Abstract
Most pancreas and islet grafts are recovered from brain-dead (BD) donors. In this study we characterized the early inflammatory response induced by brain death in pancreata and islets from nonhuman primate donors and evaluated the effect of targeted anti-inflammatory intervention in the protection of pancreatic islets prior to transplantation. BD donors were monitored for 6 h and assigned to three experimental groups: group 1: BD-untreated donors (BD-UT) (n = 7), group 2: BD + donor pretreatment with IL-1ra (n = 6), and group 3: non-BD animals serving as controls (n = 7). We observed an IL-1ra-dependent reduction in the mobilization and activation of neutrophils from bone marrow and a significantly reduced accumulation of CD68+ leukocytes in the pancreas and islets after brain death induction. Donor treatment with IL-1ra significantly decreased chemokine mRNA expression (MCP-1, IL-8, and MIP-1a) and attenuated the activation of circulating neutrophils and intraislet macrophages as demonstrated by a reduction in intracellular IL-1β, IL-6, MCP-1, and MIP-1α expression. As a result, IL-1ra dramatically improved viability, mitochondrial membrane polarity, and islet engraftment in mice transplanted using a minimal islet mass. These results suggest that early immunomodulation targeting inflammation in the BD donor may represent an effective therapeutic strategy to improve islet quality and function prior to transplantation.
Keywords
Introduction
It is currently accepted that severe brain injury resulting in brain death triggers a generalized inflammatory response that is detrimental to organ quality and function in the posttransplant period. Studies analyzing different donation modalities have consistently shown superior long-term function and graft survival in organs obtained from living donors when compared to those recovered from brain-dead (BD) donors (2,12,32,34). However, the pathophysiological mechanisms responsible for the discrepancies observed in graft survival between living and deceased donors remain poorly understood. In the last decade, several groups have reported that generalized inflammation triggered by brain death plays a key role in enhancing donor tissue immunogenicity and results in a rapid and aggressive alloresponse by the host associated with increased risk of delayed graft function and rejection after transplantation (23,24,34).
There is substantial evidence demonstrating that early expression of proinflammatory mediators is partly responsible for inducing cell migration and secondary tissue injury during the inflammatory cascade triggered by ischemia/reperfusion in various animal models (33). Interleukin-1β (IL-1β) is of particular interest due to its central role in the orchestration and amplification of the acute inflammatory response to tissue injury (9). Recent work by our group and others suggest that the IL-1β pathway is highly upregulated during the acute phase response and the inflammatory process triggered by brain death (7). IL-1β regulates the production and release of proinflammatory cytokines, chemokines, and cellular elements that further potentiate the immune response and prompt tissue infiltration by inflammatory cells of the innate and adaptive immune system. This in turn leads to production of reactive oxygen species, edema, necrosis, and apoptosis, which are considered to be the main driving forces behind acute ischemic and inflammatory damage to visceral and thoracic organs destined for transplantation (25).
In pancreatic islets, the role of IL-1β in the pathogenesis of diabetes mellitus (types 1 and 2), as well as islet dysfunction after pancreatic islet transplantation, has been extensively studied (1,10,13). In the pancreas, IL-1β is produced and secreted mainly by activated macrophages, neutrophils, endothelial cells, and β-cells within the islet (15). Numerous reports indicate that secreted IL-1β mediates the activation of nuclear factor κ-light-chainenhancer of activated B cells (NF-κB)-dependent proinflammatory pathways within the islet cells and has been linked to impaired insulin secretion, mitochondrial damage, and increased oxidative injury that ultimately result in increased levels of apoptosis and necrosis of β-cells (4,13,28).
In this study, we tested the protective effects of organ donor pretreatment with IL-1 receptor antagonist (IL-1ra) on pancreas and islet quality and function. For this purpose, we used a nonhuman primate (NHP) model of brain death to simulate the initial cascade of inflammatory events that follow acute explosive neurological injury. Our results provide evidence indicating that blockade of the IL-1β pathway at the donor level leads to improved quality and functional potency of isolated pancreatic islets by preventing innate immune activation and attenuating the inflammatory response to brain death-induced ischemia/reperfusion injury of the pancreas.
Materials and Methods
Animals and Animal Care
Male rhesus macaques (n = 20; age range 4–16 years, weight 3.0–14.0 kg) used in this study were obtained from the Wisconsin National Primate Research Center, University of Wisconsin-Madison, Madison, WI, USA. All animals were prescreened negative for TB, herpes B, SRV, SIV, and STLV-1 and housed in accordance with NIH and USDA guidelines. All donor/recipient transplant protocols were approved by Institutional Animal Care and Use Committee at the University of Wisconsin–Madison.
