Abstract
Motoneurons (MNs) derived from mouse embryonic stem cells (ESCs) begin to express low levels of polysialic acid (PSA) at the time when they acquire an ability to migrate and extend neurites. PSA is known to promote cell migration and process outgrowth/guidance in the developing nervous system. To test if experimentally enhanced expression of PSA would augment these cellular events, the PSA-synthesizing polysialyltransferase was introduced into ESCs. In culture, the resulting higher PSA expression specifically increased neurite outgrowth and cell migration from differentiated embryoid bodies. In addition, the MN population obtained after sorting for HB9::GFP expression showed enhanced survival as well as extensive neurite outgrowth. Following transplantation of ESC-derived MNs into an adult sciatic nerve devoid of endogenous axons, the PSA augmentation increased the numbers of axons growing toward the denervated muscles. Migration of some transplanted cells inside the nerve toward muscle was also enhanced. Moreover, higher PSA expression selectively affected target innervation. It produced greater numbers of neuromuscular junctions in a predominantly fast twitch muscle and had no effect in a slow twitch muscle. These findings suggest that engineering of PSA expression in ESC could serve as an enhancement for MN cell therapy.
Introduction
Polysialic acid (PSA) is a long homopolymer of α2,8-linked sialic acid primarily attached to the neural cell adhesion molecule (NCAM). Its abundance and large hydrated volume serve to hinder membrane apposition and thus can weaken interactions among cells. This reduction in cell interactions creates an environment that is conducive to formation, remodeling, and repair of tissue (10,22). For example, in the developing nervous system, PSA facilitates cell migration, as well as growth and path finding of long axonal tracts (10,22). PSA is essential for appropriate fasciculation of developing motor axons, their correct nerve and target selection, and motoneuron (MN) survival (23,25–27). In the adult, PSA was again found to be valuable in target selection by regenerating motor axons (11,12).
MNs can be derived from mouse embryonic stem cells (ESCs) using differentiation factors that operate during development (28). This offers the potential for replacement cell therapy in MN diseases, such as amyotrophic lateral sclerosis (ALS), which is characterized by a debilitating loss of MNs and atrophy of their denervated muscle targets. However, success with transplantation of ESC-derived MNs into the adult mammalian spinal cord has been very limited (6). Even when a useful source of MNs has been achieved, there remain two main challenges to this approach: i) positioning and survival of cells transplanted into adult CNS tissues and ii) adequate integration of the cells into the spinal cord and growth of their axons to distant extremities to innervate muscles. The first may be addressed in part by refinements in differentiation protocols and surgical methods to achieve successful delivery and survival in the local spinal environment. However, the actual integration of the cells and their processes into the local neural circuitry, as well as acquisition of appropriate muscle targets, is likely to require maximization of the intrinsic cellular capabilities of the grafted cells.
This study is primarily directed at the target acquisition challenge. To circumvent the primary obstacles facing injection of cells into the spinal cord, it adopts a strategy of grafting into the sciatic nerve, using a proximal ligature and transection to eliminate endogenous axons. The ability of ESC-derived MNs to survive after transplantation into a peripheral nerve and to reinnervate muscles has already been well demonstrated (18,30). The main goal here is to demonstrate that the introduction of the PST gene into ESCs can have a potentially beneficial effect on survival of ESC-derived MN cells and on the ability of the expressed PSA to improve the innervation process observed previously by others. The results suggest that engineered PSA expression may be useful for cell-based therapies for MN diseases.
Materials and Methods
The HB9::GFP mouse ESC line (HBG3 ESC) was isolated from a transgenic C57BL/6J mouse strain (mHB9:: GFP1b) that expresses the enhanced green fluorescent protein (GFP) under control of the HB9 homeodomain gene promoter (gift from Dr. Wichterle, Columbia University, New York, NY, USA) (28). Expression of HB9 is typical of differentiated somatic MNs; therefore, GFP upregulation is used to indicate MN differentiation. The undifferentiated HB9::GFP mouse ESCs have a very faint GFP background, but its expression becomes much stronger in differentiated MNs. There is complete absence of GFP in cells of other fates (Fig. 1A, B).
