Abstract
G-CSF-mobilized peripheral blood stem cells (gm-PBSCs) offer a convenient cell source for treatment of hematopoietic and vascular disorders. Whether gm-PBSCs provide beneficial effects on skeleton diseases, such as osteoarthritis (OA), remains unknown. This study was undertaken to address the hypothesis that gm-PBSCs promote articular regeneration in OA. Here we studied the effect of single-dose intra-articular injection of gm-PBSCs from male donors delivered in hyaluronic acid (HA) on papain-induced OA in the knee joints of female Sprague—Dawley (SD) rats. Contralateral OA knee joints received single-dose HA alone and served as vehicle controls. We evaluated the histologic changes in glycosaminoglycan, type II collagen, type X collagen, modified Mankin score, and cell apoptosis rate in the articular cartilage of rat knees. We demonstrated that gm-PBSCs were mobilized to the peripheral blood via G-CSF infusion for 5 days in SD rats with increasing CD34+ percentage up to 55-fold. We showed that gm-PBSCs inhibit progression of papain-induced OA via reducing articular surface irregularity, fibrillation, and erosion, preventing cellular necrosis and loss of chondrogenic proteins, such as glycosaminoglycan and type II collagen, at both 3 and 6 weeks after treatment. Moreover, gm-PBSCs reduced modified Mankin scores and cellular apoptosis rates compared with HA alone. Our findings demonstrate that HA plus gm-PBSCs, rather than HA alone, inhibits progression of OA in rats in vivo. Thus, intra-articular injection of gm-PBSCs is a convenient protocol for treating OA with consistent beneficial effects.
Keywords
Introduction
Osteoarthritis (OA) is an increasingly common joint disease with a growing number of affected elderly people in many countries. In the US, for example, 10% of the population aged above 60 years has OA (23). Owing to the limited capacity for self-repair of the articular cartilage, treatment of focal or diffused cartilage defects is still a difficult and troublesome problem for orthopedic surgeons. Currently, the treatment options of OA primarily involve the use of anti-inflammatory drugs, analgesics, and lubricating supplements or surgery including drilling, microfracture, and mosaicplasty for repair or reconstruction of the lesions. More recently, cell-based therapy, that is, implantation of cells or engineered cartilage, has been developed to aid in the repair of articular cartilage defects. Brittberg et al. reported that transplantation of autologous chondrocytes cultured under a monolayer culture condition has successfully treated cartilage defects in the knee (3). However, this approach was expensive and time consuming. Hence, progenitor or stem cells have emerged as an alternative solution to this problem.
Recent evidence indicates that bone marrow (BM), the “home organ” of hematopoietic stem cells (HSCs), contains additionally a heterogeneous population of nonhematopoietic stem cells, which have also been described as endothelial progenitor cells (2,29), mesenchymal stem cells (24,26), multipotent adult progenitor cells (11), or marrow-isolated adult multilineage inducible (MIAMI) cells (5). With different experimental strategies applied to detection of stem cells in BM, there may be similar or overlapping stem cells being assigned different names.
These various stem cell populations of BM origin are hypothesized to be the result of the “developmental migration” of stem cells when ontogenesis happens, which are chemoattracted to the permissive microenvironment of the BM tissue. Stromal-derived factor-1 (SDF-1) and hepatocyte growth factor/scatter factor (HGF/SF) (21,30) are the most important chemotactic factors secreted by BM. α-Chemokine Gαi protein-coupled seven transmembrane-spanning receptor (CXCR4) and the tyrosine kinase receptor c-met, the corresponding receptors for these factors, are expressed on migrating stem cells. It is widely accepted that SDF-1-CXCR4 or HGF/SF-c-met are involved in the mobilization of HSCs and other non-HSCs to peripheral blood (10,17,31). Moreover, several exogenous hematopoietic cytokines, such as granulocyte colony-stimulating factor (G-CSF), have been employed to mobilize stem cells of BM origin into circulation with accepted adverse effects (19).
In the current report, we hypothesize that G-CSF-mobilized peripheral blood stem cells (gm-PBSCs) contain a population of primitive stem cells, which are retained in developing organs or BM of adult bodies. These cells have the capacity for high mobility and can be released from the stem cell niches into peripheral blood for tissue regeneration (12,15). We developed standardized procedures for enriching, isolating, freezing/thawing, and application of these enriched gm-PBSCs from Sprague—Dawley (SD) rats for preclinical studies on the in vivo effect of gm-PBSCs on articular cartilage regeneration using a papain-induced model of OA in rats (4).
