Abstract
Stroke is one of the disorders for which clinically effective therapeutic modalities are most needed, and numerous ways have been explored to attempt to investigate their feasibilities. However, ischemic- or hemorrhagicinduced inflammatory neuron death causes irreversible injuries and infarction regions, and there are currently no truly effective drugs available as therapy. It is therefore urgent to be able to provide a fundamental treatment method to regenerate neuronal brain cells, and therefore, the use of stem cells for curing chronic stroke could be a major breakthrough development. In this review, we describe the features and classification of stroke and focus on the benefits of adipose tissue-derived stem cells and their applications in stroke animal models. The results show that cell-based therapies have resulted in significant improvements in neuronal behaviors and functions through different molecular mechanisms, and no safety problems have so far arisen after transplantation. Further, we propose a clinical possibility to create a homing niche by reducing the degree of invasive intracerebroventricular transplantation and combining it with continuous intravenous administration to achieve a complete cure.
Keywords
Introduction
On the basis of World Health Organization statistics, cerebrovascular disease is second only to cancer diseases in the top 10 leading causes of death in developing or developed countries (43); thus, cerebrovascular disease is another intractable disease. The pathogenesis of stroke, an extremely common form of cerebrovascular disease, involves the disruption of the blood circulation, which causes injury and hampers the brain's metabolism, eventually leading to cell death and impaired brain function (13,47). In clinical diagnosis, the majority of patients will gradually recover some of the dysfunctions caused by the acute phase of the disorder, but the irreversible injuries to the brain cause partial loss of nerve and brain function (22). Therefore recovery from a stroke often involves chronic symptoms, together with a loss of independent capacity and paralysis, resulting in a burden on both the family and society.
Stroke arises mainly from the blockage of blood vessels in different parts of the brain, resulting in different degrees of loss of nerve function with sudden hemiplegia, aphasia, and numbness in hands and feet (60,62). The current treatment for the acute phase is to impose anticoagulant, antiplatelet aggregation agents, or thrombolytic agents (12). One of the most commonly used is recombinant tissue plasminogen activator (rt-PA) that is used to breakdown blood clots (23,51). However, only 9% of patients are alive within 3 h of the onset in acute stroke, and the benefits of this treatment are not significant over that point in time (71,72). The ischemic or hemorrhagic inflammatory neuronal death causes irreversible damage and an infarct site leading to a chronic stroke. The applicability of stem cells has been widely investigated, and the medical community has begun to develop regenerative therapy for chronic stroke (6). Preliminary experimental results indicate that treated patients have significant progress in restoration of neurobehavioral symptoms, as well as no abnormal cell proliferation during the postoperative follow-up period (34,35).
In recent years, clinicians expect that autologous stem cells could be used to cure chronic stroke, if they could prevent carcinogenicity, immune rejection, and overcome ethical controversies (13,14). One proposed method is the use of granulocyte colony-stimulating factor (G-CSF) to stimulate the proliferation of hematopoietic stem cells and their migration from the bone marrow to the blood, from where they can be separated in a noninvasive manner via withdrawing blood (62). The results showed that autologous stem cells transplanted into the brain are safe and have beneficial effects in chronic stroke. Nevertheless, the source of hematopoietic stem cells is inadequate, and thus a search for an alternative source of autologous tissues for preparing a large number of stem cells is underway (29). According to the literature, adequate quantities of adipose tissue-derived stem cells (ADSCs) can be separated from fat tissue with minimal invasive procedures (24,77). In addition, we focus on the efficacy of ADSC treatment for chronic stroke and explore their potential mechanisms and effectiveness, as well as discuss future expected human clinical trials.
