Abstract
Neurogenesis in the subgranular zone of the hippocampal dentate gyrus may act as an endogenous repair mechanism in Alzheimer's disease (AD), and the Wnt signaling pathway has been suggested to closely modulate neurogenesis in amyloid-β (Aβ)-related AD models. The present study investigated whether mesenchymal stem cells (MSCs) would modulate hippocampal neurogenesis via modulation of the Wnt signaling pathway in a model of AD. In Aβ-treated neuronal progenitor cells (NPCs), the coculture with MSCs increased significantly the expression of Ki-67, GFAP, SOX2, nestin, and HuD compared to Aβ treatment alone. In addition, MSC treatment in Aβ-treated NPCs enhanced the expression of β-catenin and Ngn1 compared to Aβ treatment alone. MSC treatment in Aβ-treated animals significantly increased the number of BrdU-ir cells in the hippocampus at 2 and 4 weeks compared to Aβ treatment alone. In addition, quantitative analysis showed that the number of BrdU and HuD double-positive cells in the dentate gyrus was significantly higher in the MSC-treated group than in controls or after Aβ treatment alone. These results demonstrate that MSC administration significantly augments hippocampal neurogenesis and enhances the differentiation of NPCs into mature neurons in AD models by augmenting the Wnt signaling pathway. The use of MSCs to modulate endogenous adult neurogenesis may have a significant impact on future strategies for AD treatment.
Introduction
Ample evidence has demonstrated that the adult mammalian brain has the potential to generate new neurons and incorporate them into brain areas affected by a disease process (8,23). Neurogenesis in the subventricular zone and the subgranular zone (SGZ) of the hippocampal dentate gyrus may act as an endogenous repair mechanism (7). Thus, the modulation of endogenous neurogenesis may have a significant impact on future therapeutic strategies for neurodegenerative diseases such as Alzheimer's disease (AD) (30). Neuronal progenitor cells (NPCs), referred to as type 1 cells in the SGZ of the hippocampus, are multipotent progenitors capable of self-renewal and producing neurons and glia. NPCs have been identified as the dividing three cell types in the hippocampus: the radial type 1 cells that have astrocytic properties and are characterized by both expression of the glial cell marker [glial fibrillary acidic protein (GFAP)] and the neuronal progenitor markers (SOX2 and nestin); type 2 transit-amplifying cells, which are thought to stem from type 1 cells; and type 3 cells that differentiate from type 2 cells (47).
AD, the most common neurodegenerative disorder, is characterized pathologically by synaptic loss and the presence of amyloid-β (Ab) plaques and tau tangles (43). Growing evidence has demonstrated that neurogenic activity in the adult SGZ of the hippocampus is altered in the early stages of AD, although conflicting results have been reported depending on disease stage or animal model (30). Various key molecules involved in AD pathogenesis seem to modulate adult hippocampal neurogenesis. Of these, Aβ itself or metabolites of amyloid precursor proteins (APP), including soluble APPa, a carboxyterminal fragment, or APP intracellular domains, are shown to play unique roles in the modulation of neurogenesis (51). Regarding the mechanisms that underlie the fate of NPCs in AD, an interruption of Wnt/b-catenin signaling by Aβ may play a key role in the modulation of neurogenesis in NPCs. Several in vitro and in vivo studies have shown that Wnt3a is expressed in the hippocampal neurogenic niche and modulates the generation of newborn neurons in the SGZ of the hippocampus (19,49). Reduced Wnt/β-catenin signaling has been reported in the AD brain (29), and decreased expression of β-catenin has also been found in AD patients carrying presenilin-1-inherited mutations (12,21). Furthermore, Shruster et al. (41) demonstrated that disrupted β-catenin signaling is closely coupled with decreased hippocampal neurogenesis in the human AD brain.
Mesenchymal stem cells (MSCs) are multipotent stem cells that are capable of differentiating into various cell types under appropriate conditions (36). Additionally, MSCs secrete various cytotropic factors that, in turn, exert neuroprotective effects (48). Our series of animal studies have demonstrated that MSCs have neuroprotective effects through complex mechanisms, including the modulation of neuroinflammation, enhancement of cell survival signals, increased neurogenesis, and modulation of ubiquitinated protein (3,9,16,17,20,35). In the present study, we investigated whether MSCs would modulate hippocampal neurogenesis in AD models through modulation of the Wnt/b-catenin signaling pathway.
Materials and Methods
Aβ Peptide Preparation
Aβ1—42 was prepared as described previously (38). Briefly, Aβ1—42 (>97% ultra pure, HFIP; Millipore, Temecula, CA, USA) was initially dissolved in anhydrous dimethyl sulfoxide (Sigma-Aldrich, St. Louis, MO, USA) to a concentration of 200 μM. Aβ25—35 and Aβ1—40 were also prepared according to the manufacturer's instructions (Abcam, Cambridge, UK). The Aβ peptides were stored at −70°C until further processing. Before use, they diluted in Dulbecco's modified Eagle medium (DMEM; HyClone, Irvine, CA, USA) to the indicated concentration.
