Abstract
Autologous engineered skin substitutes (ESS) have been shown to close excised, full-thickness burns, but are consistently hypopigmented due to depletion of human melanocytes (hM) during culture of keratinocytes. Hypothetically, addition of hM to ESS may restore uniform pigmentation, but may also promote neoplasia and tumor formation. To evaluate this risk, 16 strains of hM were isolated and propagated in selective culture medium, then injected subcutaneously into athymic mice (1 χ 107 hM/animal; n = 6/strain) and followed for 24 weeks. Human melanoma cells (SK-Mel-2, SK-Mel-5) served as positive controls. No detectable tumors formed from hM strains derived from normal skin. In contrast, SK-Mel-2 formed tumors in 50% of mice, and SK-Mel-5 formed tumors in 83% of mice. Histopathology confirmed the tumorigenic anatomy of the controls and the presence of hM that were not tumorigenic in the test groups. These results support the safety of cultured hM for transplantation to restore uniform skin pigmentation in wounds closed with ESS.
Keywords
Introduction
Autologous cell therapy has become an alternative for a variety of medical conditions, including leukemia, diabetes, coronary disease, and skin wounds, such as burns and chronic wounds (2,21,24). For skin wounds, cell suspensions, partially stratified cell sheets, or combinations of cells and biopolymer scaffolds have been described that typically involve propagation of cells into large populations in selective culture (5,17,26). Among the cell types from skin that have been transplanted clinically are keratinocytes, fibroblasts, endothelial cells, and melanocytes. Rare skin cancers have been reported at sites of chronic ulceration of cultured epithelial autografts (15), but their occurrence is not known to be greater than burn scar carcinoma in split-thickness autografts (18). Another possibility is that exogenous stimulation of rapid cell division with growth factors or mitogens may promote neoplastic transformation of cells in culture before transplantation for therapeutic purposes. Each type of skin cell may become transformed, but the most aggressive and lethal form of skin cancer is melanoma (16).
Transplantation of cultured human melanocytes (hM) for restoration of skin color was reported by Lerner et al., after incubation in medium containing 12-O-tetradecanoyl-phorbol-13-acetate (TPA), a known tumor promoter (20). Other studies of hM transplantation have used suction blisters of autologous epidermis (37), uncultured epidermal suspensions (19,22,23,35), suspensions of cultured hM on membrane carriers (11,25), or cultured hM incorporated into composite skin substitutes together with other cutaneous cells (3,27,30). Most of these studies have addressed treatment of vitiligo on an elective basis. However, hypo-pigmentation has also been observed frequently after treatment of acute, full-thickness burns with grafts of cultured human epidermal keratinocytes (hK) (6,32). The deficiency of pigmentation after treatment with cultured hK results from a relative dilution of hM during selective propagation of hK. Selective propagation of hM in the absence of phorbol esters was reported by Swope et al. by activation of the ERK signaling pathway with a combination of α-melanocyte-stimulating hormone (α-MSH), endothelin-1 (ET-1), and basic fibroblast growth factor (bFGF) (29). By elimination of TPA or other tumor promoters, this medium was a crucial advance in reduction of risk in hM propagation prior to clinical transplantation.
Selective cultures of hM propagated without phorbol esters have been transplanted successfully in preclinical studies after addition into engineered skin substitutes (ESS, previously referred to as “cultured skin substitutes”) consisting of cultured human fibroblasts and hK attached to a collagen-glycosaminoglycan scaffold (8,31). Transplantation of ESS containing hM has restored uniform pigmentation at a relatively low hM density (1.0 χ 104/cm2) (30). Before translation to clinical studies, the cultured hK were subjected to tumorigenicity testing by injection into immunodeficient mice, and no palpable tumors were detected (4). In the current study, selective cultures of hM as used for inoculation of the ESS model were tested for formation of detectable tumors by subcutaneous injection into athymic mice. Satisfactory results of this testing may facilitate translation of the ESS model with hM for restoration of skin pigmentation to dermatologic conditions such as vitiligo, excised burns after grafting, or reconstructive surgery of cutaneous scars.