Experimental Design
Donor animals were allocated to one of three groups: group 1: brain death induction and standard donor management for 6 h prior to organ recovery (BD-UT, n = 7); group 2: brain death induction, standard donor management, and continuous administration of IL-1ra for 6 h prior to organ recovery (BD + IL-1ra, n = 6); group 3: Non-BD primates served as the control group (NBD, n = 7). Control animals were sedated, anesthetized, and ventilated for 1 h prior to organ recovery. A subset of control animals (n = 3) were anesthetized, intubated, and monitored for 6 h in order to document changes in hemodynamic parameters. All animals in this study were euthanized by exsanguination under anesthesia during in situ flush with cold UW preservation solution (SPS-1 Static Preservation Solution; Organ Recovery Systems, Chicago, IL, USA).
Brain Death Induction and Donor Management
A detailed description of the brain death protocol used in this study can be found elsewhere (7). In brief, animals were anesthetized, mechanically ventilated, and monitored. A 16F Foley catheter (Bard Medical, Covington, GA, USA) was placed in the extradural space of the cranial fossa and gradually inflated until hemodynamic and neurologic signs of brain stem herniation were documented (35). BD animals were monitored for 6 h and received IV fluid resuscitation to achieve a stable mean arterial pressure and urinary output. Arginine vasopressin (AVP; 0.01–0.04 U/min; American Regent, Shirley, NY, USA) was given IV when mean arterial blood pressure dropped below 45, and dopamine (0.5–5.0 U/kg/min, IV; American Regent) was started when hypotension refractory to AVP treatment was documented.
Study Drug
IL-1ra (Anakinra; Amgen, Inc., Thousand Oaks, CA, USA) was administered to BD animals as an IV bolus (10 mg/kg) at brain death induction and then maintained by continuous infusion (3.3 mg/kg/h) throughout the 6-h experimental period.
Islet Isolation, Purification, and Culture
Pancreatic islet isolation was performed using a modification of the semiautomated method previously described by Ricordi et al. (27). The pancreas was digested using Liberase CI (Roche, Indianapolis, IN, USA), mechanically disrupted, and islets were purified by Biocoll gradient centrifugation in a COBE 2991 cell processor (COBE, Lakewood, CO, USA) (29). Purified islets were then cultured in CMRL 1066 (Mediatech, Herndon, VA, USA) supplemented with 1% penicillin/ streptomycin (Thermo Scientific, Waltham, MA, USA), 10% FBS (Thermo Scientific) at 25°C in an incubator with 5% CO2 for 24 h prior to assessment.
Histology and Pathology
Pancreas biopsies were fixed in 10% buffered formalin (Sigma-Aldrich, St. Louis, MO, USA), embedded in paraffin, and mounted onto slides for staining with anticluster of differentiation 68 (CD68; clone KP1; Dako North America, Carpinteria, CA, USA) at 1:1,000 dilution, guinea pig anti-insulin 1:2,000 (Sigma-Aldrich), and hematoxylin and eosin (H&E) (Newcomer Supply, Middleton, WI, USA) for evaluation by microscopy. Slides were analyzed blindly, and quantification was performed using ImageJ software (NIH, Bethesda, MD, USA). Color separation, background subtraction, and automatic thresholding and particle analysis algorithms were used to quantify positive staining area and cell counts for each image.
RNA Extraction and Reverse Transcription
A 1-mm3 sample of pancreatic tissue was homogenized 1 ml TRIzol (Invitrogen, Carlsbad, CA, USA) and purified using the Qiagen RNeasy Kit (Qiagen, Germantown, MD, USA) following instructions for RNA cleanup and including the on-column DNase digestion step. RNA (0.5 μg) was reverse transcribed using Omniscript Reverse Transcription kit (Qiagen) following the manufacturer's instructions.
Quantitative PCR and Data Analysis
Quantitative PCR was performed using Taqman Universal PCR Master Mix and a Taqman gene expression assay (Applied Biosystems, Inc., Foster City, CA, USA) with human and primate gene-specific primers (Applied Biosystems, Inc.) on a GeneAmp 5700 or 7500 Sequence Detection System (Applied Biosystems, Inc.). We used commercially available rhesus macaque validated primers (Life Technologies, Grand Island, NY, USA) to determine changes in gene expression for chemokine C-C motif ligand 2 [CCL2; monocyte chemotactic protein 1 (MCP-1), Rh02621753_m1], IL-8 [chemokine C-X-C motif ligand 8 (CXCL8); Rh02789781_m1], and CXCL10 [interferon γ-induced protein 10 (IP-10); Rh02788358_m1], and used β-actin (Rh03043379_gH) as the endogenous control. Fold change was calculated using the DDCt method relative to untreated (UT) with hypoxanthine-guanine phosphoribosyltransferase as a second endogenous control. Error was calculated as fold change ranges within one standard deviation of the mean.