NPC and MSC Cultures
Parasagittal sections were taken from the medial surface of each hemisphere of postnatal day 1 Sprague—Dawley rats (Samtako Bio Korea, Seoul, South Korea), and a wedge of tissue was microdissected from a portion of the hippocampus that included the dentate gyrus (34). The isolated NPCs were expanded in DMEM supplemented with 10% fetal bovine serum (FBS; Hyclone) and 1% penicillin/streptomycin (P/S; Hyclone). Frozen vials of characterized human MSCs at passage 2 were obtained by written informed consent from all persons who agreed to the use of their cells for research purposes from the Severance Hospital Cell Therapy Center (Seoul, South Korea). MSCs were maintained in DMEM supplemented with 10% FBS and 1% P/S. These cells were cultivated in a humidified incubator at 37°C and 5% CO2 before use. For in vitro experiments, NPCs were plated at a density of 1 × 104/cm2 and treated with 20 μM of Aβ for 24 h. In addition, Aβ-treated NPCs were treated with Dkk1 (20 ng/ml) as a Wnt inhibitor (R&D Systems, Minneapolis, MN, USA) for 24 h (37). MSCs were plated at a density of 5 × 103/cm2. For in vivo experiments, after passage 6, MSCs were injected into mice via the tail vain (1 × 106 cells/mouse).
Cocultures of MSCs and NPCs Treated with Aβ
The effects of MSCs were tested in NPCs that were cocultured without direct contact using a Costar Transwell (Corning, Big Flats, NY, USA). The MSCs were cultured on the permeable membrane of Costar Transwell insert, and the NPCs were maintained on the bottom of a plate in a humidified incubator at 37°C and 5% CO2 for either 2 days or 2 weeks. In the control and Aβ group, NPCs were cultured by the same method without MSCs. The NPCs were then collected for assay. All experiments were replicated three times.
Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
Total RNA was extracted from the NPCs using Trizol reagent (Invitrogen, Carlsbad, CA, USA) in accordance with the manufacturer's protocol. RNA concentration was determined by absorption at 260 nm using NanoDrop Lite Spectrophotometer (Thermo Scientific, Wilmington, DE, USA), and cDNA templates were synthesized from 1 mg RNA using M-MLV Reverse Transcriptase (Invitrogen). The PCR reaction was performed using 10 pmol each of the primers for Ki-67 (Fwd: CTATTCAAAGTGGGTGTGGA; Rev: AACTTGCTCTGGTTGTAGAG), GFAP (Fwd: CT TCTCAACCTGGAAGAGTC; Rev: ACACTAATACGA AGGCACTC), SOX2 (Fwd: CCTCTTTAAGACTAGGG CTG; Rev: GAGATCTGGCGGAGAATAGT), nestin (Fwd: CCGAGCTGGAGCGGGAGTTA; Rev: GCCTGACCCA GCGAGCTTTC), HuD (Fwd: TCTGTAAGAAGGGAAT GTCG; Rev: TGGGTCATATTCTGGGGTAA), β-catenin (Fwd: ATGACGCGGAACTTGCCACC; Rev: AGCGCA TGATGGCGTGTCTG), Ngn1 (Fwd: AAGCTCACCAA GATTGAGAC; Rev: GGGGCAGGAAAGAAGTATTA), GAPDH (FwCACCTTTGATGC; Rev: GATGGTATTCG AGAGAAGGG). After an initial denaturation at 95°C for 2 min, 30 cycles of PCR were performed, consisting of denaturation (1 min, 95°C), annealing [1 min, 47°C (GFAP), 48°C (Ki-67), 49°C (SOX2), 61°C (nestin), 53°C (HuD), 62°C (β-catenin), 49°C (Ngn1), 51°C (GAPDH)], extension (1 min, 72°C) followed by a final extension (5 min, 72°C). The PCR products were separated by electrophoresis on 2% agarose gel (Intron, Seongnam-si, Kyungki-do, South Korea) and stained with ethidium bromide (Sigma-Aldrich). Gels were examined under UV illumination (Bio Imaging Systems, Jerusalem, Israel). Gene expression was normalized to the expression of GAPDH. Density was measured using Image Gauge V4.0 software (FUJIFILM Science Laboratory, Tokyo, Japan).