The Application of ADSCs
ADSCs are mesenchymal stem cells (MSCs) that can be isolated from a wide variety of sources by minimally invasive procedures, such as from abdominal subcutaneous, intra-articular, and visceral depots, as well as ectopic adipose tissues (21). The characteristics of ADSCs mean that it is easy to produce large numbers of multipotent stem cells from a small amount of tissue as well as stabilized proliferation without a loss of stemness in vitro, two essential requirements for regenerative medicine and tissue engineering (2,15). It has been shown that ADSCs express cell surface markers such as cluster of differentiation 29 (CD29), CD44, CD73, CD90, CD105, and human leukocyte antigen (HLA) class I (76). Under defined conditions, ADSCs can differentiate into various tissue-specific lineages, including adipocytes, osteoblast, hepatocytes, chondrocytes, and myocytes, as well as epithelial and neuronal cells (29,66).
For the implementation of clinical trials, the safety issues of ADSCs have been of widespread concern in extensive investigations of its therapeutic capacity for various diseases (54). Karyotype analysis of ADSCs for chromosomal deletion, translocation, duplication, inversion, and insertion showed that no chromosomal mutations occur after 12 in vitro subcultures (53). Expression of the tumor protein 53 (p53) gene was found to be the same between long-term cultured ADSCs (20 passages; P20) and normal cells (57). Telomerase activity could not be detected over 20 in vitro generations (57). The most direct evidence is that subcutaneous transplantation into nude mice of different quantities of ADSCs resulted in no abnormal proliferation after several months of follow-up (58). Moreover, ADSCs have been applied in autoimmune disease, multiple sclerosis, polymyositis, dermatomyositis, and rheumatoid arthritis; the results indicate that treated patients demonstrate no adverse side effects (53,67). Based on their safety and suitability for the theoretical basis of autologous transplantation, ADSCs have become one of the most popular items in MSC therapy research and is a potential future regenerative medicine for the clinic.
Classification of Stroke
Stroke can be classified as either ischemic or hemorrhagic based on the type of blood injury in the subventricular zone (SVZ) (28,36). Ischemic stroke arises from an endovascular occlusion leading to loss of normal blood supply, which causes immediate damage and a large number of dead nerve cells (55). Ischemic strokes account for 85% of all stroke patients and can be divided into cerebral embolism and thrombosis (1). Embolisms can arise due to rupture of atherosclerosis plaques, and the high-risk group includes three kinds of patients with a high incidence of hypertension, high cholesterol, and/or high blood sugar (40). Hemorrhagic stroke is commonly known as hyperemia and accounts for 15% of all stroke patients; however, there is a higher degree of risk than ischemic stroke (65). Vascular brain lesions, congenital (aneurysm) and acquired (hypertension), lead to changes in blood vessel fragility as well as an increased incidence of vascular rupture (19,73).
The ischemic penumbral region promotes the release of glutamate, an excitatory neurotransmitter in the central nervous system, which then binds and activates N-methyl-d-aspartate (NMDA) receptors (44,45). This causes large amounts of sodium, calcium, and water to flow into the nerve cells, which activate endogenous lysis enzymes to digest the cell components (69,74). The unbalanced flow of calcium ions also causes mitochondrial dysfunction leading to activation of proteins associated with the death of nerve cells (46,64). Ischemia-induced free radicals and other reactive oxygen species also can induce the apoptosis signals within nerve cells (48,64).
Neurological deficits of vascular origin that occur within a few minutes to hours are defined as acute stroke, whereas those that occur over an extensive duration of time are known as chronic stroke (68). Although acute stroke patients can be treated in a hospital with thrombolysis, it can cause irreversible damages to the injured brain (27). Acute therapy usually only partially restores the wounded area, and so the injury will evolve into the main problems of chronic stroke. Therefore, clinical treatments of chronic stroke are urgently required to restore the original nerve cells and their functions in the brain niche.
