Abstract
Dendritic cells (DCs) are the major specialized antigen-presenting cells for the development of optimal T-cell immunity. DCs can be used as pharmacological targets to monitor novel biological modifiers for the cure of harmful immune responses, such as transplantation rejection. Dryopteris crassirhizoma Nakai (Aspiadaceae) is used for traditional herbal medicine in the region of East Asia. The root of this fern plant has been listed for treating inflammatory diseases. Dryocrassin is the tetrameric phlorophenone component derived from Dryopteris. Here we tested the immunomodulatory potential of dryocrassin on lipopolysaccharide (LPS)-stimulated activation of mouse bone marrow-derived DCs in vitro and in skin allograft transplantation in vivo. Results demonstrated that dryocrassin reduced the emission of tumor necrosis factor-α, interleukin-6, and interleukin-12p70 by LPS-stimulated DCs. The expression of LPS-induced major histocompatibility complex class II, CD40, and CD86 on DCs was also blocked by dryocrassin. Moreover, LPS-stimulated DC-elicited allogeneic T-cell proliferation was alleviated by dryocrassin. In addition, dryocrassin inhibited LPS-induced activation of IkB kinase, JNK/p38 mitogen-activated protein kinase, and the translocation of NF-κB. Treatment with dryocrassin noticeably diminished 2,4-dinitro-1-fluorobenzene-reduced delayed-type hypersensitivity and extended skin allograft survival. Dryocrassin may be one of the potent immunosuppressive agents for transplant rejection via the destruction of DC maturation and function.
Keywords
Introduction
Dryopteris crassirhizoma Nakai (Aspiadaceae) is used for traditional herbal medicine in the region of East Asia. The root of this fern plant is an element of the Yupingfeng Asia soup formulation derivative and has been listed for treating various viral (48), infectious (3,22), and inflammatory diseases (49). Dryocrassin (Fig. 1) is the tetrameric phlorophenone component derived from Dryopteris crassirhizoma (17).

Chemical structure of dryocrassin.
Dendritic cells (DCs) are highly potent professional antigen-presenting cells, acting as crucial messengers for communication between innate and adaptive immunity (15). Up to now, DCs have been used for treating cancerous tumors and infectious diseases (4). Moreover, DCs play a key role in immune modulation, including induction of tolerance and prevention of autoimmunity (25,30). DCs have two functional types. Immature DCs are generated from hematopoietic bone marrow progenitor cells and extend in blood or tissues in contact with the outside environment. These cells exhibit high endo-cytic competence and low T-cell activation potential. Following antigen capture, DCs process selected foreign peptides, transport them to the surface, and transform into mature DCs. They then reduce their capacity for antigen capture and move to the peripheral lymph organs where they activate T-cells. In the innate immunity, DCs can use pathogen-associated molecular patterns—regular constituents of microbes—to sense pathogenic components. Lipopolysaccharide (LPS), which is located in the outer layer of the outer membrane of gram-negative bacteria, is one pathogen-associated molecular pattern and has been reported as a key cause of septic shock (32). LPS interacts with circulating LPS-binding protein and cluster of differentiation 14 (CD14) and then specially binds to tolllike receptor 4 (TLR4) on DCs. After TLR4 activation, DCs undergo the process of maturation, including release of proinflammatory cytokines [e.g., tumor necrosis factor (TNF)-α, interleukin (IL)-6, and IL-12], augmentation of the appearance of surface effector molecules [e.g., major histocompatibility complex (MHC) class II, CD40, CD80, and CD86], reduction of endocytotic/phagocytic capability, and upregulation of the expression of chemokine (C-C motif) receptor type 7 (CCR7), which increases their antigen-presenting competence and migration to secondary lymphoid organs and then triggers specific T-cell immunity (12,35).
Several signal transduction cascades are required to engage TLR4 activities, including the inhibitor of κ B (IkB) kinase (IKK)/nuclear factor κ-light-chain-enhancer of activated B-cell (NF-κB) pathway (11,42); three mitogenactivated protein kinase (MAPK) pathways (6,26), c-Jun N-terminal kinase (JNK), extracellular signal-regulated kinase (ERK), and p38; and the phosphatidylinositol 3-kinase (PI3K)/Akt pathway (1,27), all of which upgrade the expression of various genes connected to DC maturation (16,24).
Prevention of allograft rejection without long-term immunosuppression is important in the field of transplantation. DCs are primary immunomodulators, and so controlling their maturation may be a functional approach for treating transplant rejection. Many data showed that immature DCs may be useful for the establishment of antigen-specific peripheral tolerance in the absence of costimulatory signals. The employment of immature DCs in a cell-based treatment to induce immune tolerance following tissue or organ transplantation has been an active field of study, and the transfer of immature DCs has been disclosed to work synergistically with pharmacotherapy to support long-term allograft survival (21,23,36,39).
To date, no report has examined the immunomodulatory characteristics of dryocrassin in DCs. In the current research, we assessed whether dryocrassin could affect the phenotypic and functional maturation of DCs by using a model of LPS-stimulated mouse bone marrow-derived DCs (mBM-DCs). We investigated the linked signal pathways and examined the in vivo effects of dryocrassin with a skin transplantation model.
