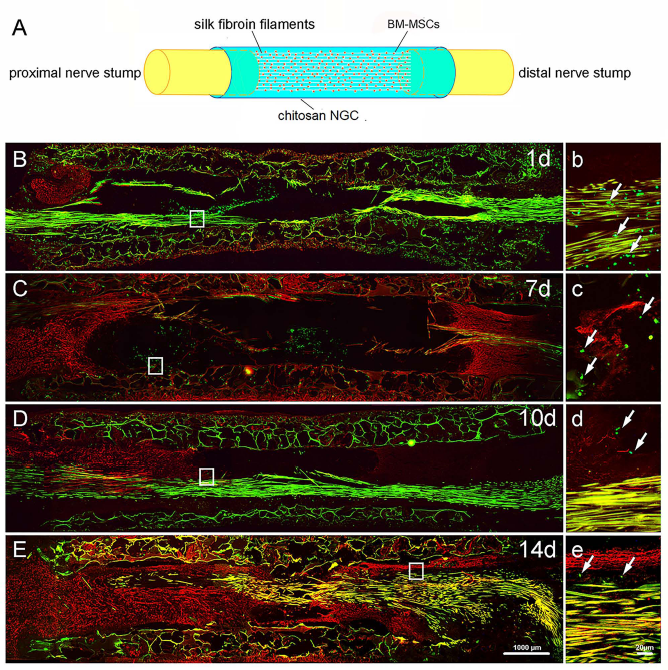
A schematic diagram (A) showing the composition and configuration of GFP-BM-MNC-containing, chitosan/SF-based TENGs. Immunohistochemistry with anti-NF200 (red) showed that axonal growth was observable at 1 (B), 7 (C), 10 (D), and 14 (E) days after nerve grafting by GFP-BM-MNC-containing, chitosan/SF-based TENGs. The GFP fluorescence (green) was monitored by microscopy. The higher magnifications of boxed areas in (B–E) are shown in (b–e), respectively. Scale bars: 1,000 μm (B–E) and 20 μm (b–e). Arrows indicate the GFP-BM-MNCs.
Functional Recovery
To assess the recovery of locomotor function in rats, CatWalk gait analysis was assessed in three animal groups. At 12 weeks after nerve grafting, the animals in both the TENG and autograft groups showed better metatarsophalangeal joint plantar flexion and toe spreading than those in the scaffold group (Fig. 2A–C). The SFI value varies from 0 to −100, with 0 corresponding to normal function and −100 to complete dysfunction. Both the TENG and autograft groups had a greater SFI value than the scaffold group (Fig. 2D).
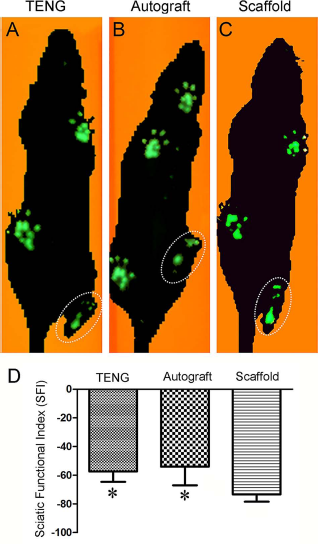
Footprints of animals in TENG (A), autograft (B), scaffold (C) groups, respectively, as detected by CatWalk gait analysis system at 12 weeks after nerve grafting. For representative footprints, see the region indicated by dotted ellipse. Histogram (D) comparing the sciatic function index (SFI) value in the three groups detected at 12 weeks after nerve grafting. *p < 0.05 versus scaffold group.
At 12 weeks after nerve grafting, CMAP recordings revealed the electrophysiological properties of animals in different groups (Fig. 3A–D). The CMAP amplitude value or motor nerve conduction velocity in the TENG or autograft group was significantly greater than that in the scaffold group. The CMAP amplitude or motor nerve conduction velocity in the BM-MNC group was close to those of the autologous group with no significant difference (p > 0.05). As expected, the CMAP amplitude or motor nerve conduction velocity was significantly lower on the injured side than on the contralateral, uninjured side (Fig. 3E, F).

Representative CMAP recordings at 12 weeks after nerve grafting, obtained from the injured side of animals in TENG (A), autograft (B), and scaffold (C) groups, respectively, or on the contralateral, uninjured side of animals (D), which is labeled as normal. The traces were recorded after stimulating the distal and proximal nerve portions, respectively. Histograms showing the CMAP amplitude (E) and the motor nerve conduction velocity (F) detected on the injured side of animals in the TENG, autograft, and scaffold groups as well as on the contralateral, uninjured side of animals, respectively. *p < 0.05 versus scaffold group, and #p < 0.05 versus the contralateral, uninjured (normal) side.