Animal Study
All procedures were performed in accordance with the Laboratory Animals Welfare Act, the Guide for the Care and Use of Laboratory Animals, and the Guidelines and Policies for Rodent Experiments provided by the Institutional Animal Care and Use Committee at the Yonsei University Health System. Male C57BL/6 mice (Orient Bio, Seongnam-si, Kyungki-do, South Korea) were acclimated in a climate-controlled room with a constant 12 h light/dark cycle (12 h on, 12 h off) for a week prior to the initiation of drug administration. At 6 weeks of age, the mice received Aβ1—42 (400 μM/mouse) or control solution (PBS) administered by intracerebroventricular injection. ***Intracerebroventricular administration was carried out in accordance with the procedure described previously (10,45) with minor modifications. Briefly, mice were anesthetized with isoflurane (Baxter, Deerfield, IL, USA), and Aβ (400 μM/mouse, intracerebroventricular injection, n = 5, each group) or control solution (PBS) was slowly injected bilaterally into the lateral ventricles (0.2 mm posterior to bregma, 1.0 mm lateral to midline, and 3.0 mm ventral to the brain surface) using a stainless-steel injection needle (26 gauge) connected to a 10-μl Hamilton microsyringe (Hamilton, Reno, NV, USA). The needle was left in place for 10 min before it was slowly withdrawn. The mice were randomly divided into three groups (n = 5; each group): (1) Sham, (2) PBS, (3) MSCs. Mice in the MSC group were subjected to MSC (1.0 × 106 cells/mouse) delivery on postoperative day 3. MSCs were suspended in 200 μl PBS and carefully injected in the tail vein using a 26-gauge syringe, and BrdU [50 mg/kg, intraperitoneal (IP) injection; Roche, San Francisco, CA, USA] was also injected consecutively for 5 days. All mice were sacrificed on postoperative weeks 1, 2, and 4.
Brain Sample Preparation
For immunochemical analysis, all mice were deeply anesthetized with chloral hydrate (0.4 g/kg, IP; Fluka, Steinheim, Germany) and then perfused with 4% paraformaldehyde (Sigma-Aldrich) in 0.1 M phosphate buffer (PB; pH 7.4). The brains were then sectioned on a sliding microtome Leica SM 2010R (Leica Biosystem, Wetzlar, Germany) to obtain 4-μm-thick and 40-μm-thick coronal sections. All sections were stored in tissue stock solution (30% glycerol; Wako, Osaka, Japan; 30% ethylene glycol; DUKSAN, Ansan-si, Kyungki-do, South Korea; 30% 3rd D.W., 10% 0.2 M PB; pH 7.2; Sigma-Aldrich).
Immunocytochemisty and Immunohistochemistry
Brain tissues and cocultured NPCs were washed twice in PBS and incubated in 0.2% Triton X-100 (Sigma-Aldrich) for 30 min at room temperature (RT). Then they were blocked with 0.5% bovine serum albumin (BSA; Sigma-Aldrich) for 30 min. After blocking, they were rinsed three times with 0.5% BSA and incubated overnight at 4°C with specific primary antibodies. The primary antibodies were used as follows: mouse anti-GFAP and rabbit anti-GFAP (1:1,000 for immunocytochemistry; Chemicon, Temecula, CA, USA), mouse anti-neuronal nuclei (NeuN; 1:100 for immunocytochemistry; Millipore), rabbit anti-nestin (1:200 for immunocytochemistry; Millipore), rabbit anti-Ki-67 (1:500 for immunocytochemistry; Abcam), rabbit anti-ELAVL4 (HuD; 1:120 for immunocytochemistry and immunohistochemistry; Abcam), rabbit anti-neurogenin 1 (Ngn1; 1:200 for immunocytochemistry; Mybiosource, San Diego, CA, USA), mouse anti-BrdU (1:200 for immunohistochemistry; Roche), CD90, CD44, CD34, CD45 (1:100 for immunocytochemistry; Chemicon), and mouse anti-NuMA (1:200 for immunohistochemistry; Millipore).
Immunofluorescence labeling was carried out by incubating the cells in goat anti-mouse IgG Alexa Fluor 488 (1:500; Invitrogen), anti-rabbit IgG Alexa Fluor 555 (1:500; Invitrogen), anti-mouse IgG Cy-3 (1:500; Chemicon), or anti-rabbit IgG FITC (1:500; Chemicon). The cell nuclei were counterstained with 4′,6-diamidino-2-phenylindole (Invitrogen). The BrdU immunohistochemistry was detected with 0.05% diaminobenzidine (DaKo, Carpinteria, CA, USA), and 0.03% H2O2 (Sigma-Aldrich). The immunostained cells were analyzed using bright-field microscopy (BX41, Olympus, Shinjuku, Tokyo, Japan) and viewed under a Zeiss LSM 700 confocal imaging system (Zeiss, Heidelberg, Germany). To analyze the localization of different antigens in double-stained samples, immunofluorescence images were created from the same tissue section and merged using Zeiss ZEN software (Zeiss).