Materials and Methods
Cell Culture
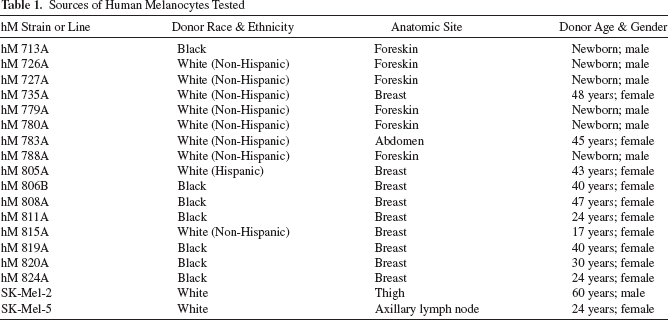
Three different sources of epidermal melanocytes included in the study are shown in Table 1: 1) primary human epidermal melanocytes (hM), which were derived from discarded tissue obtained as deidentified samples from either neonatal foreskin (n=6), reduction mammoplasty (n=9), or panniculectomy (n=1) with approval of the University of Cincinnati Institutional Review Board; 2) SK-MEL-2 cells, which were obtained from American Type Culture Collection (ATCC) and were originally derived at the Sloan-Kettering (SK) Memorial Institute from malignant melanoma of the donor's thigh; and 3) SK-MEL-5 cells, which were obtained from ATCC and were originally derived from a metastatic site at donor's axillary lymph node. SK-Mel-2 and SK-Mel-5 served as positive controls for tumorigenicity. Primary melanocytes were isolated and cultured as previously described in serum-free and phorbol-ester-free melanocyte growth medium, consisting of basal nutrient medium mMCDB 153 formulated on-site as previously reported (9), and supplemented with: 0.4% v/v bovine pituitary extract (Hammond Cell Tech, Winsor, CA, USA); 1 ng/ml bFGF (Sigma-Aldrich, St. Louis, MO, USA); 0.5 μg/ml hydrocortisone (Sigma-Aldrich); 5 μg/ml insulin (Sigma-Aldrich); 2 χ 10−11 M triiodothyronine (Sigma-Aldrich); 1 χ 10−9 M α-MSH (Sigma-Aldrich); 5 χ 10−10 M ET-1 (Sigma-Aldrich); and penicillin G, streptomycin, and Fungizone (Life Technologies, Grand Island, NY, USA) (32). Melanocytes were released selectively from primary keratinocyte cultures using 0.0025% trypsin-0.001% EDTA (w/v) (Sigma-Aldrich) for 3-1 min, were propagated in selective culture without cryopreservation, or were recovered from cryopreservation after storage in liquid nitrogen. Cryopreservation medium contained 90% fetal bovine serum (FBS; Life Technologies) and 10% v/v dimethyl sulfoxide (Sigma-Aldrich). SK-MEL-2 and SK-MEL-5 were recovered from cryopreservation and propagated in culture with Earle's Minimum Essential Medium (Life Technologies) supplemented with 10% v/v FBS as recommended by the ATCC. Stocks of SK-MEL-2 and SK-MEL-5 cells were frozen with cryopreservation medium. When preparing cells for injection, SK-MEL-2 and SK-MEL-5 were cultured in Dulbecco's modified Eagle's medium (Life Technologies) with 10% v/v FBS.
Animal Procedures
All animal care and use was performed with approvals from the Institutional Animal Care and Use and Committee of the University of Cincinnati and from the Animal Care and Use Research Office of the US Army Medical Research and Materiel Command. Athymic female mice (homozygous Foxn1nu/nu) were obtained from Harlan Laboratories and were injected subcutaneously between the scapulae with 1.0 χ 107 cells in 0.2 ml of buffered saline solution (4). Sixteen strains of hM were propagated from deidentified surgical discard skin, and two hM cell lines served as positive controls (see “Cell Culture” above and Table 1). For each cell source, six animals (n=6) were injected once. Animals were monitored weekly over a period of 24 weeks.
