Abstract
Vascular apoptosis plays a pivotal role in the development and progression of a myriad of cardiac dysfunctions, but has yet to be investigated in doxorubicin-induced cardiomyopathy (DIC). Additionally, the neovascularization potential and resulting functional consequences of embryonic stem (ES) cells and factors released from these cells in the chronic DIC myocardium remain largely unknown. To this end, we transplanted conditioned media (CM) and ES cells in the DIC-injured heart and evaluated their potential to inhibit vascular cell death, activate endogenous c-kit+ and FLK-1+ cells, enhance neovascularization, and augment left ventricular dysfunction. Data presented suggest transplanted CM and ES cells significantly blunt vascular cell apoptosis consequent to DIC. Quantitative immunohistochemistry data demonstrate significantly increased c-kit+ and FLK-1+ cells, as well as enhanced differentiated CD31+ cells in the CM and ES cell groups relative to DIC controls. Heart function, including fractional shortening and ejection fraction, assessed by transthoracic echocardiography, was significantly improved following CM and ES cell transplantation. In conclusion, our data suggest that transplantation of CM and ES cells inhibit vascular apoptosis, activate endogenous c-kit+ and FLK-1+ cells and differentiate them into endothelial cells, enhance neovascularization, and improve cardiac function in the DIC-injured myocardium.
Introduction
Doxorubicin (DOX) is an antineoplastic chemotherapeutic drug used to treat a diverse spectrum of malignancies (20,24). However, clinical applications utilizing DOX are restricted due to the time and dose-dependent cardiotoxicity inferred following treatment with doxorubicin [doxorubicin-induced cardiomyopathy (DIC)] and heart failure potential end points. Evidence provided suggests that pathophysiological mechanisms of DIC are complex and multifactorial. Although the precise mechanisms of DIC remain obscure, the following anomalies in the injured myocardium have been noted: 1) cardiac cell-type death, 2) myofibril loss and vacuolar degeneration, 3) fibrosis, 4) cardiac myocyte hypertrophy, 5) contractile dysfunction, and 6) rhythm disturbances (13,20,24,26,34). As the incidence of cancer continues to rise, causing significant morbidity and mortality worldwide, the need to identify novel therapeutic approaches is imperative.
Regenerative medicine, using stem cell transplantation as well as the use of factors released from such cells to promote advantageous paracrine mechanisms, continues to be at the forefront of translational research for the treatment of an array of pathological disorders, including DIC. Some of these factors are antiapoptotic, such as osteopontin, clusterin, cystatin-c, tissue inhibitor of metalloproteinase 1, and insulin-like growth factor-1 (IGF-1), and proangiogenic, such as IGF-1, hepatocyte growth factor, and vascular endothelial growth factor (1,9,26,29,35). Current data available suggest that these therapeutic options are capable of improving cardiac function consequent to myocardial repair and regeneration (7,10,25,27,31). Specifically, transplanted conditioned media (CM) and embryonic stem (ES) cells inhibited a host of acute DIC-induced cardiac anomalies, including cardiac myocyte apoptosis, fibrosis, cytoplasmic vacuolization, and myofibril loss, as well as improved cardiac function (26). However, the effects of transplanted CM and ES cells have never been challenged with regard to vascular cell death and neovascularization in DIC.
Accordingly, we hypothesized that transplanted ES cells and their released paracrine factors would inhibit vascular apoptosis and enhance neovascularization, leading to subsequent cardiac function augmentation. To test this hypothesis, cultured ES cells and CM generated from these ES cells were transplanted into a DIC mouse model. Four weeks post-DIC induction, data presented demonstrate that transplanted CM and ES cells inhibit vascular cell death, activate v-kit Hardy—Zuckerman 4 feline sarcoma viral oncogene homolog positive (c-kit+) and fetal liver kinase 1 positive (FLK-1+) progenitor cells, enhance neovascularization, and improve cardiac function in chronic DIC.
