Abstract
Recently, research on stem cells has been receiving an increasing amount of attention, both for its advantages and disadvantages. Genetic and epigenetic instabilities among stem cells have been a recurring obstacle to progress in regenerative medicine using stem cells. Various reports have stated that these instabilities can transform stem cells when transferred in vivo and thus have the potential to develop tumors. Previous research has shown that various extrinsic and intrinsic factors can contribute to the stability of stem cells. The extrinsic factors include growth supplements, growth factors, oxygen tension, passage technique, and cryopreservation. Controlling these factors based on previous reports may assist researchers in developing strategies for the production and clinical application of “safe” stem cells. On the other hand, the intrinsic factors can be unpredictable and uncontrollable; therefore, to ensure the successful use of stem cells in regenerative medicine, it is imperative to develop and implement appropriate strategies and technique for culturing stem cells and to confirm the genetic and epigenetic safety of these stem cells before employing them in clinical trials.
Introduction
Over the years, there have been a substantial number of studies on stem cells. The media attention associated with the “miracle” of stem cells has assisted in advancing this field of research through increased funding, thereby indirectly increasing the interest in stem cell research. Practically, however, there have been few studies that have progressed from laboratory studies to clinical trials, and gradual progress on the application of cell therapy has been recently observed.
A majority of the clinical trials have involved hematopoietic stem cells (24), with 91% of clinical trials involved in various stem cell therapeutic treatments for conditions such as multiple sclerosis, systemic sclerosis, rheumatoid arthritis, juvenile idiopathic arthritis, chronic graft versus host diseases, genetic blood diseases, type 1 diabetes (32), various histiocytic disorders, and cardiac repair. Mesenchymal stem cells (MSCs) are the second-highest category of stem cells studied in clinical trials, with 7% of studies conducted on bone and cartilage repair, graft-versus-host disease, and cardiac repair, as well as developing an aid in cure of various disorders and diseases such as neurological diseases, type 1 diabetes, liver cirrhosis, cardiomyopathy, spinal cord injury (48), multiple sclerosis (25), osteoarthritis, Parkinson's disease (112), and Crohn's disease (Fig. 1). Other types of adult stem cells, such as neural stem cells (NSCs), have also become a research focus for the treatment of lysosomal storage diseases, Parkinson's disease, and Batten's disease. Endothelial stem cells are useful options for the stimulation of angiogenesis and the treatment of atherosclerotic cardiovascular diseases. The final categories with the least number of clinical trials are the embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs). The ESC-derived oligodendrocytic progenitor cell line developed by Geron was the first ESC-derived cell to undergo clinical trials. The clinical trial has since been halted due to financial reasons. iPSCs have also progressed to the clinical trial stage with clinical trials being conducted in Japan, wherein iPSCs are being used for the regeneration of new retinal pigmented epithelium in the treatment of age-related macular degeneration (24). iPSCs have great potential in the field of regenerative medicine, and so any difficulties can hopefully be resolved after various challenges have been addressed.
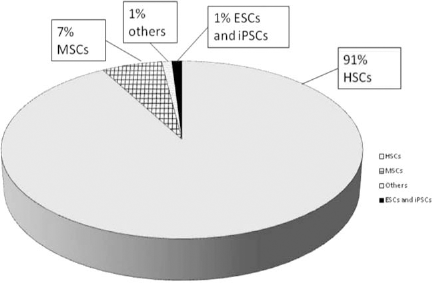
Ongoing clinical trial data for various stem cells (24). Abbreviations: HSCs, hematopoietic stem cells; MSCs, mesenchymal stem cells; ESCs, embryonic stem cells; iPSCs, induced pluripotent stem cells.
Another concern for stem cells is that the majority of these cells obtained from various sources are typically negligible in quantity; hence, it becomes a necessity to promote the long-term culturing of these cells. However, such long-term expansion can provide additional stress for stem cells and contributes to the formation of numerous genetic and epigenetic abnormalities within the cells. This article addresses whether this is an alarming issue that should concern researchers and whether it is necessary to develop appropriate protocols to ensure the genetic safety of stem cells. Furthermore, specific factors influencing the safety of stem cells are also identified and discussed.
Genetic and Epigenetic Instabilities
Embryonic Stem Cells
As ESCs possess the unique capability to differentiate into various cell types and have the capacity for self-renewal, they have great potential in regenerative medicine, thus forming the foundation for promising studies in various fields of regenerative medicine, disease models, and drug testing. However, the obvious advantages of pluripotency and plasticity are accompanied by numerous disadvantages, such as ethical concerns and genetic instability. Hayflick and Moorhead showed that all cells possess a certain limit after which they cease proliferation and enter a state of arrest; this limit is called “Hayflick's limit.” However, ESCs are one among the exceptions to this limitation because they appear to bypass senescence similar to that of transformed cells (46). Although previous research has shown that the majority of human embryonic stem cells (hESCs) are relatively stable in long-term cultures, certain studies have questioned their reliability, specifically regarding genomic purity (44). During the past few decades, numerous studies have observed genomic aberrations on chromosomes 12, 17, 20, and X (89), and a gain has been observed in either the entire chromosome 12 or parts of chromosome 12 (23,27,36,75,102) in hESCs and also similar changes in mouse ESCs (mESCs) (42,65,104). A previous study showed that, because hESCs undergo spontaneous apoptosis and differentiation at a rate that is higher than that of mESCs, it is highly difficult to maintain hESC cultures (86). Another major challenge associated with hESCs is the formation of teratoma in long-term cultures, which could be attributable to genetic changes that develop because of the cell's need to adapt to long-term expansion (44).
