Abstract
The release of proangiogenic cytokines into the circulation after stem cell (SC) therapy and compensatory increase of angiogenesis inhibitors may reflect local vasculogenesis but also can increase the risk of side effects. The aim of our study was to evaluate serum levels of angiogenic cytokines with regard to the assessment of local and systemic vasculogenesis in diabetic patients with no-option critical limb ischemia (NO-CLI). Twenty-five diabetic patients with NO-CLI treated with SCs isolated from bone marrow or stimulated peripheral blood were included in the study. Serum levels of proangiogenic cytokines (VEGF, bFGF, Ang-1, PDGF-AA, and PDGF-BB) and an antiangiogenic cytokine (endostatin) were assessed 6 months after cell treatment, compared to baseline values, and correlated with the number of injected CD34+ cells. The clinical effect of SC therapy (assessed by changes in TcPO2) and potential systemic vasculogenesis (assessed by eye fundus examination) were evaluated after 6 months. Serum levels of angiogenic inhibitor endostatin increased significantly after 1 and 3 months (p = 0.0003), but no significant increase in serum levels of proangiogenic cytokines was observed. A significant correlation between number of injected CD34+ cells and serum levels of endostatin was observed (r = 0.41, p < 0.05); however, proangiogenic cytokines did not correlate with CD34+ cells. No correlation between increase in TcPO2 after treatment and serum levels of any of the angiogenic cytokines were seen, and no signs of systemic vasculogenesis in the retina were observed after 6 months. Despite the significant increase in the levels of the angiogenic inhibitor endostatin following SC treatment, there was no risk of systemic vasculogenesis after SC therapy as documented by serum levels of proangiogenic cytokines or changes in the retina.
Keywords
Introduction
Patients with chronic, critical limb ischemia (CLI), defined as chronic ischemic rest pain, ulcers, or gangrene with objectively proven arterial occlusive disease, have a 20% mortality in the first year after presentation (17). Appropriate revascularization in those patients can be performed through endovascular procedures such as percutaneous transluminal angioplasty (PTA) or stenting or through open procedures such as a bypass surgery or thromboendarterectomy (21). Despite rapidly evolving endovascular approaches, many patients are not eligible for standard revascularization procedures [no-option CLI (NO-CLI)], and treatment with new techniques, such as autologous stem cell (SC) therapy, may be a treatment option (7,13). Autologous vascular precursor for SC therapy can be harvested from bone marrow [e.g., bone marrow-derived mononuclear cells (BMMNCs)], or separated from peripheral blood after stimulation by granulocyte-colonystimulating factor (G-CSF) [e.g., peripheral blood progenitor cells (PBPCs)].
The mechanism of action of SCs after intramuscular injection is probably paracrine, but the exact role of serum levels of angiogenic cytokines produced by these cells in connection with local and systemic vasculogenesis remains unclear. Possible mechanisms of vasculogenesis (angiogenesis and arteriogenesis) can involve the stimulation of chemotaxis and migration of mononuclear cells and the release of angiogenic cytokines contributing to proliferation of endothelium and smooth muscle cells (30). Other possible mechanisms are increased shear stress in preexisting collateral arterioles, which leads to enlargement of collaterals and/or increase in tissue microperfusion at capillary level or remodeling of fibrotic tissue to allow new capillary growth (18).
The release of proangiogenic cytokines, such as vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), angiopoietin-1 (Ang-1), plateletderived growth factors (PDGF-AA and PDGF-BB), and the endogenous inhibitor of angiogenesis (endostatin), is a possible modulator of vasculogenesis produced by SCs. Increased VEGF and bFGF levels have been shown to correlate with collateral growth of vascular cells (4); VEGF and other proangiogenic factors bind to receptors with intrinsic tyrosine kinase activity that leads to intracellular activation of protein kinase cascades (32). Endostatin, the c-terminal cleavage fragment of collagen XVIII, on the other hand, inhibits VEGF- and bFGF-induced migration of endothelial cells and has been shown to reduce intimal neovascularization (22,23).
Pro- and antiangiogenic cytokines are produced by different types of cells. The most studied vascular precursors are cluster of differentiation 34-positive (CD34+)-enriched cell population, an immature population of bone marrowderived progenitors (14,31). The number of injected CD34+ cells is usually used as a quality control during the concentration process in different SC therapy studies and is usually between 0.6% and 2.4% of the total implanted BMMNCs or PBPCs (13). However, the CD34- cell populations have an angiogenic effect similar to CD34+ cell concentrates (24).
Transcutaneous oxygen pressure (TcPO2) is a standard method for noninvasive measurement of limb ischemia, especially in diabetic patients with CLI, where ankle–brachial index is not reliable because of medial sclerosis. It serves as the main parameter for therapeutic effect in studies on SC therapy in CLI (1,7,13,27).
