Abstract
Doxorubicin (DOX) is an effective chemotherapeutic drug used for the treatment of a variety of malignancies. Unfortunately, time and dose-dependent DOX therapy induces cardiotoxicity and heart failure. We previously reported that transplanted embryonic stem (ES) cells and the conditioned medium (CM) can repair and regenerate injured myocardium in acute DOX-induced cardiomyopathy (DIC). However, the effectiveness of ES cell and CM therapeutics has not been challenged in the chronic DIC model. To this end, the long-term impact of ES cells and CM on apoptosis, fibrosis, cytoplasmic vacuolization, oxidative stress, and their associated mediators were examined. Four weeks post-DIC, ES cells and CM-transplanted hearts showed a significant decrease in cardiac apoptotic nuclei, which was consequent to modulation of signaling molecules in the Akt pathway including PTEN, Akt, and mTOR. Cytoplasmic vacuolization was reduced following treatment with ES cells and CM, as was cardiac fibrosis, which was attributable to downregulation of MMP-9 activity. Oxidative stress, as evidenced by DHE staining and lipid peroxide concentration, was significantly diminished, and preservation of the antioxidant defense system was observed following CM and ES cell transplantation. In conclusion, our data suggest that transplanted ES cells and CM have long-term potentiation to significantly mitigate various adverse pathological mechanisms present in the injured chronic DIC heart.
Introduction
Doxorubicin (DOX) is an antineoplastic therapeutic agent widely used for the treatment of various malignancies (22,29). However, clinical applications utilizing this drug are restricted due to the severe, dose-dependent, acute cardiotoxicity that may occur, leading to irreversible, chronic cardiomyopathy [doxorubicin-induced cardiomyopathy (DIC)] and congestive heart failure (22,29). Although the exact mechanism of DIC has yet to be established, the antitumor activity of DOX is thought to be independent from the mechanisms of induced cardiomyopathy (22,29). The development of post-DIC heart failure includes 1) cardiac myocyte and nonmyocyte myocardial cell apoptosis, 2) intrinsic architectural degeneration, and c) exacerbated collagen infiltration. These adverse changes promote the rearrangement of heart tissue, which in turn contributes to increased wall stress and insufficient contractility of the cardiac myocytes (17,22,29,36). Noteworthy, the exaggerated collagen synthesis in DIC coincides with extracellular matrix degradation consequent to activation of matrix metalloproteinases (MMPs) (17,34,36). Previous studies have demonstrated the ability of various factors and therapeutics to promote cytoprotection in acute DIC (16—18,30). However, investigation into the long-term potential of these approaches relative to chronic DIC is warranted.
Over the past decade, cell transplantation studies have provided invaluable insight into the potential of cellular therapy for the treatment of diverse heart anomalies including DIC (1,13,37). Published data suggest a significant improvement in cardiac function results following adult stem cell transplantation into acute DIC-afflicted myocardium (1,13,37). Of interest, these studies admittedly acknowledged minimal to no engraftment of transplanted cells (1,37). More recently, the ability of embryonic stem (ES) cells and their released factors to repair and regenerate the injured DIC myocardium was assessed (30). Evidence provided suggested that ES cells and their conditioned media (CM) were capable of blunting acute DIC adverse conditions (30). However, the long-term therapeutic potential of ES cells and CM in chronic DIC remains unknown. Therefore, in the present study, I hypothesized that transplanted ES cells and their CM containing cytoprotective released factors would provide sustainable cytoprotection in chronic DIC. I present data indicating transplanted ES cells and CM inhibit myocardial apoptosis, fibrosis, cytoplasmic vacuolization, and oxidative stress (OS), all of which are typical DIC characteristics. I also elucidated mechanisms of cell survival, fibrosis inhibition, and OS regulation, which include modulation of phosphatase and tensin homolog (PTEN), v-akt murine thymoma viral oncogene homolog (Akt), mechanistic target of rapamycin (mTOR), MMP-9, and pro- and antioxidant expression.
