Abstract
This study examined the role of bone mesenchymal stem cell (BMSC) and olfactory ensheathing cell (OEC) cografting on neural function and underlying molecular mechanisms in acute stage of traumatic brain injury (TBI) rats. Eighty Sprague-Dawley (SD) female rats were randomly divided into five groups (n = 16 per category): sham operated group (Sham), weight-drop-induced TBI group (TBI), BMSC transplantation group (BMSC), OEC transplantation group (OEC), and cotransplantation group (CO). Eight rats were randomly selected from each group for behavioral and morphological assessment. Another category (n = 8 rats) was employed in the genetic expression detection. BMSCs were isolated from GFP mice and identified by CD44 antibody. OECs were isolated from the SD rats, identified by P75 antibody and labeled by Hoechst 33342. They were then transplanted into the surrounding tissue of the epicenter of TBI rats. The result of neurological severity scores revealed that BMSC or OEC transplantation alone and BMSC and OEC cografting significantly ameliorated the neurological deficits of TBI rats. Quantitative immunohistochemical analysis showed that graft-recipient animals possessed dramatically more neurons and regenerated axons and smaller amounts of astrocytes than controls 14 days posttransplantation (p <0.05). However, the expressional level of ciliary neurotrophic factor significantly decreased in the cografting group as determined by RT-PCR (p <0.05), and the Janus kinase/signal transducer and activator of transcription pathway was significantly activated at 7 days after cell transplantation (p <0.05). This study is the first to report the role of cotransplantation of BMSCs and OECs in the therapy of TBI and explore its potential molecular mechanisms, therefore providing the important morphological and molecular biological evidence for the clinical application of BMSC and/or OEC transplantation in TBI.
Keywords
Introduction
Traumatic brain injury (TBI) is caused by a blow or penetrating injury to the head that disrupts normal brain function and is becoming increasingly prevalent in society. The more severe forms of TBI have worse prognoses that are attributed to various pathological changes, such as neuronal death (40), axonotmesis (26), degeneration, and formation of scars (10). All of this brings about a heavy burden to the patients' family and society. Therefore, it is necessary to search for effective therapeutic strategies for TBI. To date, the biological therapy based on stem cell transplantation is generally used in experimental research involving central nervous system (CNS) diseases (2,8,11,19-21,40). However, the underlying mechanisms of stem cell transplantation are unclear.
Bone mesenchymal stem cells (BMSCs), a kind of multipotent somatic stem cells, can be induced to differentiate into neurons and glial cells in vitro and in vivo. Meanwhile they secrete various neurotrophic factors, cytokines, and other factors that promote an environment conducive to neural repair and regeneration (2,8,39). Recent evidence indicates that BMSCs had been transplanted into the mammalian nervous system (19,21) and were able to facilitate the regeneration of injured axons, differentiate into neurons, and promote the functional outcome after spinal cord injury (SCI) (11,20,24,33,34). Moreover, they can be used in an autograft without the concern of immunologic rejection or ethical and moral issues. So, BMSCs are potentially an excellent resource of seed cells that have been extensively applied in the therapeutic research of CNS diseases, including CNS injury.
Olfactory ensheathing cells (OECs) exist in the olfactory bulb and are a special kind of glial cell that has the ability to penetrate the barrier of peripheral nerves and central nerves. Furthermore, various neurotrophic factors, such as nerve growth factor (NGF), brain-derived nerve growth factor (BDNF), neurotrophin-3 (NT-3), NT-4, platelet-derived growth factor (PDGF), neuropeptide-Y, and S-100 can be secreted by OECs to promote axonal myelination (16,29), ultimately facilitating the functional recovery of injured nerves. Consequently, OECs are considered a novel cellular resource for CNS disease therapy in recent years (36).
Ciliary neurotrophic factor (CNTF) has a special molecular structure and biological activity in contrast to other members of the NGF family. Combined with its receptor, CNTF receptor (CNTFR), CNTF plays a neuroprotective role via the Janus kinase/signal transducer and activator of transcription 3 (Jak/STAT3) cellular transduction pathway (30,31), which is purported to contribute to neuronal survival (17), cellular apoptosis inhibition (32), and neurite regeneration (25). Furthermore, in recent years, CNTF has been found to promote the proliferation of glial cells that could induce the formation of scars and ultimately impeded CNS injury restoration. Therefore, it is necessary to elucidate whether CNTF plays a negative or positive role in the recovery of CNS injury.
In this study, we investigated the role of BMSC and OEC cograft on neurological functional recovery in TBI rats and the regulatory mechanism of CNTF expression associated with the transplantation. Our primary aim was to determine effective strategies for the therapy of TBI based on cell transplantation.