Sources of Human Melanocytes Tested
Tumor Measurement and Histopathology
Each week following injection, the external size of any tumor was measured with calipers (Thermo-Fischer Scientific, Waltham, MA, USA) along its shortest (width, “W”) and longest (length, “L”) axes. An estimate of tumor volume in situ was calculated from the measurements, as the volume of a cylinder with hemispherical ends, by the equation:
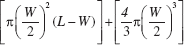
If tumors formed, final tumor volume was determined directly by fluid displacement of the resected tumor at the time of euthanasia. If one animal in a group formed tumors of approximately 1 cm in diameter, all of the animals in that group were euthanized at the same time point. Following euthanasia, tumors and/or injection sites were excised and cut into four pieces. Two pieces were fixed in 10% formalin (Fisher Scientific, Fairlawn, NJ, USA), and two were embedded in M1 medium (Richard-Allen Scientific, Kalamazoo, MI, USA) for cryomicro-tomy. One of the pieces in formalin was processed for histopathology at an independent diagnostics laboratory (ANTECH Diagnostics, Irvine, CA, USA), and the other was embedded in paraffin for routine histologic sectioning in the investigators' laboratories. Standard histology was used to identify cells with phenotypes of melanocytes or melanoma. Paraffin sections were stained with the Fontana-Masson technique (American Mastertech Scientific, Inc., Lodi, CA, USA), which enhances identification of pigmented hM. The biopsy of each injection site that was submitted to the diagnostics laboratory received an independent review by a veterinarian diplomate of the American of College of Veterinary Pathologists.
Statistical Analyses
Data were analyzed using SAS® version 9.4. Tumor frequencies were compared by chi-square analysis for overall differences, followed by Fisher's exact test for pairwise differences. A comparison of tumor progression in the cultured hM, SK-Mel-2, and SK-Mel-5 was performed by a repeated-measures analysis of variance (ANOVA) followed by least squares means comparisons. Tumor volumes at euthanasia were evaluated among experimental and control groups by analysis of variance and followed by Tukey's studentized range test. Statistical significance was accepted at p < 0.05. Sample sizes of tumor frequency for the experimental group in comparison to the reference general population were estimated by a power analysis using Fisher's exact conditional test for two proportions and performed for powers between 0.80 and 0.99 at a significance level of 0.05.
Results
Figure 1 shows the clinical presentation of animals after injection with hM from lightly pigmented donors (Fig. 1A), darkly pigmented donors (Fig. 1B), cultured melanoma line SK-Mel-2 (Fig. 1C), or SK-Mel-5 (Fig. 1D). No tumors or pigment were visible externally in mice injected with primary hM from lightly pigmented donors. Pigment without tumors was visible at the skin surface in athymic mice injected with primary hM from darkly pigmented donors. Palpable tumors were readily detected after injection of SK-Mel lines that were derived from clinical melanomas.

Photographs of athymic mice injected with cultured hM. (A) Melanocytes from a white donor were not detectable (dashed circle) on macroscopic examination. (B) Injected hM from a black donor generated a visible area of pigment beneath the dermis by 12 weeks (dotted line) but no palpable tumors. (C) SK-Mel-2 human melanoma generated palpable tumors in 17 weeks. (D) SK-Mel-5 human melanoma cells generated palpable tumors in 6 weeks. Scale in cm.
Histological analyses revealed the presence of pigmented (Fontana-Masson positive) melanocytes in mice injected with hM from normal skin (Fig. 2A, B). Pathology reports from injection sites of cultured hM described “well-differentiated” melanocytes as represented by the expression of pigment. Cultured hM from lightly pigmented donors were stained well in the subcutaneous tissue (Fig. 2A). Darkly stained melanin in hM from dark-skinned donors was also observed in the subcutaneous tissue after labeling with Fontana-Masson silver stain (Fig. 2B). Nodular tumors were observed after injection of either SK-Mel-2 (Fig. 2C) or SK-Mel-5 (Fig. 2D) beneath the overlying murine skin.
