Abstract
Granulocyte colony-stimulating factor (G-CSF) mobilizes peripheral blood stem cells (PBSCs) derived from bone marrow. We hypothesized that intraspinal transplantation of PBSCs mobilized by G-CSF could promote functional recovery after spinal cord injury. Spinal cords of adult nonobese diabetes/severe immunodeficiency mice were injured using an Infinite Horizon impactor (60 kdyn). One week after the injury, 3.0 μl of G-CSF-mobilized human mononuclear cells (MNCs; 0.5 × 105/μl), G-CSF-mobilized human CD34-positive PBSCs (CD34; 0.5 × 105/μl), or normal saline was injected to the lesion epicenter. We performed immunohistochemistry. Locomotor recovery was assessed by Basso Mouse Scale. The number of transplanted human cells decreased according to the time course. The CD31-positive area was significantly larger in the MNC and CD34 groups compared with the vehicle group. The number of serotonin-positive fibers was significantly larger in the MNC and CD34 groups than in the vehicle group. Immunohistochemistry revealed that the number of apoptotic oligodendrocytes was significantly smaller in cell-transplanted groups, and the areas of demyelination in the MNC- and CD34-transplanted mice were smaller than that in the vehicle group, indicating that cell transplantation suppressed oligodendrocyte apoptosis and demyelination. Both the MNC and CD34 groups showed significantly better hindlimb functional recovery compared with the vehicle group. There was no significant difference between the two types of transplanted cells. Intraspinal transplantation of G-CSF-mobilized MNCs or CD34-positive cells promoted angiogenesis, serotonergic fiber regeneration/sparing, and preservation of myelin, resulting in improved hindlimb function after spinal cord injury in comparison with vehicle-treated control mice. Transplantation of G-CSF-mobilized PBSCs has advantages for treatment of spinal cord injury in the ethical and immunological viewpoints, although further exploration is needed to move forward to clinical application.
Keywords
Introduction
The central nervous system, including the spinal cord, is a delicate tissue that cannot tolerate damaging physiological conditions. Thus, poor recovery following injury is generally attributed to the hostile local milieu created at the site of injury. Spinal cord injury (SCI) is characterized by so-called secondary injuries that are triggered by the initial mechanical insult (29). These include hemorrhage, destruction of the blood–spinal cord barrier, and infiltration of inflammatory cells. The damage is mediated by a complex cascade of deleterious events that lead to further degenerative damage, including spinal cord ischemia beyond the site of initial injury (8). Those events suggest that angiogenesis-promoting treatments might be valuable for SCI treatment in both the acute and subacute phases.
Granulocyte colony-stimulating factor (G-CSF) is a 19.6-kDa glycoprotein that is best known as a growth factor for hematopoietic progenitor cells. It is clinically used to treat neutropenia and to mobilize peripheral blood-derived hematopoietic stem cells for transplantation (20,24). Furthermore, G-CSF mobilizes peripheral blood stem cells (PBSCs) derived from bone marrow. The mobilized PBSCs reach the injured spinal cord by migrating through the disrupted blood–spinal cord barrier. However, only modest numbers of mobilized PBSCs reach the site of spinal cord injury (17). Thus, we hypothesized that intraspinal transplantation of PBSCs mobilized by G-CSF could promote better functional recovery after contusive SCI.
In the current study, we tested whether intraspinal transplantation of human G-CSF-mobilized mononuclear cells or CD34-positive PBSCs promote angiogenesis and accelerate hindlimb functional recovery in mouse contusive SCI.
Materials and Methods
All experimental procedures were performed in accordance with the Chiba University School of Medicine guidelines pertaining to the treatment of experimental animals. The approval of the Animal Committee of Chiba University Graduate School of Medicine was obtained before we started the experiments (the approval number was 25-72).
Spinal Cord Injury
A total of 48 adult female nonobese diabetes/severe immunodeficiency (NOD/SCID) mice (8 to 9 weeks old, average weight 25 g; Charles River Japan, Yokohama, Japan) were used in this study. Animals were anesthetized with inhaled 1.0-1.2% halothane (Wako, Osaka, Japan) in 0.5 L/min oxygen. Laminectomy was performed at the thoracic (T) 9 vertebral level, leaving the dura intact. The T8 and T10 spinous processes of the vertebra were clamped to fix the spine. Then their spinal cords were injured using an Infinite Horizon impactor (60 kdyn; Precision Systems and Instrumentation, Lexington, KY, USA). After that, muscles and skin were sutured layer to layer, and the mice were placed in warm cages overnight. Food and water were given ad libitum. All animals were given saline with antibiotics (Cefmetazon; Daiichi-Sankyo Pharmaceuticals, Tokyo, Japan) by subcutaneous administration to avoid dehydration and infection once a day for 3 days. Manual bladder expression was performed twice a day until recovery of the bladder reflex.