Materials and Methods
Animal Model Groups
Eighty 2-month-old (220-250 g) female Sprague-Dawley (SD) rats were purchased from the Animal Experimental Center of Sichuan University (Chengdu, Sichuan Province, China). They were randomly divided into five groups, with 16 rats in each group: sham operated group (Sham), weight-drop-induced TBI group (TBI), BMSC transplantation group (BMSC), OEC transplantation group (OEC), and BMSC and OEC cotransplantation group (CO).
Isolation, Culture, and Purification of BMSCs
The femur was obtained from adult male C57BL/6 green fluorescent protein (GFP) mice purchased from the Animal Experimental Center of Sichuan University. The two ends of the femur were cut to open. Dulbecco's modified Eagle's medium (DMEM; Gibco, Grand Island, NY, USA) was utilized to wash the marrow cavity three times using a 5-ml injector (BD Biosciences, San Jose, CA, USA). The cellular suspension of bone marrow was collected and a 200-μm mesh sieve (BD Biosciences) was used to filter and form the cellular suspension. The cellular suspension was seeded into a culture flask (Corning, Grand Island, NY, USA) and placed in a CO2 incubator containing 5% CO2 at 37°C. DMEM Nutrient Mixture F-12 (DMEM/F12; Gibco) supplemented with 10% fetal bovine serum (Gibco) was used to maintain cell growth. The fifth day after seeding, half of the complete medium was replaced with fresh medium. Then the complete medium was routinely changed every 3 days. The cells reached confluence at 10-14 days. Subsequently, the complete medium was removed, and 0.125% trypsin (Gibco) containing 0.02% EDTA (Sigma-Aldrich, St. Louis, MO, USA) was added to digest cells for 3 min at 37°C. The supernatant was removed by centrifugation (#3331 rotor, diameter = 10 cm; Thermo-Scientific, Waltham, MA, USA) at 1,000 rpm for 10 min. Then cells were resuspended in fresh complete medium. The cellular density was adjusted to 2 χ 105/ml and seeded with fresh complete medium into a new culture bottle. BMSCs were identified by phenotype/marker expression described in the immunohistochemistry below.
Isolation, Culture, and Purification of OECs
OECs were isolated and propagated using methods described previously (23). The olfactory bulb was harvested from 60-day-old male SD rats provided by the Experimental Animal Center of the Sichuan University. With the help of a dissecting microscope (Leica, Wetzlar, Germany), the cerebral meninges that covered the olfactory bulb were gently peeled. The tissue of the olfactory bulb was cut into 1-mm3 pieces and blended with the pumping action of a Pasteur pipette (BD Biosciences), forming a cellular suspension. About 0.4% trypan blue (Sigma-Aldrich) was utilized to detect the viability of the cells. Cells were seeded in 25-cm2 tissue culture flasks (Corning) at a concentration of 1 χ 106 cells in 1 ml of fresh complete medium and incubated at 37°C in a moist 5% CO2, 95% air atmosphere for 12 h. The cellular suspension was transferred into another 25-cm2 culture flask for 24 h (the first adherence). After that, the contents were aseptically poured into 24-well culture plates (NEST) coated by polylysine (Sigma-Aldrich) (the second adherence), followed by another week of culturing. Cells were treated with 5 χ 10−5 mol/L arabinofurano-sylcytosine (AraC; Invitrogen, Carlsbad, CA, USA) for 36 h. Then culture medium including AraC was replaced by the complete medium and added to 2 μmol/L forsko-lin (Sigma-Aldrich) and 20 μg/ml bovine pituitary extract (Sigma-Aldrich). After approximately 10 days of culturing, the cells were transplanted and identified by immunohistochemistry staining.
Identification of BMSCs and OECs by Immunohistochemistry
A two-step immunohistochemistry method was utilized to identify the BMSCs and OECs by cluster of differentiation 44 (CD44) antibody and low-affinity nerve growth factor receptor (P75) antibody, respectively. In brief, cultured and purified cells were placed on polylysine (40 g/L)-coated glass slides (Shitai, Jiangsu, China). Cells were fixed with 4% paraformaldehyde (Sigma-Aldrich) for 15-20 min at 4°C, followed by three washes with phosphate-buffered saline (PBS; 0.01 mol/L; Sigma-Aldrich), each for 5 min. Then endogenous peroxidase was inactivated by 3% H2O2 (Zhongshan Jinqiao, Beijing, China) treatment for 30 min, followed by three 0.01 mol/L PBS washes, each for 5 min. Then, 0.3% Triton X-100 (Amresco, Solon, OH, USA) was added and incubated with the glass slides for 30 min at 37°C. Five percent goat serum (Zhongshan Jinqiao) was added to block the nonspecific binding. Slides were further incubated at 37°C for 30 min. After that, CD44 antibody (1:100; Chemicon, Temecula, CA, USA) and P75 antibody (1:50; Chemicon) were added and incubated overnight at 4°C, followed by three 0.01 mol/L PBS washes, each for 10 min. Subsequently, HRP-conjugated anti-rabbit secondary antibody (1:100, Chemicon) was added, and slides were incubated for 30 min at 37°C, followed by three 0.01 mol/L PBS washes, each for 10 min. Then, 3,3′-diaminobenzidine (Zhongshan Jinqiao) was used for coloration three times, each for 5 min. The slides were dehydrated, cleared, coverslipped, observed, and photographed on a light microscope (CTR6000, Leica). Primary antibody was substituted for 0.01 mol/L PBS as negative control.