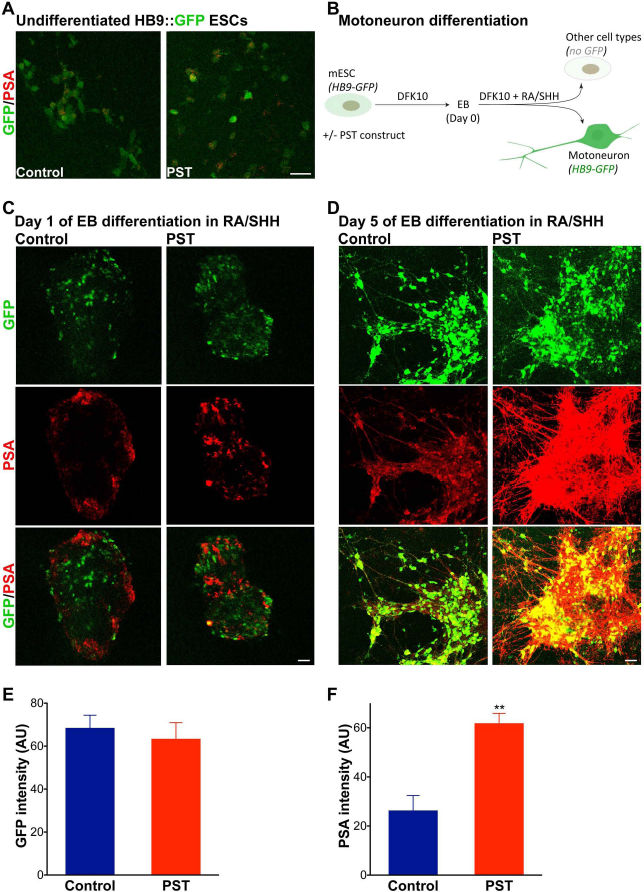
PSA expression in ESCs emerges during motoneuronal differentiation. (A) Immunostaining of cultures of undifferentiated HB9::GFP ESCs shows that these cells have undetectable levels of PSA. Only a few cells have a weak and atypical staining. Transduction with a PST construct did not induce PSA expression in the undifferentiated cells (scale bar: 20 μm). Note that the undifferentiated HB9::GFP ESCs have a very faint GFP expression. (B) Their differentiation into MNs includes two major steps. A first culture in DFK10 medium to form EBs; then the EBs are incubated with RA and SHH analog (on day 0) to induce differentiation into MNs, which is associated with an increased activity of the HB9 promoter and therefore upregulation of GFP. Expression of GFP is turned off in cells other than MNs. Immunostaining of control EBs shows that PSA appears on some cells after 1 day of differentiation (C) and becomes more abundant later (D). PSA occurs on cell bodies as well as the emerging neurites. PSA is clearly present on GFP-positive MNs and their axons. With PST cells, while the pattern is similar to controls after 1 day (C), PSA expression becomes much more widespread in later stages of differentiation compared to controls (D). Almost the entire surface of PST EBs is PSA positive by day 5 (scale bars: 50 μm), and the staining intensity is much higher than in controls (N = 2× triplicates each time point). Quantification of the intensity of GFP (E) and PSA (F) staining in day 5 cultures shows a significant increase in PSA levels with PST modification (p < 0.01, two-tailed t-test, mean ± SEM).
The cells were cultured on gelatin-coated dishes in ES medium [Knockout Dulbecco's modified Eagle medium (Knockout DMEM), 1% minimum essential medium, nonessential amino acids, 1% L-glutamine, 1% penicillin– streptomycin (all from Invitrogen, Carlsbad, CA, USA), 2000 U/μl leukemia inhibitory factor, 1% nucleoside (both from Millipore, Billerica, MA), 15% fetal bovine serum (FBS) (Hyclone, Logan, UT, USA), and 55 μM β-mercarptoethanol (Gibco, Grand Island, NY, USA)].
PST Modification
The HB9::GFP ESC line was virally transduced using a lentiviral vector (pLenti, 6.5 × 105 IU/ml at 1:250; Invitrogen) carrying the mouse PST gene under control of the ubiquitous cytomegalovirus promoter (7,8,15). Blasticidin-resistant cells were selected, propagated, and stocks stored in liquid nitrogen.
Differentiation Into MNs
Differentiation of HB9::GFP ESC into MNs consists of two major steps and was carried out as detailed by Wichterle and colleagues (28,29). Briefly, there is a first incubation of cells in DFK10 medium 50:50 of DMEM HG/ Ham-F12, 10% knockout serum replacement (Invitrogen), 1% penicillin–streptomycin, 1% L-glutamine, 1 U/μl heparin, β-mercaptoethanol, and 2.4% N2-supplement (Invitrogen) to initiate formation of embryoid bodies (EBs), which occurs after 1 day in culture. The second step starts after 2 days, when the EBs are treated with retinoic acid (RA: 1 μM; Sigma-Aldrich, St. Louis, MO, USA) and sonic hedgehog (SHH) analog 1.5 μM purmorphamine (Santa Cruz Biotech, Dallas, TX, USA) (16) to induce differentiation into MNs. The day of addition of SHH analog and RA is considered day 0 of differentiation. Cells differentiating into MNs upregulate GFP, start to grow neurites, and emigrate from the EBs.
Immunostaining
Cultures were fixed in 4% paraformaldehyde (Sigma-Aldrich) in phosphate buffer 0.1 M, 7.2 pH at indicated days and immunostained for choline acetyl-transferase (ChAT, goat IgG, 1:200), neurofilament-M (NF-M, mouse IgG, 1:200), GFP (rabbit IgG, 1:500) (all from Millipore), or PSA (5A5, mouse IgM, 1:2,000 (our own product). Appropriate Cy3, Cy5, or Alexa 488-conjugated secondary antibodies (1:800; Jackson, West Grove, PA, USA) were used to visualize staining (all buffer reagents are from Sigma). Cultures were examined and imaged using a Zeiss 510 LSM confocal microscope (Carl Zeiss, Jena, Germany).