Quantification of Serum Cytokine Levels
Serum levels of IL-1ra were determined by enzyme-linked immunosorbent assay (ELISA; Cell Sciences, Canton, MA, USA) following the manufacturer's instructions. Circulating levels of IL-2, IL-4, IL-5, IL-6, tumor necrosis factor-α (TNF-α), and interferon-γ (IFN-γ) were determined using a NHP Th1/Th2 cytokine cytometric bead array (BD Biosciences, San Diego, CA, USA) analyzed using a FACSCalibur flow cytometer (BD Biosciences).
In Vitro and In Vivo Assessment of Islet Function
For the glucose-stimulated insulin secretion assay, islets were handpicked into oxygen-saturated basal Krebs-Ringer Bicarbonate Buffer (KRB; 137 mM NaCl, 4.7 mM KCl, 1.2 mM KH2PO4, 1.2 mM MgSO4–7H2O, 2.5 mM CaCl2–2H2O, 25 mM NaHCO3; Sigma-Aldrich), 0.25% bovine serum albumin (BSA; Sigma-Aldrich), and 3.3 mM glucose (Acros Organics; Thermo Scientific), followed by incubation at 37°C for 30 min in a 5% CO2/95% air incubator. In each group, 10 islets from the equilibration cultures were then transferred to fresh oxygen-saturated KRB containing either 3.3 mM or 16.7 mM glucose and incubated an additional 60 min in a 37°C water bath with gentle shaking. Secreted insulin in the media was measured by ELISA (Millipore, Billerica, MA, USA) and values normalized to extracted islet DNA (Quant-iT Picogreen; Invitrogen). Islet viability was evaluated by fluorescence microscopy as previously described in other publications (18).
Nonobese diabetic/severe combined immunodeficient (NOD/scid) male mice (>8 weeks of age), purchased from The Jackson Laboratory (Bar Harbor, ME, USA), were used as islet recipients. Mice were rendered diabetic by a single intraperitoneal injection of streptozotocin (150 mg/ kg) (Sigma-Aldrich), and only animals with sustained blood glucose level >300 mg/dl for 3 consecutive days were used in this assay. Islets (1,000 IEQ) were transplanted under the kidney capsule of diabetic mice, and blood glucose levels followed daily for a period of 30 days using an Ascensia Elite glucometer (Bayer, Burr Ridge, IL, USA).
Multiparametric Flow Cytometry
For assessment of islet cell populations, purified islets were dispersed into single-cell suspensions by incubation at 37°C in 0.05% trypsin/0.53 mM ethylenedi-aminetetraacetic acid (Mediatech) for 5 min, followed by gentle trituration through a narrow-gauge pipette tip. Dispersed cells were then resuspended in staining buffer. For intraislet assessments, cells were suspended in 1× phosphate-buffered saline (PBS; Mediatech) with 1% BSA (Sigma-Aldrich).
For viability assessment of islet cell populations, dispersed islet cells were suspended in modified KRB containing 3.3 mM glucose and 0.25% BSA and stained with probes for viability [fluorescein diacetate (FDA); Sigma-Aldrich], apoptosis [R-phycoerythrin annexin V, Invitrogen; benzyloxycarbonyl-Val-Ala-Asp (OMe) fluoromethylketone-fluorescein isothiocyanate (VADFMK-FITC); Promega, Madison, WI, USA], and mitochondrial membrane polarity (JC-1; Invitrogen). TO-PRO3-iodide (Invitrogen) was added immediately prior to acquisition in order to identify necrotic cells. Samples were analyzed on a BD FACS Calibur with Cytek upgrade.
For assessment of leukocyte populations, peripheral blood mononuclear cells were generated from blood that was processed in lymphocyte separation medium (Mediatech) and resuspended in 1× PBS with 1% BSA. They were then incubated with 10 μl FC-Block (Biolegend, San Diego, CA, USA) and stained for surface: human leukocyte antigen-DR, CD11b, CD3, CD4, CD8, CD20 (BD Biosciences), CD14, and CD11c (Biolegend). Cells were then fixed and permeabilized using the BD Cytofix/CytoPerm kit, and stained for internal antigens IL-6, IL-8, TNF-α, CCL2/MCP-1, macrophage inflammatory protein 1a (MIP-1a/CCL3; BD Biosciences), and IL-1β (Abcam, Cambridge, MA, USA). Samples were analyzed on a BD LSRII and analyzed using FlowJo (Treestar Inc., San Carlos, CA, USA).