Morphological Observation and Morphometric Analysis of Regenerated Nerves
At 1 and 2 weeks after nerve injury, immunohistochemistry with anti-NF200 was performed to evaluate nerve regeneration across the nerve defect. The axonal growth showed obvious differences in three different groups (Fig. 4A–F). Histologically derived data provided the following interesting comparison: at 1 week after nerve grafting, the length of regenerated nerve fibers was significantly greater in the TENG group and the autograft group than in the scaffold group and significantly greater in the TENG group than in the autograft group (Fig. 4G). At 2 weeks after nerve grafting, however, the length of the regenerated nerve fibers was still significantly greater in the TENG group and the autograft group than in the scaffold group, but there was no significant difference between the TENG group and the autograft group (Fig. 4H). At 12 weeks after nerve grafting, double immunohistochemistry with anti-NF200 and anti-S100β was applied to check the distribution of regenerated nerve fibers at the place of the original defect (Fig. 5A–I). Quantitative comparison showed that the density of regenerated nerve fibers was significantly greater in the TENG group and the autograft group than that in the scaffold group, and no significant difference was present between the TENG group and the autograft group (Fig. 5J).
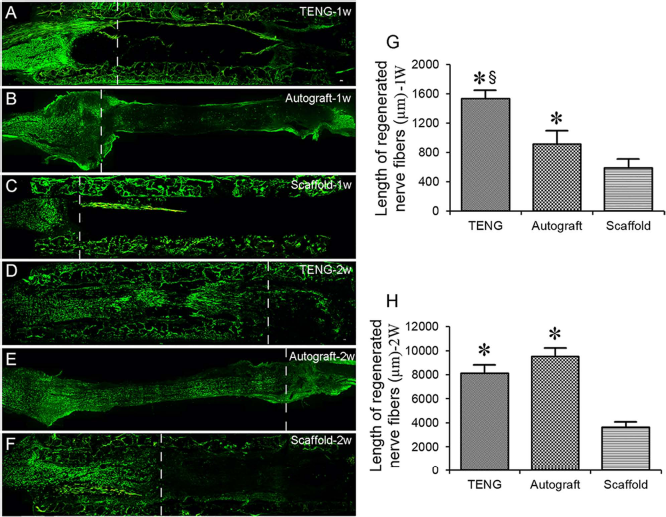
Immunohistochemistry with NF200 (green) of the longitudinal sections of regenerated nerves, which were harvested at 1 week (A–C) and 2 weeks (D–F) after nerve grafting in the TENG group, autograft group, and scaffold group, respectively. The dotted line in micrographs marks the frontline of axon outgrowth. Histograms (G, H) comparing the length of regenerating nerve fibers at 1 week (A–C) and 2 weeks (D–F) after nerve grafting. *p < 0.05 versus scaffold group, §p < 0.05 versus autograft group.

Immunohistochemistry with anti-NF200 (green, A, D, G) and anti-S100β (red, B, E, H) and their merge (C, F, I) for regenerated nerves, which were harvested at 12 weeks after nerve grafting in the TENG group, autograft group, and scaffold group, respectively. Scale bar: 20 μm. Histogram (J) comparing the number of NF200-positive nerve fibers in the distal portion of the nerve defect among three groups. *p < 0.05 versus scaffold group.
Transmission electron microscopy showed that at 12 weeks after nerve grafting, the regenerated, myelinated nerve fibers were observable in the distal stump of the injured nerve (Fig. 6A–C), and they had a clear sheath and basal SC membrane (Fig. 6D–I). Morphometric analysis further indicated that each of the three parameters (the number of the regenerated myelin layer, the diameter of regenerated myelinated fibers, and the thickness of regenerated myelin sheath) was significantly greater in the TENG group and the autograft group than in the scaffold group, and no significant difference was present in each parameter between the TENG group and the autograft group (Fig. 6J–L).

Transmission electron micrographs (A–I) of transverse sections of distal regenerated nerves taken at 12 weeks after nerve grafting from three groups. Scale bars: 5 μm (A–C) and 0.2 μm (D–I). Histograms comparing the diameter of myelinated fibers (J), the thickness of myelin sheath (K), and the number of myelin sheath layers (L) among three different groups. *p < 0.05 versus scaffold group.
Target Muscle Reinnervation and Motor Endplate Analysis
At 12 weeks after nerve grafting, α-bungarotoxin staining and NF200 immunostaining for motor endplates showed that motor endplates within the target gastrocnemius muscle were relatively smaller in the scaffold group than in the TENG group or autograft group (Fig. 7A–C). The wet weight ratio of the target gastrocnemius or tibialis anterior muscles were significantly greater in the TENG group and the autograft group than in the scaffold group, respectively, and each ratio showed no significant difference between the TENG group and the autograft group (Fig. 7D, E).