Materials and Methods
Chemicals and Antibodies
Synthesized dryocrassin (mol. wt. 820.83, 98% purity) was purchased from Fusol Material Co., Ltd. (Tainan, Taiwan), dissolved in dimethyl sulfoxide (DMSO; Sigma-Aldrich, St. Louis, MO, USA) to 100 mM, and stored at −20°C as a master stock solution. Roswell Park Memorial Institute (RPMI) 1640 l-glutamine medium, fetal bovine serum (FBS), 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid (HEPES; pH 7.4), sodium pyruvate, penicillin–streptomycin, nonessential amino acids, and β-mercaptoethanol were all purchased from Invitrogen (Grand Island, NY, USA). Recombinant mouse granulocyte-macrophage colony-stimulating factor (rmGM-CSF), recombinant mouse IL-4 (rm-IL4), and secondary lymphoid-tissue chemokine/chemokine (C-C motif) ligand 21 (SLC/CCL21) were purchased from Prospec (Ness-Ziona, Israel). LPS (from Escherichia coli 055:B5), fluorescein isothiocyanate (FITC)-dextran (42 kDa), mitomycin C, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), 2.4-dinitro-1-fluorobenzene (DNFB), and all other chemicals were purchased from Sigma-Aldrich unless specified otherwise. Phycoerythrin (PE)-conjugated antibody to mouse CD11c and FITC-conjugated antibody to mouse MHC class II, CD40, CD86, and isotypematched control antibodies were purchased from GenWay Biotech (San Diego, CA, USA). The antibodies for IKKa/b, IkBa, JNK, ERK1/2, p38, Akt, and their phosphorylated forms (p) were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). The antibody for mouse β-actin was purchased from Millipore (Billerica, MA, USA).
Generation of mBM-DCs
This research was performed in strict accordance with the recommendations in the Guide for the Care and Use of Institutional Animals of China Medical University and the Care and Use of Laboratory Animals of the National Institutes of Health. All animal experiments were conducted according to a protocol approved by the Institutional Animal Care and Use Committee of the China Medical University (Permit Number: 101-32-N). Four male C57BL/6 mice (8 weeks old, 20 g, from the National Laboratory Animal Center, Taiwan) were maintained in a specific pathogen-free area at the Animal Center of China Medical University (Taichung, Taiwan) and housed in temperature-controlled rooms with a 12-h light/dark cycle and were given food and water ad libitum. mBM-DCs were acquired as described previously (8,9,50). Briefly, bone marrow cells from the femurs and tibias of mice were first depleted of red cells with ammonium chloride–potassium (ACK) lysis buffer. Then 1 × 106 cells were placed in 24-well plates (Corning Inc., Corning, NY, USA) in 1 ml complete RPMI 1640 medium containing 2 mM l-glutamine, 10% FBS, nonessential amino acid, sodium pyruvate, HEPES, 50 μM β-mercaptoethanol, 100 U/ml penicillin and 100 mg/ml streptomycin, rmGM-CSF (20 ng/ml), and rmIL-4 (10 ng/ml) at 37°C, 5% CO2. On day 3, 1 ml complete RPMI 1640 medium containing rmGM-CSF and rmIL-4 was added to the plates to give a total of 2 ml. On days 5 and 7, 1 ml of the cell-free supernatant was removed, and 1 ml fresh medium containing rmGM-CSF and rmIL-4 was added. On day 8 of culture, nonadherent and loosely adherent cells (immature mBM-DCs) were collected. At this point, we determined by BD LSR II flow cytometry (BD Biosciences, San Jose, CA, USA) the percentage of CD11c+ cells, and for all experiments, this was ≥80%.
Cell Viability Assay
mBM-DCs were pretreated with serially diluted dryocrassin for 24 h and then stimulated with or without 100 ng/ml LPS for 20 h. The final concentration of DMSO in all dryocrassin-treated cultures was 0.1% (v/v). Cells were then harvested and stained by using 5 μg/ml propidium iodide or annexin V kit (Invitrogen). Cell viability and apoptosis was analyzed by BD LSR II flow cytometry (BD Biosciences).
Cell Activation and Treatment
Depending on the results of the cell viability assay, mBM-DCs were pretreated with 10 or 20 μM dryocrassin for 1 h. The final concentration of DMSO in all dryocrassin-treated cultures was 0.1% (v/v). After 1 h of incubation, the cells were washed twice using phosphatebuffered saline (PBS; Amresco, Solon, OH, USA), followed by stimulation with 100 ng/mlLPS for the indicated time points. Media and cells were collected for subsequent evaluation of DC activation and analysis of protein expression. Three replicates were included in each experiment.
Cytokine Assay
Dryocrassin-pretreated mBM-DCs were stimulated with LPS for 20 h (6 h for TNF-α). The secretion of TNF-α, IL-6, and IL-12p70 in cultured cell media was measured by enzyme-linked immunosorbent assay (ELISA) kits purchased from Invitrogen. The cytokine concentration was evaluated according to the manufacturer's protocol.