Materials and Methods
ES Cells and Preparation of CM
ES cells (CGR8, a mouse embryonic stem cell line provided by Michel Puceat, Centre de Researches, INSERM, U390 France) were cultured and maintained on gelatin-coated plates (Sigma-Aldrich, St. Louis, MO, USA and BD Biosciences, San Jose, CA, USA, respectively) in Dulbecco's modified Eagle medium (Invitrogen, Grand Island, NY, USA) containing leukemia inhibitory factor (LIF; Millipore, Billerica, MA, USA), glutamine (Millipore), penicillin/streptomycin (Millipore), sodium pyruvate (Millipore), β-mercaptoethanol (Sigma-Aldrich), and fetal bovine serum (Invitrogen). ES cell-derived CM was generated as previously described (28,30). In brief, 5 × 105 ES cells were cultured in a 100-mm Petri dish for 24 h followed by a media exchange with LIF-excluded media. After 48 h, cell media containing ES cell-released factors was removed, filtered (0.2-um filter; Millipore), labeled ES-CM, and retained for future use.
Animals and Experimental Protocol
All protocols involving animals were reviewed and approved by the University of Central Florida Animal Care Committee. Male and female C57BL6 mice, with no sex discrimination, between 8 and 10 weeks of age, were divided into the following five study groups: Control (C), DOX (D), DOX + cell culture (CC) media (D + CC), DOX + conditioned media (D + CM), and DOX + ES cells (D + ES Cells); n = 6—8 mice/group.
Intraperitoneal (IP) DOX (Sigma-Aldrich) injections were administered on day 1 (D1), D3, and D5 with each mouse receiving a cumulative dose of 12 mg/kg divided into three separate, equal injections, each containing 4 mg/kg. For treatment groups, CC media (400 μl/injection × three injections), ES-CM (400 μl/injection × three injections), or ES cells (5 × 105 ES cells/injection × three injections) were injected IP on D2, D4, and D6. Twenty-eight days post-D6, heart function was assessed using echocardiography (Sonos 5500 Ultrasound System with a 15-6 L hockey-stick transducer; Philips Sonos, Andover, MA, USA), and animals were sacrificed by pentobarbitol (40 mg/kg IP; Henry Schein, Lake Mary, FL, USA) and cervical dislocation.
Preparation of Sections and Histopathology
Upon removal, hearts were fixed in 4% paraformaldehyde (Fisher Scientific, Pittsburgh, PA, USA) and embedded in paraffin. Hearts were cut into 5-μm serial sections, deparaffinized in xylene (Fisher Scientific), and rehydrated by sequential incubation in 100%, 95%, 70%, and 30% ethanol (Fisher Scientific). To visualize the vessels and quantify vascular fibrosis, hearts were stained with Masson's trichrome (Fisher Scientific) as previously described (3). Next, collagen accumulation around vessels [vascular fibrosis (VF)], as well as total vessel area (VA), were measured using ImageJ software (NIH, Bethesda, MD, USA), and a ratio of VF/VA was obtained.
Apoptotic Vessels
To identify apoptotic vasculature, heart sections were deparaffinized as aforementioned and permeabilized with proteinase K (Sigma-Aldrich; 25 μg/ml in 100 mM Tris HCl; Fisher Scientific). Apoptotic nuclei were identified using a terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) assay (TMR red; Roche Applied Bio Sciences, Indianapolis, IN, USA) as previously published (3). Heart sections stained with TUNEL were costained with primary antibodies against rabbit anti-cluster of differentiation 31 (CD31; Cat# 250589, 1:100 dilution; Abbiotec, San Diego, CA, USA) followed by incubation with goat anti-rabbit IgG-fluorescein isothiocyanate secondary antibodies (1:30 dliution; Santa Cruz, Santa Cruz, CA, USA). Sections were then mounted with Vectashield (Vector Laboratories, Burlingame, CA, USA) and viewed under Olympus (Center Valley, PA, USA) fluorescent and Leica (Buffalo Grove, IL, USA) confocal microscopes. Left ventricular vessels containing colocalized CD31 and TUNEL were considered apoptotic. Apoptotic vasculature was quantified in one to two heart sections from n = 5—6 hearts.