Changes in eye fundus after SC treatment were recommended as a marker for assessment of possible systemic vasculogenesis. However, no severe adverse events were observed in published meta-analyses (2,13,15,27).
In our previous study in patients with CLI, we reported a beneficial effect of autologous stem cell therapy measured by an increase in TcPO2 on the dorsum of the affected foot (5). The aim of our present study was to evaluate the relevance of serum levels of angiogenic cytokines with regard to their role in local vasculogenesis. We also assessed the risk of systemic side effects and the relation of serum levels of cytokines to the number of injected precursor cells in diabetic patients with NO-CLI.
Materials and Methods
Twenty-five patients treated with autologous stem cells in our foot clinic from January 2008 to June 2012 were recruited into the study (Table 1). Inclusion and exclusion criteria for autologous stem cell therapy in patients with NO-CLI were published in our previous study (5). The study was approved by the local ethics committee (Ethics Committee of Institute for Clinical and Experimental Medicine and Thomayer's Hospital), and all patients gave written informed consent.
Baseline Characteristics of Patients Treated by Autologous Stem Cells

After stem cell treatment, we assessed dynamics of serum levels of both proangiogenic (VEGF, bFGF, Ang-1, PDG-AA, and PDGF-BB) and antiangiogenic cytokines (endostatin) measured after 1 day, 1 week, and 1, 3, and 6 months and compared these levels to the baseline values. Serum samples were diluted 1:4, and the levels of VEGF, bFGF, Ang-1, PDGF-AA, PDGF-BB, and endostatin were assessed using the commercial kit Fluorokine MAP Human Angiogenesis Custom Premix Kit A (R&D Systems, Minnesota, MN, USA) and Luminex device (Luminex Corporation, Austin, TX, USA). The minimum detection levels for each cytokine were VEGF 11.5 pg/ml, bFGF 11.8 pg/ ml, Ang-1 185.2 pg/ml, PDGF-AA 10.2 pg/ml, PDGFBB 28.4 pg/ml, and endostatin 123.5 pg/ml. The procedure was described previously by Thorburn et al. (28). Briefly, 50 μl of diluted serum sample was mixed with an analyte-specific antibody-coated microparticle and incubated for 3 h at room temperature with a gentle, continuous agitation. After the first wash, a detection antibody for each analyte was added and incubated again at room temperature for 1 h on a horizontal vortex. After the second wash, phycoerythrin-conjugated streptavidin (from Fluorokine kit) was added and incubated for 30 min at room temperature on the vortex. After a final wash, samples were immediately measured in the Luminex device. The sample results were calculated by interpolation from particular standard curves.
Fluorescence-activated cell sorting (FACS) analysis was done with FACS FC 500 (Beckman Coulter, Brea, CA, USA), and the number of injected CD34+ cells in the BMMNC suspension was calculated based on the detected CD34+ concentration and the volume of injected cell suspension.
Stem cell treatment was performed by two methods: BMMNCs (14 patients) or peripheral blood progenitor cells obtained by apheresis of peripheral blood stimulated by G-CSF (PBPCs; 11 patients). Since we found no difference in outcomes between those methods of isolation of stem cells in our previous study (5), data from both groups were pooled for analysis. Bone marrow was harvested from the iliac crest of both sides by the use of Jamshidi needle (B Braun Melsungen, Melsungen, Germany) in the operating theater, and afterwards BMMNCs were separated by using a Smart PReP2 (Harvest Technologies Corporation, Plymouth, MA, USA) or sedimentated using succinated gelatine (Gelofusin; B. Braun Melsungen) (25). PBPCs were separated by leukapheresis on Haemonetics MCS+ (Haemonetics, Braintree, MA, USA) using 16–20 cycles for achievement of minimal concentration of CD34+ cells in peripheral blood 2 × 104/ml after prior stimulation for 3–6 days by subcutaneous injection of 5–8 μg/ kg/day of G-CSF (filgrastim, Neupogen™, Amgen, CA, USA). A final cell suspension of 40–90 ml gained by both methods was injected into the calf and foot muscles of the affected lower limb (deep injection close to the obstructed artery trunks) in a series of about 40–50 punctures of 1–2 ml each.
The clinical effect of treatment of ischemia was assessed by TcPO2 measurement. The TcPO2 measurement was done by standardized method using a TCM400 monitor (Radiometer Medical ApS, Aarhus, Denmark) and was assessed by the same foot care specialist.
Possible systemic vasculogenesis as evidenced by increased retinal vascularization was evaluated by fundoscopic examination of both eyes at baseline and after 6 months of SC therapy by an experienced ophthalmologist.