Morphometry
To quantify PSA levels at day 5 of differentiation, the stained areas were selected, and the mean intensity of staining was measured using NIH ImageJ 1.45 (NIH, Bethesda, MD, USA). For comparison, GFP intensity was also measured. Values are presented as mean ± SEM in an arbitrary unit, and a two-tailed t-test was used for statistical comparison (Prism6, GraphPad, La Jolla, CA, USA).
The PST modification dramatically increased neurite outgrowth and cell dispersion, which by day 6 produced a widespread green mat-like structure so dense that it became difficult to trace axons and identify cell bodies. Therefore, to quantify effects, the induced spreading on the dish floor was estimated by measuring the density of GFP fluorescence per surface area (NIH ImageJ 1.45). A two-tailed t-test was used for comparison, and values are presented as mean ± SEM.
Sorting for GFP Enrichment
On day 6 of differentiation, the cells were dissociated using a papain kit (Worthington, Lakewood, NJ, USA) (28,29), resuspended in DMEM HG without serum, filtered through a 5-ml polystyrene round-bottom tube with cell strainer cap (BD Falcon, Franklin Lakes, NJ, USA), then sorted by fluorescence-activated cell sorting (FACS) for the HB9-driven GFP enrichment at the Institution Flow Core Facility using a MoFlo2 cell sorter (Beckman-Coulter, Lakeview, IN, USA). Naive HB9::GFP ESCs were used as a baseline reference. The ratio of cells that differentiated into MNs was determined and presented as mean ± SEM. A two-tailed paired t-test was used for data comparison. The cells were then allowed to settle in DMEM without serum, supplemented with 20 ng/ml of glial-derived neurotrophic factor (GDNF) and 10 ng/ml of ciliary neurotrophic factor (CNTF) (both from Millipore). Small samples were used to check postsort viability and cell density.
Postsort Culture
Monoculture
The isolated MNs with or without PST were plated at 30,000 cells per 35-mm Matrigel (BD Biosciences, Franklin Lakes, NJ, USA)-coated dish (2× triplicates) in DFK10 medium supplemented with CNTF (10 ng/ml) and GDNF (20 ng/ml). The cultures were fixed and examined for cell survival after 1, 5, 7, 11, and 15 days. A two-way ANOVA with Bonferroni posttest (Prism6, GraphPad) was used for comparison, and values are presented as mean ± SEM. Additional cultures of PST MNs (30,000 cells per 35 mm) were incubated in the presence or absence of the endoneuraminidase-N [endo-N, our own product (9), 30 U/ml in culture medium, triplicates] for 5 days, then fixed and immunostained for PSA. Untreated control HB9-MN cultures were included for comparison.
Coculture
The MNs ± PST were seeded (~50,000 cells/ 35 mm dish) on top of a monolayer of primary Schwann cells (SCs) ± PST (2× triplicates). The SCs were isolated from adult female Fischer rat sciatic nerves (15) and cultured in DMEM with 10% inactivated FBS, bovine pituitary extract (20 μg/ml; Invitrogen), forskolin (2.5 μM), and heregulin (2.5 nM; Genentech, San Francisco, CA, USA) (culture purity ~95-98%). As the unmodified SCs had an insignificant amount of PSA (7,15), they were then infected with lentiviral vectors (Invitrogen) carrying GFP alone or GFP and PST (15). The cocultures were fixed on day 5 and immunostained for NF-M to visualize MNs and their neurites. For each culture, MNs were counted in three equal fields to assess survival. Fluorescence of NF-M staining was imaged with a confocal microscope (Carl Zeiss), and its density per surface area was measured (NIH ImageJ 1.45). The analyzed areas were not overlapping and not located in specific positions in the dish; that is, each time, the stage was moved to a different random position. Individual cells and neurites were readily visualized under a 40× objective. To account for the difference in survival, densities were normalized to survival data. All values were compared using a two-way ANOVA with Tukey's posttest, and results are presented as means ± SEM.