Flow Cytometry
Dryocrassin-pretreated mBM-DCs were stimulated with LPS for 20 h. The expression of surface molecules on DCs was determined by flow cytometry as described previously (7). Cells were harvested and resuspended in ice-cold FACS washing buffer (0.1% sodium azide and 2% FBS in PBS). Cells were then blocked with 10% (v/v) normal rat serum (Santa Cruz Biotechnology) for 10 min at 4°C and stained with FITC-conjugated anti-MHC class II, anti-CD40, and anti-CD86 with PE-conjugated anti-CD11c antibody for 30 min at 4°C. The appropriate isotype-matched immunoglobulins were used as negative controls. Stained cells were analyzed by flow cytometry. The data were collected for 1 × 104 cells per sample.
Allogenic Mixed Lymphocyte Reaction
Splenocytes from the spleens of BALB/c mice (8 weeks old) were isolated by using a mouse T-cell isolation kit (Miltenyi Biotec, Bergisch Gladbach, Germany). Over 90% of cells expressed CD3+, as determined by the FITC-conjugated CD3 antibody (GenWay Biotech) using BD LSR II flow cytometry (BD Biosciences). Dryocrassinpretreated mBM-DCs were stimulated with LPS for 20 h. Cells were then harvested, washed, and incubated with 25 mg/ml mitomycin C for 30 min at 37°C. Finally, the cells were washed and diluted with the prepared splenocytes in a ratio of 1:50 and 1:100 in U-bottomed 96-well culture plates (Corning Inc.) for 3 days. Cell proliferation was determined by MTT assay. The absorbance of the dissolved solutions was detected by using a SpectraMax M2 Microplate Reader (Molecular Devices, Silicon Valley, CA, USA) at 570 nm (7,9).
NF-κB Assay
Dryocrassin-pretreated mBM-DCs were stimulated with LPS for 40 min. The nuclear protein of the cells was extracted by using the Nuclear Extraction Kit (Affymetrix-Panomics, Santa Clara, CA, USA). NF-κB p65 binding activity was determined with the Universal EZ-TFA Transcription Factor Assay Colorimetric kit according to the manufacturer's instructions (Millipore) and quantified by absorbance (450 nm) with a microplate reader.
Immunofluorescence Assay
Cells were fixed with 4% paraformaldehyde for 15 min and then permeabilized with 0.3% Triton X-100 for 20 min. After three washes with PBS, cells were blocked with 5% bovine serum albumin for 1 h. NF-κB p65 subcellular location was determined by immunostaining with rabbit anti-mouse NF-κB p65 antibodies (1:200 dilution; Santa Cruz Biotechnology) overnight at 4°C and then with Alexa Fluor-488-conjugated goat anti-rabbit IgG antibody (1:100 dilution; Abcam, Cambridge, MA, USA). 4′,6-Diamidino-2-phenylindole (DAPI) (Abcam) staining indicates the location of cell nucleus. Fluorescence signals were observed under an Axio Observer inverted fluorescence microscope (Carl Zeiss MicroImaging GmbH, Göttingen Germany).
Western Blot Analysis
Dryocrassin-pretreated mBM-DCs were stimulated with LPS for 40 min and then lysed in radioimmunoprecipitation assay buffer (Millipore) to extract proteins. The concentration of whole cell lysates was calculated by using the RC DC Protein Assay Kit (Bio-Rad Life Science, Hercules, CA, USA). Fifty micrograms of protein per sample was loaded onto a 10% sodium dodecyl sulfate (Sigma-Aldrich)-polyacrylamide (Amresco) gel electrophoresis (SDS-PAGE) gel and analyzed by Western blotting for IKKa/b, IkBa, JNK, ERK1/2, p38, Akt, and their phosphorylated forms as previously described (7,9). The blots were developed using an Amersham-enhanced chemiluminescence system (Piscataway, NJ, USA). Signals were assessed by using a UVP BioSpectrum Imaging System (Upland, CA, USA).
Inhibitor Treatment
IKK-2 inhibitor IKK-16, JNK inhibitor SP600125, and p38 MAPK inhibitor SB202190 were purchased from Selleck Chemicals (Houston, TX, USA) and dissolved in DMSO. Inhibitors were titrated at concentrations ranging from 0.1 to 50 mM. A dose of 5 mM for IKK-16, 10 mM for SP600125, or 10 mM for SB202190 was used for experiments, as this was the lowest concentration that could completely block kinase activity. For inhibitor analysis, mBM-DCs were preincubated with the indicated inhibitors for 30 min and then incubated with or without 20 mM dryocrassin. After 1 h of incubation, the cells were stimulated with 100 ng/ml LPS for 16 h. Inhibitors were removed by twice washing mBM-DCs with 2% FBS/PBS. Cells were collected, and an allogenic mixed lymphocyte reaction was performed.
Contact Hypersensitivity Assay
A contact hypersensitivity (CHS) test induced by DNFB was described previously (10). Briefly, the shaved bellies of mice were painted with 20 ml of vehicle (acetone/olive oil = 4/1), 0.5% (w/v) DNFB, 0.5% DNFB plus 0.1% (v/v) DMSO, or 0.5% DNFB plus dryocrassin (100 mg) for sensitization. Five days later, all mice were challenged by painting on the backs of their shaved ears with 10 ml 0.2% DNFB (~1 cm diameter). Unsensitized mice were challenged with DNFB to act as negative controls. Animals were sacrificed 24 h later, and the CHS response was measured by hematoxylin and eosin staining. Ear swelling was calculated by subtracting from the thickness before challenge.