Materials and Methods
Animals
The study protocol was approved according to the Institutional Animal Welfare guidelines of Taipei Veterans General Hospital (Taipei, Taiwan). Skeletally mature female SD rats, obtained from the animal core of National Yang-Ming University (Taipei, Taiwan), were used at 12 weeks of age with weight ranging from 300 to 400 g as recipients. The harvesting of tissues and all operations were performed under anesthesia induced by intramuscular (IM) injection of 25 mg/kg ketamine hydrochloride (Ketalar; Pfizer, Taipei, Taiwan) or intraperitoneal injection of 45 mg/kg sodium pentobarbital (Abbott Laboratories, North Chicago, IL, USA).
G-CSF Mobilization of PBSCs
For preparation of gm-PBSCs, 10 normal male SD rats at 4 weeks of age were IM injected with recombinant human G-CSF (50 mg/kg/day; Kyowa Hakko Kogyo Co., Tokyo, Japan) for 5 consecutive days. Immediately before first, third, and fifth G-CSF infusion, approximately 200-500 μl peripheral blood was collected by tail vein puncture from each animal using EDTA-coated vacutainer tubes (BD Biosciences, San Diego, CA, USA). One day after the fifth G-CSF infusion, total peripheral blood was obtained by intracardiac puncture into heparin-coated tubes (BD Biosciences). Mononuclear cells (MNCs) were separated using Ficoll-Paque PLUS gradient centrifugation according to the manufacturer's instructions (GE Healthcare, Uppsala, Sweden). MNCs were counted, resuspended at a density of 107 cells/ml in 10% DMSO (Sigma-Aldrich, St. Louis, MO, USA)/90% SD rat plasma from the donor rats, and cryopreserved in liquid nitrogen until required.
Cell Preparation and Cryopreservation
For recovery of cryopreserved MNCs, cells were thawed at 37°C and resuspended in 400 Kunitz units DNase I/ml PBS (260913; Merck Millipore, Darmstadt, Germany) and incubated for 5 min at 37°C. Dead cells and residual granulocytes were then removed by centrifugation at 200 × g for 5 min, and MNCs were washed once with warm PBS. For cell transplantation, MNCs were resuspended at a density of 5 × 106/100 μl in hyaluronan (HA; SciVision Biotech, Kaohsiung, Taiwan).
Identification of CD34+ Stem Cells in MNCs by FACS
MNCs (5 × 105/sample) were stained with a PE-Cy7-conjugated mouse antibody against rat CD34 (sc-7324; Santa Cruz Biotechnology, Santa Cruz, CA, USA) and a FITC-conjugated mouse antibody against rat CD45 (561867; 1 μg per 1 × 106 cells; BD Biosciences). Cells were visualized on a FACSCantoII flow cytometer (BD Biosciences) and analyzed using “Cell Quest” software. Controls were stained with isotype-matched control antibodies. 7-Amino-actinomycin D (7-AAD; BD Biosciences) was added to distinguish cells that had lost membrane integrity from live cells. For each sample, 100,000 events were analyzed.
Osteoarthritis Model
Knee joints of female rats at 12 weeks of age were divided into the following four groups (Fig. 1): OA (OA induction without HA or gm-PBSC treatment) (n = 10) and contralateral control (PBS vehicle), OA with HA (HA treatment followed by OA induction), and contra lateral OA with HA plus gm-PBSCs (HA plus gm-PBSC treatment followed by OA induction) (n = 18). Each left knee, which served as the contralateral control joint, was injected with PBS (vehicle). OA was induced in the OA group (right knee), OA with HA group (left knee), and OA with HA plus gm-PBSCs group (right knee) by intra-articular injections of 20 μl of 4% papain (Sigma-Aldrich) solution and 20 μl of 0.03 M cysteine (Sigma-Aldrich). The injections were given using an insulin needle (Terumo, Tokyo, Japan) via the patellar tendon on days 1, 4, and 7 of the experiment (8). At 1 week after the last injection of papain/cysteine solution, the knee joints of the female OA with HA group were injected with 100 μl of HA, while those of the OA with HA plus gm-PBSCs were injected with 5 × 106 viable MNCs from the male donors in 100 μl of HA. Half of the rats in each group were sacrificed by CO2 inhalation at 3 weeks, and half were sacrificed at 6 weeks after HA or HA plus gm-PBSC treatment.