In Vivo Transplantation
The sorted MNs were diluted to the desired density (3,000 or 15,000 cells/μl) in DMEM without serum, supplemented with 20 ng/ml of GDNF and 10 ng/ml of CNTF (Millipore). Adult male C57BL/6J mice (Jackson Laboratory, Bar Harbor, ME, USA) were anesthetized (pentobarbital 50 mg/kg, IP; Sigma), and their left sciatic nerve was exposed and ligated (7-0 Ethicon; Bridgewater, NJ, USA) at the level of the upper thigh (point of emergence from under the gluteus superficialis muscle). The MNs were injected (1 μl) in the nerve distal and in close proximity to the ligation using a Hamilton syringe (RN, 3PK fine needle; Hamilton, Reno, NV, USA) as described previously (6,30). Each transplant contained 3,000 or 15,000 control or PST-modified cells. The nerve was then transected ~3 mm proximal to ligation to completely eliminate regrowth of endogenous axons within the stump. The proximal stump of the nerve was also ligated. The skin was closed using 4-0 Nylon sutures (Ethicon) and tissue glue (Vector Laboratories, Burlingame, CA, USA), and the animals were observed for recovery from anesthesia. Each group/time point included 8 to 10 animals. Animal care and experimentation were carried out according to institutional and NIH guidelines and were approved by Memorial Sloan-Kettering Cancer Center and Cornell University Animal Care and Use Committee.
At endpoint, the animals were transcardially perfused under deep anesthesia (pentobarbital 250 mg/kg, IP; Sigma), with 25 ml of a 7.4 pH phosphate-buffered saline solution followed by 25 ml of a 4% paraformaldehyde, 0.1 M, 7.4 pH phosphate-buffered solution (all perfusion reagents were from Sigma). The distal sciatic stump was dissected, postfixed in the same fixative overnight, and processed for longitudinal cryostat (Leica CM1850; Leica, Buffalo Grove, IL, USA) sections (20 μm) that were immunostained for ChAT (goat IgG, 1:200), NF-M (mouse IgG, 1:200) and GFP (rabbit IgG, 1:500) (all from Millipore). Sections were examined on a confocal microscope. High-resolution images were enlarged (to full screen) to facilitate fiber identification. The number of axon profiles that crossed a 100-μm line perpendicular to the nerve axis, located 1 cm from the graft, was counted. Values were compared using a two-tailed unpaired t-test and presented as mean ± SEM.
Two muscle targets [the soleus and extensor digitorum longus (EDL)] were also dissected, longitudinally sectioned (cryostat: 40 μm), and processed for immunohistochemistry or labeling with red fluorescent α-bungarotoxin (1:200; Invitrogen). Faint and intensely red-labeled neuromuscular junctions (NMJs) were observed. The faint ones were considered noninnervated, and the numbers of intensely labeled ones per sections were counted. A one-way ANOVA with Tukey's posttest was used, and values are presented as mean ± SEM. Muscles from a few additional injected animals were sectioned and stained following the method of Pestronk et al. (2,20) that visualizes nerve terminals as dark fibers inside the blue cholinesterase staining of NMJs. In brief, sections were incubated for 12 min at 37°C in 0.55 mM 5-bromoindoxyl acetate, 5 mM potassium ferricyanide and potassium ferrocyanide, and 7.5 mM calcium chloride in 10 mM, pH: 7.2 tris buffer. They were then treated in 10% chloral hydrate and 1% pyridine for 30 min at 37°C, then incubated in 20% silver nitrate, 0.1% cupric sulfate for 60 min at 37°C (all chemicals were from Sigma). The diameter of the muscle was also measured at the level of the muscle belly. Numbers of ChAT and GFP-positive cells that reached muscles were counted in a 100 μm × 100 μm square at the level of the nerve/muscle entry zone and compared using a two-tailed unpaired t-test. Results are presented as mean ± SEM.
Results
PSA Expression on HB9 ESCs During Differentiation Into MNs
The undifferentiated HB9::GFP ESC line has a very weak GFP background, and immunostaining indicated that it did not express PSA (Fig. 1A). When these cells were allowed to form EBs and induced to differentiate into MNs with RA/SHH (N = 2× triplicates each time point) (Fig. 1B), some cells in the EBs began to show PSA after 1 day (Fig. 1C, Control). By 5 days of differentiation, a large number of cells were GFP positive (intense) and PSA positive (moderate) (Fig. 1D, Control). When PST was introduced into the HB9::GFP cells (PST-HB9), PSA staining in PST cells was comparable to control levels in undifferentiated cells and to 1 day of differentiation (Fig. 1A, C). However, by day 5, the resulting level of PSA on PST cells was substantially increased (p < 0.01, two-tailed t-test) (Fig. 1D, F), while GFP was unchanged (Fig. 1E).
After Differentiation, PST-HB9 Cultures Display Enhanced Neurite Outgrowth and EB Disintegration
MN differentiation reaches its peak by day 6 (28,29) (Fig. 2), and immunostaining showed that both control and PST-HB9 cells were positive for ChAT, confirming a relatively mature MN phenotype (data not shown). At this stage, a few GFP-positive cell bodies can be seen leaving the HB9 EBs and translocating along axon bundles or between fibers (Fig. 2A, C, E). This continuing process was more evident in PST cultures, resulting in near complete disintegration of EBs (N = 3× triplicates each) (Fig. 2B, D). Coincidental with the cell migration was outgrowth of neurites (Fig. 2), which, with PST enhancement, produced a dense mat of GFP and PSA-positive processes (Fig. 2B, D, F). Quantification of the spread of GFP over the dish surface confirmed the effect of PST on dispersion of cells and extension of neurites (p < 0.001, two-tailed t-test) (Fig. 2G).