Materials and Methods
ES Cells and Preparation of CM
Mouse CGR8 ES cells (provided by Michel Puceat, Centre de Researches, INSERM, U390 France) were maintained as previously described in Dulbecco's modified Eagle medium (Invitrogen, Grand Island, NY, USA) containing leukemia inhibitory factor (LIF; Millipore, Billerica, MA, USA), glutamine (Millipore), penicillin/streptomycin (Millipore), sodium pyruvate (Millipore), β-mercaptoethanol (Millipore), and fetal bovine serum (Invitrogen) (32,33). To prepare ES-CM, ES cells were cultured for 24 h on gelatin-coated plates (Sigma-Aldrich, St. Louis, MO, USA and BD Biosciences, San Jose, CA, USA, respectively), after which cell culture media was replaced with fresh cell culture media excluding LIF. Post-48 h, cell supernatant containing released factors was removed, filtered (0.2 um filter, Millipore), and labeled ES-CM for future use.
Animals and Experimental Protocol
All mice and animal protocols used in the study were reviewed and approved by the University of Central Florida Animal Care Committee. C57BL/6 mice (The Jackson Laboratory, Bar Harbor, ME, USA) were divided into five study groups: Control (C), DOX (D), DOX + cell culture (CC) medium (D + CC), DOX + CM (D + CM), and DOX + ES cells (D + ES cells); n = 6—8 mice were in each of the five study groups.
DOX (Sigma-Aldrich) was administered intraperitoneally (IP) on day 1 (D1), D3, and D5. Each day DOX was given, the cumulative dose of 12 mg/kg was divided into three separate, equal injections each containing 4 mg/kg. For treatment groups, CC medium (400 μl/injection × three injections), ES-CM (400 μl/injection × three injections), or ES cells (5 × 105 ES cells/injection × three injections) was injected IP on D2, D4, and D6. Four weeks post-D6, animals were sacrificed by pentobarbitol (40 mg/kg IP; Henry Schein, Lake Mary, FL, USA) and cervical dislocation. Upon heart removal, half of the heart tissue was fixed in 4% paraformaldehyde (Fisher Scientific Company, LLC, Kalamazoo, MI, USA), whereas the other half was preserved in RNA later for future biochemical and ELISA assays.
Identification of Apoptotic Nuclei and Caspase 3 Staining
Apoptotic nuclei in heart sections from control and experimental groups were evaluated using a terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) assay (TMR red; Roche Applied Biosystems, Indianapolis, IN, USA) following instructions provided in the kit and that have been previously published by our group (31). Additionally, TUNEL-positive apoptotic nuclei were costained with proapoptotic caspase 3, as well as with sarcomeric cardiac α-actin to identify cardiac myocyte cell death. Briefly, TUNEL-stained heart sections were incubated with primary antibodies; active anti-caspase 3 rabbit polyclonal (1:1,000; Cell Signaling, Danvers, MA, USA) and sarcomeric cardiac α-actin monoclonal antibodies (1:30; Sigma-Aldrich, St. Louis, MO, USA). Heart sections were washed and incubated with appropriate secondary antibodies: Alexa 568 (Invitrogen) or Alexa 488 (MOM kit; Vector Laboratories, Burlingame, CA, USA). Sections were mounted with Vectashield containing 4′,6-diamidino-2-phenylindole (DAPI; Vector Laboratories), which stained total nuclei blue. Heart sections were examined, and photomicrographs were prepared using an Olympus fluorescence microscope (Center Valley, PA, USA) and a Leica laser scanning confocal microscope (Buffalo Grove, IL, USA). Left ventricular apoptotic nuclei, examined under 20× magnification, was quantified by two blind observers in four to five random areas in one to two heart sections from n = 5—6 hearts as previously published (31). The percentage of apoptotic nuclei per section was calculated using the following formula: (number of apoptotic nuclei/total number of nuclei per section) × 100.
Caspase 3 Activity
Caspase 3 activity was quantitated using a kit from BioVision (Milpitas, CA, USA) as reported previously (5). Heart tissue from all groups was homogenized in radioimmunoprecipitation assay (RIPA) buffer (Enzo Life Sciences, Inc., Farmingdale, NY, USA). Protease inhibitor cocktail, 1 mM phenylmethylsulfonyl fluoride, 2 mM sodium orthovandate, and 5 mM sodium fluoride (all Sigma) were added into the RIPA buffer before homogenization. Following centrifugation and isolation of supernatant, protein concentrations were estimated using a Bio-Rad assay (Hercules, CA, USA). The activity assay was completed following the manufacturer's instructions, and the developed blue color was measured at 405 nm in a Bio-Rad microplate reader. Caspase 3 activity was measured in n = 4—6 animals/group and plotted as arbitrary units (AU).