Western Blot Analysis
Brain tissues was dissolved in ice-cold lysis buffer (20 mM Tris-HCl, pH 7.5, 1 mM EDTA, 5 mM MgCl2, 1 mM dithiothretol, 0.1 mM phenylmethylsulfonyl fluoride plus protease inhibitor cocktail) (Sigma-Aldrich). The lysates were centrifuged (20 min, 14,000 × g, 4°C), and supernatants were transferred to fresh tubes. Proteins were analyzed using the Bio-Rad Protein Assay Kit (Hercules, CA, USA). Equal amounts of protein were loaded in each lane with loading buffer containing 0.125 M Tris-HCl, pH 6.8, 20% glycerol, 4% sodium dodecyl sulfate, 10% mercaptoethanol, and 0.002% bromophenol blue. Samples were boiled for 15 min before gel loading. Proteins were transferred electrophoretically to polyvinylidiene difluoride membranes (Millipore). Membranes were washed in Tris-buffered saline solution with 2.5 mM EDTA (TNE) and then blocked in TNE containing 5% skim milk for 1 h. Membranes were incubated overnight at 4°C with primary antibodies specific for rabbit anti-β-catenin (1:1,000; for Western blot; Cell Signaling, Danvers, MA, USA) and mouse anti-actin (1:2,000; for Western blot; Santa Cruz, Santa Cruz, CA, USA). After washing, the membranes were incubated with secondary antibodies (1:2,000; Amersham, Piscataway, NJ, USA) for 1 h at RT, and washed again. The blots were finally developed with the ECL Western blotting detection reagents (Amersham). For semiquantitative analysis, the density of immunoblot bands was measured with the computer imaging (Fugi film).
Radial Arm Maze
The radial arm maze used in the present study consisted of eight arms, numbered from 1 to 8 (35 × 5 cm), extending radially from a central area (20 cm in diameter), with a 5-cm edge around the apparatus. Each animal was placed individually in the center of the maze and subjected to a reference and working memory task for 28 days, where the same four arms (numbers 1, 3, 5, and 7) were baited for 5 days on training trials. The other four arms (numbers 2, 4, 6, and 8) were never baited. Before the actual training began, the animals were shaped for 5 days to run to the end of the arms and consume the water. The water was initially available throughout the maze, but gradually was restricted to the food cup. The training trial continued until all four baits in the food cups had been consumed or until 5 min had elapsed. We measured the number of reference memory errors (entering an arm that was not baited) and the time taken to consume all four waters.
Stereological Cell Counts
To determine the number of BrdU- and HuD-positive cells in the granule cell layer and hilus of brains, an average of five sections per mouse were used, and each experimental group consisted of three to five mice. Adjacent sections were not used for cell counting to avoid double counting. All of the counting was performed under a confocal laser-scanning microscope (Zeiss) using a 40× objective in stacks of five optical sections.
Statistical Analysis
Data are expressed as mean ± SEM. Statistical analyses were performed with Prism 5.0 software (GraphPad Software, La Jolla, CA, USA). Comparisons between groups were made using the one-way ANOVA followed by a Dunnet post hoc test. Values of p < 0.05 were considered statistically significant.
Results
Characterization of NPCs and MSCs
To identify the immunocytochemical characteristics of hippocampal NPCs, radial type 1 cell markers (GFAP), neuronal progenitor markers (nestin), proliferation markers (Ki-67), and neuronal markers (NeuN) were utilized. Immunocytochemical labeling showed that cultured NPCs were positive for GFAP, nestin, and Ki-67, but negative for NeuN (Fig. 1A). In addition, immunofluorescent labeling demonstrated that MSCs expressed CD44 and CD90, which are positive markers for MSCs, but did not express CD34 and CD45, which are negative markers for MSCs (Fig. 1B).
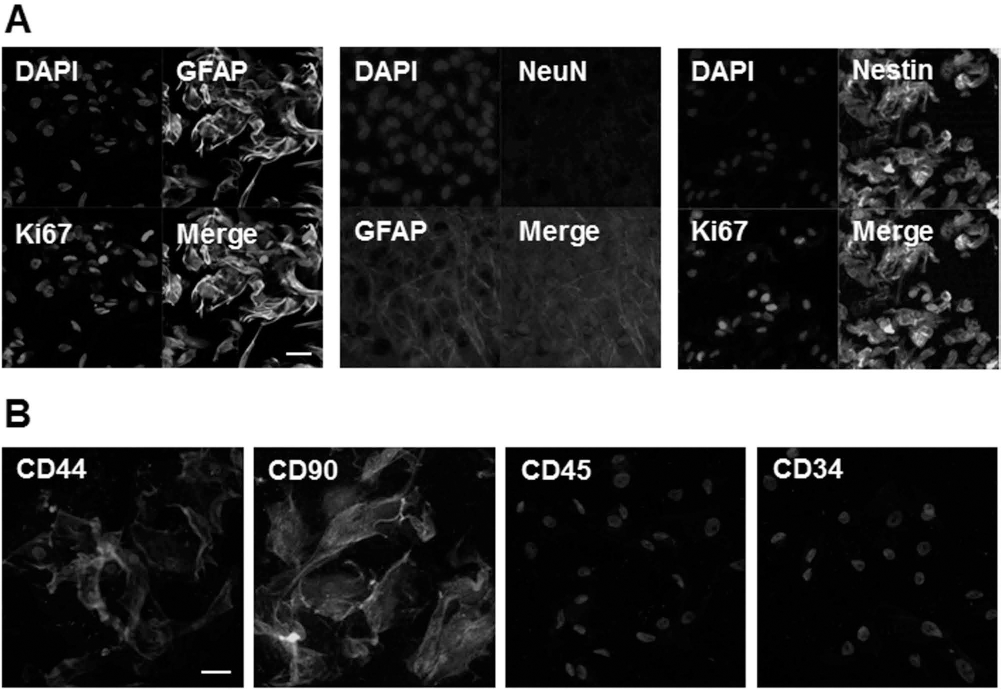
Phenotypic characterization of NPCs and MSCs. NPCs isolated from the SGZ of the hippocampus were positive for GFAP, nestin, and Ki-67, but negative for NeuN (A). Immunofluorescent labeling showed that MSCs were positive for CD44 and CD90, but negative for CD34 and CD45 (B). Scale bar: 50 μm.
