Abstract

Dear editor:
Identifying neuronal derivatives of stem cells is essential for both basic research and future applications in regenerative medicine targeting neurodegenerative diseases. Erroneous identification could result in the transplantation of inappropriate cell types, thus directly affecting clinical outcome.
Stem cell and neurobiology researchers widely regard the class III β-tubulin protein (TUBB3), a member of the microtubule family, as being selectively expressed in neuronal cells. Antibodies against this protein are therefore often used to identify cells of the neuronal lineage or to quantify neuronal cells within cell cultures. The most renowned anti-TUBB3 antibody is the monoclonal TUJ1, with other examples including clones TU-20 and 2G10.
Nevertheless, we propose reconsidering the use of TUBB3 as a marker for neurons when melanocytes are potentially present or being formed. Why? Research in our own lab indicated that melanocytes were formed in studies on neuronal differentiation. Intrigued, we investigated and detected high TUBB3 expression during melanocyte differentiation in vitro and in vivo (3) in both human fetal and adult melanocytes (see Fig. 1A for an example in the human skin). In agreement, two other articles (1,2) have described the expression of TUBB3 in chick and mouse melanoblasts, indicating that TUBB3 expression is clearly not restricted to neurons and should therefore not be regarded as a standard for neuronal identification, but rather as a broader marker of the neural lineage (both neural crest and neural tube derivatives).
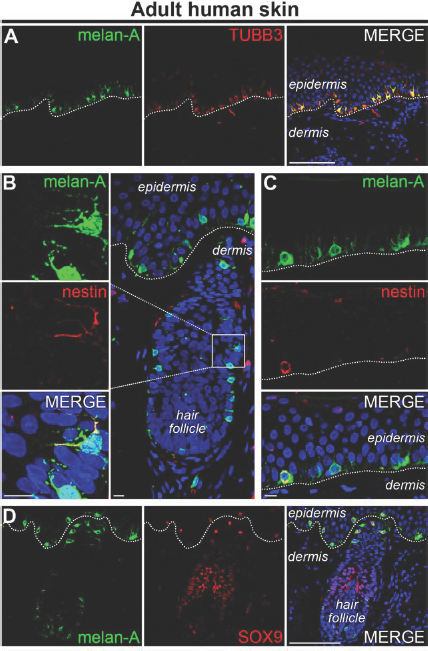
In vivo expression of melanocytic and neuronal markers in the human skin. Human facial skin was obtained from a 54-year-old male after informed consent in accordance with the WMA Declaration of Helsinki guidelines was provided. The use of material was approved by the Medical Ethical Committee of the Leiden University Medical Center (Netherlands). For immunofluorescence, tissue was deparaffinized and stained according to standard protocols, as previously described (3). Primary antibodies used in this study: rabbit anti-melanoma antigen (melan-A; 1:200, NBP1–30151, Novus Biologicals, Cambridge, UK), mouse anti-melan-A (1:20, ab731, Abcam, Cambridge, UK) mouse anti-class III β-tubulin protein (TUBB3; 1:200, 2G10, ab78078, Abcam), rabbit anti-nestin (1:100, ab28944, Abcam), and rabbit anti-sex-determining region Y box 9 (SOX9; 1:200, ab5535, Millipore, Amsterdam, Netherlands). Secondary antibodies (all 1:500, Life Technologies, Grand Island, NY, USA) used in this study: Alexa Fluor 488 donkey anti-mouse (A-21202), 488 donkey anti-rabbit (A-21206), 568 donkey anti-mouse (A-10037), and 568 donkey anti-rabbit (A-10042). As antibody specificity controls, primary antibodies were omitted. Nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI, Vector Laboratories, Burlingame, CA, USA). Confocal images of immunohistochemistry performed in 5-μm skin sections against various melanocytic and neuronal proteins. (A) Melanocytes express both melanoma antigen (melan-A) and TUBB3. TUBB3, but not melan-A, is also expressed by dermal and epidermal nerve fibers. (B, C) Although most melanocytes do not express nestin, a few exceptions can be found, both in the hair follicle (B) and in the epidermis (C). (D) Melanocytes, but not neurons, express both melan-A and SOX9. The dashed line indicates the border between the dermis and the epidermis. In the merges, nuclei were stained blue with DAPI. Scale bar: 100 μm (A and D) or 10 μm (B and C).
Furthermore, double-labeling combinations of microtubule-associated protein 2 (MAP2) and TUBB3 (used to identify cells as “neurons”) or tyrosine hydroxylase (TH) and TUBB3 (used to identify cells as “dopaminergic neurons”) are not fail-safe as melanocytes can express both MAP2 and TH. Melanocytes can also express nestin (considered to be a neural stem cell marker, although its presence in endothelial cells has also been reported), which together with TUBB3 is often used to identify “neuronal precursor cells” (Fig. 1B and C). Thus, using these combinations of markers may result in cells being incorrectly identified as neurons. The same is true for neurotrophins [nerve growth factor (NGF), brain-derived growth factor (BDNF), neurotrophin 3 (NT3), or NT4] and neurotrophic receptors [tropomyosin-receptor-kinases (Trks) and low-affinity nerve growth factor receptor (NGFR; p75NTR)] (5). Even functional ion channels (sodium, potassium, or calcium) of various types have been identified in the melanocyte (4).
Melanocytes and neurons are both derived from the neural plate (more specifically, all neurons from the peripheral nervous system and melanocytes are derived from the neural crest), share morphological features (e.g., an extensive dendritic tree), and to a large extent express the same biomarkers. If melanocytes are potentially present in the experimental system of interest, another method of identification should be sought. One option is to rule out melanocytes, for example, by taking advantage of markers present in melanocytes but not in mature neurons such as melan-A or sex-determining region Y box 9 (SOX9) (Fig. 1D). The other option is to irrefutably demonstrate successful derivation of neurons by their true neuronal hallmark: the action potential.
Or is it?