Phosphorylated PTEN Activity
Levels of phosphorylated PTEN (p-PTEN) were quantified using a Phospho-PTEN Sandwich ELISA kit (No. 7285; Cell Signaling) as previously published (10). In brief, heart tissue from all groups was homogenized as aforementioned, and protein estimation was performed using the Bio-Rad assay. The p-PTEN ELISA was performed according to the instructions provided with the kit (Cell Signaling). There was a development of color reaction following 20—30 min incubation. The color proportional to p-PTEN expression was determined at 450 nm in an ELISA plate reader (Bio-Rad). p-PTEN activity was corrected for the total protein concentration measured for each sample. Data were collected from n = 3—5 heart samples in duplicates and plotted as AU.
Phosphorylated Akt Activity
Phosphorylated Akt (p-Akt) activity was examined using a commercially available phospho-Akt1 (PAN) ELISA kit (Exalpha Biologicals, Inc., Maynard, MA, USA). In brief, tissues were homogenized as described beforehand, and p-Akt was quantified according to manufacturer's instructions. The developed color reaction was measured at 450 nm using a microtiter plate reader (Bio-Rad). p-Akt data were obtained from n = 4—6 animals/group and plotted as AU.
mTOR Activity
Levels of phosphorylated mTOR (p-mTOR) were assessed using a commercially available Phospho-mTOR Sandwich ELISA kit (No. 7976S; Cell Signaling Technology) according to manufacturer's instructions. In brief, hearts from each experimental group were homogenized as previously described, and protein concentration was estimated. The developed reaction, proportional to p-mTOR expression within left ventricular tissue, was measured at 450 nm, and resulting data were corrected for protein concentration of each sample. p-mTOR data were obtained from n = 3—4 animals/group in duplicates and plotted as AU.
Determination of Vascular Vacuolization and Fibrosis
Hearts were removed, fixed in 4% buffered formalin (Fisher Scientific, Kalamazoo, MI, USA), and embedded in paraffin as previously published (31). Five-micrometer serial sections were prepared, followed by deparaffinization in xylene (Fisher Scientific, Fair Lawn, NJ, USA). Sections were rehydrated by sequential incubation in 100%, 95%, and 70% ethanol for 5 min each at room temperature. To identify vascular vacuolization and fibrosis, heart sections were then stained with hematoxylin and eosin (H&E) or Masson's trichrome (both Fisher Scientific), respectively. Two to three H&E-stained heart sections from each group were examined for the evaluation of cytoplasmic vacuolization. The qualitative data scale used to determine cytoplasmic vacuolization ranged from 0 to 2. Left ventricular heart sections demonstrating no cytoplasmic vacuolization were graded 0, whereas if a significant portion of the section had cytoplasmic vacuolization, then the section was graded 2. Cardiac fibrosis, predominantly present in the left ventricle, was examined in Masson's trichrome-stained sections and quantified by measuring the total blue area per square millimeter using the NIH program, ImageJ (NIH, Bethesda, MD, USA).
MMP-9 Expression
MMP-9 concentration in heart homogenates from each experimental group was quantified using an enzyme-linked immunoassay (MMPT90; R&D Systems, Minneapolis, MN, USA) following the manufacturer's instructions. In brief, mouse hearts from n = 6—8 animals were homogenized in RIPA buffer, and protein concentrations were estimated. Samples were added into MMP-9 antibody-coated wells for 2 h, followed by incubation with a polyclonal antibody for mouse MMP-9 (provided with the kit) and a substrate with washings in between each step. Following the addition of a stop solution, the color reaction was measured at 450 nm in a microtiter plate reader. Data obtained was corrected for protein concentration of each sample. The MMP-9 data were obtained from n = 5—6 animals/group, and the graph was plotted as AU.
Dihydroethidium Staining
Superoxide generation within DIC-injured myocardium was quantified using dihydroethidium (DHE; Invitrogen, Eugene, Oregon, USA) fluorescence staining as previously described (10). In brief, sections were stained with 1 μM/ml DHE for 25 min at room temperature following deparaffinization and hydration. Images were visualized and DHE measured under Olympus and confocal microscopes (Leica). DHE fluorescence intensity data were obtained from four to five randomly selected regions in the heart from n = 5—6 animals/group. The average integrated density following background correction was calculated for each group and plotted as arbitrary fluorescence units.