Cell Transplantation
One week after the initial injury, the mice were randomly allocated to three groups, and the spinal cords were reexposed. In the mononuclear cell (MNC) group (n = 16), 3.0 μl of G-CSF-mobilized human MNCs (0.5 × 105/μl) was injected into the lesion site with a fine glass pipette attached to a Hamilton syringe (Hamilton Company, Reno, NV, USA). In the CD34-positive group (n = 16), 3.0 μl of G-CSF-mobilized human CD34-postive PBSC (0.5 × 105/μl) were injected into the lesion site. The cell dosage was determined according to the previous studies (16,18).
In the vehicle group (n = 16), normal saline (3.0 μl) was injected in the same fashion as the transplanted groups. We purchased human cells mobilized by G-CSF (MNC: mPB004F; CD34-positive PBSCs: mPB015F; All Cells, Alameda, CA, USA). The injection speed was set to 1 μl/min for each group with an air-driven syringe pump (PUMP 11 ELITE; Harvard Apparatus, Holliston, MA, USA). After surgery, animals were treated the same way as after the initial surgery.
Tissue Preparation
Mice were sacrificed 1 week (n = 3/group), 3 weeks (n = 3/group), and 7 weeks (after the termination of locomotor assessment, n = 10/group) after the transplantation. The animals were deeply anesthetized with intraperitoneal sodium pentobarbital (Kyoritsu Seiyaku, Tokyo, Japan) and perfused transcardially with 4% paraformaldehyde (Wako) in phosphate-buffered saline (PBS, pH 7.4; Life Technologies Japan, Tokyo, Japan). Spinal cords including the lesion site were removed and postfixed in the same fixative for 24 h. Then tissues were immersed in 20% sucrose in PBS at 4°C. After that, the tissue was embedded in OCT compound (Tissue-Tek, Sakura Finetech. Tokyo, Japan), frozen on dry ice, and sectioned on a cryostat. The sagittal serial frozen spinal cord sections (25 μm in thickness) were mounted onto poly-l-lysine-coated glass slides (Matsunami, Tokyo, Japan). Each slide contained five sliced sections at 100-μm intervals, and the sections for each slide were offset by 25 μm from the previous slide in the set. In this way, we were able to cover the central 500 μm of the lesion at 25-μm intervals in five slides.
Immunohistochemistry
We performed immunohistochemistry as previously described (7,11). Sections of mouse spinal cord were rehydrated with 0.3% Triton-X (Wako) in PBS for 1 h and washed three times with PBS. Slides were then incubated with blocking solution (Block Ace; Yukijirushi, Sapporo, Japan) for 1 h at room temperature. The primary antibodies were as follows: mouse monoclonal anti-human mitochondria antibody (1:100; Merck Millipore, Billerica, MA, USA), which is a marker for transplanted human cells; mouse anti-myelin basic protein (1:400; Merck Millipore), which is a marker for residual myelin sheath, and rat monoclonal anti-CD31 antibody (CD31, 1:400; Merck Millipore), which is a marker for endothelial cells of murine and human origin. Double immunofluorescence for cleaved caspase 3 (1:400; R&D Systems, Minneapolis, MN, USA), which is a marker for apoptotic cells, and adenomatous polyposis coli (APC, clone CC-1, 1:800, Merck Millipore), which is a marker for oligodendrocyte, was performed to evaluate the effect of cell transplantation on apoptosis. To assess axonal regeneration/ sparing, immunohistochemistry for serotonin and growth-associated protein-43 (GAP-43) was performed using rabbit anti-serotonin polyclonal antibody (1:800, S5545; Sigma-Aldrich, St. Louis, MO, USA) and mouse anti-GAP-43 monoclonal antibody (1:400; Abcam, Cambridge, UK). Serotonergic fibers have been previously reported to contribute to hindlimb motor control in rodents (4,6,25). The sections were reacted overnight at 4°C. After three 10-min washes with PBS, the sections were reacted with Alexa Fluor 488-conjugated or Alexa Fluor 594-conjugated donkey anti-mouse IgG (1:800; Life Technologies, Carlsbad, CA, USA) and with Alexa Fluor 488-conjugated or Alexa Fluor 594-conjugated goat anti-rabbit IgG (1:800; Life Technologies).