All data are expressed as median and interquartiles. The statistical significance was analyzed using Mann–Whitney analysis and Wilcoxon pair test (using Bonferroni correction on exact significance levels of Wilcoxon test). The correlation between the number of total injected CD34+ cells, changes in TcPO2, and serum levels of angiogenic cytokines was done using Spearman correlation coefficient (MedCalc Software, Ostend, Belgium).
Results
Serum levels of the angiogenic inhibitor endostatin increased significantly after 1 and 3 months (p = 0.0003) (Fig. 1a). We did not observe any increase in serum levels of proangiogenic cytokines (Fig. 1b, c); however, PDGF-AA decreased significantly after 6 months (p = 0.026), and PDGF-BB decreased significantly after 3 and 6 months (p = 0.034 and p = 0.002) (Fig. 1d).
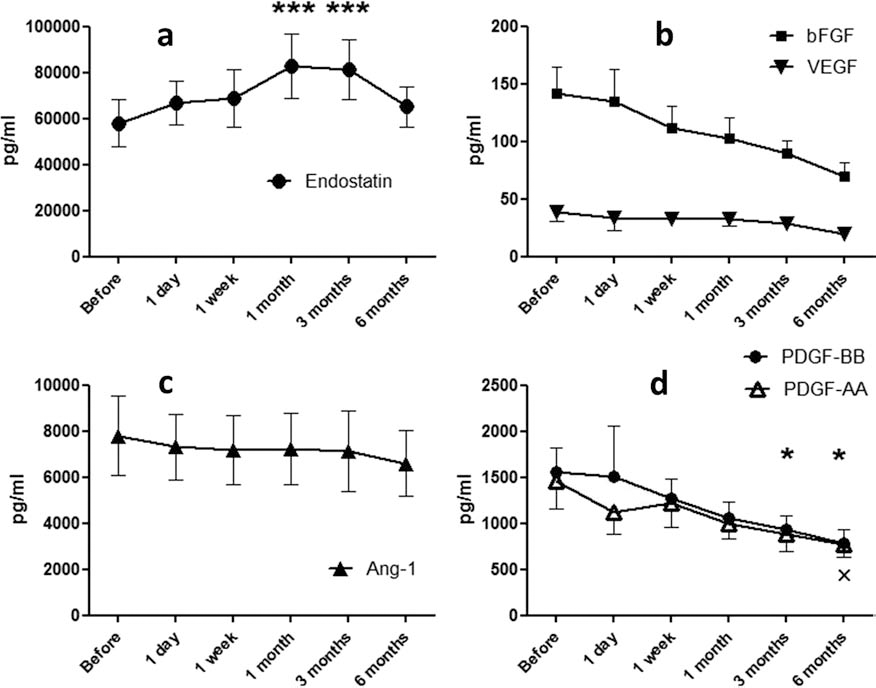
Comparison of levels of endostatin and proangiogenic cytokines with baseline values. (a) Levels of endostatin: significant increase after 1 and 3 months compared to baseline. ***p < 0.001. (b) Levels of vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF): no significant difference in levels of VEGF and bFGF compared to baseline. (c) Levels of angiopoietin-1 (Ang-1): no significant difference in levels of Ang-1 compared to baseline. (d) Levels of platelet-derived growth factors (PDGF-AA and PDGF-BB) compared to baseline: *p < 0.05 in PDGF-BB, xp < 0.05 in PDGF-AA. Values are median ± interquartile pg/ml over time (n = 25).
The median number of injected CD34+ cells was 1.4 (0.81–2.36) × 107. We observed a significant positive correlation between levels of antiangiogenic cytokine endostatin after 3 months and total number of injected CD34+ cells (r = 0.41; p < 0.05) (Fig. 2), but the correlation between the latter and levels of proangiogenic cytokines was not significant.
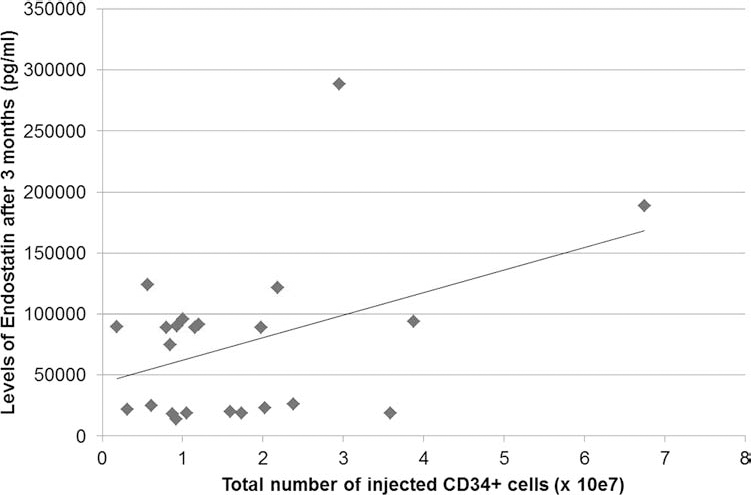
Correlation between total number of injected CD34+ cells and levels of endostatin after 3 months (r = 0.41; p < 0.05). Values are median ± interquartile pg/mls of endostatin versus the total number of cluster of differentiation 34-positive (CD34+) cells injected (×107; n = 25).