Protocol for TBI Model
This research was approved by the Sichuan University Animal Care and all procedures according to the Chinese Academy of Sciences guidelines. The modified Feeney method (35) was used to prepare the strike injury on the cerebral cortex of rats. In brief, 3.6% chloral hydrate (1 ml/100 g; Sigma-Aldrich) was utilized to anesthetize rats by intraperitoneal (IP) injection. The right parietal bone was exposed. A bore was drilled into the skull 2.5 mm away from the sagittalis suture and 1.5 mm away from the arcuate suture. The skull was removed until the “bone window” was enlarged to a 5.0 mm χ 5.0 mm area. A sterilized clout was placed on the exposed cerebral cortex. A ferric cylinder weighing 50 g was dropped along with the vertical metal pole to strike the clout, which resulted in a moderate contusion injury of the right cerebral cortex. Rats were attentively cared for and received 5 IU penicillin (Sigma-Aldrich) by IP injection daily to prevent postinjury infection. Manual evacuation of the bladder was performed. The rats were given food and drink ad libitum.
Cellular Labeling, Transplantation, and Application of Immunosuppressive Agents
BMSCs, isolated from GFP adult mice, emitted green fluorescence under the microscope. Cultured OECs were treated with Hoechst 33342 (10 μg/ml, prepared by DMEM/F12; Sigma) for 30 min in an incubator causing them to emit blue fluorescence. After the labeled cell suspension was prepared, it was stored at 4°C and transplanted into the host within 4 h. Briefly, 5 μl OECs, BMSCs, and BMSC + OEC cellular suspensions were prepared for transplantation. Cell transplantation was performed immediately after TBI. A glass capillary tube (Tykofiber, Shenzhen, China), connected between the stereotaxic apparatus (Alcott Biotech Co., Shanghai, China) and animals, was inserted respectively into four locations: 3 mm rostral, caudal, left, and right from the epicenter of the injured site. For each location, the cell suspension was injected at 1 μl/min at three depth points, viz., 2 mm, 1.5 mm, and 1 mm from the surface of the cortex. The volumes of cell suspension given were 1.5 μl, 2 μl, and 1.5 μl, respectively. The micropipette was left for 1 min to minimize efflux of the inoculum after pipette withdrawal.
Rats were administered a daily IP injection of cyclosporine A (diluted 10χ with normal saline; BioVision, San Francisco, CA, USA) starting 3 days before transplantation and continuing until the end of the experiments in order to suppress the immunologic rejection of the transplants.
Sample-Harvesting for RT-PCR and Immunohistochemistry
At sampling times, the rats were anesthetized IP with 3.6% chloral hydrate (1 ml/100 g; Sigma-Aldrich). Samples, 7 and 14 days postoperation (dpo), were harvested in the living body of rats for RT-PCR; however, it was fixed by paraformaldehyde for immunohistochemistry after 14 days posttransplantation. The injured cerebral cortex was completely exposed. The brain tissue round area diameter 4 mm away from epicenter was extracted and placed on ice. The samples were rinsed with dieth-ylpyrocarbonate (Sigma-Aldrich)-treated water in order to remove blood, hair, and other impurities. Then, tissues were placed into 1.5-ml Ependoff (Axygen) tubes and were stored in a −70°C freezer immediately to be used later for an RT-PCR experiment. Samples were put into 4% paraformaldehyde phosphate buffer and postfixed until processed for immunohistochemistry.
Quantitative Analysis of Survival, Migration, and Differentiation of Implanted Cells and Proliferation of Host Glial Cells, Neuronal Survival, and Axonal Regeneration
The postfixed samples were dehydrated with 20% sucrose (Sigma-Aldrich) at 4°C overnight, followed by sectioning into 15-μm sections. Then a two-step immunohistochemistry method was used to detect the number of differentiated and proliferated neurons from implanted cells in the host brain using neuronal nuclei (NeuN) antibody (1:1,000; Chemicon). Glial fibrillary acidic protein (GFAP; 1:500; Chemicon), a specific antibody for astrocytes, was used to evaluate the differentiated astrocytes from implanted cells and host proliferative astrocytes. Growth-associated protein 43 (GAP-43) antibody (1:1,000; Chemicon) was used to assess the axonal regeneration in the host brain. After mounting, the sections were placed on a fluorescence microscope (Leica DM I6000B) to observe the morphology, density, and distribution and determine the survival and migration of implanted cells. Five sections selected at equal intervals from every sample were used for observation under a light microscope. Five high-power visual fields randomly selected from every section were used for the counting of cells and axons.