Lipid Peroxides Assay
Levels of lipid peroxides (LPOs) were quantitated in isolated supernatant collected from homogenized control and experimental heart tissue as aforementioned. Per the manufacturer's instructions provided in the LPO-CC kit (Kamiya Biomedical, Seattle, WA, USA), reactions were developed and measured at 655 nm. Lipid peroxide values were calculated according to the following formula: (Esample − Eblank) × 50.0/(Ecalibrator − Eblank) and plotted as AU. Data was collected from heart homogenates of n = 5—8 animals/group.
MnSOD Activity Assay
Manganese-containing mitochondrial superoxide dismutase (MnSOD) activity was quantified in control and experimental hearts using a SOD-560 colorimetric assay kit (Applied Bioanalytical Labs, Sarasota, FL, USA). The MnSOD activity assay was performed following manufacturer's instructions and measured at 560 nm. MnSOD activity was calculated according to the following formula: 125 × (100% − Ratesample/Rateblank) × dilution factor. MnSOD activity was plotted as AU. Data were collected from heart homogenates of n = 6—8 animals/group in duplicates.
Catalase Activity Assay
Catalase activity was quantified using a Catalase assay kit (Abcam, Cambridge, MA, USA) according to manufacturer's instructions. The coloric reaction was measured at 570 nm, and results were normalized to total protein concentration as determined by the Bradford assay. Catalase activity was plotted as AU. Data were collected from heart homogenates of n = 6—8 animals/group.
Total Antioxidant Capacity Assay
The total antioxidant capacity for control and experimental groups was assessed using a Total Antioxidant Capacity (TAC) Colorimetric Assay Kit (Cat# K274-100; BioVision). The TAC assay was performed according to manufacturer's instructions, and absorbance was measured in a microplate reader at 570 nm. Obtained values were normalized to loaded protein concentrations, and resulting data was plotted as AU.
Data Analysis
Statistical analysis was performed using SigmaStat 3.5 software (Statcon, Witzenhausen, Germany). Values are presented as means ± SEM. One-way analysis of variance was performed followed by Tukey's significant difference post hoc test. Values were considered statistically significant when p < 0.05.
Results
Effects of Transplanted ES Cells and CM on Apoptosis in DIC
Figure 1A shows total nuclei stained with DAPI in blue (A, D, G, J, and M), apoptotic nuclei in red (B, E, H, K, and N), and merged images in pink (C, F, I, L, and O). Bottom panel histogram (B) shows there was a significant (p < 0.05) increase in apoptotic nuclei in D, D + CC, and D + CM treated hearts compared with controls. This significant increase in TUNEL-positive nuclei was attenuated in D + CM and D + ES cell groups relative to D and D + CC, suggesting CM and ES cells provide antiapoptotic cytoprotection in DIC (p < 0.05) (Fig. 1B). To demonstrate cardiac myocyte apoptosis, heart sections from all groups were triple labeled with TUNEL and antibodies for sarcomeric cardiac α-actin and caspase 3. Representative photomicrographs are shown for all groups in Figure 2A demonstrating TUNEL-positive nuclei stained red (A, F, K, P, and U), active caspase 3 in yellow (B, G, L, Q, and V), cardiac myocytes stained green (C, H, M, R, and W), total nuclei stained with DAPI in blue (D, I, N, S, and X), and colocalization of TUNEL, caspase 3, and sarcomeric cardiac α-actin (E, J, O, T, and Y). The boxes in the far right panel of Figure 2A are magnified single cardiac myocytes positive for TUNEL, caspase 3, and sarcomeric cardiac α-actin, suggesting apoptosis does occur in cardiac myocytes in DIC. Moreover, a caspase 3 activity assay was performed, and resulting data are shown in Figure 2B. Significant caspase 3 activity in the DOX alone-treated group compared with controls was attenuated following CM or ES cell transplantation (Fig. 2B). Additionally, hearts transplanted with ES cells also had significantly diminished caspase 3 activity compared to the D + CC group (p < 0.05) (Fig. 2B). Of note, apoptosis and caspase 3 activities were not significant between D + CM and D + ES cells groups.

Transplanted CM and ES cells inhibit cardiac myocyte apoptosis at 4 weeks post-DIC. (A) Representative photomicrographs of total nuclei stained with DAPI in blue (A, D, G, J, and M) and terminal deoxynucleotidyl TUNEL-stained apoptotic nuclei in red (B, E, H, K, and N) and merged nuclei in pink (C, F, I, L, and O). Scale bar: 100 μm. (B) Bottom histogram shows quantitative apoptotic nuclei per section from control and experimental groups: C (control), D (DOX), D + CC (DOX + CC), D + CM (DOX + CM), and D + ES cells (DOX + embryonic stem cells). Data are from the n = 5—6 animals. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC. DIC: DOX-induced cardiomyopathy.