MSCs Enhance NPC Proliferation and Neuronal Differentiation in Aβ-Treated NPCs
Short-term exposure to Aβ did not lead to changes in the characteristics of NPCs, while long-term Aβ exposure resulted in altered patterns of RNA expression. Compared to control NPCs, the expressions of a proliferation marker (Ki-67), neuronal progenitor markers (GFAP, SOX2, and nestin), and a neuronal marker (HuD) were significantly decreased in Aβ-treated NPCs (Fig. 2A, B). To evaluate the modulatory effect of MSCs on NPC proliferation and neuronal differentiation in Aβ-treated NPCs, Aβ-treated NPCs were cocultured with MSCs for either 2 days or 2 weeks. Short-term treatment with MSCs in Aβ-treated NPCs slightly increased the expression of the proliferation marker and neuronal progenitor markers without changes in the expression of HuD, whereas long-term treatment significantly increased the expression of the proliferation marker, neuronal progenitor markers, and the neuronal marker compared to Aβ treatment alone (Fig. 2A, B). To evaluate whether a Wnt inhibitor modulates neurogenic activity of MSCs, Dkk1 was added in Aβ-treated NPCs that were cocultured with MSCs. Dkk1 treatment decreased significantly the expression of neuronal progenitor markers and the neuronal marker that was enhanced in MSC treatment (Fig. 2A, B). This indicates that MSCs may not only increase the survival of NPCs against Aβ toxicity but may also induce neuronal differentiation of the NPCs to commit to a neuronal fate. The modulatory effect of MSCs on neuronal differentiation was further supported by morphological analysis. An immunocytochemical analysis showed that, compared to control NPCs or Aβ-only treated NPCs, NPCs cocultured with MSCs for 2 weeks exhibited multiple outgrowing neurites as well as stronger HuD expression (Fig. 2C). In addition, we evaluated whether MSCs modulate neurogenic activity against a different form of Aβ. Long-term MSC treatment significantly increased the expressions of both neuronal progenitor markers and the mature neuronal marker compared to either Aβ25—35 or Aβ1—40 treatment alone (Fig. 2D, E).

MSCs enhance NPC proliferation and neuronal differentiation of Aβ-treated NPCs. Long-term exposure of NPCs to Aß led to a significant decrease in the expression of Ki-67, GFAP, SOX2, nestin, and HuD. However, when Aβ-treated NPCs were cocultured with MSCs, long-term treatment of MSCs significantly increased the expression of the proliferation marker, neuronal progenitor markers, and the neuronal marker in Aβ-treated NPCs compared to Aß treatment alone. Dkkl treatment decreased significantly the expressions of both neuronal progenitor markers and the neuronal marker that were enhanced in MSC treatment (A and B, *p< 0.05, **p < 0.01; n = 3, each group). An immunocytochemical analysis showed that compared to control NPCs or Aβ-treated NPCs, NPCs cocultured with MSCs for 2 weeks exhibited multiple outgrowing neurites and stronger HuD expression (C). Likewise Aβ1—42 treatment, long-term MSC treatment significantly increased the expressions of neuronal progenitor markers and neuronal marker compared to either Aβ25—35 or Aβ1—40 treatment alone (D and E, *p < 0.05, **p < 0.01; n = 3, each group). Scale bar: 50 μm.
Wnt/β-Catenin Signaling Is Involved in MSC-Induced Neuronal Differentiation From NPCs
Wnt ligands are known to modulate neurogenesis in the SGZ of the hippocampus, and thus, we evaluated whether MSCs modulate neuronal differentiation via the Wnt/β-catenin signaling pathway. Short-term Aβ treatment did not lead to any changes in RNA expression involving Wnt/β-catenin signaling, but long-term Aβ exposure significantly decreased the expression of β-catenin and neurogenin-1 (Ngn1) compared to control NPCs. When Aβ-treated NPCs were cocultured with MSCs, long-term MSC treatment significantly increased the expression of β-catenin and Ngn1 compared to Aβ treatment alone. In addition, when Dkk1 was added in Aβ-treated NPCs that were cocultured with MSCs, expressions of β-catenin and Ngn1 were significantly decreased compared to cocultured MSCs in the Aβ-treated NPCs (Fig. 3A, B). This indicates that Wnt/β-catenin signaling is involved in the MSC-induced increase in survival and neuronal differentiation in NPCs. An immunocytochemical analysis showed that NPCs cocultured with MSCs for 2 weeks exhibit a marked increase in immunoreactivity of Ngn1 compared to control NPCs or Aβ-only treated NPCs (Fig. 3C).