Skin Transplantation Model
Dryocrassin-pretreated, LPS-stimulated mBM-DCs (1 × 106) were injected intravenously into six male BALB/c mice (8 weeks old, 20 g, from the National Laboratory Animal Center, Taiwan) in PBS with 0.5% bovine serum albumin. After 7 days, skin transplantations were performed with protocols modified from those described previously (47). In brief, a 1-cm× 1.5-cm full-thickness skin graft was removed from the C57BL/6 donor mouse. A graft bed of right dorsal flanks on the anesthetized BALB/c recipient was surgically prepared with fine scissors (Fine Science Tools Inc., Foster City, CA, USA) by removing an area of epidermis and dermis down to the layer of intrinsic muscle. The skin graft was fixed to the recipient's graft bed and covered with sterile protective tape (3M Health Care, St Paul, MN, USA). The mice were raised individually in cages. Graft survival was recorded by daily visual examination. Scoring was based on redness, crust forming, and loss of hairs by comparison with syngeneic grafts. Rejection was defined as more than 80% necrosis of the graft surface. Skin graft survival rates were statistically analyzed by the log rank test.
Statistical Analysis
All statistical analyses are expressed as mean ± standard deviation (SD) from three independent tests. Three replicates were done of each test. The differences between two means were determined by Bonferroni-corrected Student's t test. Values of p < 0.05 were determined to be statistically significant.
Results
Effects of Dryocrassin on Cell Viability
In the present study, the immunomodulatory effects of dryocrassin were determined by using mBM-DCs. A propidium iodide staining/flow cytometry analysis and an annexin V-fluorescein staining/flow cytometry analysis were used to evaluate the cytotoxicity of dryocrassin. With or without LPS stimulation, cell viability and apoptosis were not significantly changed by 24-h treatment with up to 20 mM dryocrassin (Fig. 2). In the experiments following this assay, cells were therefore treated with dryocrassin at concentrations of up to 20 mM.
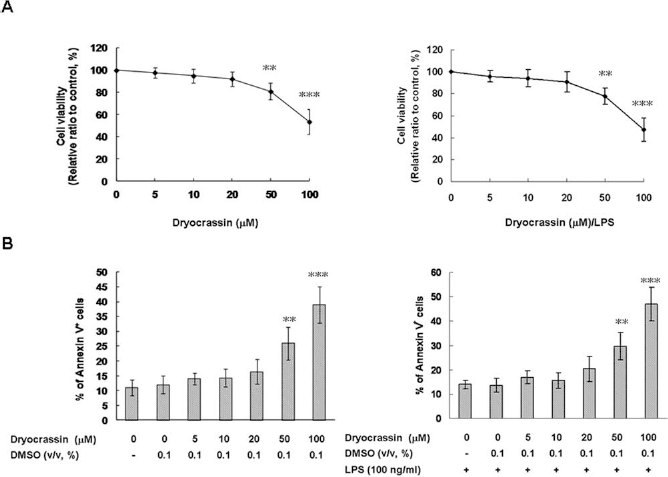
Effects of dryocrassin on the viability of mouse bone marrow-derived dendritic cells (mBM-DCs). The mBM-DCs were treated with serially diluted dryocrassin for 24 h and then stimulated with or without 100 ng/ml lipopolysaccharide (LPS) for 20 h. The viability of cells was determined by propidium iodide staining/flow cytometry assay (A). The apoptosis of cells was determined by annexin V-fluorescein staining/flow cytometry assay (B), as described in the text. The data represent the mean ± SD (n = 3). **Differences between dryocrassin-untreated control samples and dryocrassin-treated samples, p < 0.01. DMSO, dimethyl sulfoxide (vehicle).
Inhibitory Effects of Dryocrassin on LPS-Induced TNF-a, IL-6, and IL-12p70 Secretion
During maturation, DCs change their phenotype and functional classification. TNF-α, IL-6, and IL-12 are three essential proinflammatory cytokines that motivate the expression of costimulatory/accessory molecules on DCs and promote DC-mediated T-cell responses (38). Cytokine release was measured by ELISA. In unstimulated mBM-DCs, 20 mM dryocrassin did not alter TNF-α, IL-6, and IL12p70 secretion. Treatment with LPS caused a 20-fold (p < 0.001), 16-fold (p < 0.001), and 14-fold enhancement (p < 0.001) in the production of TNF-α, IL-6, and IL-12p70, respectively. Dryocrassin decreased the production of TNF-α, IL-6, and IL12p70 in a concentration-dependent manner. At 20 mM dryocrassin, LPS-stimulated TNF-α release was reduced by about 56% (p < 0.01), IL-6 secretion by about 63% (p < 0.001), and IL-12p70 secretion by about 62% (p < 0.001) (Fig. 3).