c-kit, FLK-1, and CD31 Immunostaining
Heart sections were deparaffinized in xylene, rehydrated in ethanol, and washed in distilled water and PBS (Fisher Scientific) as described above. c-kit, FLK-1, and CD31 immunostainings were performed according to previously published protocols (3). In brief, nonspecific binding sites were blocked by incubation in 10% normal goat serum (NGS; Vector Laboratories) for 1 h. Sections were incubated with mouse anti-c-kit, anti-FLK-1, or anti-CD31 monoclonal antibodies (Santa Cruz Biotechnologies, Santa Cruz, CA) diluted 1:25 in 10% NGS for 60 min followed by three PBS washes. Next, sections were incubated with Alexa Fluor 568- or 488-conjugated goat anti-mouse IgG secondary antibodies (Invitrogen) diluted 1:50 in PBS for 60 min and mounted with antifade Vectashield medium containing 4′,6-diamidino-2-phenylindole (DAPI) (Vector Laboratories) for nuclear visualization. Quantitative analysis of c-kit+, FLK-1+, and CD31+ cells was performed on heart sections from three to five different hearts from each group under Olympus and Leica confocal microscopes. In brief, the total number of c-kit+ cells per section was corrected with DAPI and quantified under an Olympus microscope. Next, c-kit cells positive for FLK-1 and CD31 were quantified, and a ratio of double c-kit+/FLK-1+ and c-kit+/ CD31+ cells over total c-kit+ cells were obtained and multiplied by 100. Finally, CD31+ cells were quantified in the same fashion as c-kit and corrected with total DAPI to obtain a ratio of percent CD31+ cells per section.
Vascular Smooth Muscle Immunostaining
To quantify the number of vessels per heart section, deparaffinized and rehydrated sections were incubated overnight with mouse anti-smooth muscle α-actin antibody (1:100; Sigma-Aldrich), followed by anti-mouse antibody (4:1,000, M.O.M. kit; Vector Laboratories) for 1 h over an orbital shaker. Sections were then mounted with antifade Vectashield mounting medium containing DAPI to detect total nuclei. Total number of vessels per slide was quantified under Olympus microscope, and representative pictures for each group were obtained using a Leica scanning confocal microscope.
Echocardiographic Analysis
Cardiac function was assessed at 4 weeks post-D6 by transthoracic echocardiography in n = 6—8 mice/group as previously published (8). Mice were anesthetized with 2% isoflurane (Butler Animal Health Supply, Dublin, OH, USA) and placed in the supine position on a temperature-controlled heating pad (Gaymar Industries, Inc., Orchard Park, NY, USA). Echocardiography was performed using a Philips Sonos 5500 Ultrasound system with a 15-6 L hockey-stick transducer. Left ventricular internal dimension-diastole (LVIDd), left ventricular internal dimension-systole (LVIDs), fractional shortening [FS, (LVIDd − LVIDs)/LVIDd × 100], left ventricular volume at end diastole [EDV = 7/(2.4 + LVIDd) × LVIDd3], left ventricular volume at end systole [ESV = 7/(2.4 + LVIDs) × LVIDs3], and ejection fraction [EF = (EDV —ESV)/EDV × 100] were measured in the short-axis view at the midpapillary muscle level.
Data Analysis
Statistical analysis was performed using SigmaStat 3.5 software (Statcon, Witzenhausen, Germany). All values are presented as mean ± SEM. Analysis of data was performed using the one-way ANOVA and the Tukey ad hoc test. Statistical relevance was assigned when p < 0.05.
Results
CM and ES Cells Prevent Vessel Apoptosis in DIC
Representative photomicrographs demonstrating vascular apoptosis are shown in Figure 1A with CD31+ cells stained red (A—E), TUNEL+ nuclei in green (F—J), and total nuclei stained blue with DAPI (K—O). The lower panels in Figure 1A (P—T) are merged images from each group. The smaller boxes (U—Y) are enhanced images shown to demonstrate the colocalization of CD31, TUNEL, and DAPI within a single vessel. Quantification of apoptotic vessels suggest vascular apoptosis is significantly elevated in D and D + CC hearts relative to controls (p < 0.05) (Fig. 1B). Importantly, CM and ES cell transplantation dramatically mitigate vessel apoptosis relative to D and D + CC groups (p < 0.05) (Fig. 1B). However, no statistical significance was obtained between CM and ES cell-transplanted hearts (Fig. 1B).