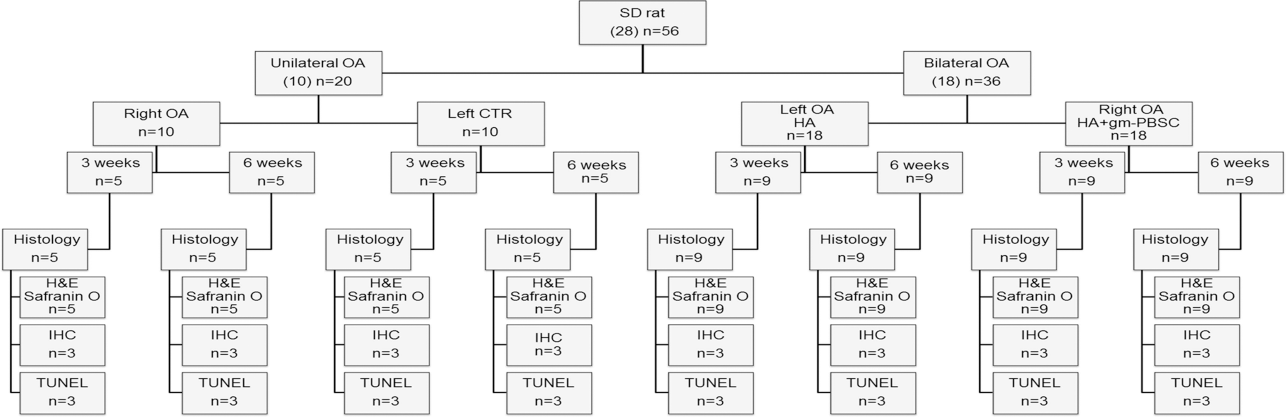
Experimental protocol and animal allocation details. OA, osteoarthritis; IHC, immunohistochemistry; HA, hyaluronic acid; gm-PBSCs, G-CSF-mobilized peripheral blood stem cells.
Histologic Analysis
After rats were sacrificed, the knees were harvested, and the tibia plateaus with articular cartilage were collected and fixed with 10% neutral-buffered formalin (Tonyar Biotech, Taipei, Taiwan) prior to histologic preparation. The samples were then decalcified in 10% formic acid (Sigma-Aldrich)/PBS (Gibco/BRL, Grand Island, NY, USA). The decalcified tibia articular samples were paraffin embedded, and 5-μm microsections in the sagittal plane were prepared. Glycosaminoglycan was stained with Safranin O fast green (1% Safranin O counterstained with 0.75% hematoxylin and then 1% fast green; Sigma-Aldrich), and the total and red-stained areas in the articular cartilage of each proximal tibia were measured using Image-Pro Plus software, version 5.0 (Media Cybernetics, Bethesda, MD, USA). The ratio of red-stained area to total area (red/total) in each group was calculated.
Modified Mankin Score
The levels of articular cartilage degeneration in the knees were evaluated and recorded using the modified Mankin score (1), including four variables: surface (0 = normal, 1 = irregular, 2 = fibrillation or vacuoles, 3 = blisters or erosion), hypocellularity (0 = normal, 1 = small decrease in chondrocytes, 2 = large decrease in chondrocytes, 3 = no cells), clones (0 = normal, 1 = occasional duos, 3 = duos or trios, 3 = multiple nested cells), and alcianophilia (0 = normal, 1 = small decrease in color, 2 = large decrease in color, 3 = no color). In this evaluation system, the higher the score, the higher the level of OA. The entire histological evaluation was performed by three investigators. The investigators were blind to group allocation when this analysis was made.