Inhibitory effects of dryocrassin on TNF-α, IL-6, and IL-12p70 secretion in LPS-stimulated mBM-DCs. mBM-DCs were pretreated with 10 or 20 mM dryocrassin. After 1 h of incubation, the cells were washed, followed by stimulation with 100 ng/ml LPS for 20 h (6 h for TNF-α). Media were collected and assayed for TNF-α, IL-6, and IL-12p70 levels by using an enzyme-linked immunosorbent assay (ELISA) kit. The data represent the mean ± SD (n = 3). #Differences between LPS-stimulated and unstimulated cells, p < 0.001. *Differences between the LPS-stimulated control samples and dryocrassin-pretreated, LPS-stimulated samples, *p < 0.05, **p < 0.01.
Inhibitory Effects of Dryocrassin on LPS-Induced Surface Marker Expression
Interactions between surface effector molecules on DCs and their ligands are necessary for the full activation of T-cells. MHC class II presents the processed fragments of foreign antigen peptide to a CD4+ T-cell. CD40 associates with CD154 on the helper T-cell to obtain an activation signal, augmenting antigen presentation and the expression of other costimulatory molecules. CD86 binds CD28 on the CD4+ T-cell for T-cell priming and survival (31). The effect of dryocrassin on surface-specific molecules of mature DCs was resolved by flow cytometry and the expression level of MHC class II, CD40, and CD86 measured by fluorescence intensity. Dryocrassin treatment of unstimulated mBM-DCs did not influence the expression of MHC class II, CD40, and CD86. In contrast, LPS-stimulated mBM-DCs evidently raised the expression of each of these molecules (p < 0.001), which was then repressed by treatment with 20 mM dryocrassin (all values of p < 0.001) (Fig. 4).

Inhibitory effects of dryocrassin on MHC class II, CD40, and CD86 expression in LPS-stimulated mBM-DCs. mBM-DCs were pretreated with 10 or 20 mM dryocrassin. After 1 h of incubation, the cells were washed, followed by stimulation with 100 ng/ml LPS for 20 h. The expression of major histocompatibility complex (MHC) class II, cluster of differentiation 40 (CD40), and CD86 on CD11c+ cells was determined by flow cytometry. The data are represented as the mean fluorescent intensity ± SD (n = 3). #Differences between LPS-stimulated and unstimulated cells, p < 0.001. *Differences between the LPS-stimulated control samples and dryocrassinpretreated, LPS-stimulated samples, *p < 0.05, **p < 0.01.
Inhibitory Effect of Dryocrassin on Allostimulatory Capacity
To define the effect of dryocrassin on the allostimulatory capacity of mBM-DCs, we used a mixed lymphocyte reaction/MTT assay and splenocytes from BALB/c mice as responder T-cells. As shown in Figure 5, dryocrassinpretreated, LPS-stimulated mBM-DCs had a smaller stimulatory capability compared with untreated, LPS-stimulated mBM-DCs (20 mM, p < 0.001), indicating that dryocrassin treatment prevented the allostimulatory ability of stimulated mBM-DCs.

Inhibitory effect of dryocrassin on the proliferation of naive allogeneic T lymphocytes by LPS-stimulated mBM-DCs. mBM-DCs were pretreated with 10 or 20 mM dryocrassin. After 1 h of incubation, the cells were washed, followed by stimulation with 100 ng/ml LPS for 20 h. DCs were then washed and incubated with 25 mg/ml mitomycin C for 30 min at 37°C. Finally, the cells were washed and diluted with the prepared splenocytes in a ratio of 1:50 and 1:100 in culture plates for 3 days. The proliferation of T-cells was assessed by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. DCs were sufficiently fixed with mitomycin C according to a DC-only control (data not shown). The values of unstimulated DCs served as control values in calculation of the percentage of proliferation. The data represent the mean ± SD (n = 3). #Differences between LPS-stimulated and unstimulated cells, p < 0.001. *Differences between the LPS-stimulated control samples and dryocrassin-pretreated, LPS-stimulated samples, *p < 0.05, **p < 0.01.
Inhibitory Effect of Dryocrassin on LPS-Induced NF-κB p65 Translocation
Translocation of NF-κB from the cytosol to the nucleus is important for LPS-stimulated activation of DCs (37). Given that dryocrassin hindered the LPS activation of DCs in our studies, we evaluated the effects of dryocrassin on NF-κB p65 levels in the nucleus. As shown in Figure 6, LPS-stimulated DCs obviously enhanced the NF-κB p65 levels in the nucleus (p < 0.001), whereas 20 mM dryocrassin treatment reduced nuclear NF-κB p65 levels in the LPS-stimulated DCs in a concentration-dependent manner (p < 0.001).