Effects of transplanted CM and ES cells on vascular cell apoptosis in the DIC injured heart. (A) Representative photomicrographs demonstrating vascular apoptosis are shown with CD31+ cells stained red (A—E), TUNEL+ nuclei in green (F—J), and total nuclei stained blue with DAPI (K—O). The lower panels (P—T) are merged images from each group. The smaller boxes (U—Y) are enhanced images shown to demonstrate the colocalization of CD31, TUNEL, and DAPI within a single vessel. Scale bar: 100 μm. (B) Right histogram demonstrates quantitative vessel apoptotic data from n = 5—6 animals per group. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC. D + CM: DOX + CM; D + ES cells: DOX + ES cells. DIC, doxorubicin-induced cardiomyopathy.
CM and ES Cells Activate c-kit+/FLK-1+ Cells in Chronic DIC
To determine the impact of transplanted CM and ES cells on cardiac endogenous vascular progenitor cells and their contribution to neovascularization in chronic DIC, sections were double-stained with c-kit and FLK-1. Representative images in Figure 2A demonstrate c-kit+ cells in green (A, F, K, P, and U), FLK-1+ cells in red (B, G, L, Q, and V), total nuclei in blue (C, H, M, R, and W), and merged images (D, I, N, S, and X). The smaller boxes (E, J, O, T, and Y) are enlarged images shown to demonstrate the colocalization of c-kit, FLK-1, and DAPI. Statistical analysis revealed that a significant decline in the number of c-kit+/FLK-1+ cells resulted in the D and D + CC groups relative to the non-DIC hearts (p < 0.05) (Fig. 2B). Importantly, a significant increase in the number of cells with combined c-kit and FLK-1 expression was quantitated in the CM and ES cell-transplanted groups suggesting that CM and ES cells may promote new artery formation in the DIC injured heart (p < 0.05) (Fig. 2B).

Effects of transplanted CM and ES cells on c-kit and FLK-1 activation. (A) Representative images demonstrate v-kit Hardy—Zuckerman 4 feline sarcoma viral oncogene homolog-positive (c-kit+) cells in green (A, F, K, P, and U), fetal liver kinasepositive (FLK-1+) cells in red (B, G, L, Q, and V), total nuclei in blue (C, H, M, R, and W), and merged images (D, I, N, S, and X). Enlarged merged images are presented in E, J, O, T, and Y. Scale bar: 80 μm. (B) Right histogram reveals a significant increase in c-kit+/FLK-1+ activation following CM and ES cell transplantation. n = 5—6 animals/groups. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC.
CM and ES Cells Enhance c-kit+/CD31+ Cells in the Chronic DIC Myocardium
To determine whether c-kit+ cells were present in mature coronary artery sites, sections were double-labeled with c-kit and CD31, a mature endothelial marker. Representative photomicrographs in Figure 3A show c-kit+ cells in green (A, F, K, P, and U), CD31+ cells in red (B, G, L, Q, and V), total nuclei stained with DAPI in blue (C, H, M, R, and W), and merged images (D, L, N, S, and X). Enlarged images demonstrating colocalization of c-kit, CD31, and DAPI are presented in (E, J, O, T, and Y). Data provided suggest that the amount of c-kit+ cells localized with CD31+ cells is significantly diminished in the D and D + CC groups compared to the control group (p < 0.05) (Fig. 3B). However, following CM and ES cell transplantation, c-kit+ cells colocalized with CD31+ cells were significantly enhanced relative to the D and D + CC groups, implying CM and ES cells activate c-kit+ cells, which, in turn, differentiate into mature endothelial cells (p < 0.05) (Fig. 3B).