Statistical Analysis
Statistical analysis was performed using the GraphPad Prism V4.03 (San Diego, CA, USA). All data are shown as mean ± standard error of the mean. Comparison of more than two groups was performed by one-way ANOVA or Kruskal–Wallis test with either Bonferroni's or Dunn's posttest correction where appropriate. Differences between treatment groups were considered significant with a value of p < 0.05. Survival analysis was performed with Kaplan– Meier survival analysis using the log-rank test.
Results
Hemodynamic Alterations Induced by Brain Death and Drug Delivery to BD Nonhuman Primates (NHPs)
Following the inflation of the intracranial catheter, we observed a sharp increase in systolic and diastolic blood pressure (Fig. 1A) along with sustained tachycardia (Fig. 1B) and a progressive increase in urinary output suggestive of diabetes insipidus (Fig. 1C). BD-UT and BD + IL-1ra-treated animals evidenced a decrease in blood pressure at 30 min post-brain death induction followed by a return to hemodynamic stability comparable to that of NBD donors for the remaining experimental period.

Hemodynamic alterations induced by brain death induction in nonhuman primates. Mean arterial (A) blood pressure, (B) heart rate, and (C) urinary output were monitored for a 6-h period following brain death induction. (D) Serum interleukin-1 receptor antagonist (IL-1ra) levels measured at 0, 60, 180, and 360 min after brain death induction. Non-brain-dead (NBD) controls (dashed line, n = 3), brain-dead untreated (BD-UT) group (black line, n = 7), and BD + IL-1ra group (gray line, n = 6). Data are expressed as mean values ± SEM (*p < 0.05, **p < 0.01, and ***p < 0.001 between NBD controls and BD-UT group and †p < 0.05, ††p < 0.01, and †††p < 0.001 between NBD and BD + IL-1ra group).
We measured serum levels of circulating IL-1ra in the serum of donors at 0, 60, 180, and 360 min post-brain death induction and documented a significant increase to desired therapeutic levels in IL-1ra-treated animals (p < 0.001) (Fig. 1D).
IL-1ra Mitigates the Brain Death-Induced Recruitment of Innate Immune Cells Into Peripheral Circulation
We examined the phenotype and activation state of circulating innate immune cell populations by flow cytometry. To assess the time-dependent changes in circulating leukocyte populations, we measured the relative change from baseline to organ recovery. At 6 h post-brain death induction, the BD-UT group showed a significant increase in polymorphonuclear neutrophil (PMN) levels (374.33 ± 187.70%) when compared to baseline PMN counts (p < 0.01). Conversely, levels of PMN decreased in NBD and IL-1ra-treated animals (50.5 ± 38.2% and 39.8 ± 11.2%, respectively). Monocyte levels showed a similar trend, with a negligible increase observed in the BD-UT group at 6 h and relatively no change observed in the NBD and IL-1ra-treated groups; however, these results were not statistically significant (Fig. 2A). To determine the level of activation, we analyzed intracellular cytokine staining and documented an increase in IL-1β, IL-8, and MIP-1α expression in circulating neutrophils (Fig. 2B) and IL-1β, IL-6, and MCP-1 expression in circulating monocytes obtained from the BD-UT group (Fig. 2C). However, these changes did not achieve statistical significance. The frequency of positive-staining cells from T0 to T6 in animals in the BD + IL-1ra group demonstrated a decrease that paralleled changes observed in the NBD group. In addition, we observed a significant increase in serum IL-6 levels from BD-UT donors (p < 0.05), which was suppressed to control levels by IL-1 blockade. Circulating levels of IL-2, IL-4, IL5, TNF-α, and IFN-γ were not statistically different among all three groups (Fig. 2D).