Preparation of Primers, RNA Extraction, and RT-PCR
Primers of CNTF were designed by Primer Premier 5.0 software (Premier, Palo Alto, CA, USA) and synthesized by Takara Biological Company (TaKaRa, Shiga, Japan). The following set of primers was used: CNTF forward, 5′-CTTTCGCAGAGCAAACACCT-3′, reverse, 5′-CATC CCATCAGCCTCATTTT-3′; β-actin forward, 5′-GTAAA GACCTCTATGCCAACA-3′, reverse, 5′-GCTCAGTAA CAGTCCGCCTA-3′. The size of the PCR product was 422 bp and 277 bp, respectively. The total RNA of samples was extracted using the TRIzol (Invitrogen) method. Then reverse transcription was performed, and 20 μl cDNA was obtained. The PCR reaction system (Fermentas, Vilnius, Lithuania) was established: 2χ PCR Master Mix, 12.5 μl, forward primer: 0.5 μl, reverse primer: 0.5 μl, sample cDNA: 1 μl, nuclear-free water, 10.5 μl. Amplification was performed using the ABI 7000 Sequences Detector System (Applied Biosystems, Foster City, CA, USA) with 2 min at 94°C for initial denaturing, followed by optimized cycle numbers of 94°C for 45 s, annealing at 52°C to 65°C for 45 s, and 72°C for 30 s. β-Actin was used as an internal quantitative control.
PCR amplified products were detected by Agarose Gel Electrophoresis (Biowest, Barcelona, Spain) and analyzed by Bio-Rad (Hercules, CA, USA) gel automatic imaging system. The optical density value of the electrophoresis strip was measured using ImageJ 2.1.4.7 software (NIH, Bethesda, MD, USA). The ratio of optical density value of the target gene/β-actin was used for statistical analysis.
Western Blot Analysis
Brain tissues were rapidly isolated from rats and immediately transferred into freshly prepared NP-40 lysis buffer (Biyuntian, Shanghai, China). An equal amount of protein (20 μg) from each sample was separated by sodium dodecyl sulfate-polyacrylamide gel (Sigma-Aldrich) electrophoresis and blotted on polyvinylidene fluoride membranes (Millipore, Billerica, MA, USA). The membranes were blocked with 5% milk (Wandashan, Heilongjiang, China) and incubated overnight at 4°C with primary antibodies specific for β-actin (loading control, 1:1,000; Santa Cruz Biotechnology, Santa Cruz, CA, USA), STAT3 (1:1,500; Cell Signaling Technology, Danvers, MA, USA), and phospho-STAT3 (1:1,000; Cell Signaling Technology). Then the membranes were incubated with peroxidase-conjugated IgG (1:10,000; Santa Cruz Biotechnology) diluted in 5% milk for 2 h at room temperature. The ratio of pSTAT3/STAT3 was calculated to assess the activation state of this signaling pathway.
Neurological Severity Score
The method reported by Chen et al. (5) was used to evaluate the neurological severity score (NSS), that is, the degree of impaired cerebral function of TBI rats. The rats were trained before NSS evaluation, and the rats scored 0 were selected as experimental animals. The NSS was obtained by blind evaluation of professional researchers. All five groups of rats were subjected to NSS at 7 days and 14 days dpo.
Statistical Analysis
All experiments in vitro were performed at least three times independently. Data were analyzed by Bonferroni's multiple comparison test following a one-way ANOVA (SPSS 13.0; IBM, Armonk, NY, USA) and expressed as mean ± SEM. A value of p <0.05 was considered statistically significant.
Results
Identification for In Vitro Culture of BMSCs
At 12 h after primary culture, the cells began to adhere, and the number of adherent mononuclear cells increased gradually with the extending of culture time. Simultaneously, the endoparticles of these cells augmented gradually. At 48 h, the BMSCs presented a spindle shape and deplanate morphology under microscope, with the features of fibroblasts. Ninety-six hours later, the cells predominantly turned out to be of aliform growth type. After subculturing for five passages, the morphology of the cells became relatively uniform. At this stage, the morphology of cells was mainly a radiated shape (Fig. 1A). In order to identify BMSC features, a CD44 antibody was chosen, and the cells showed brown staining (Fig. 1B), confirming that they were BMSCs. Furthermore, BMSCs isolated from GFP mice could be observed under a microscope emitting green fluorescence, which can be traced in vivo after transplantation (Fig. 1C).