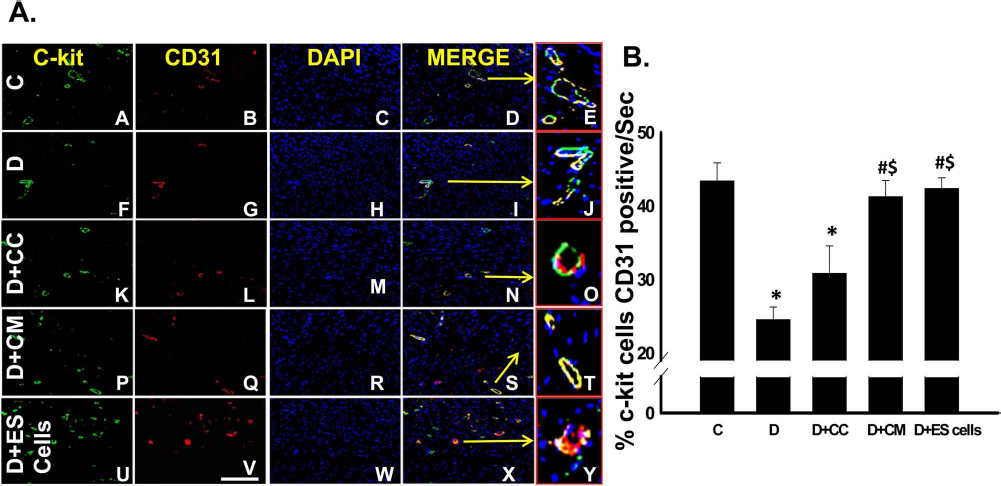
Transplanted CM and ES cells enhance c-kit+/CD31+ cells in DIC injured heart. (A) Representative photomicrographs show c-kit+ cells in green (A, F, K, P, and U), CD31+ cells in red (B, G, L, Q, and V), total nuclei stained with DAPI in blue (C, H, M, R, and W), and merged images (D, I, N, S, and X). Enlarged merged images are presented in E, J, O,T, and Y. Scale bar: 100 μm. (B) Right histogram demonstrates c-kit+/CD31+ cell quantitative data. n = 5—6 animals/groups. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC.
CM and ES Cells Enhance CD31+ Cells in the DIC Heart
Representative images are shown in Figure 4A demonstrating CD31+ cells in red (A, E, I, M, and Q), total nuclei in blue (B, F, J, N, and R), and merged images (C, G, K, O, and S). Enlarged images of CD31+ cells are depicted in (D, H, L, P, and T). The number of CD31+ cells in heart sections from each group was quantified, and our data reveal a significant decrease in the number of these cells in the untreated DIC hearts compared to non-DIC myocardium (p < 0.05) (Fig. 4B). Notably, CM and ES cell-transplanted groups contained dramatically more CD31+ cells compared to the D and D + CC groups (p < 0.05) (Fig. 4B). Although not statistically significant, a trend of increased CD31 expression in D + CM and D + ES cell groups relative to control was observed, suggesting CM and ES cells promote vessel formation in response to acute DIC injury (Fig. 4B).
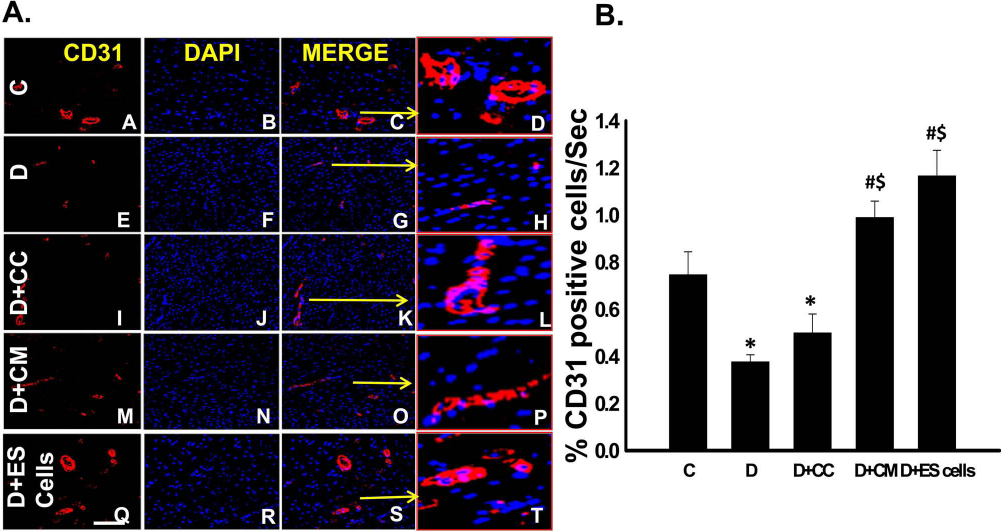
Effects of CM and ES cell transplantation on cardiac CD31 expression. (A) Representative images are shown demonstrating CD31+ cells in red (A, E, I, M, and Q), total nuclei in blue (B, F, J, N, and R), and merged images (C, G, K, O, and S). Enlarged merged images are presented in D, H, L, P, and T. Scale bar: 100 μm. (B) Right histogram reveals a significant increase in CD31 expression in CM and ES cell groups compared to D and D + CC groups. n = 5—6 animals/groups. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC.