Wnt signaling is involved in MSC-induced stemness and the neuronal differentiation of NPCs following Aβ toxicity. Long-term treatment of NPCs with Aβ significantly decreased the expression of β-catenin and Ngn1. However, when Aβ-treated NPCs were cocultured with MSCs, the expression of these compounds was significantly increased following long-term MSC treatment compared to Aβ treatment alone. When Dkk1 was added in Aβ-treated NPCs that were cocultured with MSCs, expressions of β-catenin and Ngn1 were significantly decreased compared to cocultured MSCs in the Aβ-treated NPCs (A and B, *p < 0.05, **p < 0.01; n = 3, each group). NPCs cocultured with MSCs for 2 weeks exhibited a marked increase in immunoreactivity of Ngn1 compared to control NPCs or Aβ-treated NPCs (C). Scale bar: 20 μm.
MSCs Increase Hippocampal Neurogenesis in Aβ-Treated Animals
To evaluate whether MSC treatment modulates hippocampal neurogenesis, brain tissue in the dentate gyrus of the hippocampus was immunostained with BrdU, a marker of cell proliferation. At 1, 2, and 4 weeks following Aβ treatment, the number of BrdU-ir cells in the hippocampus was profoundly decreased in Aβ-only treated animals; however, MSC treatment in Aβ-treated animals significantly attenuated the neuronal loss of BrdU-ir cells in the hippocampus at 1, 2, and 4 weeks (Fig. 4A). Stereological analysis revealed that MSC treatment in Aβ-treated animals significantly increased the number of BrdU-ir cells in the hippocampus at 2 and 4 weeks compared to Aβ treatment alone (35 ± 5 vs. 17 ± 0.8, 26 ± 7 vs. 8.5 ± 0.5, p < 0.05), whereas the difference in BrdU-ir cells between these groups was not significant at 1 week following Aβ treatment (Fig. 4B).

MSCs increase hippocampal neurogenesis in Aβ-treated animals. The number of BrdU-positive cells in the hippocampus significantly decreased in Aβ-only treated animals, and MSC treatment in Aβ-treated animals significantly attenuated the neuronal loss of BrdU-ir cells in the hippocampus at 1, 2, and 4 weeks (A). Stereological analysis revealed that MSC treatment in Aβ-treated animals significantly increased the number of BrdU-ir cells in the hippocampus at 2 and 4 weeks compared to Aβ treatment alone (B). *p < 0.05; n = 3, each group. Scale bar: 20 μm.
MSCs Increase Neuronal Differentiation From BrdU-Positive Cells in the Hippocampus of Aβ-Treated Animals
Finally, to examine whether MSCs would enhance neuronal differentiation in NPCs, the ratio of HuD-ir cells costained with BrdU to total BrdU-ir cells was determined in AD animal models. The degree of neuronal differentiation from BrdU-ir cells was similar between Aβ-only treated animals and MSC-treated animals 1 week after Aβ treatment. However, about 73% of BrdU-ir cells were coimmunostained with HuD antibody in MSC-treated animals 4 weeks after Aβ treatment, which indicates a significant difference in the ratio of neuronal differentiation compared to Aβ-treated animals (Fig. 5A, B). Furthermore, we evaluated whether MSCs may enhance the expression of β-catenin and Ngn1 in AD animal models. Western blot analysis showed that expression of β-catenin was significantly increased in MSC-administrated AD mice compared with that in Aβ-only treated mice (Fig. 6A). Double-stained cells with BrdU and Ngn1 in the hippocampus was frequently observed in MSC-treated AD animals; however, these double-merged cells were not detected in Aβ-only treated animals (Fig. 6B). In addition, we attempted to identify MSCs in the temporal region using human-specific NuMA immunostaining. The immunohistochemical analysis showed that NuMA-positive cells were observed in the entorhinal cortex and hippocampal areas, whereas these cells were not detected in control or Aβ-only treated animals (Fig. 6C). Finally, behavioral analysis showed that MSC treatment significantly decreased the number of reference memory errors in Aβ-treated animals (Fig. 6D).