Increased PSA expression in differentiating MNs produces more neurite outgrowth and cell migration. (A) Day 6 control cultures had some growing neurites as well as cells emigrating from the EBs (arrows). (B) The PST modification was associated with a dramatic increase in neurite outgrowth, and the cell migration was so important that the EBs became flat and more spread (scale bar: 100 μm; N = 3× triplicates). (C, D) Higher magnification images showing the larger amount of GFP-positive axons (stars) and cells (arrows) leaving the EBs in PST cultures (scale bar: 50 μm). (E, F) Examples from day 6 cultures showing the difference in PSA staining on GFP-positive MNs between control and the axon/cell migration-rich PST cultures (scale bar: 100 μm). In PST cultures, the growing axons and migrating cells formed a much denser mesh that covered the dish floor, and quantification of this GFP spread is shown in the graph (G) (p < 0.001, two-tailed t-test, mean ± SEM).
The Degree of MN Differentiation Is Not Altered by PSA Overexpression
The ratio of cells that differentiated into MNs was determined by FACS sorting for HB9-driven GFP expression (Fig. 3). Typically, control HB9::GFP ESC yielded 2–8% GFP-positive cells. The introduction of PST did not change the yield of MNs (p = 0.8; two-tailed paired t-test; N = 5× triplicate cultures each) (Fig. 3A). Although some PSA-NCAM may have been lost during the enzymatic digestion used to dissociate cells, Western blotting of the sorted cells showed that PST MNs continued to have higher PSA levels than controls (Fig. 3D, E).

PSA overexpression did not change differentiation ratio but improved survival and sprouting of differentiated MNs. (A) Quantification of the yield of differentiation as represented by the percentage of cells that differentiated into MNs determined during sorting for GFP enrichment. The PST modification did not change this ratio (p = 0.8; two-tailed, paired t-test; N = 5× triplicate cultures each; mean ± SEM). (B) Differentiated and sorted MNs were able to survive in culture. Some control cells grew neurites in the first days of culture. Thin protrusions were present on some neurites. PST MNs grew more neurites and protrusions, and their arborization was more extensive after 1 day in culture. Neurites were present on almost all cells (scale bar: 20 μm). (C) Counts of differentiated MNs at different stages of postsort culture (N = 2× triplicates/time point). The PSA overexpression significantly improved MN survival (p < 0.001, two-way ANOVA; Bonferroni posttest p < 0.05), and the effect was maximal after 5–7 days (mean ± SEM). (D) Sorted ESC-derived MNs were lysed and probed for NCAM by immunoblotting. In control cells, both NCAM-140 and NCAM-180 isoforms are polysialylated, and the PSA smear extends as high as in the positive reference (newborn brain). With PST modification, the polysialylation smear was much more extensive. Newborn brain treated with endo-N was used as negative reference, showing NCAM-140 and NCAM-180 bands. (E) Densitometry of the PST and control smears (area indicated as smear in D). (F) Sorted MNs cultured for 5 days with or without endo-N are stained for PSA. There are more cells with extensive neurites in PST cultures than controls. PSA removal with endo-N reverses the enhancing effect, which confirms that the augmentation is PSA specific (scale bar: 100 μm).
Sorted PSA-Enhanced MNs Exhibit Increases in Axon Outgrowth and Cell Survival in Culture
After sorting for GFP, HB9-MNs ± PST were cultured sparsely and observed for process outgrowth on the substrate. After 1 day in culture, control HB9-MNs had extended one or more simple or ramified neurites with thin protrusions on some fibers. Many cells had no or very short neurites (Fig. 3B). In the PST-HB9 MN cultures, almost all MNs grew neurites with elaborate branching patterns and fiber–fiber networks. There were also multiple protrusions from each cell body or fiber (Fig. 3B). This neurite outgrowth effect persisted for several days and was not found for PST-HB9 MNs that were cultured in the presence of the PSA-specific endoneuraminidase-N (endo-N) (Fig. 3F).
In the HB9-MN control cultures, the total number of cells gradually declined to a very low density by day 7. These changes were less pronounced in the PST-HB9-MN cultures, with significantly more cells remaining by days 5, 7, and 11, (p < 0.001, two-way ANOVA; N = 2× triplicates/ time point) (Fig. 3C).
Cocultures of HB9-MNs with Schwann Cells
The in vivo portion of this study involves grafting of the sorted MNs into the SC-rich peripheral nerve. To provide information about the action of PSA in the presence of SCs, the sorted HB9-MNs were cultured (50,000 cells/35-mm dish; N = 2× triplicates) on a monolayer of primary SCs that were themselves modified with a GFP-PST or GFP construct (7,15). NF-M-positive cells (Fig. 4A) were counted and their neurites examined on day 5. As above, the PST-HB9 MNs exhibited a significant improvement in survival. Remarkably, the same increase was observed when only the SCs were modified with PST, and the enhancement of PSA expression on both cell types resulted in an additive effect on MN survival (p < 0.0001) (Fig. 4B).