Transplanted CM and ES Cells Promote Vessel Formation in the Injured DIC Heart
To determine the effects of transplanted CM and ES cells on coronary vessel formation, total myocardial vessels were identified using smooth muscle α-actin immunostaining and Masson's trichrome staining as depicted in Figure 5A and B, respectively. Quantitative analysis revealed a significant increase in the number of vessels in the CM and ES cell group myocardium relative to the DOX-alone injured heart (p < 0.05) (Fig. 5C). However, no statistical significance (p > 0.05) was observed between the CM and ES cell groups (Fig. 5C).
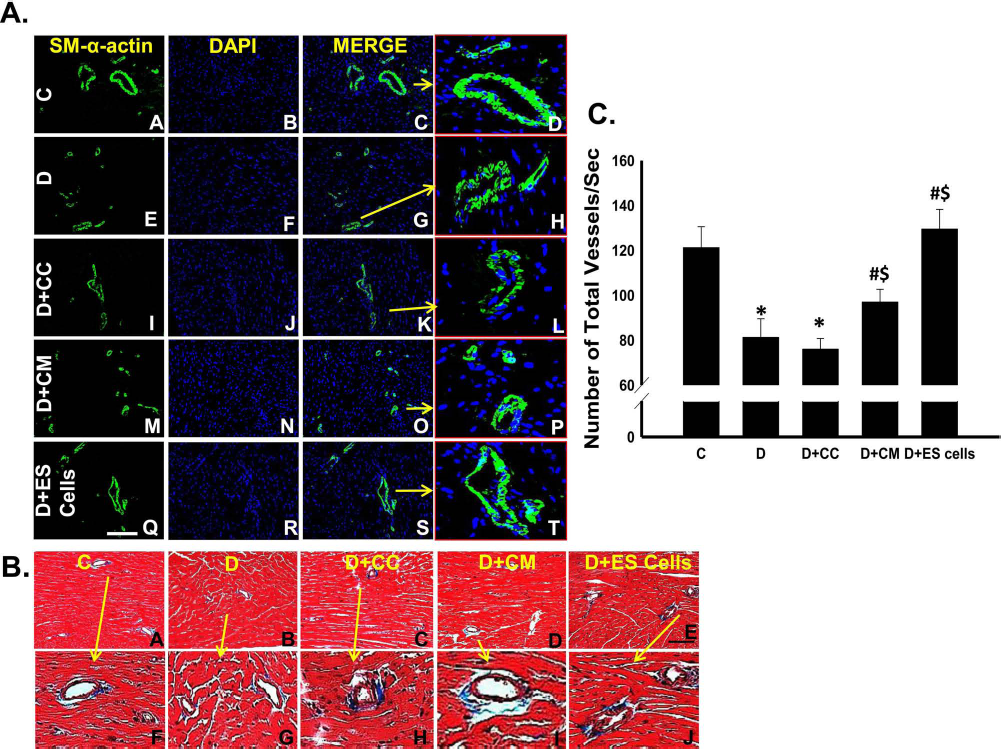
Effects of transplanted CM and ES cells on coronary artery formation in the DIC-injured myocardium. (A) Representative images are shown demonstrating vascular smooth muscle cells (A, E, I, M, and Q), total nuclei in blue (B, F, J, N, and R), and merged images (C, G, K, O, and S). Enlarged merged images are presented in D, H, L, P, and T. (B) Representative photomicrographs demonstrating Masson's trichrome-stained sections from control and experimental groups (A—E). Enlarged vessels from (A—E) are depicted in (F—J). Arrows represent the areas enlarged from (A—E) to (F—J), respectively. (C) Histogram demonstrating quantitative analysis of total vessel counts. n = 5—6 animals/groups. Scale bar: 100 μm. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC.