Immunohistochemistry
Localized type II collagen and type X collagen were immunostained. The tibia articular sections were rehydrated, and the endogenous peroxidase in tissue was blocked with 3% hydrogen peroxide (Sigma-Aldrich). The method for epitope retrieval was modified from that described previously (22). In brief, type II collagen was retrieved with a mixture of 2.5% hyaluronidase (Sigma-Aldrich) and 1 mg/ml of pronase in PBS (pH 7.4; Sigma-Aldrich) at 37°C for 1 h, while type X collagen was retrieved by treatment with 0.1 units/ml of chondroitinase ABC (Sigma-Aldrich) at 37°C for 1 h, followed by treatment with 1 mg/ml of pepsin (Sigma-Aldrich) in Tris HCl (pH 3.0; MDBio, Taipei, Taiwan) at 37°C for 15 min. Sections were then blocked with Ultra V block (Thermo Scientific, Fremont, CA, USA) for 10 min and incubated with primary antibodies against type II collagen (mouse monoclonal antibody; CP18; Calbiochem, La Jolla, CA, USA) and type X collagen (rat polyclonal antibody; ab58632; Abcam, Cambridge, MA, USA) at 4°C overnight. After that, the sections were incubated at room temperature with secondary polymeric antibody following the manufacturer's instructions (Super Sensitive™ IHC Detection Systems kit; BioGenex, Milan, Italy). Finally, sections were counterstained with hematoxylin (Sigma-Aldrich) and observed with a microscope.
TUNEL Staining
Apoptotic cells in each section were measured by TUNEL staining using the in situ Cell Death Detection Kit, fluorescein (Roche Diagnostics, Basel, Switzerland). Sections were rehydrated and incubated with proteinase K (10 μg/ml in Tris HCl, pH 7.4; Sigma-Aldrich) for 20 min. After permeabilization, sections were incubated with pepsin (0.25% in HCl, pH 2.0) for 30 min at 37°C. Slides were rinsed twice with PBS. The TUNEL reaction mixture containing terminal deoxynucleotidyl transferase and fluorescein (the labeling dye) was added to the slides and incubated at 37°C for 60 min. The reaction was stopped with PBS. Cells were mounted with mounting medium containing 4,6-diamidino-2-phenylindole (Gentaur Molecular Products, Santa Clara, CA, USA). The slides were observed under a fluorescence microscope. Stained cells were counted in five microscopic fields on each slide. The rate of apoptosis in chondrocytes was defined as the ratio of green-stained cells (apoptotic cells) to blue-stained cells (total cells).
Statistical Analysis
All data are expressed as mean and standard deviation (SD). Statistical comparisons of the histopathological grade among the OA PBSC group, the OA HA group, and the nontreated group were performed with nonparametric tests, such as the Wilcoxon test for paired two groups, to analyze the statistical power. Differences were considered significant when the value of p < 0.05.
Results
Consecutive Infusion of G-CSF Increases the Percentage of CD34+ in Peripheral Blood
The MNC concentration in peripheral blood before G-CSF infusion was 1.6
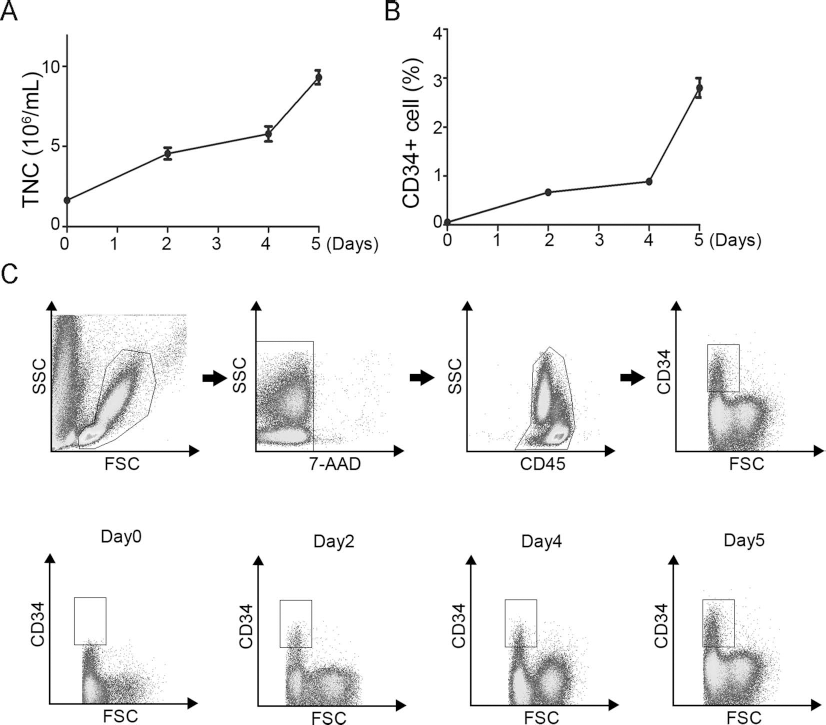
Consecutive infusion of G-CSF increases peripheral MNC concentration and CD34+ percentage. (A) Graph shows (left) the MNC concentration and (right) the CD34+ percentage of peripheral blood before (0 day) and indicated days after consecutive IM infusion of G-CSF. Data are shown as mean ± SD (n = 10). (B) Gating strategy to detect CD34+ stem cells by whole blood flow cytometry; one representative plot is shown. A first gate is set on the lymphocyte—monocyte—granulocyte region in a forward (FSC) and sideways scatter plot. A second gate identifies the single cells in an FSC area plot. Only viable 7-AADs are included in the analysis. The percentage of CD34+ stem cells is measured within the CD45+ population. Isotype-matched controls and unstained cell samples are processed as negative controls. (C) Representative flow cytometry profiles show CD34+ percentage before and after consecutive IM infusion of G-CSF.