In vivo studies have also shown that euploid and anueploid stem cells are capable of developing into tumorous cells. Moon et al. employed red fluorescence proteintransfected animal models to elucidate the fate of abnormal hESC karyotypes. The transformed cells exhibited trisomy 12, and they developed tumors in animal models after 6 weeks of transplantation (78). Over time, there has been an increase in the number of techniques applied to identify these abnormalities. Karyotyping with G-banding has been the traditional approach to testing genomic integrity, although advancements in technology have provided additional options such as comparative genomic hybridization (CGH), fluorescent in situ hybridization (FISH), and microarray analysis, all of which improve the accuracy of information for detecting diminutive changes in the genomic integrity of cells. The never-ending identification of novel abnormalities indicates the need for further research in this field, and the development of sophisticated techniques could assist in ensuring the safety of stem cells before undergoing in vivo transplantation. Such studies have expressed caution regarding the need for conducting thorough research to validate the application of ESCs, even though other researchers have maintained ESCs in long-term cultures for more than a year (116). Thus, the increase in the unpredictability of such cells only jeopardizes innovations in cell therapy (Table 1).
Commonly Observed Abnormalities in Embryonic Stem Cells

CNV, copy number variation; hESCs, human embryonic stem cells; iPSCs, induced pluripotent stem cells; mESCs, mouse embryonic stem cells; mPSCs, mouse pluripotent stem cells.
The epigenetic instabilities are usually accompanied along with genetically abnormal karyotypes among modified hESCs, differential expressions of various genes related to differentiation and proliferation, different pluripotency markers, and suboptimal conditional requirements are observed when compared with unmodified hESCs. Numerous studies have reported that the various genes involved in the proliferative pathways are upregulated, and the genes involved in apoptosis and the genetic stability of cells are downregulated. Compared with previous findings, novel genes, such as Human Krüppel-related 3 (HKR3), lymphocyte-specific protein tyrosine kinase (LCK), JUN, and T-cell acute lymphocytic leukemia 1 (TAL1), have been shown to be involved in the maintenance of plasticity and pluripotency. The increased expression of specific genes in these cells may be the result of both increased genomic amplification and elevated transcriptional activation, thus the amplified signal (118).
Cowan et al. (27) were among the first to report a trisomy in chromosome 12, which was accompanied by an increased proliferation rate and lower population-doubling level (PDL), which was consistent with characteristics that are typical of tumorigenic cells. Draper et al. (36) conducted FISH and observed an additional gain of both chromosome 12 and trisomy 17q on three cell lines in five independent cases. The gain in chromosome 12 could be directly related to STELLAR, NANOG, and growth differentiation factor 3 (GDF-3) overexpression (located correspondingly in the chromosome 12), which are stem cell pluripotency indicators, thereby indirectly upregulating pluripotency (23,102). Chromosome 17 also possesses vital indicators of proliferation, such as baculoviral inhibitor of apoptosis protein (IAP) repeat containing 5 (BIRC5; antiapoptotic protein) (12) and Homo sapiens microRNA 21 (hsa-mir 21) mRNA (involved in cancer progression) (15). In addition, Yang et al. observed the abnormal duplication of 1p32–36, and they subsequently observed the upregulation of two oncogenes (LCK and HKR3) on the same gene, which is typically observed among transformed cells (118). Certain critical genes in the wingless-type mouse mammary tumor virus (MMTV) integration site family (Wnt) signaling pathway and survival pathways have been mapped on 1p32–36, and a direct and significant relationship was identified between the increased proliferation rate and tumorigenesis; the obvious presence of the Wnt-4 gene in the 1p region increases the gene's importance in controlling and regulating the cellular mechanisms (29).
Previous studies have shown that hESCs typically undergo apoptosis earlier than mESCs do. This early apoptosis hinders the progress of the application of these cells in cell therapy. Tumor protein 53 (p53; a tumor-suppressor gene) plays a vital role in apoptosis initiation, and it is considered a critical marker for elucidating the tumorigenesis of transformed ESCs (Fig. 2). However, numerous contradictory theories have been proposed relating to its function in transformed ESCs. Typically, p53 accumulates as a stress response that can activate apoptosis in two pathways: either through the mitochondrial pathway or the direct activation of the target genes involved in apoptosis. Qin et al. reported that p53 activates apoptosis in various types of ESCs differently (86). Under stressful conditions and DNA damage in hESCs, p53 accumulates in the cells. Although it cannot further activate the target genes, it can bind to the mitochondrial membrane and activate apoptosis. However, p53 can also control apoptosis in mESCs directly by activating the target genes, thereby indirectly also activating the feedback control for p53 gene, the mouse double minute 2 (mdm2) gene, which is absent in hESCs; hence, a higher degree of apoptosis is observable among the target genes in mESCS than among those in hESCs. Furthermore, p53 activates p21 (cyclin-dependent kinase inhibitor 1), B-cell CLL/lymphoma 2 (BCL2)-associated X protein (bax), p53 upregulated modulator of apoptosis (puma), and phorbol-12-myristate-13-acetate-induced protein 1 (noxa), but suppresses octamer-binding transcription factor 4 (oct4) and nanog (key factors in the self-renewal and differentiation of ESCs). Previous research has shown that oct4 and nanog suppression activates differentiation (86). In human gliomas, the core regulatory genes oct4, nanog, and sex-determining region Y box 2 (sox2) appear to be overexpressed, and there is a simultaneous increased proliferation rate and improved maintenance of the undifferentiated state of cells compared with normal cells (43). In abnormal ESCs, the G1 and G2 phases are shortened, causing the vital cell cycle arrest points to be missed, hence truly questioning the role of p53 in cell cycle control. In addition, the apoptotic indices were indistinguishable for both positive and negative p53 cells when treated using adriamycin (2). Similar studies (13,66,68,93) have also expressed the questionable role of p53, thus increasing the serious need for a proper quality assurance testing before the in vivo transplantation of human ESCs, and it is necessary to thoroughly understand the molecular nuances of certain cell cycle markers.