Transplanted CM and ES Cells Reduce Vascular Fibrosis in the Injured DIC Heart
Collagen accumulation around the vessels was detected on Masson's trichrome-stained heart sections (Fig. 6). The ratio of VF over VA demonstrated significantly increased fibrosis around vessels in DOX alone and DOX-CC animals (p < 0.05). Nonetheless, this increase was significantly reduced following CM and ES cell transplantation (p < 0.05).
CM and ES Cells Preserve Cardiac Function in Chronic DIC
Echocardiographic analysis was used to examine the effects of transplanted CM and ES cells on cardiac function at 4 weeks post-D6. LVIDd, LVIDs, FS, EDV, ESV, and EF were significantly dysregulated in the D and D + CC groups compared to the nonpathological mice (Fig. 7A—F). However, in CM and ES cell-transplanted mice, heart function for all parameters evaluated were significantly improved including 1) decreased LVIDd (Fig. 7A); 2) decreased LVIDs (Fig. 7B); 3) increased FS (Fig. 7C); 4) decreased EDV (Fig. 7D); 5) decreased ESV (Fig. 7E); and 6) increased EF (Fig. 7F) relative to D and D + CC mice (p < 0.05). Overall, the echocardiographic data suggest significant left ventricular functional improvement following CM and ES cell transplantation consistent with neovascularization.

Transplanted CM and ES cells reduce vascular fibrosis following DIC. (A) (A—E) Representative images of VF for all experimental groups. (F) Histogram demonstrating quantitative analysis of VF/VA percent. n = 5—6 animals/groups. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC.

Transplanted CM and ES cells improve cardiac function following DIC. (A) LVIDd, (B) LVIDs, (C) FS, (D) EDV, (E) ESV, and (F) EF. LVIDd, left ventricular internal dimension-diastole; LVIDs, left ventricular internal dimension-systole; EDV, left ventricular volume at end diastole; ESV, left ventricular volume at end systole. n = 6—8 animals/groups. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC.
Discussion
DOX is one of the most conventionally used chemotherapeutic drugs presently available for the treatment of variegated neoplastic disorders. Although its efficacy is indisputable throughout the medical community, dose-dependent, adverse cardiotoxicity accompanies DOX therapy (6,11,20,23). Alterations to endogenous myocardium consequent to DOX include, but are not limited to, myocardial cell apoptosis, hypertrophy, adverse cardiac remodeling, and impaired cardiac function (6,11,20,23). To this end, the need to identify novel approaches to reduce the cardiotoxic side effects of DOX treatment remains a priority.
Throughout the past decade, an abundance of data has been presented, highlighting the efficacy and potential of stem cells to treat various cardiovascular dysfunctions including DIC (1,9,26,29,35). Specifically, transplanted adult stem cells significantly improved cardiac function in DIC-afflicted rodent models with limited to no engraftment of transplanted cells reported (1,35). Additionally, the ability of ES cells and their released factors to repair and regenerate the acute DIC-injured myocardium was recently challenged (26). Data presented suggest ES cells and the CM 1) inhibit apoptosis, fibrosis, cytoplasmic vacuolization, and myofibrillar loss; 2) activate c-kit+ cardiac stem cells; and 3) improve cardiac function (26). However, the sustained neovascularization potential and resulting functional consequences of transplanted CM and ES cells in the chronic DIC myocardium remain largely unknown. In the present study, we have evaluated the effects of transplanted CM and ES cells on vessel apoptosis, vascular progenitor cell (VPC) stimulation, new vessel formation, and cardiac function in chronic DIC. To the best of our knowledge, this is the first investigation into the vascular impact propagated by CM and ES cells in the prolonged DIC-injured myocardium.