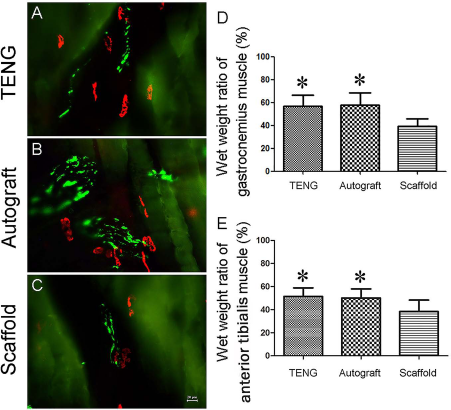
Micrographs (A–C) of longitudinal sections of gastrocnemius muscles at the injured side after α-bungarotoxin staining of motor endplates and immunohistochemistry with anti-NF200 (green) of regenerated axons were applied to animals in three groups at 12 weeks after nerve grafting. Scale bar: 20 μm. Histograms (D, E) comparing the wet weight ratio of gastrocnemius and anterior tibialis muscles among three groups. *p < 0.05 versus scaffold group.
Discussion
In this study, we developed a new design of TENGs, which were formed by introduction of BM-MNCs into chitosan/SF-based nerve scaffolds as a cell component of TENGs. As is well known, BM-MNCs are a mixed population of hematopoietic and nonhematopoietic cells (mesenchymal cells). Their safety has been tested and confirmed in animal models. They have few adverse effects in the human body and exert beneficial effects on the nervous system (8,17,26,27,30). Importantly, BM-MNCs represent a cell source that can be harvested in sufficient numbers and transplanted on the same day (23). In comparison to BM-MSCs, which have proven a promising source of support cells for TENGs, use of BM-MNCs may enable avoidance of some puzzling problems, including the standardization and optimization of cell treatment as well as the increased cost, time, and risk associated with cell treatment (19). As to chitosan/SF-based nerve scaffolds, their usefulness for peripheral nerve repair has been evaluated in our previous studies and was found to possess good biocompatibility, suitable degradation kinetics, and other favorable properties of the related biomaterials and a well-established scaffold configuration.
Density gradient centrifugation, as a common method for isolation of BM-MNCs, was used in this study, and then trypan blue exclusion was adopted for a cell viability assay. Flow cytometry-based cell phenotype analysis was used for cell characterization.
A well-defined rat model of sciatic nerve transaction was established to evaluate the feasibility of BM-MNC-containing, chitosan/SF-based TENGs in treating peripheral nerve injury, and the autologous nerve graft and chitosan/SF-based scaffold served as positive and negative controls, respectively.
Immunohistochemistry and morphometric analysis demonstrated that the injured sciatic nerve was regenerated after nerve grafting by our developed TENGs with an efficacy similar to that observed with autografts, but better than that attained with a chitosan/SF-based scaffold. Electrophysiological assessments provided an important index for the conductive function of sciatic nerves, while behavior examination with CatWalk gait analysis tracked the changes in the motor function of sciatic nerves. The analysis of some parameters in these two measurements confirmed that sciatic nerve regeneration was accompanied by functional recovery due to BM-MNC transplantation, compared to controls. Target muscle reinnervation is an important measure for peripheral nerve regeneration. The data from immunostaining for motor endplates in gastrocnemius muscle and determination of the wet weight ratio of gastrocnemius and anterior tibialis muscles collectively confirmed that BM-MNCs enhanced target organ reinnervation.
Moreover, we used GFP-BM-MNCs, which had been isolated from GFP transgenic rats, to track the location of BM-MNCs within the injured sciatic nerve and track the fate of cells within the 2-week period after nerve grafting. The results suggested that BM-MNCs are able to survive for at least 2 weeks after transplantation.
As a matter of fact, the therapeutic potential of BM-MNCs has already been shown in the treatment of blood disorders (1,6,7,18), musculoskeletal disorders (11), and cardiovascular disease (24,36). In contrast, the use of BM-MNCs for nervous system disorders is still in its infancy stage.
This study showed two major features compared to our previous studies. First, an adequate number of BM-MNCs could be obtained from autologous bone marrow and the use of BM-MNCs as support cells induced few adverse effects on the body. Second, GFP-BM-MNCs from transgenic rats were used to trace the cell fate after cell transplantation, thus contributing to gaining a preliminary insight into the promoting action of BM-MNCs on peripheral nerve regeneration.
In conclusion, a new design of TENGs was used to bridge a 10-mm-long sciatic nerve defect in rats. A series of histological and functional assessments provided evidence that BM-MNC-containing, chitosan/SF-based TENGs may hold a promise for future therapies for peripheral nerve injury.
Footnotes
Acknowledgments
We thank Professor Jie Liu for his help in manuscript revision. This work was graciously supported by the Hi-Tech Research and Development Program of China (863 Program, Grant No. 2012AA020502), National Key Basic Research Program of China (973 programs, Grant No. 2014CB542202), National Natural Science Foundation of China (Grant No. 81130080 and 81402447), and Technology Project of Nantong (HS2014053). The authors declare no conflicts of interest.