Quantification
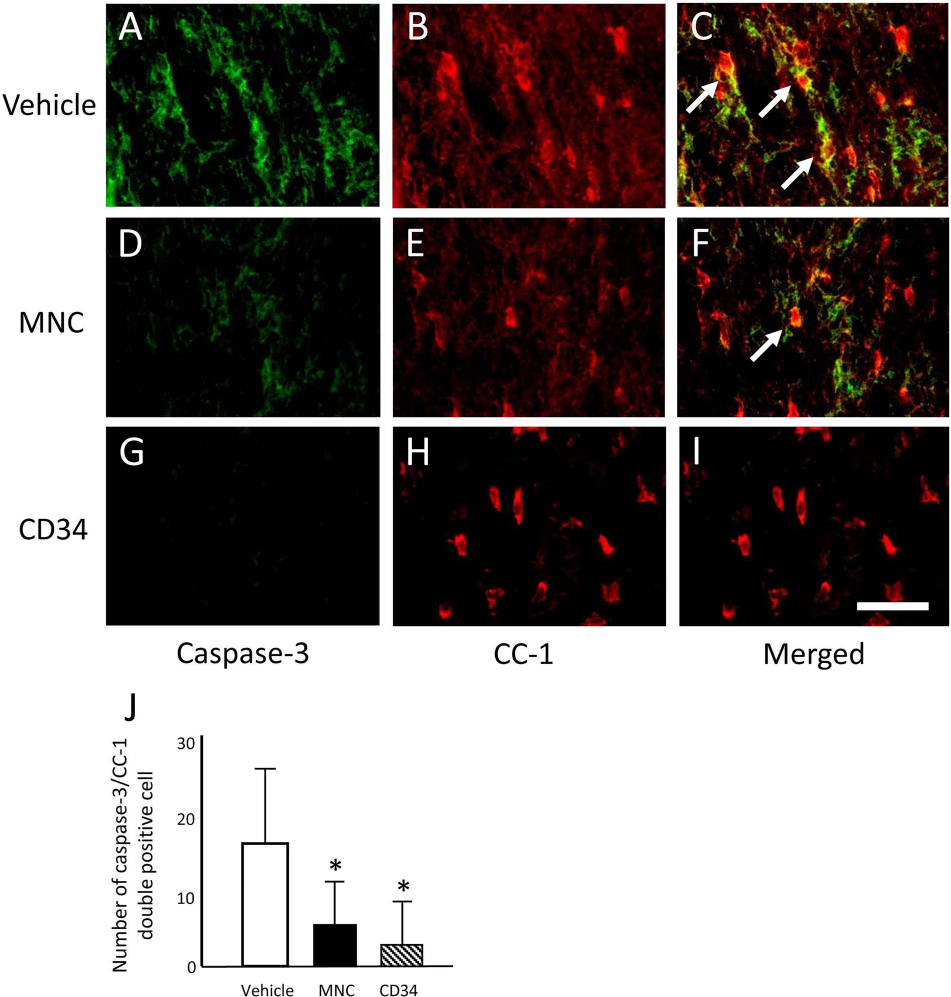
Quantification of histological and immunohistochemical results was performed by blinded observers kept unaware of treatment groups. The numbers of transplanted cells were counted in the lesion epicenter as human mitochondria-positive cells. MBP-negative area, which indicates the degree of tissue destruction, was measured with ImageJ software and expressed as the ratio to the total white matter area. CD31- and serotonin-positive areas were measured by ImageJ software and were expressed as the ratio of those immunopositive areas to the total spinal cord area observed. The number of double-positive cells for cleaved caspase 3 and CC-1, which indicates the number of apoptotic oligodendrocytes, was counted.
Locomotor Function and Behavioral Testing
Hindlimb functional recovery was assessed using the Basso Mouse Scale (BMS) (2). Mice were observed individually for 5 min each in an open field by two blinded investigators. Hindlimb motor function was recorded and scored according to the BMS guidelines once per week. All open-field BMS score episodes were recorded using a video camera and were reviewed later to confirm the accuracy. We followed the BMS score for 8 weeks after the transplantation (n = 10 per group). After 8 weeks, we performed movement analysis using the SCANET MV-40 (Melquest, Toyama, Japan) (Fig. 6B). The mice were allowed to move freely in the attached wide plastic box. The SCANET system consists of a cage equipped with two crossing infrared sensor frames arranged at different heights, with which small (M1) and large (M2) horizontal movements were monitored. We assessed locomotor function by determining the M1 scores for 30 min. The quantity of motions was analyzed automatically (19).