MSCs increase neuronal differentiation of BrdU-positive cells in the hippocampus of Aβ-treated animals. Brain tissue in the dentate gyrus double immunostained with BrdU and HuD. BrdU-positive cells expressing HuD are infrequently observed in the dentate gyrus of control (Aa—c), Aβ- (Ad—f), and MSC-treated (Ag—i) mice, and the ratio of neuronal differentiation in BrdU-ir cells was similar between Aβ-treated animals and MSC-treated animals 1 week after Aβ treatment. However, BrdU-ir neurons that were coimmunostained with HuD antibody 4 weeks after Aβ treatment were frequently identified in MSC-treated AD mice (Ap—r, white arrows) compared to controls (Aj—l) and Aβ-treated mice (Am—o). Quantitative analysis showed that the number of BrdU and HuD double-positive cells in the dentate gyrus 4 weeks after Aβ treatment was significantly higher in the MSC-treated group (73.3%) than in controls (45%) and the group that received Aβ treatment alone (27.2%) (B). *p < 0.05; n = 5, each group. Scale bar: 40 μm.
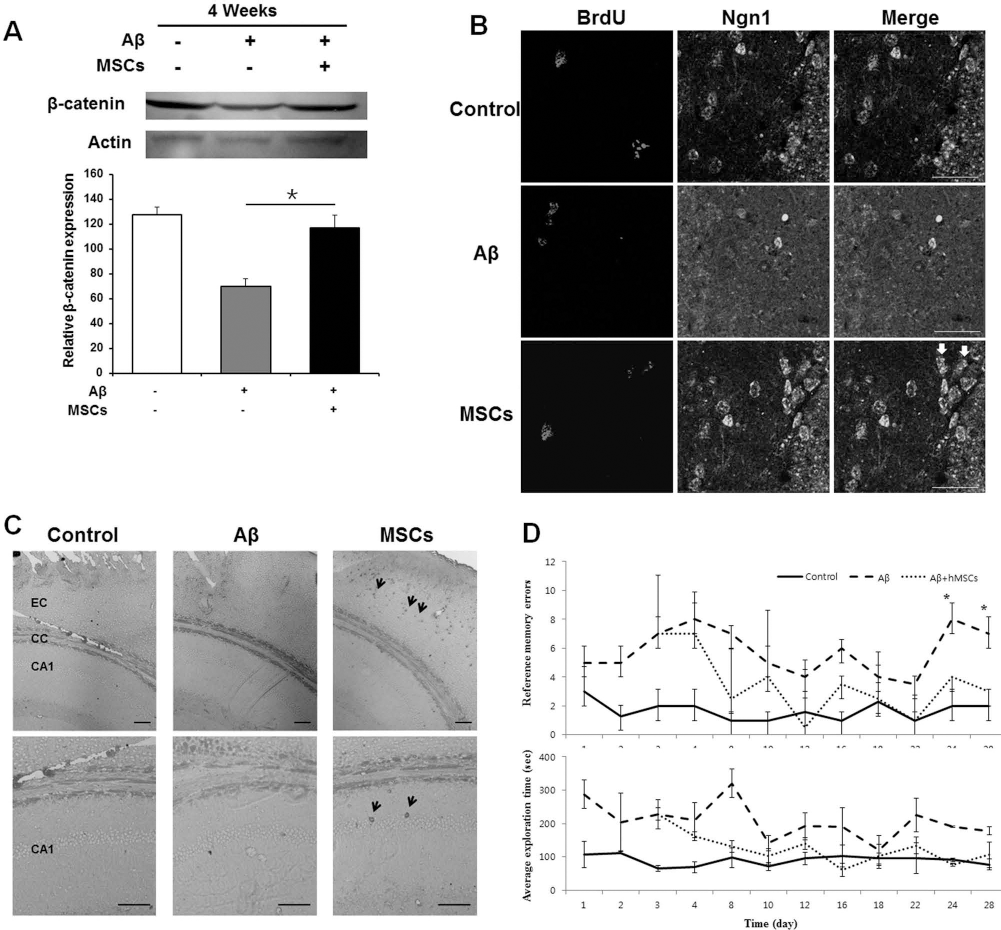
The consequence of MSC treatment in Aβ-treated animals. Western blot analysis showed that expression of β-catenin was significantly increased in MSC-administrated AD mice compared with that in Aβ-only treated mice (A). Double-stained cells with BrdU and Ngn1 were frequently observed in MSC-treated animals (white arrows); however, these double-merged cells were not noted in Aβ-only treated animals (B). The immunohistochemical analysis to identify recruited MSCs showed that NuMA-positive cells were observed in the entorhinal cortex (EC) and CA1 area of the hippocampus (black arrows), whereas these cells were not detected in control or Aβ-only treated animals (C). In addition, behavioral analysis showed that MSC treatment significantly improved cognitive performance of memory in Aβ-treated animals (D). CC, corpus callosum; CA1, cornu ammonis area 1 of the hippocampus. *p < 0.05 (n = 5, each group). Scale bar: 40 μm (A), 10 μm (upper lane in B), and 20 μm (lower lane in B).
Discussion
The present study demonstrated that MSC treatment significantly increased hippocampal neurogenesis and enhanced neuronal differentiation from NPCs in Aβ-treated cellular and animal models of AD. Furthermore, we found that the neurogenic modulation of MSCs was mediated by an augmentation of the Wnt signaling pathway against Aβ-toxicity in AD models. Hence, our data suggest that the modulation of endogenous adult neurogenesis and neuronal differentiation to repair the damaged AD brain using MSCs would have a significant impact on future strategies for AD treatment.