Vascular apoptosis plays a role in the manifestation and progression of a myriad of cardiac anomalies but has yet to be investigated in DIC (17). To this end, we sought to establish a relationship between DOX and myocardial vasculature cell death. Colocalization of CD31, a vascular endothelial cellular marker, and TUNEL were significantly elevated in the D and D + CC hearts relative to saline control hearts suggesting a novel mechanism of cardiac dysregulation following DOX treatment. However, vessel apoptosis was significantly abrogated in groups transplanted with CM and ES cells relative to the diseased models. Of note, a comparative analysis involving vascular cell death cannot be made between acute and chronic DIC at this time, as vessel apoptosis in DIC has never been examined.
Several studies have suggested that activation of the stem cell factor/c-kit signaling pathway enhances the survival, migration, and tube formation potential of endothelial cells as well as significantly contributes to neovascularization and myocardial regeneration in ischemic tissue (2,4,12). Similarly, the efficacy of transplanted c-kit+ stem cells into the injured myocardium has been evaluated with results including enhanced cardiac repair and regeneration, neovascularization, and improved cardiac function (14,22). In this context, we demonstrate the number of c-kit+ cells, indicative of cardiac progenitor cells, also expressing FLK-1, the earliest known marker of endothelial cell lineage, were significantly enhanced in the chronic DIC myocardium following CM and ES cell transplantation. Our data are concurrent with earlier studies suggesting 1) CM generated from ES cells stimulated c-kit+ cardiac progenitor cells and circulating FLK-1 cells in the infarcted heart and 2) transplanted CM and ES cells increase levels of c-kit+ cardiac progenitor cells in acute DIC (3,26).
The generation and maturation of cardiac vasculature is a complex, multifaceted process involving various cell types (33). To this end, although the increased presence of c-kit+/FLK-1+ cells in the CM and ES cell groups indicates early onset of neovascularization, identification of later vessel maturation is warranted. Consistent with the elevated concentration of myocardial c-kit+/FLK-1+ cells in the CM and ES cells groups, mature vessels derived from the c-kit+ progenitor cells, as demonstrated by colocalized c-kit and CD31 staining, were dramatically increased following CM and ES cell transplantation in the DIC-injured myocardium. Additionally, CD31+ cells and total vessel counts were abundant following CM and cell therapy. Furthermore, our data also suggest decreased fibrosis around existing vessels in animals subjected to CM and ES cells treatment. Overall, our data not only suggest that CM and ES cells promote neovascularization in DIC via paracrine mechanisms, by activation of vascular progenitor cells, but also through maturation mechanisms not yet identified.
CM and cellular therapy has been widely tested in a host of animal and human cardiac dysfunctions (9,15,16, 18,19,21,22,26). Data from animal studies have demonstrated significant improvement in left ventricular function following transplantation of stem cells and their released factors (21,22). Transplanted human ES cell-derived vascular cells, bone marrow-derived multipotent progenitor cells, and c-kit+ stem cells significantly impacted the functional capacity of the infarcted myocardium consequent to neovascularization and myocardial repair (14,22,32). Moreover, bone marrow-derived stem cells, ES stem cells, and CM have been shown to inhibit heart failure and augment impaired contractility in various acute models of DIC (5,26). Congruent with these earlier studies, transplantation of CM and ES cells significantly blunted impaired FS and EF consequent to chronic DIC.
In conclusion, reported for the first time, the data presented suggest that CM and ES cells transplanted into the DIC-injured heart have the potential to stimulate the differentiation and maturation of endogenous VPCs and improve cardiac function. However, further studies are warranted to answer critical questions, including identification of the molecular mechanisms by which neovascularization occurs.
Footnotes
Acknowledgments
The authors would like to thank Carley Glass, Binbin Yan, Reetu Singla, and Aisha Ahmed for their assistance with the article. This work was supported, in part, by grants from the National Institutes of Health (1R01HL090646-01 and 5R01HL094467-02 to D.K.S.). The authors declare no conflicts of interest.