Mononuclear Cell Concentration in Peripheral Blood (106/ml)

Day n indicates the day number after the first dose of G-CSF infusion.
The CD34+ Percentage in Peripheral Blood

Effects of Intra-Articular Infusion of gm-PBSCs on OA Progression
The MNCs were collected and cryopreserved in liquid nitrogen for up to 1 month. The MNC viability as analyzed by staining with 7-AAD was 98.8 ± 1.0%. Thawed MNCs were injected intra-articularly into the knee joints without any leakage of cells as confirmed by methyl blue injection with repeated preexperimental practices.
Histologic and Histomorphometric Findings in Rat Articular Cartilage Sections
Representative photomicrographs of H&E-stained articular cartilage from the OA joints and the contralateral control joints in the control studies of rats, as well as those from the OA treated with HA joints, and the contralateral OA treated with HA plus gm-PBSC joints in the experimental studies of rats are shown in Figure 3. Compared with the contralateral control joints, the OA joints showed increase in occurrence of surface irregularity, erosion, or blister; in loss of chondrocytes in middle and deep layers; and in cloning with duos, trios, or multiple nested cells in the superficial layer (Fig. 3). Compared with OA joints, those treated with HA plus gm-PBSCs showed a significant decrease, while those treated with HA alone showed only slight decrease in occurrence of surface irregularity, erosion, or blister; in loss of chondrocytes in middle and deep layers; and in cloning with duo, trio, or multiple cells in the superficial layer (Fig. 3). These data revealed that OA joints treated with HA plus gm-PBSCs increased joint regeneration compared with those treated with HA alone, suggesting that gm-PBSCs had an effect on the regeneration of articular cartilage.

Injection with gm-PBSCs reduces surface irregularity, fibrillation, and erosion in papain-induced OA. H&E staining in sections of articular cartilage in the proximal tibia from the knee joints of OA, control (CTR), OA with HA, and OA with HA plus gm-PBSC groups. Scale bar: 100 μm.
Representative photomicrographs of Safranin O-stained articular cartilage from the OA joints and the contralateral control joints in the control studies of rats, as well as those from the OA treated with HA joints, and the contralateral OA treated with HA plus gm-PBSC joints in the experimental studies of rats are shown in Figure 4A. The ratio of Safranin O-stained area to total area (red/total) was measured (Fig. 4B) and compared among groups (Fig. 4B). The red/total ratios in the control, OA, OA treated with HA, and OA treated with HA plus gm-PBSCs were 82.64 ± 2.38%, 47.86 ± 18.54%, 65.84 ± 13.82%, and 82.60 ± 3.93% for 3 weeks, and 73.19 ± 9.42%, 53.43 ± 7.14%, 41.80 ± 18.31%, and 69.54 ± 13.58% for 6 weeks, respectively. These data demonstrate the superior effect of HA plus gm-PBSCs in retaining Safranin O staining compared with HA alone, suggesting the effect of gm-PBSCs on the maintenance of glycosaminoglycan content.