Morphology of bone mesenchymal stem cells (BMSCs) and olfactory ensheathing cells (OECs) in vitro. Representative photomicrographs showed the morphology of BMSCs. (A-C) The cells were mainly radiated shaped and relatively uniform after subculturing for five passages (A). Cultures of BMSCs showed strong brown staining of CD44, a marker of BMSCs (B). GFP was expressed in BMSCs, isolated and cultured from GFP mice, and was observed under a microscope (C). Characteristics of the OEC preimplantation (D-F) showed that the bipolar and multipolar cells were predominant with a strong three-dimensional, interlacing network (D). Immunohistochemistry shows OECs were positive for low-affinity nerve growth factor receptor (p75) (E) and could be labeled with Hoechst 33342 in vitro (F). GFP (green) and Hoechst 33342 (blue) were shown in all figures. The arrows depict the BMSCs (A-C) and OECs (D-F). Scale bars: 100 μm, shown in D.
Identification for In Vitro Culture of OECs
The cultured cells began to adhere 12 h after seeding. They formed a monolayer after fusion and showed a mosaic arrangement. After the second adherent culture (24 h), the cell attachment rate was about 70%. Most of the cells had relatively bigger cell bodies, shorter processes, and showed scattered growth under the microscope. The fifth day after the second adherent culture, three classical cellular shapes, bipolar, multipolar, and oblateness emerged. The number of fibroblasts was significantly decreased by AraC treatment after culturing for 36 h. The bipolar and multipolar cells were predominant with long and thin processes, interlacing in a network. At this time, the cellular morphology became clear with strong three-dimensional shape and brightness (Fig. 1D). OECs were identified by an immunocytochemical method, and the results showed a great number of bipolar and multipolar OECs were positive for P75, which was located in cytoplasm and processes (Fig. 1E). Moreover, Hoechst 33342 was used to label and trace the fate of OECs in vivo, which emitted a visible blue fluorescence (Fig. 1F).
Survival, Migration, and Differentiation of Implanted Cells
Fluorescence was found in the BMSC group, suggesting that implanted BMSCs could survive in the host cerebral cortex (Fig. 2A). Similarly, we observed fluorescence in nuclei, implying that transplanted OECs labeled by Hoechst 33342 had also survived in the host brain (Fig. 2B). In order to investigate the profile and differentiation of implanted cells, tissues were stained by immunofluorescence at 14 days after cell transplantation. The results of immunofluorescence suggest that differentiation to neuron-like cells and astrocytes was not observed in the BMSC group (data not shown). However, a few NeuN-immunopositive cells (arrows) were observed in the cotransplantation group(Fig. 2C, D). These cells integrated well with local brain tissue and migrated to surrounding brain tissue.

Survival, migration, and differentiation of implanted cells in vivo. BMSCs implanted in host cerebral cortex can survive and emit fluorescence (A). Similarly, fluorescence can be observed in nuclei, implying that transplanted OECs previously labeled by Hoechst 33342 had survived in host brain (B). Immunofluorescent analysis showed that a few NeuN-immunopositive cells were observed in the BMSC and OEC cotransplantation group (CO), suggesting the differentiation of neuron-like cells from the BMSCs (C, D). Moreover, these cells integrated well with local brain tissue and migrated to the surrounding brain tissue. The arrows show the surviving BMSCs (A), OECs (B), and differentiated NeuN neurons from the BMSCs (C, D). Scale bars: 100 μm, shown in C.
Changes in Host Cortical Cell Number
To analyze the number of neuronal and glial cells that proliferated and differentiated in host cells of cortical tissue, NeuN antibody and GFAP antibody were used in immunohistochemistry. In the sham operation group, the cortical neurons were healthy, and cellular atrophy was not observed. However, the number of host neurons decreased, and the cell bodies of residual cells showed obvious atrophy in TBI group. All of these changes were reversed by cell transplantation therapy. Following statistical analysis, the numbers of NeuN-positive cells in the host cortical tissue following surgery exhibited a significant decrease, compared with the sham group (p <0.01)(Fig. 3A-C). NeuN-positive cells began to proliferate rapidly in the BMSC group, OEC group, and CO group (p <0.05 vs. TBI) (Fig. 3A, C-F), indicating that cortical neurons proliferated rapidly, and cellular atrophy diminished after BMSC and/or OEC transplantation. Moreover, this beneficial effect was most obvious in the CO group (Fig. 3A, C, F). In contrast, in the TBI group, GFAP-positive cells were significantly increased compared to the sham operated group (p <0.01) (Fig. 3A, G, H). GFAP-positive cells significantly decreased following BMSC or OEC transplantation and were the least in the BMSC and OEC cograft group (p <0.05 vs. TBI) (Fig. 3A, H-K). These results demonstrated that cell grafting could enhance cortical neuron proliferation and inhibit cortical astrocyte growth.