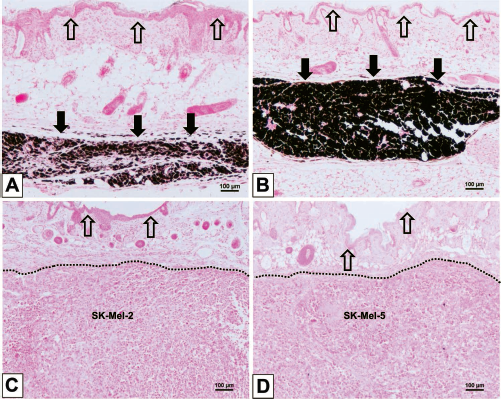
Photomicrographs of subcutaneous injection sites. Fontana-Masson staining of pigmented hM located beneath the epidermis in the deep dermis, adjacent to the panniculus carnosus. Injected cultured hM from either lightly pigmented donors (A) or darkly pigmented donors (B) expressed pigment and aggregated (solid arrows) but did not form tumors. Injection of SK-Mel-2 (C) or SK-Mel-5 (D) formed palpable tumors within 17 or 6 weeks, respectively. The epidermal surfaces are indicated with hollow arrows, and the tumor masses for SK-Mel-2 (C) or SK-Mel-5 (D) are located beneath the dotted black line as indicated. Scale bar: 100 μm.
Quantitative analysis of progression of tumor volumes over time and final tumor volumes were determined (Fig. 3). Figure 3A shows that no palpable tumors were observed after injection of cultured hM (0/96) during 24 weeks of observation. Injection of SK-Mel-2 generated tumors in 50% (3/6) of animals after 17 weeks, and SK-Mel-5 produced tumors in 83% (5/6) of animals after 6 weeks. Tumor frequencies of either SK-Mel-2 or SK-Mel-5 were significantly different from hM strains. Furthermore, the rate of tumor progression was markedly faster in SK-Mel-5 than SK-Mel-2. Repeated-measures ANOVA and pairwise comparisons by differences of least squares means showed statistical differences (p < 0.05) between the cultured hM and each of the SK-Mel lines. Determination of mean tumor volume by fluid displacement after euthanasia and resection is shown in Figure 3B. No tumors were found at hM injection sites. Mean tumor volumes (±SEM) were 0.47 ± 0.39 ml for SK-Mel-2 at 17 weeks after injection, and 1.18 ± 0.39 ml for SK-Mel-5 at 6 weeks after injection. ANOVA followed by pairwise comparisons using Tukey's Test found that tumor volumes for SK-Mel-2 and SK-Mel-5 at the time of euthanasia (17 weeks and 6 weeks, respectively) were statistically greater (p <0.05) than cultured hM cell strains (tumor volume=0) at the same time points.

Plots of calculated tumor volume and tumor displacement volume. (A) Tumor volumes in situ showed no detectable tumor formation (0/96) after injection of primary cultured hM; rapidly growing tumors (3/6) after injection of cultured melanocytes from the clinical tumor SK-Mel-2, and aggressive tumors (5/6) after injection of SK-Mel-5. Chi-square analysis showed tumor frequencies of cultured hM to be statistically different from both of the SK-Mel groups. Repeated-measures ANOVA and pairwise comparisons by differences of least squares means showed differences in calculated tumor volume (p <0.05) between the cultured hM and each of the SK-Mel lines. (B) Tumor volume at the time of euthanasia was determined by fluid displacement of resected tumors. SK-Mel-2 tumors had a mean volume of 0.47 ml, and SK-Mel-5 tumors had a mean volume of 1.18 ml. Cultured hM were statistically different (p <0.05) from both SK-Mel groups by ANOVA followed by pairwise comparisons using Tukey's test.
Discussion
The results of this study indicate that culture of hM for a limited number of population doublings, in medium that does not contain TPA or other tumor promoters, does not induce tumorigenicity in athymic mice after 24 weeks of incubation. These results support the safety of cultured hM for autologous transplantation for wound repair and correction of hypopigmented cutaneous lesions. These findings are consistent with observations that no neoplastic lesions have been reported after clinical grafting of autologous engineered skin substitutes to more than 100 human subjects over 15 years of study (6). The safety of transplanted hM is also supported by grafting of ESS containing melanocytes in numerous studies showing partial or complete skin pigmentation but without observation of any melanocytic lesions (8,30-32).