Statistical Analysis
The results were evaluated by multiple comparisons among control, mononuclear, and CD34 groups. BMS scores were analyzed by repeated-measures analysis of variance (ANOVA) followed by the post hoc Tukey–Kramer test. The amounts of spontaneous motion measured by the SCANET-40 as well as immunohistochemical data were subjected to ANOVA. Data are presented as mean values ± standard errors of the mean (SEM). Differences were considered as significant at p < 0.05.
Results
Immunohistochemical analyses of injured spinal cords were conducted 1, 3, and 7 weeks after the transplantation [2 weeks (n = 3/group), 4 weeks (n = 3/group), and 8 weeks (n = 10/group) after the injury]. We assessed the frequency and distribution of transplanted human mitochondria-positive cells. One week after the transplantation (2 weeks after the injury, transplanted human cells were detected in segments 2 mm rostral and 2 mm caudal to the lesion site in both the MNC and CD34 groups) (Fig. 1A, B). There was no significant difference between the numbers of detected human cells in the two groups. Three weeks after the transplantation (4 weeks after the injury), the numbers of transplanted cells significantly decreased in both groups (Fig. 1C). No transplanted human cell was detected 7 weeks after the transplantation (8 weeks after the injury) (Fig. 1C).
Transplanted peripheral mononuclear cells (MNCs) or CD34-positive cells mobilized by granulocyte colony-stimulating factor survived in injured spinal cord. Survived CD34-positive cells were located around the epicenter (A, B). MNC and CD34-positive cells were stained with anti-human mitochondria antibody. The number of transplanted cells decreased over time (C). Scale bars: 1 mm (A) and 100 μm (B). Asterisks indicates significant difference between 2, 4, and 8 weeks after the injury (*p < 0.05, **p < 0.01, C).
We measured the CD31-positive area at the lesion epicenter, an indicator of neovascularization, and compared the results among the three treatment groups. The CD31-positive area was significantly larger in the MNC and CD34 groups compared with the vehicle group (Fig. 2). There was no significant difference between the MNC and CD34 groups in the CD31-positive area (Fig. 2B, C, E F, ***-G). We carried out immunohistochemical analysis for serotonin, a marker for raphe-spinal serotonergic fibers that constitute important tracts for hindlimb motor control. The data revealed that the area of serotonin-positive fibers was significantly larger in the MNC and CD34 groups than in the vehicle group at the rostral and caudal segments (Fig. 3A–G). The serotonin-positive area at the lesion epicenter was larger in the CD34 group compared with that in the vehicle group, whereas there was no significant difference between the MNC and the vehicle group (Fig. 3G). Moreover, a part of those serotonin-positive fibers was also positive for GAP-43, which is a marker for growth cone indicating regenerating axons (Fig. 3H–J). We performed double immunofluorescence study for cleaved caspase 3 (a marker for apoptotic cells) and CC-1 (a marker for oligodendrocytes) to assess the effect for cell transplantation on apoptosis of oligodendrocytes. The number of double-positive cells for cleaved caspase 3 and CC-1 was significantly larger in the MNC and CD34 groups than that in the vehicle group (Fig. 4). We used immunohistochemistry for MBP to investigate the area of the demyelinated sheath 7 weeks after transplantation (8 weeks after the injury, n = 4/group). Immunohistochemistry for MBP revealed that the areas of demyelination in the MNC- and CD34-transplanted mice was significantly smaller than that in the vehicle group, indicating that cell transplantation suppressed demyelination (Fig. 5A–D).