Role of p53 in transactivation of other genes and apoptosis. Abbreviations: bax, B-cell CLL/lymphoma 2 (BCL2)-associated X protein; mdm2, mouse double minute 2; noxa, phorbol-12-myristate-13-acetate-induced protein 1; oct4, octamer-binding transcription factor 4; p16, cyclindependent kinase (CDK) inhibitor 2A; p19, CDK 4 inhibitor 2D; p21, CDK inhibitor 1A; Rb, retinoblastoma; sox2, sex-determining region Y box 2.
Adult Stem Cells
Adult stem cells are highly similar to ESCs, except for the critical factor of self-renewal, and their differentiation potential is substantially lower than that of ESCs. Among all stem cell types, hematopoietic stem cells (HSCs) are the most successfully studied and exploited, and they have been the subject of the greatest number of clinical trials (24) and are capable of self-renewal and differentiation (110). MSCs are progenitor cells with the potential to differentiate into multiple lineages, such as adipocytes, osteoblasts, and chondrocytes; thus, they have been shown to have a vital role in tissue engineering using bone, cartilage, muscle, ligament, tendon, and adipose tissues (16,37,57,74). Although they were addressed as nonhematopoietic stem cells during the early 1960s, they have also been defined as colony-forming unit fibroblasts (CFU-Fs). MSCs are not immortal, although they have the capacity to expand multiple times in vitro, while simultaneously maintaining growth and multiple lineage potential. Cluster of differentiation 105 (CD105; SH2) and CD73 (SH3/4) act as standard identification markers for MSCs, whereas CD34, CD45, and CD14 act as negative markers because they are vital hematopoietic markers (18).
Bone-Marrow-Derived MSCs
In the 1970s, Till and McCulloch reported that bone marrow-derived MSCs (BM–MSCs) can be considered as a useful source of cells (105). Subsequently, they have been employed in various treatment strategies for repairing spinal cord injuries, bone and cartilage injuries, and damaged cartilage tissue. Røsland et al. reported the first observed case of spontaneous malignant transformation in hBM–MSCs (92). These cells were indicated as transformed mesenchymal stem cells (TMCs), and they exhibited characteristics that were typical of tumorigenic cells (i.e., increased proliferation rate, altered morphology and phenotype, proficient growth in soft agar assays, inability to undergo complete differentiation, and rapid growth rate). However, a recent publication from the Røsland et al. group stated that their previous findings might have been the result of cross-contamination with other cell lines (i.e., from gliomas, human fibrosarcomas, and osteosarcomas), and they emphasized the need to adopt precise techniques, such as DNA fingerprinting, to affirm the purity of the stem cell lineage, thus averting any opportunity for cross-contamination (107).
Another study on hMSCs transduced with human telomerase gene (hTERT) reported extended growth caused by the ectopic expression of the telomerase gene along with a PDL of 256 with normal karyotype; however, many multiple epigenetic changes and morphological changes were observed. The cells became anchorage independent, exhibited contact inhibition, and all 10 mice developed tumors when transferred in vivo. The transformed cells exhibited a deletion in the p16 inhibits cyclin-dependent kinase 4 (ink4a)/alternate reading frame (ARF) of CDKN2A (cyclin-dependent kinase inhibitor 2A) locus, and certain cells lost the expression of the cell cycle-associated protein deleted in bladder cancer chromosome region candidate 1 (DBCCR1), which the authors asserted to be the key point in the transformation of the normal cell line (100). Houghton et al. showed that epithelial cancers can originate from BM–MSCs, thereby proving the persistent hazard of cells transforming into tumorous cells (49).
Previous studies also reported that hMSCs are relatively stable in long-term cultures (11), although other studies have shown that murine and rhesus BM–MSCs (rhBM–MSCs) exhibit poor stability in long-term cultures (1,55,76,106). Rat BM–MSCs have also been employed as a useful source for elucidating the dynamics of stem cells. In a previous study (38), rats with acute myocardial infarction were injected with BM–MSCs, and abnormal colony development was observed within passage 3 and an abnormal karyotype was later also noted (i.e., an increase in number of chromosomes ranging from 49 to 221 was observed). But the status of the myocardial infarction in the rat models was unaltered. Similarly, Zhou et al. showed that mouse BM–MSCs also lose contact inhibition at approximately passage 3, and they observed an increase in the chromosomal number and Robertsonian translocation. When these cells were injected intramuscularly into nude and severe combined immunodeficient (scid) mice, they formed invasive carcinomas (121).
Izadpanah et al. (55) observed that both BM–MSCs and adipose-derived stem cells (ADSCs) from rhesus monkeys (rh) became tetraploid at passage 20. At passage 90, approximately 20% of the metaphase chromosome spreads were diploid 42, XY; 40% had an 84, XXYY karyotype; and the remaining 40% were aneuploid. A microarray analysis showed irregularities in both rhBM– MSCs and rhADSCs when compared to hBM–MSCs and hADSCs. Various pathways involved in cell cycle regulation, cell proliferation, and tumorigenesis appeared to be modified among various cells.
Adipose-Derived Stem Cells
ADSCs have been considered advantageous, as they can be obtained with relative ease and cause only minor discomfort to patients; thus, these cells are excellent candidates for cell therapy. Few studies have examined the genetic stability of ADSCs, the results of which have been inconclusive and controversial. Meza-Zepeda et al. employed basic G-banding karyotyping and comparative genomic hybridization to analyze the genetic stability of ADSCs in long-term cultures, and they observed that the karyotypes of ADSCs were consistently normal (72). The high-resolution microarray-based CGH analysis showed that almost all of the cells were relatively stable in long-term cultures (approximately 6 months). One cell line exhibited a minor deletion in generich telomeric and subtelomeric regions on three chromosomes in the early passages. However, this deletion was transient and spontaneously eliminated from the culture, thus, having no major contribution to the transformation of the cells.