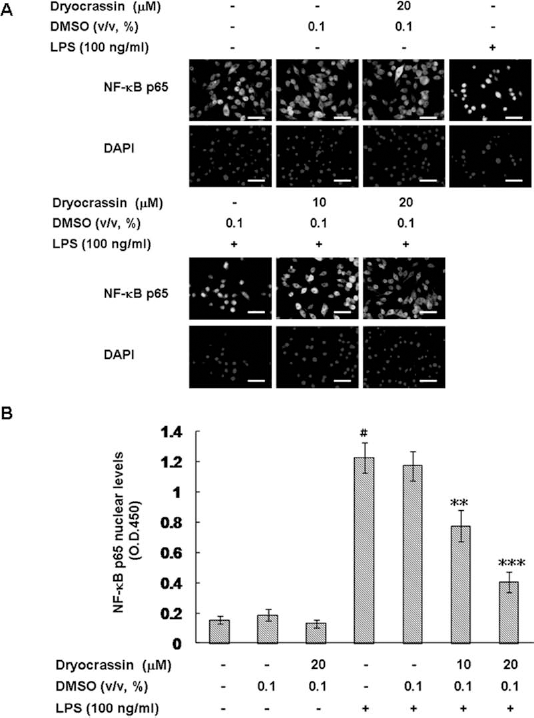
Inhibitory effect of dryocrassin on the translocation of NF-κB p65 in LPS-stimulated mBM-DCs. mBM-DCs were pretreated with 10 or 20 mM dryocrassin. After 1 h of incubation, the cells were washed, followed by stimulation with 100 ng/ml LPS for 40 min. (A) The localization of nuclear factor κ-light-chain-enhancer of activated B-cell (NF-κB) p65 was determined by immunostaining with anti-NF-κB p65 antibody. 4′,6-Diamidino-2-phenylindole (DAPI) staining indicates the location of cell nucleus. Scale bar: 50 μm. (B) Cells were lysed and the nuclear fraction determined for relative binding activity of NF-κB p65 by using the Universal EZ-TFA Transcription Factor Assay Colorimetric kit. The data represent the mean ± SD (n = 3). #Differences between LPS-stimulated and unstimulated cells, p < 0.001. *Differences between the LPS-stimulated control samples and dryocrassin-pretreated, LPS-stimulated samples, *p < 0.05, **p < 0.01.
Inhibitory Effects of Dryocrassin on LPS-Induced IKK and JNK/p38 MAPK Phosphorylation in the Cytoplasm
Several signaling cascades are connected in DC maturation (24). To determine the effect of dryocrassin on IKK/NF-κB, PI3K/Akt, and MAPK pathways, we examined the phosphorylation levels of key signaling factors involved in the activation of DC by using Western blot analysis. As shown in Figure 7, LPS induced the phosphorylation of IKKa/b, IkBa, JNK, and p38 and caused the degradation of IkBa in mBM-DCs, both of which are blocked in dryocrassin-pretreated cells in a concentration-dependent manner (20 μM,p < 0.001). LPS-induced ERK1/2 and Akt phosphorylation was not arrested by dryocrassin treatment. In addition, blocking the activities of IKK, JNK, and p38 MAPK with the inhibitors IKK-16, SP600125, and SB202190, respectively, completely abrogated the capacity of dryocrassin to retard DC-induced allogeneic T-cell proliferation (Fig. 8). Therefore, dryocrassin adjusted the activation of NF-κB and MAPK signal transduction cascades linked with DC maturation by inhibiting IKK, JNK, and p38 activity.

Inhibitory effects of dryocrassin on the activation of the IKK/NF-κB and MAPK signaling pathways in LPS-stimulated mBM-DCs. mBM-DCs were pretreated with 10 or 20 mM dryocrassin. After 1 h of incubation, the cells were washed, followed by stimulation with 100 ng/ml LPS for 40 min. Cells were lysed, and phosphorylation levels (p) of inhibitor of kappa B (IkB) kinase (IKKα/β), IκBα, c-Jun N-terminal kinase (JNK), extracellular signal-regulated kinase (ERK1/2), p38 mitogen-activated protein kinase (MAPK), and Akt were analyzed by Western blot analysis. The expression of β-actin was used as an internal control. One representative result from three independent experiments is shown. The relative fold in protein level was calculated as the level in pretreated cells relative to that in dryocrassin-untreated, LPS-unstimulated controls. The data represent the mean+SD (n = 3). #Differences between LPS-stimulated and unstimulated cells, p < 0.001. *Differences between the LPS-stimulated control samples and dryocrassinpretreated, LPS-stimulated samples, *p < 0.05, **p < 0.01.
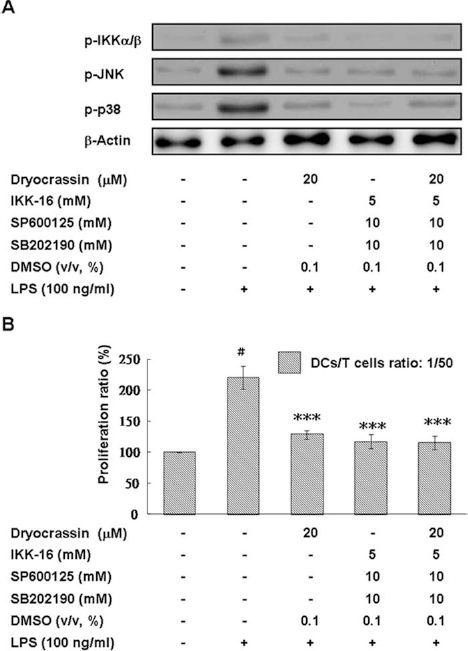
Effects of IKK, JNK, and p38 MAPK pathways on allostimulatory ability of dryocrassin-pretreated, LPS-stimulated mBM-DCs. mBM-DCs were preincubated with the indicated inhibitors for 30 min and then treated with or without 20 mM dryocrassin. After 1 h of incubation, the cells were washed, followed by stimulation with 100 ng/ml LPS for 20 h. (A) The detection of phosphorylated forms of IKK, JNK, and p38 MAPK by Western blot analysis with specific antibodies. (B) DCs were then washed and incubated with 25 mg/ml mitomycin C for 30 min at 37°C. Finally, the cells were washed and diluted with the prepared splenocytes in a ratio of 1:50 in culture plates for 3 days. The proliferation of T-cells was assessed by the MTT assay. The values of unstimulated DCs served as control values in calculating the percentage of proliferation. The data represent the mean ± SD (n = 3). #Differences between LPS-stimulated and unstimulated cells, p < 0.001. **Differences between the LPS-stimulated control samples and inhibitor-preincubated (or inhibitor/dryocrassin-preincubated), LPS-stimulated samples, p < 0.01.