Transplantation of peripheral mononuclear cells (MNCs) or CD34-positive cells mobilized by granulocyte colony-stimulating factor promoted angiogenesis in injured spinal cord. CD31-positive area around the injury epicenter was larger in both the cell-transplanted groups compared with that in the vehicle group (G). There was no significant difference between the MNC and CD34 groups. Scale bars: 1 mm (A–C) and 100 μm (D–F). Asterisks indicate significant difference compared with the vehicle group (**p < 0.01, G). Transplantation of peripheral mononuclear cells (MNCs) or CD34-positive cells mobilized by granulocyte colony-stimulating factor promoted serotonergic axonal regeneration/sparing in injured spinal cord. More serotonin-positive axons were observed in the segment rostral to the injury epicenter in both the cell-transplanted groups (A–F). Serotonin-positive area in the segments rostral and caudal to the injury epicenter was larger in both the cell-transplanted groups compared with that in the vehicle group (G). In the lesion epicenter, CD34 group showed significantly larger serotonin-positive area compared with that in the vehicle group, whereas there was no significant difference between the MNC and vehicle groups (G). There was no significant difference in serotonin-positive area between the MNC and CD34 groups (G). Double immunofluorescence study revealed a part of those serotonergic fibers was also positive for growth-associated protein-43 (a marker for regenerating axons, H–J). Scale bars: 1 mm (A–C), 100 μm (D–F, H–J). Asterisks indicate significant difference compared with the vehicle group (**p < 0.01, *p < 0.05). Transplantation of MNC and CD34 suppressed oligodendrocytre apoptosis. Double immunofluorescence study revealed that the number of double-positive cells for cleaved caspase 3 (a marker for apoptotic cells) and CC-1 (a marker for oligodendrocytes), which indicates apoptotic oligodendrocytes, was smaller in the MNC and CD34 groups than that in the vehicle group. Scale bar: 50 μm (A–I). Arrows indicate double-positive cells for cleaved caspase 3 and CC-1. Asterisks indicate significant difference compared with the vehicle group (*p < 0.05). Transplantation of peripheral mononuclear cells (MNCs) or CD34-positive cells mobilized by granulocyte colony-stimulating factor promoted tissue sparing in injured spinal cord. Myelin basic protein-negative area, which indicates demyelination, was smaller in both the cell transplanted groups compared with that in the vehicle group (A–D). There was no significant difference between the MNC and CD34 groups. Scale bar: 1 mm (A–C). Asterisks indicate significant difference compared with the vehicle group (*p < 0.05, D). Transplantation of peripheral mononuclear cells (MNCs) or CD34-positive cells mobilized by granulocyte colony-stimulating factor promoted hindlimb functional recovery after spinal cord injury. Locomotor function was assessed using Basso Mouse Scale (BMS). Repeated measures ANOVA and post hoc test showed better functional recovery in both the cell transplantation groups (A, square: MNC, triangle: CD34) than the vehicle group (A, circle). Spontaneous movement was measured by SCANET-40® (B). CD34 group showed significant increase of spontaneous movement compared with that in the other groups (C). Values are mean ± SEM. **p < 0.01 (A, C).



Both the MNC and CD34 groups showed significantly better hindlimb functional recovery compared with the vehicle group (repeated measures ANOVA, p < 0.01) (Fig. 6A). Post hoc analysis revealed significant increase of the BMS score 6 to 8 weeks after the initial injury in the MNC and CD34 groups compared with the vehicle group (p < 0.01) (Fig. 6A). There was no significant difference between the two types of transplanted cells. The recovered BMS score in cell-transplanted groups was approximately six points, which indicates “frequent or consistent plantar stepping, some coordination, paws parallel at initial contact” or “frequent or consistent plantar stepping, mostly coordinated, paws rotated at initial contact and liftoff,” whereas the recovered BMS score in the vehicle group was approximately five points, which indicates “frequent or consistent plantar stepping, no coordination” or “frequent or consistent plantar stepping, some coordination, paws rotated at initial contact and liftoff.” Those differences of BMS scores between the cell-transplanted groups and the vehicle group are simulated to the capability of weight bearing in clinical situations.
We also assessed the quantity of spontaneous locomotion with the SCANET MV-40 8 weeks after the injury. A significant increase in the quantity of locomotion in the CD34 group was detected compared with the vehicle group (p < 0.01) (Fig. 6B, C), whereas the MNC group showed no significant increase in spontaneous locomotion.
Discussion
In this study, we examined the impact of intraspinal transplantation of G-CSF-mobilized enriched human CD34-positive cells or mononuclear cells following SCI in mice. The data demonstrated that 7 weeks after treatment (8 weeks after the injury) intraspinal transplantation promoted angiogenesis, serotonergic fiber regeneration/sparing, suppression of oligodendrocyte apoptosis, and preservation of myelin, resulting in improved hindlimb function after SCI in comparison with vehicle-treated control mice.