Other studies have shown that, although ADSCs can be maintained safely in their original form during early ex vivo expansion (i.e., 6–8 weeks), the cells subsequently underwent spontaneous transformation during long-term culture (i.e., 4–5 months), and both genetic and epigenetic changes were observed. The observed chromosomal rearrangements were nonrandom and consistent, with a translocation between chromosomes 3 and 11, and the intrachromosomal rearrangement of chromosome 5, as well as the occasional presence of isochromosome 8. However, certain groups involved in the study have retracted their results because they were unable to reproduce the data, and the authors indicated that this phenomenon could be the result of cross-contamination (31,95). This uncertainty indicates the need to improve strategies that ensure the purity of stem cells before in vivo testing.
Other molecular characterization studies have asserted that the transformation of healthy ADSCs into TMCs is a complex process. Previous studies have reported that when fibroblasts from mouse embryos were cultured for extended periods, they typically underwent senescence followed by a “crisis” phase, during which many cells died (94,95). After further expansion, the cells that survived the crisis phase became immortal and, thus, tumorigenic. Current research has proven that when MSCs escape senescence, it is achieved through the upregulation of c-myc and the downregulation of p21, p53, and p16INK4A levels; thus, these are valuable markers that assist in the identification of tumorigenic cells. Furthermore, the cell crisis phase can be bypassed with increased telomerase activity, Ink4a/Arf locus deletion, and retinoblastoma (Rb) hyperphosphorylation (96).
In another study, p21 and p53 were linked directly to tumorigenesis in MSCs from adipose tissue by employing the following three MSC types: (i) p21-deficient (p21-/-) and p53 normal (p53+/+) MSCs; (ii) p21-deficient (p21-/-) and MSCs with an allele-carrying mutation in the p53 gene (p53+/-); and (iii) wild-type MSCs. When these cells were injected into immunodeficient mice, the p21-/- and p53+/- MSCs developed fibrosarcomas, whereas the p21-/-, p53+/+ group, and wild-type MSCs did not. In addition to the formation of fibrosarcomas, a substantial increase in the growth rate, unstable karyotypes, the loss of p16 expression, and a lack of senescence were observed. These findings further proved the importance of p53 in tumorigenesis; even when p21 was deficient if p53 was positive, the healthy cells can be protected from transformation unlike the allele-deficient p53 mice (91,96). Microarray analyses of the transformed cells have provided valuable information regarding the various molecular components involved in the spontaneous transformation of stem cells (Table 2).
Various Proteins and Genes Involved in the Transformation of Mesenchymal Stem Cells and Their Dynamics in the Regulation of Cell Cycle

Cdk, cyclin-dependent kinase; p16, CDK inhibitor 2A; p21, CDK inhibitor 1A; p53, tumor protein 53; Rb, retinoblastoma.
Mortality is another unexpected advantage of MSCs from various tissues; although it is a cliché, mortality reduces the probability that transformation occurs. Unlike ESCs, which are immortal, MSCs senesce after long-term expansion (PDL, 40–50), thereby reducing the probability of generating tumors, and there have been few reports of tumors occurring during early passages (77). For the successful and safe use of these MSCs in various industries, it is necessary to address certain issues. In Europe, a meeting between the Cell Products Working Party and the Committee for Advanced Therapies concluded that certain issues require attention; foremost, they emphasized that the need to increase the proliferation rate cannot be neglected, although they indicated that this should be avoided because it could only result in an increased probability of chromosomal abnormalities (7). It has generally been accepted among many researchers that MSCs are relatively more stable than other stem cells, but still care must be taken to ensure that long-term changes do not arise. CGH/single nucleotide polymorphism (SNP) arrays and FISH appear to be considered suitable high-tech options for verifying the genetic purity of stem cells, as it has been observed that the abnormality is occasionally too minuscule to be identified using karyotyping. Hence, it is advisable to confirm the stability of MSCs by using superior techniques such as CGH, microarray, or massARRAY techniques.
Other Adult Stem Cells
NSCs have been isolated from the fetal and adult brain tissues of mice, rats, and humans. NSCs possess high potentiality in numerous areas, such as drug testing and the development of various therapeutic strategies. Vukicevic et al. (111) focused on the proliferation potential and associated changes of NSCs in long-term cultures. They showed that NSCs developed sphere-forming units that correlated directly with the increased proliferation, which was indirectly aided by high-mobility group-A gene expression. This specific gene was associated with the suppression of p16Ink4a and CDK 4 inhibitor D alternate reading frame (p19ARF) gene expression, which was associated directly with an increase in proliferation. The long-term expansion also indicated the presence of specific chromosomal abnormalities; at week 16, the cells attained a gain in chromosome 1, and random structural abnormalities were also observed. The chromosome 1q abnormality appeared to be the most frequently observed abnormality among the NSCs. Furthermore, these cells also appeared to bypass senescence, which is a strong indicator of tumorigenesis. However, when transferred in vivo, they typically appeared to be incapable of developing tumors in mouse models. Sareen et al. (97) reported trisomy in chromosomes 7 and 19 to be the most prevalent abnormalities observed in human NSCs (hNSCs). However, this differs considerably from mouse NSCs, in which trisomy 13 and 14 were the most prevalent. The mechanism by which the cells from varying sources caused varying abnormalities remains unclear. Harrison discussed the findings of Varela et al. and proposed that the abnormal gain in chromosome 7 was most relevant in the case of a neural tumor that exhibited an increased amplification of genes in that particular region (9,45,109).