Effects of transplanted CM and ES cells on cardiac myocyte apoptosis and caspase 3 activity. (A) Representative photos of heart sections demonstrating TUNEL-positive nuclei stained red (A, F, K, P, and U); active caspase 3 in yellow (B, G, L, Q, and V); cardiac myocytes stained green (C, H, M, R, and W); total nuclei stained with DAPI in blue (D, I, N, S, and X); and colocalization of TUNEL, caspase 3, and sarcomeric cardiac α-actin (E, J, O, T, and Y). The far right panels in (A) demonstrate enhanced images of colocalization of sarcomeric α-actin, caspase 3, TUNEL, and DAPI. Scale bar: 50 μm. (B) Right histogram shows quantitated caspase 3 activity from n = 4-6 animals per group. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC. AU, arbitrary units.
CM and ES Cells Modulate the Akt Pathway
Previous studies have shown that apoptosis is mediated by alterations to the Akt signaling pathway in ischemic heart diseases (8,20,33). To this end, expressions of Akt, as well as mediators of the cell survival pathway, were quantitated in each of the control and experimental groups. Levels of PTEN, an upstream inhibitor of phosphoinositide-3-kinase (PI3K) phosphorylation and subsequent Akt activation, were significantly increased in D and D + CC groups (p < 0.05) (Fig. 3A). However, PTEN activation was significantly attenuated following ES cell transplantation (p < 0.05) (Fig. 3A). Phosphorylated Akt was significantly diminished in the D and D + CC groups compared with controls, suggesting decreased cardiac cell survival in these groups (p < 0.05) (Fig. 3B). Importantly, hearts transplanted with CM and ES cells had significantly increased activation of p-Akt compared to D and D + CC (p < 0.05) (Fig. 3B). Finally, levels of activated mTOR were assessed, and a significant decrease in activation of the Akt downstream mediator was observed in the D and D + CC groups (p < 0.05) (Fig. 3C). A trend of increased mTOR expression was noted in the D + CM and D + ES cell groups, but statistical significance was not obtained (Fig. 3C).

Akt pathway modulation following CM and ES cell transplantation in DIC. (A) Histogram shows quantitative analysis of P-PTEN expression. (B) Histogram examines quantitative phosphorylated Akt expression. (C) Histogram demonstrates significant p-mTOR dysregulation in chronic DIC. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC. Data set from heart homogenates of n = 3—6 animals/group. AU, arbitrary units.
CM and ES Cells Inhibit Cytoplasmic Vacuolization
Cytoplasmic vacuolization, an adverse architectural alteration commonly described in DIC, was examined within the current study (Fig. 4A). Control group hearts were comprised of full-length cardiac myocytes with intact cytoplasmic framework (Fig. 4A). Conversely, D and D + CC treated hearts demonstrated a dramatic increase in the presence of cytoplasmic vacuoles (Fig. 4B). However, following transplantation of CM and ES cells, cytoplasmic vacuolization was significantly attenuated suggesting the ability of these therapies to maintain the integrity of the ventricular architecture (p < 0.05) (Fig. 4B).

Effects of transplanted CM and ES cells on cytoplasmic vacuolization. (A) Histological sections stained with H&E of representative left ventricular tissue from each group (A—E) at 4 weeks post-DIC induction. Arrows indicate areas of vacuolization. Scale bar: 100 μm. (B) Histogram shows quantitative cytoplasmic vacuolization in post-DIC with or without treatment. Data are from n = 3—6 animals. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC.
CM and ES Cells Inhibit Fibrosis in DIC
To determine the effect of transplanted CM and ES cells on cardiac fibrosis, heart sections were stained with Masson's trichrome. In Figure 5A, interstitial fibrosis is demonstrated by the blue areas present in the control and experimental groups. Quantitative data suggest that D, D + CC, and D + CM hearts contained significantly (p < 0.05) more fibrotic area relative to controls (Fig. 5B). However, this increased fibrosis was reduced following ES cell transplantation (Fig. 5B).