ADSCs in Chronic Stroke
For treatments in various neurodegenerative diseases, there are many cells or tissues that have been transplanted into the human brain, such as aborted fetal tissue containing stem cells (4), ganglion lineage cells (32), umbilical cord blood monocytes (79), and CD34+ hematopoietic stem cells (61). In one case, neuron-committed teratocarcinoma (NT2) cells were transplanted into adult stroke rats, and they not only restored stroke symptoms but also prolonged the survival time (59). These results confirm that stem cells have the potential to be an effective therapy in brain diseases and suggest that they are safe. However, stem cells used for the treatment of stroke that are not derived from the patients themselves may need to go through the HLA-matching procedures to avoid immune rejection after transplantation (49,61,78). Therefore, it is a valuable objective to determine whether the administration of autologous ADSCs into the injured penumbra results in functional regeneration as a clinical cell-based therapy for chronic stroke (11,24). In fact, most of the available reports confirm the successful administration of ADSCs into injured brains, involving both intracerebroventricular (ICV) and intravenous (IV) transplantation for the treatment of chronic stroke (Table 1) (24). Even though the mechanisms of effective therapy are not well known, it is important to classify current evidences in a methodical manner.
ADSC Application in Chronic Stroke
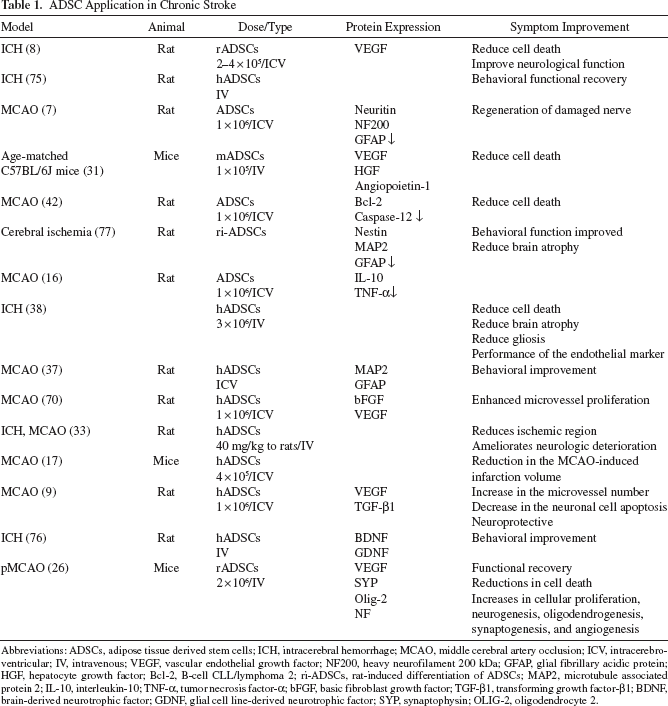
Abbreviations: ADSCs, adipose tissue derived stem cells; ICH, intracerebral hemorrhage; MCAO, middle cerebral artery occlusion; ICV, intracerebroventricular; IV, intravenous; VEGF, vascular endothelial growth factor; NF200, heavy neurofilament 200 kDa; GFAP, glial fibrillary acidic protein; HGF, hepatocyte growth factor; Bcl-2, B-cell CLL/lymphoma 2; ri-ADSCs, rat-induced differentiation of ADSCs; MAP2, microtubule associated protein 2; IL-10, interleukin-10; TNF-α, tumor necrosis factor-α; bFGF, basic fibroblast growth factor; TGF-β1, transforming growth factor-β1; BDNF, brain-derived neurotrophic factor; GDNF, glial cell line-derived neurotrophic factor; SYP, synaptophysin; OLIG-2, oligodendrocyte 2.
Angiogenesis
When hemorrhagic or ischemic stroke transforms into the chronic type of stroke, the injured niche is devoid of blood vessels to supply nutrients for maintaining cell survival; thus, vascular endothelial growth factor (VEGF) might be a possible mechanism to protect neurons from cell death (18). In a rat model of hemorrhagic stroke, immunohistochemical (IHC) staining indicated that the proportion of VEGF in the group treated with ADSCs was higher, suggesting that the secreted VEGF may repair brain damage by promoting angiogenesis (8). Reports also support the suggestion that ADSCs could restore the function of the brain through the secretion of VEGF to promote angiogenesis in the injured region (8,26,31).