Induced Pluripotent Stem Cells
Pluripotent stem cells are usually obtained from a wide variety of sources, including the inner-cell mass of blastocysts, primary germ cells, parthenogenetic oocytes, mature spermatogonia, and modified adult somatic cells. iPSCs are a potential solution to the complexities associated with ESCs, although there are persistent uncertainties that must be addressed. Reprogramming processes involve applying the concept of altering the somatic cells and causing them to produce specific factors that are similar to those observed in ESCs. However, during the past 3–4 years, there has been increasing evidence of the accumulation of both genetic and epigenetic modifications among iPSCs, which could be lethal (103). Gene expression analysis using microarray has surpassed traditional karyotyping techniques, thereby assisting researchers in analyzing large samples, and improving the accuracy and efficiency of deduction on changes in genetic makeup. The most frequently observed aberrations have been classified into two types based on a general cause of occurrence, either from the culture adaptation, or inherited directly from the parent somatic cell. However, a novel hypothesis for a third type that is related to iPSCs is based on evidence that mutations arise as a direct consequence of the reprogramming procedure. The reprogramming procedure has been divided into the following two types: (i) transgenic transcriptional factors enable the transition, such that the iPSCs resemble ESCs, and (ii) transgenic factors are silenced, implying that they are transgene independent (81). The genetic abnormalities observed among iPSCs have been shown to be similar to those observed in ESCs. In a study designed to identify the contribution of various mutations, almost 32.9% of iPSCs were of trisomy chromosome 12. Trisomy 8 and trisomy 20 were also among the list of similar abnormalities with the lesser occurrence of trisomy X, although it was more common in hESCs (70). Mayshar et al. employed transcriptional genome analysis to examine approximately 66 human iPSCs (hiPSCs) and 38 ESCs, and observed that changes in the normal genetic makeup were caused by reprogramming. Furthermore, the incidence of chromosome 12 duplication increased, and the gene-rich regions correlated with the increased proliferation, thereby rendering the cells tumorigenic (71). Genome analysis using microarray has become the most frequently applied technique for elucidating the genetic complications. One such study was conducted to determine whether DNA methylation is also one of the causes of abnormalities. A microarray analysis was conducted using hiPSCs obtained from five sources (fetal lung fibroblasts, amnion, endometrium, placental artery endothelium, and menstrual blood cells), and 24,273 CpG sites were observed to be methylated in 13,728 genes.
A strategy for improving the efficacy of genetic reprogramming strategies is the simultaneous upregulation and downregulation of c-myc and p53 genes, respectively. However, Pasi et al. argued that this drastic change induces DNA replication stress, which causes genomic deletions and amplifications, thereby causing healthy cells to become tumorigenic cells (85). Similarly, another study reported high levels of genetic mutations and increased copy number variants among iPSCs, which was attributed mainly to the replicative stress induced by reprogramming (50). Sequencing strategies have also been shown to be effective in identifying various abnormalities. In a study by Gore et al., the majority of protein coding exons were sequenced for 22 hiPSCs, and each cell was reprogrammed using various strategies including integrating methods (four-factor retroviral, four-factor lentiviral, and three-factor retroviral) and nonintegrating methods (episomal vector and messenger RNA delivery into fibroblasts). Among these cells, 124 mutations were observed, 83 of which were missense mutations, five were nonsense mutations, and four were splice variants. The occurrence of at least 50% of these was premediated because these mutations already existed in the progenitor cells, albeit in a low frequency. However, the remaining mutations arose in the later stages as a result of reprogramming, thereby providing evidence of the unpredictability of reprogramming, which further emphasizes the need to develop and implement appropriate methods to verify the genetic integrity of stem cells (39).
Numerous theories have been proposed regarding the exact time and situation at which iPSCs become abnormal. One report stated that there was no correlation between reprogramming and genetic abnormalities, although other studies have shown that hiPSCs become chromosomally abnormal during either reprogramming or early culture, and this has been attributed to the stress to the cells caused by the pressure of reprogramming (39,50,71). Finally, to resolve this contradictory theory, Gore et al. (39) and Lund et al. (67) showed that specific cells acquired their mutation from the original cell source, although others acquire it during reprogramming. Despite the accumulating volume of evidence that cautions against the use of iPSCs, there is also an increasing amount of data that indicate numerous successful and safe applications of iPSCs in various therapeutic models (Table 3) (83).
Various Abnormalities Observed Among Adult Stem Cells and Induced Pluripotent Stem Cells
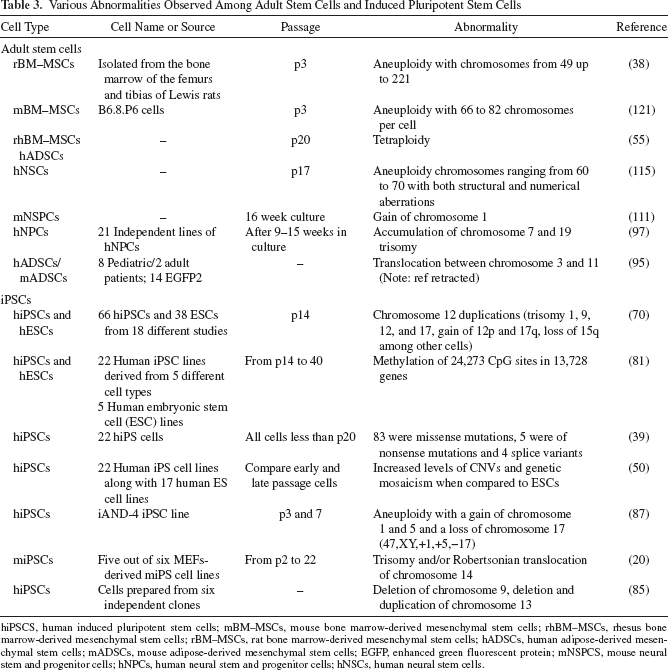
hiPSCS, human induced pluripotent stem cells; mBM–MSCs, mouse bone marrow-derived mesenchymal stem cells; rhBM–MSCs, rhesus bone marrow-derived mesenchymal stem cells; rBM–MSCs, rat bone marrow-derived mesenchymal stem cells; hADSCs, human adipose-derived mesenchymal stem cells; mADSCs, mouse adipose-derived mesenchymal stem cells; EGFP, enhanced green fluorescent protein; mNSPCS, mouse neural stem and progenitor cells; hNPCs, human neural stem and progenitor cells; hNSCs, human neural stem cells.