Although the underlying molecular mechanisms of the altered neurogenesis in AD remain unknown, the neuroinflammatory pathway and the Wnt signaling pathway have been implicated Aβ-related AD models. An in vivo study by Zheng et al. (50) found that Aβ inhibits hippocampal neurogenesis in the adult brain through downregulation of interferon-γ and transcription factor nuclear factor-κB. Other animal studies have supported the role of neuroinflammation in hippocampal neurogenesis by showing that the modulation of microglia activation or neuroinflammation is closely associated with a modulation of neurogenic activity in the hippocampus of AD (28,42). In addition, He and Shen (11) demonstrated that Aβ can modulate hippocampal neurogenesis by interrupting β-catenin signaling, whereby increased Aβ leads to an induction of GSK-3b, which, in turn, promotes the phosphorylation and degradation of β-catenin. Since the Wnt/β-catenin signaling pathway is a key player in the regulation of neural stem cell proliferation and differentiation (26,27,32,41), elevated levels of Aβ in Aβ-treated NPCs inhibit neuronal induction by interfering with the β-catenin signaling pathway, which leads to a decrease in the expression of proneural genes (46). In accordance with previous results, the present study also showed that Aβ treatment significantly decreased the self-renewal of NPCs as well as neuronal differentiation from NPCs in cellular and animal models, and decreased proliferation and differentiation of NPCs were related to the downregulation of Wnt/β-catenin signaling pathways.
Interestingly, the present study demonstrated that MSCs augmented neurogenesis and neuronal differentiation against Aβ toxicity in AD models by modulating Wnt signaling pathways. MSC treatment in Aβ-treated NPCs significantly increased the expression of β-catenin and Ngn1, which had been attenuated by Aβ treatment, and led to increased numbers of NPCs and BrdU-positive cells in cellular and animal models of AD. Moreover, MSC treatment in the present study increased significantly the proportion of mature neurons stemming from NPCs or BrdU-positive neurons in AD models and paralleled the increased expression of Ngn1, as if the Wnt/b-catenin signaling pathway regulates the endogenous expression of Ngn1, a proneural gene expressed in newly committed neuronal progenitors and immature neurons (13). Regarding the detailed molecular mechanisms of MSCs that underlie modulation of Wnt signaling pathways, recent expression profiling studies have demonstrated that MSCs express various proteins related to the Wnt signaling pathway (25,39). In addition, Salazar et al. (35) found that MSCs derived from bone marrow and cord blood produce biologically active Wnt proteins, and when inhibited by a Wnt signaling antagonist, the proliferative effects of MSCs are diminished. Accordingly, the increased Wnt proteins produced by MSC treatment may counteract the negative influence of Aβ on neurogenic activity in that Aβ binds to the cysteine-rich frizzled domain of the Wnt receptor and inhibits the Wnt/b-catenin signaling pathway (24,33).
In addition to stem cell proliferation and differentiation, ample evidence has indicated that the Wnt signaling pathway has an important role in the pathogenesis of AD (4,6). β-catenin levels are markedly reduced in AD patients with presenilin-1 mutations (15), and this mutant protein may disturb β-catenin translocation to the nuclei (18,31), which acts as a negative regulator of Wnt signaling. Activation of GSK-3β, a Wnt/β-catenin antagonist, decreases nuclear β-catenin levels (40) and leads to increase in tau hyperphosphorylation and formation of neurofibrillary tangles (2,22). Moreover, the addition of Wnt to hippocampal neurons in culture prevents Aβ neurotoxicity (1), and activation of Wnt signaling rescues neurodegeneration and cognitive impairment induced by Aβ fibrils (5,6,14,44).
Together, these studies suggest that dysregulation of the Wnt signaling pathway is one of several purported pathogenic factors in the development of AD. Therefore, in conjunction with improved cognitive performance in learning and memory tasks following enhanced hippocampal neurogenesis and neuronal differentiation, the modulatory effects of MSCs on the Wnt signaling pathway might possess neuroprotective properties against Aβ-related neurotoxicity in an AD microenvironment. From a therapeutic perspective, these modulatory effects of MSCs could be clinically applicable in future strategies for AD treatment.
In summary, MSC administration significantly augments hippocampal neurogenesis and increases differentiation of NPCs into mature neurons in AD models by augmenting the Wnt signaling pathway. Our results suggest that the modulation of endogenous adult neurogenesis to repair the damaged AD brain using MSCs would have a significant impact on future strategies for AD treatment.
Footnotes
Acknowledgments
This study was supported by a grant of the Korea Healthcare Technology R&D Project, Ministry for Health, Welfare and Family Affairs, Republic of Korea (A091159) and the Brain Korea 21 PLUS Project for Medical Science, Yonsei University. The authors declare no conflicts of interest.