Neurogenesis
Administration of ADSCs could also induce neuronal differentiation, as well as stimulate brain repair markers associated with neurogenesis to cause functional recovery of chronic stroke (8,24,56). It has been shown that the contralateral brain has a small number of newly generated neuron cells, due to a capability of differentiation with injected ADSCs. IHC staining showed that ADSCs differentiate into neuron-like or glia-like cells expressing neuronal nuclei (NeuN), nestin, and glial fibrillary acidic protein (GFAP) (26,52), which are related with the recovery of brain function (33). Moreover, synaptic integration with nerve regeneration is required for axonal plasticity and remodeling to occur in brain repair, and this implies that the newly derived cells transform into a functionally integrated neuron (25).
Gliosis
However, a glial scar is formed in the infarct boundary, when there is excess proliferation or hypertrophy of glial cells, such as astrocytes, microglia, and oligodendrocytes (3,10). The use of ADSCs for stroke treatment may also reduce the generation of GFAP+ cells, thereby inhibiting brain nerve fibrosis, which could be another effective treatment mechanism (77). Several studies also indicate that ADSC administration reduces reactive gliosis in response of enhanced neurological outcome, as well as reducing the size of the infarcted region (24,30,37,38).
Apoptosis
Moreover, neuronal recovery is accompanied with a reduction in brain injury-derived apoptosis as well as activation of the natural repair response following brain injury (24). IV administration of ADSCs showed that the apoptotic marker caspase-3 was much lower than in the control group, suggesting that stem cell treatment may inhibit apoptosis to achieve a therapeutic effect (77).
Neurotrophic Factors
In addition to reducing apoptosis signals, ADSCs also increase neuron cell survival and/or differentiation through neurotrophic factors that may play an important role in the maintenance of neural tissue (39,50). A recent report showed that ADSCs can express and secrete several neurotrophic factors and improved functional recovery in middle cerebral artery occlusion (MCAO)-treated rats (37). Administration of ADSCs is associated with an improvement in stroke deficits by increasing the presence of neurotrophic factors such as brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF), and glial cell line-derived neurotrophic factor (GDNF) (30,52).
Immune Inflammatory Response
Another possible mechanism for a protective role of ADSCs is immunomodulatory since increased interleukin (IL)-10 and decreased tumor necrosis factor (TNF)-α was observed in the MCAO-treated rat (16). ADSC therapy attenuates the inflammatory response via reducing the levels of IL-18, toll-like receptor 4 (TLR-4), and plasminogen activator inhibitor-1 (PAI-1), which modulates immune reactivity and enhances neurological outcome (41). In summary, these results indicate that neuronal behaviors and functions can be significantly improved through different mechanisms after administration of ADSCs in rodent stroke models.
Future Expectation
The main purpose of regenerative medicine studies in chronic stroke is to find out the most effective and safe treatments, which improve functional outcome after brain injury. The neurological recovery of ICV administration of ADSCs is greater than that by the IV route (52); however, invasive delivery might cause intracerebral trauma and surgical complications. In addition, chronic stroke does not express inflammatory response factors (such as IL-1, TNF-α, etc.), or the homing factors [such as stromal cell-derived factor-1 (SDF-1), chemokine C-X-C motif receptor 4 (CXCR-4), BDNF, and GDNF, etc.] to assist in the prognosis of stroke (20,63). According to the literature and our own investigations, ADSCs can secrete homing factors themselves while transplanted into the brain niche (5,20). ICV-transplanted ADSCs could therefore form a concentration gradient of homing factors to attract additional stem cells administered intravenously. Continuous IV injection of ADSCs after a minimal ICV transplant may therefore allow one to complete therapy with minimal invasive manipulation. Thus, it is therefore an approach that could be used clinically for the treatment of chronic stroke, combined therapy of ICV and IV administrations.
Footnotes
Acknowledgments
This study is supported in part by Taiwan Department of Health Clinical Trial and Research Center of Excellence (DOH102-TD-B-111-004). The authors declare no conflicts of interest.