The number of neuronal and glial cells changes in host brain cortex. The number of host neurons showed (A-F) that NeuN-positive cells in the host cortex following surgery was significantly decreased, compared with the sham group (p <0.01) (A-C). However, it increased rapidly in the BMSC, OEC, and CO groups (p <0.05 vs. TBI) (A-F). Moreover, the effect, described above, was most obvious in the CO group (A, C, F). A seemingly parallel and opposite countertrend existed in the number of GFAP-positive cells (A, G-K). The arrows depict the positive NeuN neurons (B-F) and GFAP astrocytes (G-K). Scale bars: 100 μm, shown in G. *p <0.05, #p <0.01.
GAP-43-Positive Fibers Change in Host Brain
The effect of cell grafting on the change of GAP-43-positive fibers had the same tendency with neuronal cells in the host brain. The number of GAP-43-positive axons significantly decreased following TBI, while it significantly increased and recovered after cell transplantation treatment, especially in the BMSC and OEC cograft group (p <0.05) (Fig. 4A-E).

Effects of cell grafts on axonal regeneration in host brain. The immunostaining of GAP-43 antibody in cortical sections following cell transplantation was used to assess the axonal regeneration in host brain. The number of GAP-43-positive axons significantly decreased following TBI, while it significantly increased and recovered after cell transplantation, especially in the BMSC and OEC cograft group (p <0.05) (A-E). The arrows depict the GAP-43-positive regenerated axons. Scale bars: 100 μm, shown in C. *p <0.05.
CNTF Expression After Cell Transplantation
PCR-amplified products were detected, and the optical density value of the electrophoresis strip was measured using ImageJ software. The ratio of optical density value of the target gene was accordant with the control one. The expression of mRNA for CNTF in the brain close to the lesion site significantly decreased at 7 days after cellular transplantation in the cografting group (p <0.05 vs. TBI), while there was no significant change at 14 days. Besides, no other significant differences were found in other groups at 7 dpo or 14 dpo (Fig. 5).
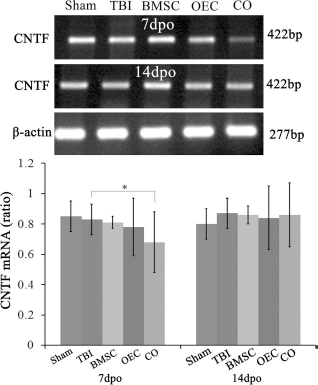
Effects of cell grafts on ciliary neurotrophic factor (CNTF) expression. The expression of mRNA for CNTF in the brain close to the lesion site significantly decreased at 7 days after cellular transplantation in the cografting group (p <0.05 vs. TBI). Besides, there was no significant difference in all of the groups at 14 dpo. The ratio of optical density value of the target gene was in accord with the control one. *p <0.05.
The Jak/STAT3 Signaling Pathway Activation After Cell Transplantation
Following cell transplantation, the expression of pSTAT3 and STAT3 was evaluated by Western blot. We calculated the ratio of pSTAT3/STAT3 as the extent of activation of this signaling pathway. Our data demonstrated the ratio was decreased greatly at day 7 of TBI treatment when compared with sham control (p <0.05), while this variation was reversed, and the ratio was significantly increased in the BMSC (p <0.05 vs. TBI), OEC (p <0.01 vs. TBI), and cotransplantation groups (p <0.01 vs. TBI). Moreover, the ratio following OEC and cotransplantation was increased when compared with that of the BMSC group (p <0.05 vs. TBI) (Fig. 6). There was no statistically significant difference among all groups at 14 days after cell transplantation (data not shown), similar to the data from CNTF mRNA expression at the same time point.

Effects of cell grafts on the Jak/STAT3 signaling pathway. Western blot analysis showed the ratio of pSTAT3 and STAT3 was decreased at 7 days following TBI treatment when compared with sham control (p <0.05), while it was significantly increased in the BMSC (p <0.05 vs. TBI), OEC (p <0.01 vs. TBI), and cotransplantation groups (p <0.01 vs. TBI). Moreover, compared with that of the BMSC group, the ratio was significantly increased in the OEC and cotransplantation groups (p <0.05 vs. TBI). #p <0.01, *p <0.05.
NSS
To determine the effect of cellular transplantation therapy on neurological function after TBI, NSS was used to assess the functional recovery. Rats exhibited movement dysfunction immediately after surgery, and the NSS was significantly higher (p <0.01 vs. Sham) at 7 dpo and 14 dpo. The NSS of the cell transplantation groups significantly decreased when compared with the TBI group at 14 days after cell transplantation (p <0.05), which suggested that the BMSC or OEC transplantation could have ameliorated the neurological function in TBI rats. The improvement was more significant in the BMSC and OEC cograft group. However, there was no significant difference at 7 days between cell transplantation groups and the TBI group (Fig. 7).