Inhibitory Effect of Dryocrassin on Contact Hypersensitivity (CHS) Responses
We confirmed a retardant effect of dryocrassin on DC maturation, which indicates that dryocrassin may prevent DC-mediated disorders. Thus, we used a DNFB-induced CHS response as a model to prove this hypothesis. Mice were sensitized by painting DNFB with or without dryocrassin directly onto their abdomens. The CHS response to DNFB was then tested. The ears were visibly swollen in DNFB-sensitized mice but not in DNFB plus dryocrassin-sensitized mice (20 mg, p < 0.001), whereas DMSO had no effect on DNFB-sensitized mice (Fig. 9), implying that dryocrassin restrains the DC-mediated sensitization in CHS. These results hint that dryocrassin has the potential to prevent delayed-type hypersensitivity disorders, such as allergic contact dermatitis.

Inhibitory effect of dryocrassin on the contact hypersensitivity response in mice. A contact hypersensitivity response was shown by hematoxylin and eosin staining (A), and the thickness of the challenged ear was calculated. Scale bar: 100 μm (B). Mice that were not sensitized but were challenged with 2.4-dinitro-1-fluorobenzene (DNFB) provided negative controls. One representative result from three independent experiments is shown. The data represent the mean ± SD (n = 3). #Differences between unsensitized and sensitized mice, p < 0.001. **Differences between the DNFB-challenged control samples and DNFB plus dryocrassin-challenged samples, p < 0.01.
Beneficial Effect of Dryocrassin on Skin Allograft Survival
Next, we tested the modulatory potential of the dryocrassin-pretreated, LPS-stimulated mBM-DCs in a fully allogeneic skin graft model. BALB/c mice were injected intravenously with unstimulated mBM-DCs; LPS-stimulated mBM-DCs; DMSO-pretreated, LPS-stimulated mBM-DCs; or dryocrassin-pretreated (20 mM), LPS-stimulated mBM-DCs. One week after treatment, these mice underwent transplantation with skin grafts derived from C57BL/6 mice. The donor skins were rejected by the mice injected with LPS-stimulated mBM-DCs with a median survival time (MST) of 14 days, which is not significantly different from the survival in mice injected with unstimulated mBM-DCs or with DMSO-pretreated, LPS-stimulated mBM-DCs (MST, 15 days) (Fig. 10). However, when mice underwent transplantation after injection with dryocrassin-pretreated, LPS-stimulated mBM-DCs, a significantly prolonged allograft survival was observed (MST, 32 days; p < 0.01). These results suggest that the dryocrassin-pretreated, LPS-stimulated mBM-DCs are capable of inducing donor-specific tolerance to prolong complete MHC-incompatible skin allograft survival.

Beneficial effect of dryocrassin on skin allograft survival. Graft survival of C57BL/6 donor skin in untreated BALB/c mice and in mice receiving an intravenous injection of 1 × 106 dryocrassin-pretreated, LPS-stimulated mBM-DCs 7 days before transplantation. A significantly prolonged graft survival was found in dryocrassin-pretreated, LPS-stimulated mBM-DC injected mice compared with LPS-stimulated mBM-DC injected mice (**p < 0.01).
Discussion
Our current data show that dryocrassin arrests the release of LPS-induced proinflammatory cytokines TNF-α, IL-6, and IL-12p70 by DCs; decreases LPS-induced expression of MHC class II, CD40, and CD86 molecules by DCs; and reduces LPS-induced, DC-triggered allogeneic T-cell proliferation. To the best of our knowledge, this is the first description of the immunosuppressive property of dryocrassin on DC maturation. Some approaches have confirmed that phytocompounds, including baicalin (18), cepharanthine (44), honokiol (20), lupanetype triterpenoids (28), piperine (2), and shikonin (19), have special immunosuppressive effects on DCs. This research reveals that dryocrassin is a new member on the list of phytocompounds with these effects. These evidences support the claims of traditional Chinese medicine practitioners about the employment of herbs containing dryocrassin in the treatment of inflammatory-related diseases (49). Using this readily obtainable drug offers a cheap, convenient, and highly effective means of regulating the immunomodulatory capacity of DCs.