Various Factors Affecting Gene Stability during Long-Term Expansion
The long-term expansion of stem cells involves a considerable number of unpredictable and unknown stress signals acting on the cells. However, the known factors involved in the long-term culture can be optimized according to the requirements of a specific cell line, and this can assist in avoiding—or at least in reducing— the probability of genetic or epigenetic abnormalities. Several factors that can affect the long-term expansion of stem cells include growth supplements, growth factors, oxygen concentrations, passaging techniques, and cryopreservation.
Growth Supplements
There are many alternative growth supplements available for the successful expansion of stem cells. The most frequently employed supplements are fetal bovine serum (FBS) and fetal calf serum (FCS), and other options include platelet lysate (PL) and autologous serum (AS). FBS and FCS have many undesirable effects because of risks associated with the transmission of the pathogens or xenogenic proteins. A consequence of the potential virus and prion transmission and the immunological complexities is that various types of cells grown in FCS seem to develop complications such as the formation of malignant ventricular arrhythmias and even sudden death when transplanted into patients (69). A high concentration of FCS without cytokines typically results in a lower proliferation rate of neural progenitor cells during long-term expansion of the stem cells (34). Doucet et al. showed that when MSCs were cultured in the presence of PL obtained from platelet-rich plasma, the cells became confluent faster, and the CFU-F size increased more rapidly in comparison with FCS media; thus, PL is a valuable alternative to FCS (35).
Although hESCs that have been grown with the support of a human serum matrix appear to remain in an undifferentiated state with a well-maintained karyotype, AS appears to be the superior option. In a study on BM–MSCs, AS outperformed FBS in maintaining both long-term genetic stability and a normal gene copy number. Numerous other studies have reported various side effects or complications arising from the application of FBS-cultured hMSCs in cell-based therapies (30). However, a major complication associated with AS is its limited availability. This is especially relevant for patients who are receiving chemotherapeutic treatment, in which case the quantity of AS available is negligible (10). Serum-free media are an attractive alternative to the other supplement options, and hESCs cultured under cell- or serum-free conditions are typically maintained under a stable condition with a normal karyotype, an appropriate expression of pluripotency markers, and appropriate differentiation into all three types of germ layers (64). However, another study reported conflicting results, stating that hESCs cultured under serum-free conditions cannot maintain an undifferentiated state. In that study, a reduced proliferation rate was observed, which was attributed to the lack of the serum necessary for intracellular communication between gap junctions (114). Other studies have reported that hESCs grown in a serum-free medium [e.g., a modification of minimum essential medium (α-MEM) without cytokines or other growth factors] obtained a higher subpopulation, longer telomeres, and increased gene expression compared with hESCs grown in FCS (41,61). These conflicting opinions show the need for further research to obtain a greater understanding of the various supplements that are available. In addition, future research would benefit from type-specific data on stem cells.
Growth Factors
Growth factors are major parameters affecting stem cell pluripotency, differentiation, and maintenance. Growth factors that have been reported as being useful for stem cells are interleukin-6 (IL-6), ciliary neurotrophic factor (CNTF), basic fibroblast growth factor (bFGF), stem cell factor, fetal liver tyrosine kinase-3 ligand, thrombopoietin, oncostatin-M, IL-6 family members (69), glial cell-derived neurotrophic factor (60), transforming growth factor-β1 (TGF-β1), activin-A, hepatocyte growth factor, β nerve growth factor, retinoic acid (99), bone morphogenetic protein (BMP) (51,58,101), epidermal growth factor (EGF), platelet-derived growth factor (PDGF) (59), and leukemia inhibitory factor (LIF). IL-6 and CNTF have been shown to affect the maintenance of pluripotency in ESCs (26,82). Among all the growth factors, bFGF has been reported the most—either alone or in combination—as possessing the capacity to maintain the undifferentiated growth of ESCs and MSCs without causing changes to their genetic character, although there was a gradual inclination of these cells to progress later down the chondrogenic lineage. Certain studies have shown that serum replacement (SR) and bFGF are suitable options for serum-free conditions for achieving the prolonged maintenance of stable ESCs (3), and another study reported that LIF is useful for the self-maintenance of ESCs (113). A comparative study examined the following three groups of growth factors to elucidate their effect on stem cell maintenance: (i) TGF-b1, LIF, and bFGF (TLF); (ii) TGF-b1 and LIF without bFGF (TL); and (iii) bFGF only (F). The results of that study showed that TL and F maintained the characteristics of the stem cells, although TLF caused approximately 40% of the cells to exhibit an abnormal karyotype (47, XXX) for 20 passages (4). Numerous other growth factors, such as the stem cell factor, fetal liver tyrosine kinase-3 ligand, thrombopoietin, and LIF required the aid of bFGF for proper stem cell growth (117).