Injection with gm-PBSCs increases glycosaminoglycan levels in papain-induced OA. (A) Safranin O/fast green staining in sections of articular cartilage in the proximal tibia from the knee joints of OA, control (CTR), OA with HA, and OA with HA plus gm-PBSC groups. Scale bar: 100 μm. (B) Quantitation with the use of Image-Pro Plus software. Data are shown as mean ± SD (*p < 0.05, as determined by Wilcoxon test, n = 5 for OA and CTR, n = 9 for OA with HA and OA with HA plus gm-PBSCs).
Modified Mankin Score in Rat Articular Cartilage Sections
The OA progression was quantified with the use of modified Mankin scores (9,32) (Fig. 5). The scores in the control, OA, OA treated with HA, and OA treated with HA plus gm-PBSCs were 0.5 ± 0.6, 9.5 ± 0.6, 6.8 ± 2.7, and 4.42 ± 2.64 for 3 weeks, and 2.7 ± 1.9, 6.2 ± 2.0, 7.0 ± 1.7, and 2.5 ± 2.6 for 6 weeks, respectively. These data suggest papain-induced OA-induced cartilage degeneration with an increase in modified Mankin scores, while HA plus gm-PBSCs, rather than HA alone, improves cartilage degeneration with a decrease in modified Mankin scores.

Injection with gm-PBSCs decreases modified Makin scores in papain-induced OA. Data are shown as mean ± SD (*p < 0.05, **p < 0.01, as determined by Wilcoxon test, n = 5 for OA and CTR, n = 9 for OA with HA and OA with HA plus gm-PBSCs).
Immunohistochemistry in Rat Articular Cartilage Sections
Representative photomicrographs of the immunohistochemical-stained articular cartilage from the OA joints and the contralateral control joints in the control studies of rats, as well as those from the OA treated with HA joints, and the contralateral OA treated with HA plus gm-PBSC joints in the experimental studies of rats are shown in Figure 6. Immunohistochemistry analysis by quantifying the relative density (Fig. 6) showed that the density of immunolocalized type II collagen in the OA joints was much smaller than that of contralateral control joints at both 3 and 6 weeks after OA induction (Fig. 6A). Immunolocalized type II collagen was obviously reduced in OA joints treated with HA alone compared with OA joints treated with HA plus gm-PBSCs at both 3 and 6 weeks. These data demonstrate the superior effect of HA plus gm-PBSCs in retaining immunolocalized type II collagen compared with HA alone, suggesting the effect of gm-PBSCs on the maintenance of type II collagen content.
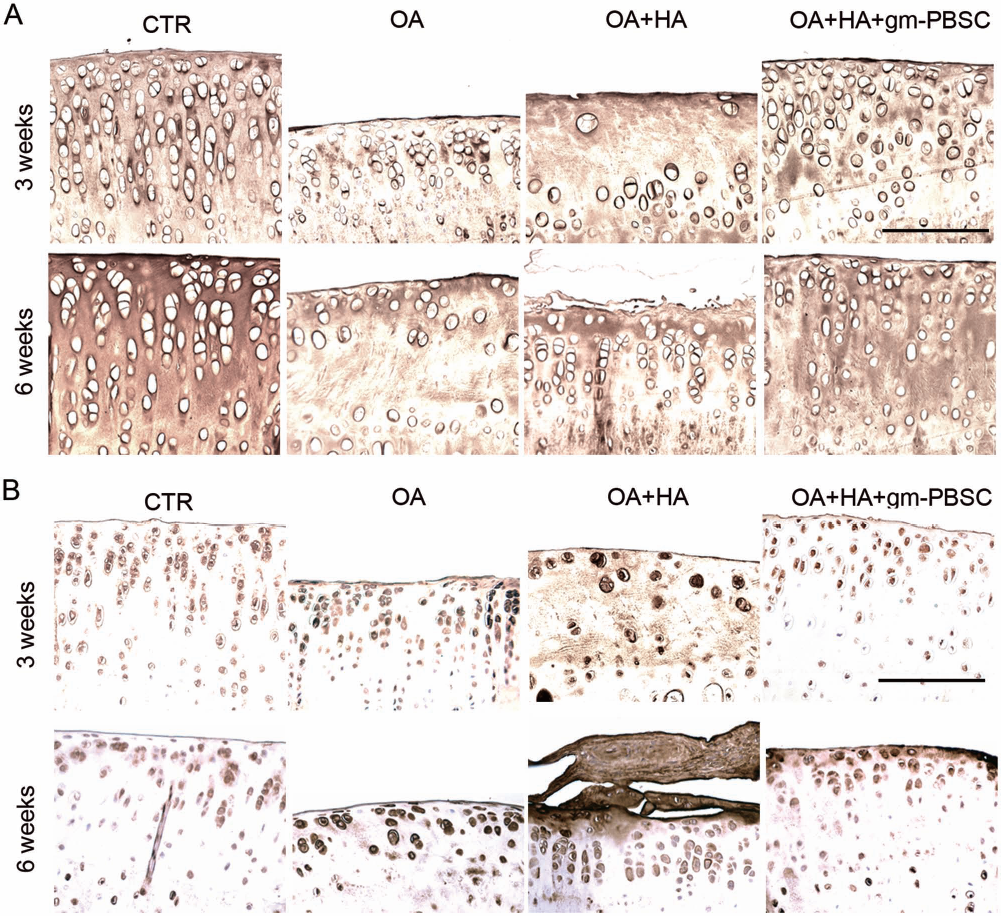
Injection with gm-PBSCs increases type II collagen expression, but reduces type X collagen expression in papain-induced OA. (A, B) Immunostaining of type II collagen (A) and type X collagen (B) in sections of articular cartilage in the proximal tibia from the knee joints of OA, control (CTR), OA with HA, and OA with HA plus gm-PBSC groups. Scale bar: 100 μm.