PST modification improves survival and neurite outgrowth of MNs cultured on a SC monolayer. Sorted MNs were cultured on a monolayer of GFP-expressing SCs (50,000 MNs/35-mm dish; N = 2× triplicates). MNs and their neurites were visualized by NF-M staining (red) after 5 days in culture as shown in this example (A) from a culture where the PST gene was introduced in both MNs and SCs. MNs appear yellow in the overlay as they coexpress GFP and NF-M (scale bar: 50 μm). (B) Quantification of surviving MNs after 5 days showed that the introduction of PST in either cell type (SC or MN) produced a significant increase in survival, and modification of both cells had an additive effect (p < 0.0001, two-way ANOVA, mean ± SEM). (C) Similarly, quantification of the density of NF-M staining and its normalization to survival values to estimate neurite outgrowth showed a significant increase by PSA enhancement (p < 0.001, two-way ANOVA, mean ± SEM).
Similarly, increased PSA expression on either HB9-MNs or on SCs promoted outgrowth of NF-M-positive neurites, and PST modification of both cell types produced a stronger effect. Quantification of the density of NF-M-positive neurites relative to survival values is shown in Figure 4C (p < 0.001).
Grafted MNs Can Migrate Inside the Sciatic Nerve, and PSA Overexpression Enhances This Migration
The sciatic nerve was ligated in the upper thigh and transected ~3 mm proximal to ligation to completely eliminate regrowth of endogenous axons within the nerve. Sorted HB9-MNs (~3,000 cells) were injected distally in close proximity to the ligation (Fig. 5A). Examination of distal parts of the nerve revealed that by 2 months some of the grafted MNs had migrated within the nerve toward muscle (Fig. 5B). When the EDL, a predominantly fast-twitch muscle, and the soleus, a predominantly slow-twitch muscle, were examined, a few of the GFP-positive grafted cells were found to have entered these targets (Fig. 5C). With the PST-HB9 MNs, there were more cells migrating inside the nerve (Fig. 5B), and accordingly, more cells arrived at muscles (Fig. 5D, E). No obvious difference was seen between the EDL and soleus.
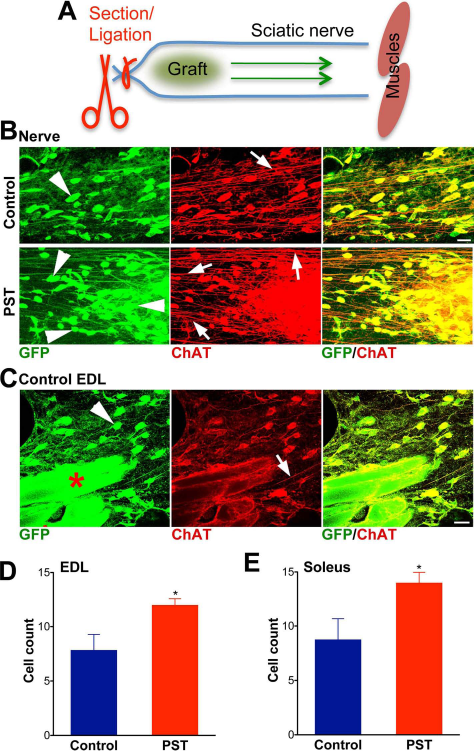
Grafted MNs migrated inside the nerve, which was enhanced by PSA overexpression. (A) Sorted MNs were grafted in the sciatic nerve immediately distal to a ligation and section that eliminate all endogenous axons. The arrows indicate the direction to denervated muscles. (B) Some GFP and ChAT-positive grafted MNs were found migrating inside the nerve (arrowheads), as shown in images ~1 cm away from the graft site. There were more migrating cells (arrowheads) in PST samples (arrows indicate axons) (scale bar: 20 μm). (C) These cells were able to reach muscles as shown in this representative image from the nerve entry zone into a control EDL muscle (arrows: axons). The inherently autofluorescent muscle fibers are indicated by red stars (scale bar: 20 μm). Counts of GFP and ChAT-positive cells per 100 μm × 100 μm square at the nerve/muscle entry zone show that more cells reached the EDL (D) and soleus (E) muscles in the PST samples than in controls (p < 0.05, two-tailed t-test; N = 3).
Grafted PST-HB9 MNs Exhibit More Robust Axonal Growth in the Adult Sciatic Nerve
Examination of control longitudinal nerve sections 2 months after grafting showed GFP and ChAT-positive axons extending from the grafted cells with large numbers of fibers being able to grow distally inside the nerve (Fig. 6A). With the introduction of PST, there was a marked increase in the density of fibers growing from the grafts (Fig. 6A), and fiber counts ~1 cm from the graft were significantly higher for the PST-HB9 MNs (p < 0.001, two-tailed t-test) (Fig. 6B). A similar PST-induced increase was initially observed in animals grafted with 15,000 cells and examined 3, 4, or 8 weeks later; however, the fiber density was too high for adequate quantification (data not shown).