Effects of transplanted CM and ES cells on cardiac fibrosis. (A) Photomicrographs show histological sections stained with Masson's trichrome of representative left ventricular tissue from each group (A—E) at 4 weeks post-DIC induction. Blue area shows extent of fibrosis in control and experimental groups. Scale bar: 100 μm. (B) Histogram shows quantitative cardiac fibrosis in post-DIC for control and experimental groups. (C) Histogram of quantitative MMP-9 analysis from each group. Data are from n = 4—6 heart homogenates/group. *p < 0.05 versus C, and #p < 0.05 versus D, and $p < 0.05 versus D + CC.
To determine whether inhibition of fibrosis was propagated by changes in MMP-9, a documented mediator of adverse cardiac remodeling, levels of MMP-9 were evaluated using an enzyme-linked immunoassay. D and D + CC hearts demonstrated significantly increased MMP-9 concentrations compared with controls (p < 0.05) (Fig. 5C). Notably, hearts transplanted with ES cells showed a significant decline in MMP-9 expression, suggesting a potential mechanism by which ES cells prevent interstitial fibrosis (p < 0.05) (Fig. 5C).
CM and ES Cells Abrogate DIC-Induced Oxidative Stress
To examine the impact of CM and ES cells on chronic DIC-induced OS, myocardial intracellular anion production in heart sections was assessed using DHE fluorescent staining (Fig. 6A). As demonstrated in the representative photomicrographs (Fig. 6E and H) and quantified summary data (Fig. 6B), superoxide was significantly more concentrated in D and D + CC groups compared with controls. Conversely, the myocardium of groups receiving CM or ES cells contained significantly less superoxide relative to D and D + CC myocardium (p < 0.05) (Fig. 6B). To confirm the presence of OS in chronic DIC, levels of LPO were evaluated. D and D + CC hearts contained significantly elevated levels of LPO compared to their respective controls (p < 0.05) (Fig. 6C), whereas following CM and ES cell transplantation, these concentrations were significantly abrogated (Fig. 6C).

CM and ES cells blunt DIC-induced OS. Representative photomicrographs of total nuclei stained blue with DAPI (A, D, G, J, and M), superoxide anions in red (B, E, H, K, and N), and merged images in pink (C, F, I, L, and O). Scale bar: 100 μm. (B, C) Histograms demonstrating quantified DHE fluorescence and LPOs, respectively. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC. AFU, arbitrary fluorescence units; AU, arbitrary units.
To elucidate potential mechanisms by which CM and ES cells ameliorate chronic DIC OS assault, catalase, MnSOD, and total antioxidant capacity were evaluated. A significant decrease in MnSOD and catalase was observed in the D and D + CC groups relative to controls (p < 0.05) (Fig. 7A, B). However, transplantation of CM and ES cells dramatically blunted the diminished antioxidant levels observed in the D and D + CC groups, suggesting a mechanism by which CM and ES cells inhibit DIC-induced OS (Fig. 7A, B). Finally, the total antioxidant capacity in D, D + CC, and D + CM hearts was significantly depreciated (p < 0.05) relative to controls. Contrarily, the total antioxidant capacity following ES cell transplantation was greatly improved.
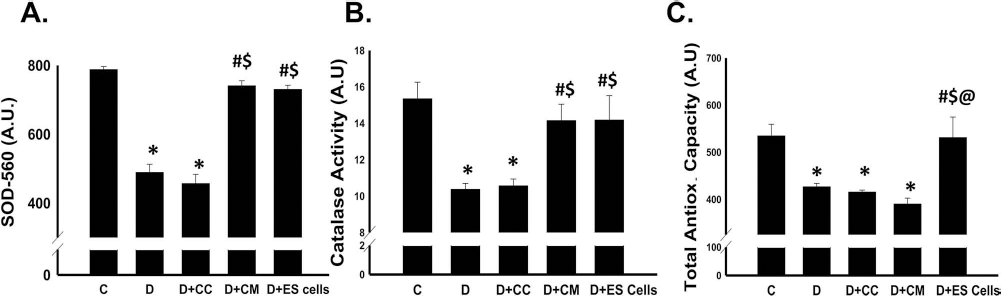
CM and ES cells protect antioxidant defense system in chronic DIC. (A) Histogram shows CM and ES cells significantly blunt diminished manganese mitochondrial superoxide dismutase (SOD-560) expression observed within D and D + CC groups. (B) CM and ES cell-transplanted hearts have significantly increased catalase activity relative to diseased groups. (C) Histogram shows total antioxidant capacity for control and experimental groups. *p < 0.05 versus C, #p < 0.05 versus D, and $p < 0.05 versus D + CC. AU, arbitrary units.