Effect of cell grafts into host brain on neurological severity score (NSS). To determine the effect of cell graft therapy on neurological function after TBI, NSS was used to assess the functional recovery. After surgery, rats were immediately paraplegic with no observable hindlimb movement. Compared with TBI group, the NSS of the BMSC, OEC, and CO groups were significantly decreased at 14 days after transplantation (p <0.05), suggesting cell transplantation could significantly ameliorate neurological deficits in TBI rats. However, there was no significant difference between cell graft groups and TBI group at 7 days after transplantation. *p <0.05, #p <0.01.
Discussion
In the present study, the TBI model was established for determining the therapeutic effects of cell transplantation, including BMSCs or OECs alone and BMSC and OEC cotransplantation. We also explored the potential molecular mechanisms behind the therapeutic benefits. We examined the fate (survival, proliferation, and differentiation) of these cells following transplantation in host brain in vivo. As expected, morphological evidence supported that not only BMSC and OEC grafting, respectively, could promote neuronal survival and axonal regeneration and inhibit astrocyte proliferation, but BMSC and OEC grafting combined showed significant beneficial effects. Moreover, we found that the rats that suffered TBI exhibited severe neurological function deficits, but the changes were completely reversed by BMSC or OEC single grafting and combined grafting, especially. The possible molecular mechanism is related to the decrease of CNTF expression, which plays an important role on the promotion of scar formation.
Purification and Identification of Cultured Cells
BMSCs, a kind of self-renewing cell, have the capacity for multilineage differentiation and may act as a promoter of tissue regeneration and repair under abnormal physiological conditions (37). This study demonstrated that the cultured BMSCs showed a state of suspension initially, and 6 h later adherent cells were observed. At 24 h after plating, the adherent cells significantly increased. These cell bodies gradually became fusiform shaped, or deplanate and polygon shaped. Primary cultured cells reached confluence and were well distributed on the culture bottle at day 4. At this stage, the cells were mainly fusiform and polygon shaped with irregular process. The two-stage adherent technique was applied to purify BMSCs for transplantation. Identified by CD44, these positive cells were confirmed as BMSCs.
In the culture system of OECs derived from the olfactory bulb, fibroblasts were the main contaminated cells. In this study, adult rats were favorable for the purification of OECs because of the mature development of olfactory bulb. Moreover, with the help of AraC, the two-stage adherent method removed the fibroblasts effectively. Therefore, compared with neonatal rats, it was easy to obtain relatively pure OECs from adult rats. Considering the strong adherent ability of fibroblasts, they could directly be adherent on the uncoated smooth glass bottle 1 h after seeding. Astrocytes commonly turn out to be adherent during 24 h, while OECs need 24-48 h to be adherent. Based on the different adherent abilities of those cells, our study used a two-stage adherent method to purify the OECs. Nash et al. (23) reported that above 90% purity of OECs could be obtained by using the two-stage method.
In the present study, AraC, a kind of periodicity-specific antimetabolite, was also used to inhibit the growth of fibroblasts, which could inhibit the activity of DNA polymerase, reduce the synthesis of DNA, and interfere with the replication of DNA (1). The cells occupying DNA synthetic phase (S phase) are most sensitive to it. Therefore, during the process of cell culture, the addition of AraC effectively removed the contamination of fibroblasts. In this study, we used a two-stage adherent combined with chemical inhibitory method to further increase the purification of OECs.
Survival, Migration, and Differentiation of Implanted Cells in Host Brain
Since nervous tissues have distinct immunologic mechanisms (4), to promote the survival of implanted cells in host brain and prolong the survival time, it is important to apply immunosuppressive agents as a practical transplantation strategy, which ensures that the host accepts transplanted cells as much as possible. Moreover, the cellular labeling before transplantation allowed the implanted cells to be observed and traced under a fluorescence microscope. Our results showed that the transplanted cells integrated well with the host brain tissues. Survival and differentiation into neurons were the two key factors for host neurological restoration (38). In the BMSC or OEC group, NeuN- and GFAP-positive cells were not observed. This is at variance with the controversial assertion that neuronal differentiation from BMSCs could occur in vivo after transplantation (22,27). Recent studies have demonstrated that OECs promoted axonal regeneration, remyelination, and synaptic formation, facilitating the amelioration of neurological function after SCI (9,13,28). BMSCs could be differentiated into neurons and glial cells, secreting neurotrophic factors, cytokines, and other factors with biological activity (6,39), and ultimately play a crucial role in SCI therapy (7). In our laboratory, BMSC and OEC cografting into the host brain following TBI was designed and performed for the first time. Interestingly, in the BMSC and OEC cografting group, the robust immune-positive NeuN cells were observed, suggesting that the cografting cells partially differentiated into neuronal cells.