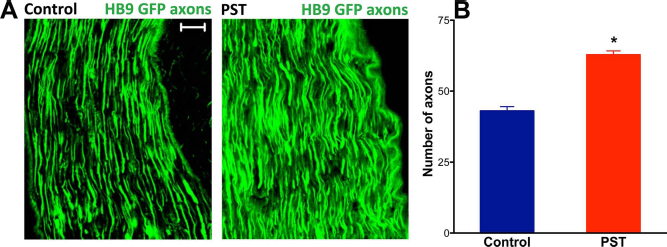
ESC-derived MNs grew more axons in the sciatic nerve following PSA overexpression. The injected cells were able to grow axons toward muscle targets as shown in pictures taken ~1 cm distal to graft limit (A), 2 months after transplantation (3,000 grafted cells). There were more GFP-expressing axons growing from PST grafts than controls (scale bar: 20 μm). (B) Quantification of axons that crossed a 100-μm transverse line located ~1 cm from the graft edge showed that PSA overexpression significantly increased this growth (p < 0.001, two-tailed t-test; mean ± SEM).
PST Modification of Grafted MNs Specifically Increases Neuromuscular Junction (NMJ) Formation in the EDL Muscle
By 3 weeks postgraft, GFP/ChAT-positive axons were found entering both the predominantly fast-twitch EDL muscle and the predominantly slow-twitch soleus muscle. Fluorescent α-bungarotoxin was applied 2 months after grafting, and NMJs were observed in both muscles, with or without PSA overexpression (Fig. 7A). Some NMJs had a faint staining and therefore were not considered reinnervated. Round as well as some elongated NMJs were observed in all samples with no visible difference in shape distribution between control and PST samples.

PSA overexpression increased reinnervation of the EDL muscle by ESC-derived MNs. Motor axons that grew inside the nerve were able to reach muscle targets. (A) Application of red-fluorescent α-bungarotoxin to 40-μm longitudinal sections of the predominantly fast-twitch EDL muscle revealed more intensely labeled NMJs with PST-MN grafts (scale bar: 100 μm). (B) Counts of these NMJs in the EDL showed that the PSA increase was significant with both 3,000 and 15,000 cell grafts (p < 0.01). The PSA augmentation was so important that the graft size could be reduced to 3,000 cells and still achieve an innervation level statistically comparable to that obtained with 15,000 cells (p = 0.12). In the slow-twitch muscle soleus, neither PSA levels nor cell numbers could significantly change NMJ counts (mean ± SEM; 5 each). (C) Section of a PST EDL stained histoenzymatically to visualize NMJs by cholinesterase activity (blue) and the nerve terminals as dark brown processes on the surface of muscle fibers and inside the NMJs. (D) Quantification of innervated NMJs confirmed the PSA effect (mean ± SEM; N = 3 each; p < 0.01) observed with the α-bungarotoxin method.
With grafts of 3,000 cells, counts of the strongly labeled NMJs did not change with PSA enhancement in the soleus (p = 0.38) (Fig. 7B). By contrast, a significantly higher number of NMJs was observed in the EDL muscle when PST MNs were grafted (p < 0.01) (Fig. 7B). When a larger graft (15,000 cells) was used, the results were similar: NMJ numbers were increased by PST modification in the EDL (p < 0.001) (Fig. 7B), but not in the soleus (p = 0.7). Notably, the number of NMJs in the PST EDL was not significantly increased by the fivefold increase in graft size (p = 0.12) (Fig. 7B), suggesting that even relatively small numbers of PST-MNs can give a maximal target innervation result. The PSA effect on EDL innervation was initially observed using a histoenzymological method that visualizes the NMJ area by cholinesterase activity and stains the nerve terminals (p < 0.01) (Fig. 7C, D) (2,20), then confirmed with the α-bungarotoxin technique. The latter method was adopted throughout the study for its consistency and simplicity. Interestingly, the change in innervation did not affect the size of the EDL muscle as shown by diameter measurements (Control: 1,321 ± 11 μm, PST: 1,244 ± 72 μm; p = 0.35).
Discussion
The present study revealed the ability of PST gene addition into ESCs to upregulate PSA expression during their subsequent differentiation into MNs. They then demonstrated that this enhanced PSA is sufficient to augment MN behavior both in culture and following transplantation in vivo.
These findings are in accord with effects of PSA expression during normal development, when its presence on differentiating neural cells coincides with cell translocation and neurite outgrowth (22). PSA appears on developing MNs when they begin projecting toward muscles (5,23,26) and on progenitors in the adult subventricular zone (SVZ) and dentate subgranular layer at the time of migration or neurite outgrowth (14,17). The fact that upregulation of the PST gene did not change PSA expression in undifferentiated HB9::GFP ESCs suggests the existence of a posttranscriptional control of PST activity.