Peripheral blood mononuclear cells were analyzed by flow cytometry prior to brain death induction (T0) and 6 h after (T6), at organ recovery. (A) Changes in neutrophil, monocyte, and myeloid dendritic cells in blood from brain death induction (T0) to 360 min (T6) postinduction. (B) Changes in neutrophil expression of IL-1β, IL-6, IL-8 [chemokine C-X-C motif ligand 8 (CXCL8)], monocyte chemotactic protein 1 [MCP-1; chemokine C-C motif ligand 2 (CCL2)], and macrophage inflammatory protein 1a (MIP-1a; CCL3) between T0 and T6 and (C) changes in monocyte expression of IL-1β, IL-6, IL-8, MCP-1, and MIP-1α between T0 and T6 in all groups. (D) Quantification of proinflammatory cytokine levels (IL-2, IL-4, IL-5, IL-6, TNF-α and IFN-γ) in serum between T0 and T6 was performed by ELISA. Data expressed as mean percent change from baseline ± SEM. NBD (n = 7), BD-UT (n = 7), and BD + IL-1ra (n = 6) (*p < 0.05 and **p < 0.01).
Brain Death Induces Pancreatic Injury That Is Attenuated by Donor Pretreatment with IL-1ra
To determine the overall impact of brain death on pancreatic injury, we analyzed tissue sections of pancreata recovered from BD donors and compared them to those obtained from NBD controls and IL-1ra-treated animals. Although pancreatic H&E sections obtained from BD donors did not exhibit gross changes in morphology and cytoarchitecture (Fig. 3A–C), immunohistochemical analysis of several sections stained with anti-insulin antibodies revealed decreased insulin-staining intensity and disruption of the cytoarchitecture and morphological integrity of the islets from BD donors when compared to those of NBD controls or IL-1β-treated animals (Fig. 3D–F). In addition, levels of serum amylase were markedly increased at 6 h post-brain death induction in the BD-UT donor group, but remained unchanged in NBD and IL-1ra-treated donors (BD-UT: 473.1 ± 86.2 U/L; n = 5 vs. NBD: 270.5 ± 43.2 U/L; n = 4 vs. BD + IL-1ra: 268.0 ± 33.4 U/L; n = 4, p = 0.028) (Fig. 3G).

Hematoxylin and eosin (H&E) and insulin staining. H&E-stained representative sections at 200× magnification of pancreas tissue in (A) NBD (n = 7), (B) BD-UT (n = 7), and (C) BD + IL-1ra (n = 6) treated donors at 6 h post-brain death induction. Insulin staining representative sections at 100× magnification (inset 400× magnification) of pancreas sections in (D) NBD, (E) BD-UT, and (F) BD + IL-1ra-treated donors. (G) Quantification of serum amylase levels at 6 h post-brain death induction. Data are expressed as mean values ± SEM (*p < 0.05).
Donor Management with IL-1ra Prevents Macrophage Migration, Accumulation, Activation, and Differentiation Within Islets Recovered From BD Donors
Next, we investigated the migration of PMNs and monocytes into pancreatic tissue. Immunohistochemical staining of the pancreas showed that a higher number of CD68+ staining cells accumulated in pancreas from BD-UT donors and in some cases exclusively surrounding pancreatic islets when compared to NBD controls and IL-1ra-treated animals (11.47 ± 0.03% vs. 2.32 ± 0.01% vs. 4.70 ± 3.63%, respectively, n = 5 for each group, p < 0.001) (Fig. 4A–D). Flow cytometric analysis also revealed an increase in activated macrophages/dendritic cells (CD11C+ cells) in dispersed isolated islets from BD-UT donors when compared to those receiving IL-1ra treatment and, more importantly, compared to NBD donors (Fig. 5A–D). These CD11C+ cells had significantly elevated expression of IL-1β (p < 0.01) and IL-8 (p < 0.01) when compared to those isolated from NBD donors and those obtained from IL-1ra-treated BD donors (Fig. 5E). Expression of MCP-1 and MIP-1α was also elevated in DCs from BD-UT islets, but this comparison did not achieve statistical significance.

Migration and infiltration of inflammatory leukocytes into pancreatic tissue 6 h after brain death induction. Representative images at 400× magnification of CD68+ cells in pancreas biopsies and islets (outlined) of (A) NBD (n = 7), (B) BD-UT (n = 7), and (C) BD + IL-1ra (n = 6)-treated animals. (D) Images were analyzed and quantification performed using image analysis software. Data are expressed as mean values ± SEM (*p < 0.05 and **p < 0.01).