Cellular Transplantation Inhibited the Proliferation of GFAP-Positive Cells, Promoted the Survival of NeuNPositive Neurons, GAP-43 Axonal Regeneration
Immunohistochemistry showed that the GFAP-positive cells proliferated with the impairment of neurons after TBI. In the BMSC or OEC group, proliferation of GFAP-positive cells was effectively inhibited, while the survival of NeuN-positive neurons and GAP-43 axonal regeneration were promoted. Notably, in the cotransplantation group, beneficial effects described previously were the most optimal (15,41) after 14 days posttransplantation. This result indicates that cografting had the better therapeutic efficiency than BMSC and OEC transplantation alone. The probable reason was that OECs facilitate the survival of BMSCs and induce BMSC differentiation into neurons. The interaction may promote our understanding of neuron survival and axonal regeneration.
Cellular Cografting Downregulated the Expression of CNTF mRNA
In this study, RT-PCR results showed that in the BMSC or OEC transplantation, CNTF expression did not significantly decrease compared to the TBI group at 7 dpo. However, in the cotransplantation group, the expression of CNTF was significantly downregulated (p <0.05 vs. TBI). Previous studies indicated that CNTF served as a signaling molecule of paracrine and autocrine secretion and had an effect on the recovery of neural injury (18). Other studies found that, on the contrary, CNTF enhanced the gummosis of astrocytes in CNS diseases and subsequently played a role in the promotion of scar formation (3,12). In this study, we found that cellular cografting was favorable for the downregulation of CNTF, suggesting that it played a crucial role in inhibiting the proliferation of astrocytes after TBI and ultimately provided a conducive environment for the regeneration of GAP-43-positive axons and local synaptic reconstruction.
The Effect on Jak/STAT3 Following Cell Transplantation Treatment of TBI
The Jak/STAT3 pathway, an evolutionary conserved signaling pathway, is closely associated with many vital cellular processes, including cell differentiation, growth, and proliferation. Previous data suggested that STAT3 phosphorylation was enhanced both in CNTF-/- and wild-type mice, but this effect was significantly more pronounced in the mutants following optic nerve crush (14). Coincidentally, our data showed CNTF gene expression was reduced significantly, and the ratio of pSTAT3/ STAT3 was increased significantly at 7 days after cell transplantation. Therefore, we reasoned that the beneficial effects of cell transplantation therapy, including stem cell differentiation, neuron proliferation, axonal regeneration, and astrocyte growth inhibition, may interact by the inactivation of CNTF and activation of the Jak/STAT3 signaling pathway.
Implanted Cells Promoting the Behavioral Amelioration
NSS is a quantified system to evaluate neurological function, such as movement, sensory, equilibrium, and nervous reflex. In NSS evaluation, the higher the scores, the more serious is the neurological impairment. So far, the NSS system is one of the most reliable methods widely accepted for the evaluation of neurological function.
In this study, NSS showed that the cerebral neuromotor function in OEC or BMSC transplantation and the BMSC and OEC cografting group was significantly ameliorated when compared with the TBI group at 14 dpo (p <0.05), which was consistent with previous studies (2,8,29,36). However, this satisfactory effect was not observed between the cografting and BMSC or OEC groups as well as between all of the cell grafting groups and TBI group at 7 days after surgery. This result suggested that (i) cell grafting could improve the neurological deficits induced by TBI, and it may be in a time-dependent manner; (ii) the effect of cografting of BMSCs and OECs was not strong enough to ameliorate the neurological function. In addition, we used moderate brain injury models, and the neurological function of rats recovered to some extent 14 days after injury. Therefore, the significant difference was difficult to find in all cell transplantation groups. Furthermore, although neither the BMSC or OEC transplantation alone or the cografting played a role in the recovery of neurological function, the differential effects on neurological function recovery can be induced in the long term. Our results provide new evidence for consideration of stem cell and a gliocyte combined therapy for the repair of traumatic CNS injury.
In this study, after the transplantation, the proliferation of glial cells was inhibited, but the survival of host neurons and regeneration of GAP-43-positive axons were promoted. We found that BMSC and OEC cografting and BMSC and OEC transplantation alone promoted the recovery of neurological function in TBI rats, which may be closely related to the decrease of CNTF expression and activation of the Jak/STAT3 signaling pathway. This study provided important morphological and molecular biological evidence for the therapy of TBI by using BMSC and/or OEC transplantation.
Footnotes
Acknowledgment
This research was supported by a grant from the China National Science Foundation (No. 81260191, 81271358, 81172828, 81171176). We thank Professor Seng-Kee Leong for his valuable comments on this manuscript. The authors declare no conflicts of interest.