Immunolocalized type X collagen (stained brown) was predominantly found in articular chondrocytes from the OA joints compared with the contralateral control joints at both 3 and 6 weeks. Moreover, immunolocalized type X collagen was less evident in the cartilage of the OA treated with HA plus gm-PBSC group compared with the OA treated with HA alone group at both 3 and 6 weeks (Fig. 6B). These data demonstrate the superior effect of HA plus gm-PBSCs in decreasing immunolocalized type X collagen compared with HA alone, suggesting the inhibitory effect of gm-PBSCs on the production of type X collagen.
Immunolocalized KI-67, which determines the proliferative status of tissues, was almost not observed in the knee articular cartilages of all groups at both 3 and 6 weeks after OA induction (data not shown). These data suggest that chondrocytes in articular cartilage do not proliferate after papain-induced damages either without or with treatment with HA or gm-PBSCs.
TUNEL Assay in Rat Articular Cartilage Sections
TUNEL staining further revealed that the percentage of TUNEL staining in OA joints was significantly greater than that in contralateral control joints at both 3 and 6 weeks after OA induction. TUNEL staining was obviously enhanced in OA joints treated with HA alone compared with OA joints treated with HA plus gm-PBSCs at both 3 and 6 weeks (Fig. 7).

Injection with gm-PBSCs prevents cellular apoptosis in papain-induced OA. (A) TUNEL staining in sections of articular cartilage in the proximal tibia from the knee joints of OA, control (CTR), OA with HA, and OA with HA plus gm-PBSC groups. Scale bar: 100 μm. (B) Quantitation of TUNEL+ cell percentage. Data are shown as mean ± SD (n = 3).
Discussion
Cell-based therapy using expanded chondrocytes, progenitor, or stem cells remains an option for OA treatment, albeit expensive and time consuming. Hence, identifying a reliable cell preparation protocol for OA treatment has attracted much interest in this field. Here we demonstrated that gm-PBSCs were mobilized to the peripheral blood via G-CSF infusion for 5 days in SD rats with increasing CD34+ percentage up to 55-fold. We further demonstrated that a single-dose intra-articular injection of a frozen aliquot of gm-PBSCs inhibits progression of papain-induced OA via reducing articular surface irregularity, fibrillation, and erosion, preventing cellular necrosis and loss of chondrogenic proteins, such as glycosaminoglycan and type II collagen, at both 3 and 6 weeks after treatment. We also showed that a single-dose injection of gm-PBSCs reduced modified Mankin scores at both 3 and 6 weeks after treatment compared with single-dose injection of vehicle alone. Since the preparation of gm-PBSCs does not rely on the expansion protocol, which requires strict regulation and monitoring, intra-articular injection of gm-PBSCs is a convenient protocol for treating OA with consistent beneficial effects.