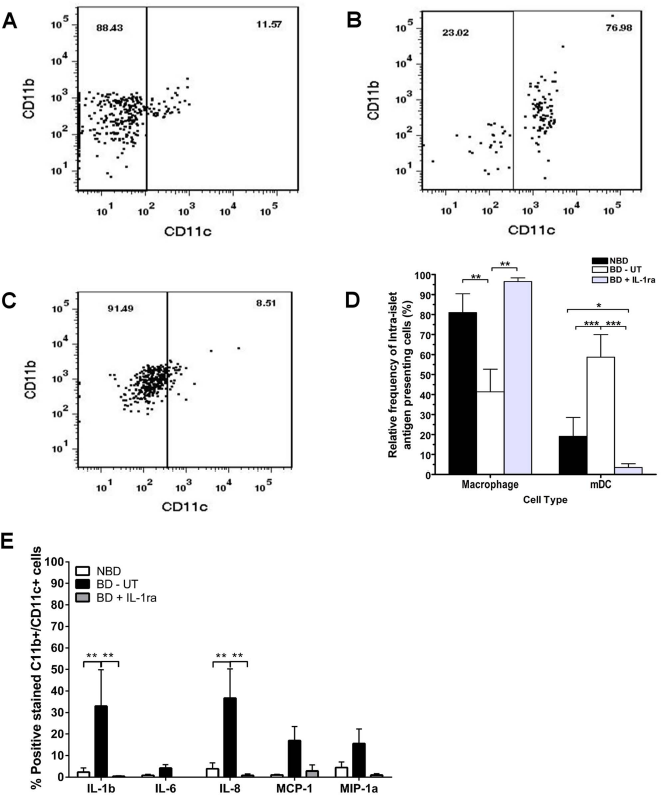
Relative frequency of immature monocytes and activated macrophages between groups. Dispersed islets were stained using mAb specific to CD11b and CD11c and analyzed by flow cytometry. The relative frequencies of immature monocytes (CD11b+, CD11c-) and activated macrophages/myeloid dendritic cells (mDCs, CD11b+, CD11c+) in islets isolated from (A) NBD (n = 7), (B) BD-UT (n = 7), and (C) BD + IL-1ra (n = 6) were measured and compared between groups (D). Intraislet activation of mDCs was then analyzed in dispersed islets stained for activation markers IL-1β, IL-6, IL-8, MCP-1, and MIP-1a. The scatter plots are representative of one experiment. Data are expressed as mean values ± SEM (*p < 0.05, **p < 0.01, and ***p < 0.001).
IL-1 Receptor Blockade Decreased the Expression of Proinflammatory Chemokines in the Pancreas of BD Donors
We measured chemokine expression to determine if systemic delivery of IL-1ra was decreasing leukocyte recruitment to tissue by interfering with these signals. We observed a significant increase in the level of CCL2 (MCP-1), CXCL8 (IL-8), and CXCL10 (IP-10) transcripts in pancreas tissue from BD-UT donors. Animals receiving IL-1ra treatment had a marked downregulation of chemokine mRNA, and the expression levels were similar to those observed in the NBD control group (Fig. 6A–C).

Analysis of chemokine mRNA expression in pancreas tissue 6 h after induction of brain death. Expression levels of (A) CCL2 (MCP-1), (B) CXCL8 (IL-8), and (C) CXCL10 (IFN-γ-induced protein 10; IP-10) were analyzed and compared in all groups. Data are shown as relative fold induction in BD-UT (n = 7) and IL-1ra (n = 6)-treated donors, compared to control UT pancreatic tissue from NBD control animals (n =7). Data are shown as mean values ± SEM (*p < 0.05 and **p < 0.01).
Donor Pretreatment with IL-1 Receptor Blockade Increased Islet Functional Potency In Vitro and In Vivo
Isolated islets were dispersed and analyzed by flow cytometry using probes designed to test cell viability (FDA and Topro3), apoptosis (annexin V and VADFMK), and mitochondrial membrane polarity (JC-1). Islets recovered from BD-UT donors showed lower viability (%), higher apoptosis (%), and significantly decreased MMP (%) when compared to their NBD counterparts. More importantly, islets from donors pretreated with IL-1ra did not exhibit a reduction in viability or MMP and maintained an apoptosis level comparable to that of islets from NBD donors (Fig. 7A–D). In addition, isolated islets from donors in all groups were transplanted into the renal subcapsular space of diabetic NOD/scid mice (1,000 IEQ/mouse) to evaluate functional potency. Recipients transplanted with islets from NBD (n = 13) and IL-1ra-treated donors (n = 11) showed a markedly reduced median time to cure (7 and 3 days, respectively) and significantly higher cure rate of 72.7% for NBD donors and 84.6% for recipients of IL-1ra-treated islets (p < 0.05) when compared to animals transplanted with islets from BD-UT donors (n = 10) in which median time to cure was 11 days and overall cure efficiency rate was 33.3% at 14 days postimplantation (p < 0.05) (Fig. 8). No differences between time to cure, cure percentage, and blood glucose were observed between recipients of islets from BD donors treated with IL-1ra and recipients from the NBD control group. We did not observe significant differences in postisolation yield and glucose-stimulated insulin release or viability by fluorescein diacetate/propidium iodide (FDA/PI) assay 24 h after isolation (Table 1).