The various cell types that make up the blood are of mesodermal origin and are derived from a common pool of PBSCs. During embryogenesis, hematopoietic and endothelial lineage cells are derived from common progenitor cells, called hemangioblasts. In adult humans, CD34-positive cells likely include the PBSC fraction, and they have been used clinically for hematopoietic stem cell transplantation and reconstitution. After G-CSF administration, leukocytes, including CD34-positive PBSCs, are mobilized from bone marrow, and they can be readily collected by apheresis (9). Recently, human peripheral blood CD34-positive cells were reported to include an endothelial progenitor cell-enriched population as well as a PBSC fraction (1). Thus, intravenous administration of G-CSF mobilized CD34-positive PBSCs has been shown to facilitate vascular regeneration of ischemic tissues, and therapeutic use of CD34-positive PBSC transplantation for ischemic tissue has been performed in many preclinical studies (10,12,26,27). In patients with ischemic diseases of lower limbs, transplanted PBSC mobilized by G-CSF are capable of enhancing neovascularization. In patients with acute myocardial infarction, transplantation of CD34-positive cells promotes vasculogenesis and cardiomyogenesis, enhancing functional regenerative recovery (10,15). Furthermore, in a cerebral stroke model, intracerebral CD34-positive PBSC implantation has enhanced neovascularization in the penumbra region of the ischemic brain and subsequently promoted marked neurogenesis (3, 26).
In SCI, the use of hematopoietic stem cells derived from human umbilical cord blood was reported to promote restoration of spinal cord tissue and recovery of hindlimb function in adult rats (21,23). Human umbilical cord blood-derived CD34-positive cells were also beneficial in restoring hindlimb function by stimulating production of glial cell line-derived neurotrophic factor (GDNF) and vascular endothelial growth factor (VEGF) (13). We have demonstrated that intraspinal transplantation of human CD34-positive PBSCs mobilized by G-CSF promoted angiogenesis in injured spinal cord and functional recovery in mice. Mononuclear cells collected following G-CSF treatment include many cell populations in addition to CD34-positive cells, including CD45-, CD133-, and CD11b-positive cells. The present results showed that there was no significant difference between MNC and CD34-positive groups in promotion of angiogenesis, axonal regeneration/sparing, tissue restoration, and hindlimb functional recovery, although the total MNC fraction contained a small number of CD34-positive hematopoietic stem cell/endothelial progenitor cells.
It has been reported that the G-CSF-mobilized MNC fraction is capable of neovascular regeneration in the ischemic brain (26). As for the spinal cord, recent reports have indicated that the G-CSF-mobilized MNC fraction can promote angiogenesis and has therapeutic effects in SCI (5), indicating that other cell populations than CD34-positive cells can promote neurological recovery.
The possible underlying mechanisms of action of MNC and CD34-positive cell transplantation are as follows. The results of immunohistochemistry for human cell markers showed that transplanted MNC and CD34-positive cells disappeared 4 weeks after injury. In spite of the poor survival of transplanted cells, angiogenesis, axonal regeneration/sparing, and tissue preservation were observed at a final follow-up time point 8 weeks after injury. A double immunofluorescence study revealed antiapoptotic effect and serotonergic axonal regeneration-promoting effect of those types of cell transplantation. These data suggest that the transplanted cells exert their therapeutic potential through early effector mechanisms, including secretion of soluble factors and immunomodulation.
We previously reported that intravenous injection of G-CSF mobilized bone marrow-derived cells to the injured spinal cord directly suppressed neuronal apoptosis and promoted angiogenesis in the injured spinal cord (14,17,22). We have completed an early phase clinical trial of neuroprotective therapy using intravenous injection of G-CSF for patients with acute SCI (28). In the present study, we demonstrated possible therapeutic effects of MNC and CD34-positive PBSC intraspinal transplantation. We suspect that a combination therapy of intravenous G-CSF injection in the acute phase to protect spinal cord tissue from secondary injury mechanisms and intraspinal transplantation of MNC and CD34-positive PBSCs mobilized by G-CSF injection in the subacute phase could improve motor and sensory function in patients with acute SCI. G-CSF is clinically used to treat hematological disorders and is essentially safe, and autologous PBSC transplantation can be performed without problems of immune rejection. Moreover, G-CSF-mobilized PBSC transplantation is less problematic because there are no major ethical concerns arising from the use of embryonic tissues as a cell source.
In conclusion, transplantation of G-CSF-mobilized peripheral MNCs or CD34-positive cells is a realistic approach for treatment of SCI in ethical and immunological viewpoints, although further exploration is needed to move forward to clinical application.
Footnotes
Acknowledgments
This work was supported by Grants-in-Aid for Scientific Research from the Ministry of Education, Science and Culture of Japan and the General Insurance Association of Japan. The authors declare no conflicts of interest.