The therapeutic effects of gm-PBSCs include treatment for postchemotherapy and irradiation, polycythemia, fulminant hepatitis, and peripheral arterial diseases (25). Recently, gm-PBSCs from a young pig have also been applied for rejuvenation of aged pig facial skin (7). This is the first study that shows the therapeutic effect of gm-PBSCs on skeleton diseases, such as OA. Advantages of PBSCs over BM aspirates include the following: (1) elimination of the need of general anesthesia for pain and other side effects of BM aspiration, (2) patients with BM metastases could be transplanted with autologous PBPCs as there is a potential for tumor cell-free collection, and (3) faster hematological recovery with PBPCs than BM, thus reducing significantly the time to transfusion independence (28). Moreover, new advances in effective mobilization of PBSCs have permitted a greater proportion of patients to benefit from autologous stem cell transplantation (6). Thus, gm-PBSCs have multiple therapeutic benefits in a lot of disorders.
The immediate side effects of G-CSF administration, such as bone pain, headache, fatigue, nausea, and splenomegaly, have been established for a long time, with most of them being transient, self-limited, and without long-term consequences. Although potential long-term adverse effects such as an elevated risk of hematological malignancies after G-CSF administration have been reported, the frequency is very low, and the relationship between these major adverse events and G-CSF administration is not clear (19). Thus, G-CSF administration is still safe and has benefits that exceed side effects.
Although the current study did not aim to identify the cellular components in gm-PBSCs for contributing to OA therapeutics, knowing which cells in BM are mobilized to exert therapeutic effects on OA is important for future application. BM contains HSCs and a heterogeneous population of non-HSCs, which have been described as endothelial progenitor cells (2,29), mesenchymal stem cells (24,26), multipotent adult progenitor cells (11), MIAMI cells (5), or very small embryonic-like (VSEL) cells (13,14). Since different experimental strategies are employed to detect those stem cells in the BM, similar or overlapping stem cells may be assigned different names. Of note, VSEL is a population of CXCR4+, stage-specific embryonic antigens (SSEA)+, Oct-4+, Nanog+ stem cells in murine BM and human cord blood, and is suggested to be deposited early in the development of marrow tissue and are descendants of epiblast-derived stem cells (27). Recent data also reveal that a great amount of VSEL cells could be mobilized to peripheral blood after infusion with G-CSF intravenously (16). Thus, we could not exclude the possibility that VSEL plays an important role in the therapeutic effects of gm-PBSCs on OA.
The effect of intra-articular injection of HA in treating OA remains controversial. Some studies reported its effect in treating OA, while some did not recognize a therapeutic effect and argued that HA only provided a lubrication effect (18). The current study did not identify a therapeutic effect of HA alone in treating OA when compared to the nontreatment group. The etiology may be attributed to single-dose HA injection, which is much less than the dosage recommended for OA treatment. The role of HA in the current protocol is a vehicle. Since HA was reported to localize at the articular surface after intra-articular infusion, gm-PBSCs delivered in HA will also be delivered to the articular surface. We did not find any evidence that transplanted cells migrated to articular cartilage and engrafted for regeneration. These data suggest gm-PBSCs enhanced cartilage repair by a mechanism not involving engraftment and differentiation into chondrocytes. A previous study showed that caprine MSCs when delivered in HA following injection into articular joint of OA only engrafted to synovium or meniscus tissues (20), suggesting a paracrine effect in reducing OA progression. Thus, gm-PBSCs may provide therapeutic effects through affecting endogenous cells or tissues, rather than directly engrafting for regeneration. For example, gm-PBSCs may induce endogenous stem cells to differentiate into chondrocytes, inhibit cartilage from being eroded by papain, or inhibit consequent inflammation. This speculation is supported by the findings that articular cartilage from knee joints treated with gm-PBSCs decreased the apoptosis rate compared to that from knee joints treated with vehicle alone.
In conclusion, our findings indicate that HA plus gm-PBSCs, rather than HA alone, inhibits progression of OA in rats in vivo. Given the advantage that the preparation of gm-PBSCs does not rely on an expansion protocol, intra-articular injection of gm-PBSCs is a convenient protocol for treating OA with consistent beneficial effects. The data presented here may be used as guidelines for clinical trials.
Footnotes
Acknowledgments
This work was assisted in part by the Division of Experimental Surgery of the Department of Surgery, Taipei Veterans General Hospital and supported by the Grants R11001-01 and R11001-02 from Taipei Veterans General Hospital to Shih-Chieh Hung. The authors declare no conflicts of interest.