Donor pretreatment with IL-1ra prevents mitochondrial dysfunction induced by brain death and the proinflammatory response. Islets cultured for 24 h after isolation were evaluated for changes in viability by (A) fluorescein diacetate (FDA) staining, (B) apoptosis by benzyloxycarbonyl-Val-Ala-Asp (OMe) fluoromethylketone (VADFMK) staining, and (C) mitochondrial integrity and function by JC-1 staining. (D) Data shown are the percentage of islet cells with that are viable, apoptotic, and have polarized Δψm indicated by high JC-1 red/JC-1 green fluorescence. Data are expressed as mean ± SEM [NBD (n = 7), BD-UT (n = 7), and BD + IL-1ra (n = 6), **p < 0.01].

Percent diabetes-free NOD/scid mice after xenogeneic islet transplantation. NOD/scid mice received 1,000 islet equivalnts (IEQs) implanted in the subcapsular space of the left kidney. Mice in the NBD control group (n = 11), BD-UT group (n = 10), and BD + IL-1ra group (n = 13) were followed for 30 days after implantation and monitored daily for return to normoglycemia (defined as random blood glucose <200 mg/dl). The differences between groups were significant (p = 0.0428 established by the log-rank test).
Islet Isolation and Quality Control Assessment of Isolated Islets

BD, brain-dead donor; IL-1ra, interleukin-1 receptor antagonist; IEQ, islet equivalent; FDA/PI, fluorescein diacetate/propidium iodide; GSIS S.I., glucose-stimulated insulin secretion stimulation index.
Discussion
In this study, we investigated the effect of early antiinflammatory intervention in the BD donor and its impact on pancreas quality and islet function following islet isolation and transplantation. Brain death is characterized by hemodynamic instability and the release of vasoactive hormones that result in tissue hypoperfusion and rapid activation of innate immunity (2,22). This systemic and organ-specific inflammatory response has been associated with increased tissue immunogenicity and host alloreactivity, known to increase the risk of delayed graft function and acute rejection (23,24).
The role of proinflammatory cytokines has been documented extensively in the pathogenesis of type 1 and type 2 diabetes mellitus as well as in the inflammatory process associated with islet isolation and subsequent intraportal transplantation (3,5). We chose to target the IL-1 receptor based on previous work showing IL-1ra-protected islets from IL-1β-induced necrosis and apoptosis triggered by cytokines in culture from and data from our own laboratory showing that during brain death the IL-1 pathway is strongly upregulated in NHPs following brain death induction (7,28). In the context of islet transplantation, Contreras et al. showed a detrimental effect of brain death in postisolation islet yields and islet functional potency both in vitro and after intraportal transplantation into diabetic immunodeficient mice (6). Although we did not observe significant differences in islet yields, our results did indicate that brain death results in a marked reduction in islet cell and exocrine tissue viability as shown by increased serum amylase levels, a substantial loss in insulin staining, and alterations in morphological integrity of the islets within the pancreas.
We documented a rapid mobilization of neutrophils into peripheral circulation 6 h after brain death induction. Interestingly, donor animals receiving treatment with IL-1ra did not show significant changes in the numbers of circulating immunocytes compared to NBD controls. In addition, intracellular staining of these cell populations showed that IL-1ra-treated animals had a reduced expression of proinflammatory cytokines and chemokines, suggesting an IL-1-dependent anti-inflammatory effect that impacted systemic cell activation and mobilization. These findings are in agreement with data from previous studies in which administration of IL-1ra was found to decrease neutrophil activation when tested in mouse models of cerebral focal ischemia and in patients after acute ischemic injury and stroke (11,36). Ischemia/reperfusion is also known to increase expression and secretion of proinflammatory mediators that modulate inflammatory responses and play a role in inflammatory injury to end organs (30,31). We observed that expression for all measured cytokines and chemokines was significantly higher in cells obtained from UT donors at 6 h postinduction. Interestingly, animals that received IL-1ra throughout the brain death period revealed no changes in the level of cytokine and chemokine expression compared to baseline levels, and these results paralleled those observed in NBD donors. Elevated levels of IL-6 in deceased donors and transplant recipients have been previously associated with increased graft immunogenicity, early allograft dysfunction, and lower recipient survival (8,20). We observed a systemic release of IL-6 that was suppressed to control levels by IL-1 blockade in our model. Our data support a generalized anti-inflammatory effect of IL-1ra during brain death through the suppression of proinflammatory cytokine and chemokine production and release. Flow cytometric data in our model suggest that IL-1ra prevented activation of monocytes and mDCs in systemic circulation and also within the pancreatic islet. The inactivation of proinflammatory leukocytes at the tissue level may likely prevent the formation of reactive oxygen species and cytokines that induce apoptosis and cell necrosis, and suggests a potential mechanism by which IL-1ra confers protection when administered after neurological injury has occurred. These data are supported by findings from studies in which steroid treatment in the donor aimed at preventing inflammatory injury led to improved organ quality and function after transplantation (14,21).