Although PSA has been shown to modulate differentiation of endogenous SVZ progenitors (21), the PSA augmentation in ESCs did not significantly alter the efficiency of differentiation into MNs. It may be that any such effects would be masked in an in vitro differentiation process driven by exogenous SHH and RA or that the fate specification by these factors occurs before PSA upregulation in the cell. Nevertheless, with PST overexpression, there was an increase in the survival of differentiated MNs, either when cultured alone or on a monolayer of SCs. This would be consistent with the fact that PSA loss of function in the embryo reduces survival of spinal MNs (25) as well as forebrain neurons (3).
The studies of MNs grown on SC monolayers revealed that PSA can augment cell survival and neurite outgrowth when present on either cell type and can act additively when expressed on both. In the adult animal, PSA facilitates axonal growth when expressed on the neurons themselves or, as shown previously, on glial cells (8,10,15). This might seem surprising but in fact is consistent with the role of PSA as a regulator of membrane–membrane interaction by filling the space between the cells; that is, it does not matter which cell is providing the space. This phenomenon suggests that direct SC–MN contact is relevant to the effect on survival and neurite outgrowth. It should be noted, however, that these parameters were also improved when the HB9-MNs were grown sparsely on the tissue culture substrate without SCs, and thus this effect could include a role for matrix (1) or growth factors (13) as well.
Previous studies using similar grafts in denervated peripheral nerves have already demonstrated the capacity of MNs to survive without immunosuppression in the presence of growth factors and their ability to reinnervate muscle targets (30). Like in the present study, the grafted MNs were able to project relatively straight axons, alone or in bundles (30), most likely along the bands of Bungner. The fact that PSA augmentation can promote reinnervation of fast-twitch muscle targets by HB9-MNs is consistent with previous observations in normal development. PSA expression is highest on developing motor axons that innervate fast muscles. With removal of PSA by endo-N, the fasciculation of these axons adopts the pattern of a slow muscle (23). Moreover, the differentiation protocol used in this study (28) primarily generates medial motor column-like MNs that normally innervate axial muscles (24). It is important, however, to remember that PSA primarily acts as a permissive factor for selective changes in axon behavior (22) rather than a specifier of fast motor innervation patterns. Thus, although PSA enhancement can stimulate axonal growth in the nerve, in the muscle it would facilitate the axon response to some local cues present in the fast-twitch muscle. The lack of a PSA effect in the soleus may reflect a difference in such cues. However, it is also possible that, in this muscle, a maximum of reinnervation could already be achieved by control grafts. Fewer cells may be used in future experiments, together with electrophysiological assessment, to ascertain these observations. Similarly, the level of reinnervation by control grafts is probably sufficient to improve muscle size, which would explain the absence of a PSA effect. A possible delay in maturation of NMJs by PSA overexpression could also slow down recovery of muscle mass. Earlier studies using MN grafts in peripheral nerves observed fewer surviving cells (30), which is probably due to procedural differences. This discrepancy in survival is also reflected in the amount of axonal growth. With a lower survival, cell migration inside the nerve would have been negligible and went therefore undetected. Such a migration has also been documented following transplantation in the auditory nerve (19).
The ability of PSA to augment path finding and target selection is not unique to MNs; it has been observed in other major axon projection systems, including sensory innervation, the retinotectal projection, and the appearance of mossy fiber terminals in hippocampus (10,22,26,27). Interestingly, we have recently shown that the same PSA enhancement improves the integration and function of ESC-derived dopamine neuron grafts in parkinsonian mice (4). Thus, it will be interesting in future work to see whether PSA enhancement can be used to improve innervation of other neuronal cell types.
Transplantation of ESC-derived MNs directly into the spinal cord of adult ALS animal models has been problematic (6) with very poor survival and integration. The ability of transplanted MNs to translocate within peripheral nerve, as shown in the present study, might make it possible to introduce the graft just outside the spinal cord, if in the absence of a nerve ligation or section they then can migrate centrally. Such a central migration has recently been reported following transplantation of ESCs in the auditory nerve and was facilitated by chondroitin sulfate digestion from the transitional zone (19).
In sum, the present results illustrate that enhanced PSA expression on ESC-derived MNs results in a number of significant changes in their behavior that could be relevant to a more efficient and effective integration into adult host tissue. As such, PSA engineering could prove to be a valuable adjuvant to cell-based therapies in the nervous system.
Footnotes
Acknowledgments
We would like to thank Hynek Wichterle (Columbia University) for kindly providing the HBG3 ESC line, and Damien Pearse and Mousumi Ghosh (University of Miami) for supplying Schwann cells. We also thank the Flow Cytometry Core Facility at MSKCC for their technical support. This project is supported by MSKCC internal funds. The authors declare no conflicts of interest.