TNF-α acts as a chief role in mediation of the inflammatory response such as inducing secretion of IL-6, IL-1, platelet-derived growth factor, and transforming growth factor and motivating expression of adhesion molecules. Furthermore, TNF-α also raises the production of nitrogen species and reactive oxygen by white blood cells (5). IL-6 has been connected in a wide range of biological functions, including fever, upregulation of acute phase protein synthesis in the liver, antibody production in activated B-cells, neutrophil production in the bone marrow, proliferation and differentiation of lymphocytes, and control of Th1-related cytokine release and IL-2 receptor (33). IL-12 is necessary for the proliferation, differentiation, and maintenance of Th1 cells that induce interferon (IFN)-γ and IL-2 production. IL-12 also plays a key role in the activities of natural killer cells and cytotoxic T-cells. Alternately, these cytokines advance T-cell responses and macrophage activation (45). Consequently, dryocrassin hindered the expression of TNF-α, IL-6, and IL-12 to arrest inflammatory response. Reducing expression of CD40 molecules on DCs could abolish antigen-presenting capability, decrease cytokine production, and diminish CD80 and CD86 expression (13).
In this investigation, we found that dryocrassin hinders multiple intracellular signaling cascades downstream of TLR4 in DCs (Fig. 11). The cytoplasmic IkB proteins control the nuclear translocation and activity of NF-κB. IkBs connect with NF-κB dimers, thus blocking their nuclear translocation. Initiation of TLR4 signaling by the LPS complex induces phosphorylation of IkBs, followed by their degradation and the release of NF-κB, which is then migrated into the nucleus. NF-κB transcription factor modulates the expression of many proinflammatory mediators, including cytokines, costimulatory molecules, as well as adhesion molecules in DCs, and is upregulated when DCs mature (51). Dryocrassin lessened the activation of NF-κB by inhibiting the degradation of IkBa and the nuclear translocation of p65 in LPS-stimulated DCs. MAPK signal transduction pathways also play a fundamental role in DC maturation. In this research, JNK and p38 activity was reduced by dryocrassin in LPS-stimulated DCs. Activation of JNK, which is linked with the inhibition of tyrosine phosphatases in DCs, is linked with the induction of DC apoptosis (14). p38 has been indicated to induce the expression of IL-12 (43) and CD83 (41) in DCs, and it also upregulates the cellular inhibitor of apoptosis protein 2 (cIAP2), indicating a role in maintaining DC survival (29). By using an inhibitor assay, we verified that dryocrassin destroys LPS-induced activation of DCs, at least in part, through the blocking of IKK and JNK/p38 MAPK pathways. Furthermore, dryocrassin may compete with DCs for LPS binding, causing inhibition of LPS-stimulated DC maturation. The result of the LPS-binding assay showed that dryocrassin does not affect LPS binding on DCs (data not shown), which implies that the blocking of LPS-stimulated DC maturation by dryocrassin is not due to a direct interaction between LPS and dryocrassin. The specific mechanisms underlying these results will require further study.
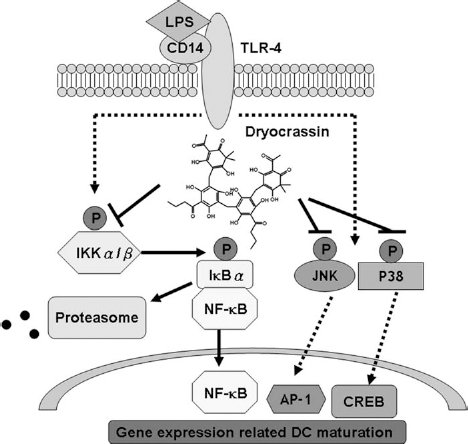
Dryocrassin reduces maturation of mouse bone marrow-derived DCs by downregulating IKK/NF-κB and JNK/p38 MAPK pathways. TLR-4, Toll-like receptor 4; AP-1, activator protein 1; CREB, cAMP response element-binding protein.
There is considerable therapeutic potential in operating DC–T-cell interactions for the treatment of complex diseases. Traditional immunosuppressants arrest T-cell activity to avoid rejection after transplantation. However, these drugs damage both T-cells targeting donor antigens and those that protect against pathogens, raising the risk of postoperative infection. An ideal immunosuppressant would impair only antigen-presenting cells that respond specifically to donor antigens while not affecting other immunological responses (34,40,46). The current study in a fully allogeneic skin graft model in mice extends our in vitro study with mBM-DCs to a clinically significant in vivo setting. Maturation stimulation after dryocrassin pretreatment promotes DCs with in vitro and in vivo tolerogenic properties. Pretreatment of recipients with these DCs leads to notably prolonged skin graft survival. This prolonged survival was shown experimentally in a fully MHC transplantation mismatched combination. In future experiments, we will test whether this pretreatment allows a reduction of the immunosuppressive drug dose after transplantation.
Footnotes
Acknowledgments
This work was supported by the National Science Council (Taiwan) (NSC 100-2314-B-039-036), the Taiwan Department of Health Clinical Trial and Research Center of Excellence (DOH102-TD-B-111-004), and China Medical University (DMR103-054), and Gwoxi Stem Cell Applied Technology Co., Ltd. (CMUH102-REC-AAA). The authors declare no conflicts of interest.