Discussion
DOX is one of the most widely utilized chemotherapeutic agents currently available for treatment of numerous neoplastic diseases. Despite the fact that its efficacy is well established in the clinical circuit, overwhelming evidence of adverse, dose-dependent cardiotoxicity post-DOX therapy has been reported (9,14,22,27). Following DOX treatments, aberrations to myocardial architecture and function include cardiac cell hypertrophy and death, heightened susceptibility to myocardial infarction, cardiomyopathy, and left ventricular dysfunction (9,14,22,27). Previous studies, although limited in number, have suggested that transplanted stem cells promote salutary effects in the acute model of DIC (7,12,25,30). However, no data have been published regarding the potential lasting effects of cellular therapy in a sustained model of DIC. In the current study, I evaluated the prolonged response of the DIC-impaired myocardium to transplanted ES cells and its CM as well as identified signaling molecules that may play a role in the cytoprotection propagated by transplanted stem cells and released paracrine factors. To the best of our knowledge, this is the first investigation exploring the potential long-term cytoprotection mediated by ES cells and CM in the DIC-injured myocardium.
Apoptosis is an elemental mediator of cardiac myocyte cell death, which contributes to the pathophysiology of various diseases, including DIC (2,17, 18). Previously published data have suggested a significant increase in apoptosis at D14 in the injured myocardium consequent to DIC (30). In agreement, the diseased mouse models (D and D + CC) demonstrated significantly elevated apoptosis relative to controls implying that DOX contributes to sustainable cardiotoxicity. Notably, in the diseased groups, there is ~40% reduction in apoptotic nuclei at D28 compared with the published study at D14 (30). I suggest that this decrease in cardiac apoptosis is due to the apoptotic cells being replaced by other reparative mechanisms including hypertrophy, fibrosis, and/or myofibrillar loss. Of significant impact, cardiac cell death was significantly attenuated following CM and ES cell transplantation compared to the diseased models 4 weeks following administration. These data suggest that CM and ES cells have the potential to propagate lasting cardioprotection in DIC.
Previously identified, CM and ES cells inhibit apoptosis in acute DIC, in part, by modulating the PI3K/Akt pathway (30). PI3K/Akt activity plays a monumental role in various cellular processes, notably, cell growth, proliferation, and survival (6,23). Taking into consideration the aforementioned capacity of CM and ES cells in acute DIC, levels of Akt and up- and downstream regulators of the cell survival pathway were investigated. PTEN, an endogenous inhibitor of PI3K/Akt activation, has been shown to be dysregulated in various models of cardiac dysfunction (10,26). Consistent with these studies, PTEN expression was dramatically elevated in the D and D + CC groups. Although a trend of inhibited PTEN was observed in the D + CM group, only when transplanted with ES cells were concentrations of PTEN significantly attenuated, suggesting a mechanism by which ES cells mediate their long-term cytoprotection. Additionally, congruent with the acute DIC study, levels of Akt were significantly diminished in the diseased state groups and considerably elevated in the CM and ES cell-transplanted groups 4 weeks postadministration (30). Our data suggest that long-term inhibition of myocardial cell type apoptosis observed in the CM and ES cells groups may be attributable to the continual upregulation and activation of the Akt pathway.
mTOR, in its nonpathological capacity, amalgamates multiple signals to stimulate growth and survival, enhance cellular metabolism, and promote transcription and translation through activation of downstream targets. Multiple reports have provided evidence of mTOR dysregulation in a host of diseases, including diabetes, cancer, hypertrophy, and heart failure (15,19,35). Importantly, Zhu et al. recently reported that inhibition of normal mTOR signaling is an influential mediator of acute DOX cardiotoxicity (38). In this regard, mTOR signaling in the sustained DIC model was evaluated. The observation that significant inhibition of mTOR activity was still evident at 4 weeks post-DOX, coupled with increased cell death in the diseased models, implies an association between mTOR dysregulation and adverse chronic DOX cardiac effects. Although CM and ES cell treatment in DIC failed to significantly impact mTOR activity, a trend of increased mTOR expression was observed in these groups, which is supported by a recent study suggesting transplanted stem cells in hindlimb ischemia significantly activated mTOR and its downstream targets (4).