Important factors in the current study that are considered to contribute to the safety of hM transplantation are the formulation of the nutrient medium, the conditions for isolation and propagation of hM, and the limited number of population doublings prior to transplantation. Primary cultures of melanocytes used for transplantation have been incubated in media containing phorbol esters without reports of mutation or neoplastic transformation after short-term follow-up (3,20,27), but this is considered to be an unnecessary and preventable risk if those compounds may be eliminated as described here. Another nonphysiologic mitogen that has been used in melanocyte culture is 3-isobutyl-1-methylxanthine (1,12), which inhibits phosphodiesterases and increases levels of cAMP and proliferation. Media formulations without these kinds of nonphysiologic compounds may reduce risks by activating mitogenic and melanogenic pathways through normal signaling mechanisms (13,29). In addition, under conditions described here, hM were propagated to approximately 15-20 population doublings to generate sufficient numbers of cells for inoculation of ESS at 1 χ 104 hM/cm2 and to generate as much as 100 times the area of ESS compared to the area of the skin biopsy (6,7). This factor of cellular expansion provides confidence that sufficient hM will be available to address patients' needs for extensive, emergent indications such as full-thickness burns. Furthermore, after addition of the cultured hM to ESS grafts, hM have been shown to develop cell-cell contacts that promote pigment transfer to their surrounding population of keratinocytes (8). Importantly, the keratinocytes and fibroblasts are known to synthesize components of the dermal-epidermal junction, including basement membrane proteins, such as collagens IV and VII, and laminins 1 and 5 (9). These extracellular matrix proteins provide the natural substrates for binding of hM in vivo through integrin receptors (14,32), serve to regulate proliferation, and promote normal pigment production and transfer to hK (10). It is likely that these natural regulatory signals promote the normalization of hM physiology. Indeed, after transplantation in ESS, the expression of pigment and the radial outgrowth of hM is confirmed by the slow progression of skin pigment from several initial foci to coalescence with other progressing pigment foci resulting in uniformly pigmented skin (8,32). Together, these procedures are believed to minimize the sources and durations of exposure to factors that may contribute to mutation or neoplastic transformation.
Although these results suggest safety of hM transplantation after addition to ESS, a limitation of the current study is that postmortem biopsies were performed only at the injection sites. In addition, no genetic analyses of cultured hM were performed to investigate whether any detectable level of mutations occurred in the cellular genomes or if any oncogenes were activated. Rather, the extended observation time (24 weeks), the number and variety of donor strains (16), the relatively large number of cells per injection site (1e7), and the number of animal subjects per cell strain (n=6) raise the level of confidence that the frequency of possible mutagenic or neoplastic events was sufficiently low as to be undetectable in the experimental population. However, the absence of tumor detection in this study sample (0/96), does not represent accurately the frequencies of melanoma in the general population, which are approximately 1 in 5,000 individuals (28,33,34,36). A statistical power analysis predicted that conclusive data would require an experimental population of greater than 1 χ 107 individuals to show statistically significant differences at the α=0.05 level and powers ranging from 80% to 99%. Such an analysis is not logistically feasible. Notwithstanding this qualification, the absence of tumorigenicity in the experimental population reported here is consistent with a transformation rate that is no greater than the rate found in the general population. Furthermore, to the authors' knowledge, this is the only tumorigenicity study of primary cultured hM to date. Therefore, no data were found that contradict the safety of transplantation of melanocytes after culture in media from which TPA or similar tumor promoters have been eliminated.
Histopathological analyses suggested that following subcutaneous injection, the integration of the hM into the subcutaneous tissue occurred without elicitation of a foreign body reaction by host macrophages or formation of a fibrous capsule. These are additional anatomic characters that are consistent with behavior of nontrans-formed cells. In contrast, control hM derived from clinical melanomas formed tumors within a few weeks and expressed no pigment, which indicates dedifferentiation and unregulated growth.
Conclusion
Collectively, these data indicate that conditions for isolation, culture, and transplantation of autologous hM in ESS present no more than minimal risks to prospective patients. Benefits to recipients are anticipated to include increased protection from solar exposure, and restoration of uniform skin color to improve personal appearance, and to promote a positive self-image.
Footnotes
Acknowledgment
The authors thank Mary Rolfes, ASCP, for histological expertise, and Laura James, MS, for assistance with statistical analyses. This study was supported by a subcontract to the University of Cincinnati from Rutgers-the State University of New Jersey, the prime contractor of the Rutgers-Cleveland Clinic Consortium of the Armed Forces Institute for Regenerative Medicine, DoD contract # W81XWH-08-2-0034. The authors declare no conflicts of interest.