The most important finding in our study was the observed increase in tissue engraftment and islet function after islet transplantation into diabetic immune-deficient recipients when using islets isolated from IL-1ra-treated donors. We chose the mouse bioassay as a simple method to monitor islet functional potency and avoid the confounding effects of alloreactivity in the primate model. The improved cure efficiency and sustained normoglycemia in mice receiving islets from IL-1ra-treated donors was comparable to that of islets transplanted from NBD donors. Since transplant recipients did not receive treatment, the protective effect of IL-1ra was likely explained by attenuation of the inflammatory response at the donor level, and more importantly, protection was sustained during the isolation process and through tissue culture. To our knowledge, this is the first report describing the use of anti-inflammatory therapy targeting IL-1 in the context of donor management. However, there is already some evidence of the beneficial effect of IL-1ra treatment in islet transplantation. We previously described the protective effect IL-1ra on isolated islets and demonstrated that blocking endogenously produced IL-1β during islet isolation and culture led to an important reduction in cell necrosis, apoptosis, and mitochondrial dysfunction (28). Moreover, a recent study by Montolio et al. reported a pivotal role for IL-1β-driven nitric oxide islet injury responsible for extensive β-cell necrosis after islet transplantation (19). In addition, recent reports have shown that blockade of IL-1β in culture through the use of IL-1 receptor antagonists prior to transplantation prevents islet injury, improves islet quality, and facilitates engraftment (16,17,28). The combined use of IL-1ra and TNF-α blockade is being used in preclinical models of rodent and human islet transplantation and has been found to improve tissue engraftment. These reports expanded on preliminary results showing that combined IL-1 and TNF-α blockade facilitates tissue engraftment and improves islet transplantation efficacy (16,17).
For the purpose of understanding the immunobiology of BD donors, we created a unique NHP model that recreates clinical brain death and constitutes a powerful tool to study the role of intervention at the organ donor level. However, the implementation of this model involves some important limitations that need to be addressed. First, we focused our efforts on understanding the effect of therapy in the organ donor and did not address the impact of treatment in an intraportal NHP allotransplantation model. We used the mouse bioassay as a simple method to evaluate islet functional potency in the absence of confounding variables commonly associated with the use of a large animal islet transplantation models. However, further investigation is needed to determine the impact of donor IL-1 blockade using a large animal allotransplant model to validate our preliminary results. Furthermore, the objective of this study was to provide a proof of concept, and the data presented here are only representative of the early events of brain death-induced acute inflammatory injury. We acknowledge that 6 h is a relatively narrow time frame, and further studies evaluating the effect of therapy in clinically relevant time frames are warranted. Lastly, we did not test the additive benefit of anti-inflammatory therapy at any other stage of the transplant process. It is possible that treatment with IL-1ra alone or in combination with other potent anti-inflammatory therapies (donor management, during cold preservation, tissue culture, and/or at the level of the recipient) may hold greater potential in the protection of islets from inflammatory injury. We did not consider the use of steroids since they are known to negatively impact islet and β-cell function in the peritransplant period and have failed to consistently show protective effects when used in donor management prior to transplantation (26).
The results from this study indicate that inflammatory tissue injury begins at the donor level and support results by others suggesting a detrimental effect of ischemia/ reperfusion injury that is triggered early in the transplantation process. More importantly, it provides evidence suggesting that early intervention in the BD donor using IL-1ra may be beneficial in the protection of the pancreas and islets prior to transplantation. Based on our results, we conclude that early donor management targeting IL-1 receptor signaling is a powerful and practical approach to improve current clinical islet transplant outcomes.
Footnotes
Acknowledgments
This study was supported in part by internal funds of the Department of Surgery/Division of Transplantation at the University of Wisconsin-Madison. We gratefully acknowledge Kimberly Maurer, Dan Consigny, and Drew Roenneburg for expert technical assistance. The authors declare no conflicts of interest.