Characteristic cardiotoxic effects sustained in DIC include cytoplasmic vacuolization in myocardial tissue (28,29). Parallel to these studies and the acute DIC study aforementioned, significant vascular vacuolization was observed within the diseased groups (28—30). Although the degree of cytoplasmic vacuolization was diminished at 4 weeks post-DOX compared to acute DIC by mechanisms not yet investigated, the CM and ES cell-transplanted hearts demonstrated a significant reduction in cytoplasmic vacuolization compared with respective controls.
Cardiac fibrosis, a major contributor to the pathophysiology of various cardiac disorders, is a consequence of apoptosis and subsequent activation of various proteinases, including MMPs and decreased expression of their inhibitors. Within acute and chronic DIC, evidence is provided demonstrating the significant existence of left ventricular collagen deposition with the concentration of cardiac fibrosis in short- and long-term DIC (30). Importantly, cardiac fibrosis in the DOX-injured heart was significantly attenuated following CM and ES cell transplantation in the acute model of DIC, whereas blunted collagen deposition remained evident in the ES cell-transplanted group at 4 weeks post-cell therapy, suggesting that ES cells may have greater long-term therapeutic potential compared with CM (30). Concurrent with earlier findings suggesting MMP dysregulation in cardiac anomalies, MMP-9 expression was dramatically altered in the diseased animal hearts (11). Coincidental with the fibrosis data, only the ES cell-transplanted group had significant augmentation to MMP-9 levels in the DIC heart. The observations suggest that mechanisms other than factors released by ES cells are the key modulators of inhibited fibrosis in chronic DIC, but identification of such processes is beyond the scope of the current investigation.
The convoluted etiology of DIC is characterized by an accumulation of metabolic dysregulations, including the inability to restrict the formation and scavenging of free radicals resulting in OS (24). As expected, levels of superoxide radicals and LPO, variant markers of OS, were significantly enhanced in the myocardium of diseased mice, whereas following CM or cell transplantation, levels were significantly reduced. Furthermore, levels of specific antioxidants were evaluated, and our data suggests that CM and ES cells ameliorate DIC-induced OS through blunted catalase and MnSOD dysregulation.
Comparison of results between CM and ES cell groups remains a pertinent component of the present study. Previous studies have suggested that CM generated from stem cells contains a host of growth factors and cytokines, including cystatin c, osteopontin, clusterin, tissue inhibitor of metalloproteinases 1, vascular endothelial growth factor, and monocyte chemoattractant protein-1, to name a few, promoting antiapoptotic and angiogenic effects (3,32). In general, ES cells may have an advantage over CM in that enhanced cardiac repair and regeneration and improved left ventricular function observed within previous studies was attributed to not only paracrine mechanisms elicited by the transplanted cells but by activation of intrinsic cardiac progenitor cells and recellularization of indigenous tissue (5,21). Within the current data, similar results were attained in the CM and ES cell groups with regard to apoptosis inhibition, Akt activation, and free radical scavenging, suggesting a similar paracrine mechanism elicited by both treaments. Variations in the results between CM and ES cells are most evident with regard to fibrosis inhibition in which only ES cells promote significant augmentation. Although not investigated within the current study, I do hypothesize that the long-term antifibrotic potential of transplanted ES cells in chronic DIC is inferable to the compounded actions of early protection promoted during immediate DOX assault and the lasting actions of the cells through autocrine and/or paracrine mechanisms.
Of brief note, cardiac functional data are not presented within the current article. At D14, significant improvement in left ventricular output is observed within the CM and ES cell-transplanted groups following DOX administration (30). I suggest that this remains evident in chronic DIC. However, these data are presented in a follow-up article.
In conclusion, our data suggest that CM and ES cells have therapeutic long-term potential for the treatment of DIC with favorable changes purported to include 1) inhibition of cardiac myocyte cell death; 2) enhanced activation of the Akt cell survival pathway through modulation of multiple members including PTEN, Akt, and mTOR; 3) diminished cytoplasmic vacuolization; 4) augmentation to post-DIC fibrosis through decreased MMP-9 signaling; and 5) inhibited OS promoted by the preservation of the antioxidant defense system.
Footnotes
Acknowledgments
The author would like to thank Carley Glass, Binbin Yan, Reetu Singla, and Asha Ahmed for assistance with the article. This work was supported, in part, by grants from the National Institutes of Health (1R01HL090646-01, and 5R01HL094467-02 to D.K.S.). The author declares no conflicts of interest.