Occasionally, there is a demand for expansion with maintenance in an undifferentiated state and without the use of an animal-based serum, while maintaining genetic and epigenetic stability; a growth factor that meets such conditions would be considered an ideal growth factor (90). Previous studies have shown that each growth factor possesses a unique capacity or effect on stem cells; TGF-β and bFGF caused an increase in the proliferation rate and an inclination in the differentiation of MSCs to chondrogenic lineage (90). bFGF aided in the self-renewal of hMSCs, whereas in hESCs, it was crucial to maintain the cells in the undifferentiated state (73), and TGF family BMP caused an increase in the differentiation toward osteogenesis. The group of EGF family growth factors has also been researched extensively, and previous studies have shown that it is the most ideal group of growth factors. Previous reports have indicated that EGF receptors and vascular endothelial growth factors improve both the survival and proliferation of MSCs and also maintain the cells in an undifferentiated state (73). The application of basic Dulbecco's modified Eagle's medium with FBS has been shown to be sufficient for producing normal BM–MSC karyotypes (120), although the appropriate choice of growth factors must be selected carefully to ensure the proper maintenance of the corresponding stem cells. PL does not appear to cause any abnormalities in the presence of bFGF, PDGF, or EGF (30). Chase et al. reported that combining PDGF-BB, bFGF, and TGF-b1 (serum-free medium) was the most positive among all available options regarding both the proliferation and differentiation of MSCs; furthermore, no genetic abnormalities have been reported using this combination as a treatment strategy (19). Therefore, it is critical to select an ideal growth factor combination strategy that facilitates either a differentiated or an undifferentiated state, while increasing the proliferation rate without causing genetic abnormalities.
Oxygen Tension
Oxygen is a highly critical component of culture systems, although only minor emphasis has been given to the oxygen concentration and its physiological role in the various components of stem cell biology. Csete stated that low oxygen tension is more effective than high oxygen tension in promoting the growth of the majority of stem cells, including central nervous system-derived multipotent stem cells, (rat) fetus-derived neural crest stem cells, adult murine skeletal muscle satellite cells, (rat) marrow-derived MSCs, and CD34+ marrow progenitor populations (29).
A low oxygen concentration might not be a consistent requirement because the oxygen concentration requirements of stem cells vary according to their region or origin. For example, the majority of marrow stem cells reside in low oxygen concentration areas, which might be a major determinant for the migration of these cells toward diseased areas (6,21). However, hypoxia (<2% oxygen tension) has been observed to cause chromosomal abnormalities among various stem cell populations. A study on human bone marrow-derived stromal cells and ADSCs reported that hypoxic conditions mediated the downregulation of numerous genes involved in DNA repair and damage-response pathways [e.g., MutL Homolog 1 (MLH1), DNA repair protein RAD51, breast cancer 1 early onset (BRCA1), and X-ray repair complementing defective repair in Chinese hamster cells 5 80-kDa subunit (Ku80)], and the ADSCs responded to a low oxygen concentration much slower than BM–MSCs did. Furthermore, genomic stability and changes in gene expression profiles were observed (84). Grayson et al. cultured human MSCs at extremely low oxygen levels (as low as 2% oxygen concentrations), while simultaneously maintaining a high level of colony-forming units and expression among proliferation genes (40). In another study, BM–MSCs were cultured in a low oxygen concentration (0.5% oxygen), and an increase in reactive oxidative species, as well as the gradual transformation of healthy cells to tumorous cells were observed with modification in the levels of c-myc, p53, and hypoxia-inducible factor-1 (28).
A previous study determined the normal mean oxygen tension level within the bone marrow to be approximately 5% (79), although the oxygen concentration varies within the bone marrow regions. Previous studies on culturing hematopoietic stem cells in various oxygen concentrations have reported that the growth of cells can be improved when the oxygen tension is low (1% oxygen). Furthermore, the growth rate was maintained in the lowest progenitor cells, although increasing the oxygen tension levels resulted in differentiation into mature hematopoietic stem cells (22,53). Thus, as proposed by Csete, the optimal level of oxygen tension varies among stem cells, and researchers should standardize the test conditions based on the type of stem cell and available laboratory conditions (29). Tsai et al. stated that hypoxic oxygen influences the biology of adult stem cells in various ways (108), including inhibited senescence, increased proliferation rate, and the increased differentiation potential of cells through various lineages. However, for the long-term ex vivo expansion of ADSCs and BM-MSCs in hypoxic conditions, Oliveria et al. (84) reported that the chromosomes appeared to develop microsatellite instability, and the genes involved in DNA repair were down-regulated. A decrease in adenosine triphosphate content and the quantity of mitochondrial genomes was also observed, which contradicted the findings of previous reports. This discussion shows that hypoxia involving an oxygen concentration as low as 2% is permissible for the culturing of hMSCs, although applying lower levels can result in various abnormalities.
Passage Technique
Passaging technique also has a highly crucial role in maintaining the genetic stability of stem cells. There are three types of passaging techniques: (i) the manual method, (ii) the enzymatic method, and (iii) the nonenzymatic method. Among these techniques, the manual method has been considered the most laborious, although numerous studies have indicated that this causes the least amount of stress to the cells. Buzzard et al. (14) argued that the manual technique is robust, supporting their observation that human ESCs cultured using this technique can be maintained for up to 100 passages. The more time-efficient techniques risk causing genetic abnormalities because they exert more stress on the cells (75). The nonenzymatic method involves using an enzyme-free dissociation buffer that is anticipated to be milder on the cells than the enzymatic method, which uses collagenase/trypsin (47). Mitalipova et al. reported that hESCs survived 105 passages by using the manual method, while the ESC line BG02 developed abnormal karyotypes and trisomy 12 and 17 as early as passage 45. When the non-enzymatic and enzymatic methods were employed, the hESCs developed abnormalities at passages 25 and 2, respectively (75). Therefore, the decision to employ the safer technique assists in maintaining the safety of cells.
Cryopreservation
Cryopreservation is a vital concept in the maintenance of stem cells, and few studies have been conducted in this area of stem cell preservation. Various studies have shown that improper cell cryopreservation can cause cellular and DNA damage (33). Draper et al. stated that chromosomal abnormalities recur in human ESCs, the frequency of which appears to increase when bulk populations are passaged (36). Repeatedly freezing and thawing cells can cause serious damage to the DNA replication and the chromatin structure, which could be the result of (i) the negative effects of stress induced by the formation of ice crystals or (ii) an increase in the formation of free radicals caused by the freezing process, which could damage the DNA (63,88). The standard method for cell preservation is slow-programmed freezing in a medium containing human serum albumin and numerous other cryoprotective molecules, such as dextran and dimethyl sulfoxide (DMSO), although this could have a toxic effect on patients when transferred in vivo (98). DMSO is known to affect the epigenetic makeup of cells through histone modifications and DNA methylation. Iwatani et al. reported that DMSO is capable of causing both hyperand hypomethylation in mouse ESCs and that the choice between hyper- and hypomethylation is dependent on the genomic areas. DMSO can also cause the upregulation of DNA methyl transferases (Dnmt)-3a, and mutations in Dnmt-3b have also been observed. This particular mutation has been associated with immunodeficiency–centromeric instability–facial anomalies syndrome (54). No other study has presented a strong case against the use of DMSO, although avoiding high concentrations of DMSO and avoiding repeated freezing and thawing can assist in maintaining the stability of stem cells over long periods.
Conclusion
The majority of stem cells can transform into tumorigenic cells during long-term expansion, and relevant studies have shown that these cells are capable of developing problems such as trisomy, aneuploidy, and duplications. Epigenetically, there have been indications that the p53-Rb pathway, p16Ink4a and p21 wild-type P53-activated fragment 1/cyclin-dependent kinase inhibitor 1 (p21Waf/Cip1), c-myc, cyclin, and cdks are critical controllers of cell apoptosis/senescence and survival. Long-term expansion has been shown to induce stress on transformed cells, thereby causing cells to bypass senescence through the downregulation of p53 and p16 genes. However, it has generally been accepted among certain researchers that short-term expansion and slow growth assists in avoiding the development of tumorous cells. Along with this, the use of a low PDL for both cryopreservation and cell therapy has been considered as the ideal option because it minimizes the occurrence of anomalies. The stress induced by long-term expansion can be reduced by employing various appropriate factors. Factors that can affect the long-term expansion of stem cells include growth supplements, growth factors, the oxygen concentration, culture surface modifications, the passaging technique, and cryopreservation. Further research is necessary to elucidate the limitations of these factors because selecting an appropriate technique based on the type of cell can assist in maintaining safe cell conditions.
The discussed factors are the various possibilities that could assist researchers in the field of regenerative medicine to understand the complexities associated with the long-term expansion of stem cells. Catalina et al. (17) reported that the karyotypes of three hESC lines cultured under feeder cultures remained stable until passage 71, although they developed abnormalities and exhibited independent behavior when transferred into a feeder-free culture. In that study, the karyotypes for two of the younger cells developed abnormalities as early as passages 10 and 17, whereas the older cell line remained stable until passage 215. Accordingly, it is pertinent to question whether stem cells possess an inherent capacity to develop genetic abnormalities, and it must be acknowledged that the answer to this question is still unclear. However, for any research to be successful, pass clinical trials, reach the patients, and ultimately cure diseases, the quality of the cells is highly crucial. Thus, developing and implementing an appropriate protocol and elucidating the relevant parameters for maintaining stem cell stability are vital tasks. A standard testing protocol should be developed to confirm the genetic and epigenetic stability of stem cells before in vivo tests are conducted. The factors discussed above from multiple sources, all of which collectively indicate the need for further research to ensure the successful transition from laboratories to clinical trials and ultimately to practical patient applications. There is also a considerable need to develop appropriate safety control measures for checking and maintaining the purity of stem cells before in vivo transfer. Few studies on adult stem cells have reported genetic abnormalities in cell lines, although subsequent studies have been unable to replicate these results because of manual errors. Therefore, there is also a strong need for further intensive research to elucidate instabilities associated with adult stem cells.
Future Research
The modernization of scientific research and advancements in technology have assisted researchers by providing advanced techniques such as CGH, M-FISH, and DNA fingerprinting, as well as metagenome analysis tools such as microarray and massARRAY, all of which facilitate an improved understanding of the complexities associated with cell behavior. These advancements have enabled the accumulation of a substantially large number of data based on the genomic profiles of various stem cells. However, further research is required to facilitate the transition from the identification of the various instabilities and the application of this knowledge in establishing a standard precautionary procedure for the production of safe clinical-grade stem cells. Researchers can benefit from the available selection of various growth factors and supplements that can assist in developing customized growth conditions for stem cells. With the aid of the above-mentioned factors, there is a justifiable cause and reason to conduct extensive research that may assist in elucidating the factors involved in these cells, as well as how these cells interact with other factors in vivo. It is the responsibility of researchers working with these cells for cell therapy to ensure the safety of these cells because stem cell technology must become more approachable. Currently, it is possible to develop a protocol for the culturing of various stem cells and also for the testing of various genetic abnormalities. Finally, such a protocol must be successfully implemented in various clinical and preclinical trials. Regular testing and further extensive research can also help researchers become aware of the novel genetic and epigenetic abnormalities in various stem cells, thereby ensuring that they proceed with a high level of caution.
Footnotes
Acknowledgments
This work was supported by grants from Gwo Xi Stem Cell Applied Technology Co., Hsinchu, Taiwan, ROC (ndhu 100A060). The authors declare no conflicts of interest.