Clinical effects were measured by an increase in TcPO2. It was significantly higher, from 19 (6–24.5) to 39 (35.25–48.5) mmHg (p < 0.001) after 6 months (Fig. 3). No significant correlation between changes of TcPO2 and number of injected CD34+ cells, or levels of any of tested angiogenic cytokines was observed.

Dynamics of levels of endostatin and changes of TcPO2 up to 6 months compared to baseline. *p < 0.05. **p < 0.01. ***p < 0.001. Values are median ± interquartile endostatin (pg/ml) and median ± interquartile transcutaneous oxygen pressure (TcPO2; mmHg) over time.
No signs of systemic vasculogenesis was observed in the retina 6 months after treatment with stem cell therapy.
Discussion
In our study, serum levels of the endogenous inhibitor of angiogenesis endostatin increased significantly after 1 month in contrast to levels of proangiogenic cytokines. Levels of endostatin also correlated significantly with the total number of injected CD34+ cells. One of the possible explanations for the increase in endostatin is that it is a compensatory mechanism—an indirect sign of increased vascularization through the pathway of a negative feedback. Endostatin is cleaved from collagen XVIII, a core protein of heparan sulfate proteoglycan in vascular basement membranes, by proteases and metalloproteases such as elastase, matricillin, or cathepsin L that are produced by endothelial cells as a reaction to induced angiogenesis (3,8). Endostatin inhibits VEGF-induced endothelial progenitor cell (EPC) synthesis but also appears to interfere directly with endothelial nitric oxide synthase (eNOS) and targets upstream of its signaling pathway (23,29). The positive correlation between the number of injected CD34+ cells and endostatin levels could reflect this theory.
We did not observe any increases in proangiogenic cytokines up to 6 months after treatment, and the levels of platelet cytokines (PDGF-AA and PDGF-BB) significantly decreased after 3–6 months. We also found no correlation between the plasmatic levels of proangiogenic cytokines and the number of injected CD34+ cells, which can support the theory of their paracrine effect during hypoxic conditions and low serum levels of these cytokines (10). This finding is in accordance with results of fundus examination of the eyes—in that we did not observe a negative effect of autologous stem cell treatment by distal systemic vasculogenesis as demonstrated by a lack of progression of diabetic retinopathy (9).
Kajiguchi et al. also assessed proangiogenic cytokines (VEGF, bFGF, and Ang-1) after intramuscular SC therapy (BMMNCs or PBPCs) in patients with CLI and did not observe any significant difference in their serum levels between the responders and nonresponders to cell therapy (11). Similar findings were also seen by Tateishi-Yuyama et al., who did not demonstrate an increase in VEGF and bFGF in the first 48 h after BMMNC therapy in patients with CLI (26). Duong Van Huyen et al. investigated amputation specimens from patients after failed stem cell therapy and found positive endothelial markers on newly formed vessels in the distal part of the ischemic limb but not in the gastrocnemius muscle, the original site of bone marrow cell injection (6); this finding supports the theory about precursor cell migration along the ischemic muscles.
We showed an improvement in the clinical effect of stem cell treatment by a significant increase in TcPO2 but did not observe a correlation between this increase and serum levels of angiogenic cytokines or number of injected CD34+ cells. Lack of correlation between levels of endostatin (a cytokine with highest serum levels of all angiogenic cytokines) and TcPO2 increase might be explained by different kinetics in the changes in both parameters (Fig. 3). Active angiogenesis, which probably influenced the level of endostatin, appeared to be shorter, based on the lack of changes in angiogenic cytokines, than the clinical effect measured by TcPO2.
No relation between number of injected CD34+ and clinical effect on CLI has been confirmed in several trials. Miyamoto et al. showed no correlation between the number of injected CD34 cells and increase in ankle–brachial pressure index (ABI) in patients with thromboangiitis obliterans (16). Kawamoto and colleagues, using three different doses of CD34+ cells/kg in patients with CLI observed no significant dose response in relation to clinical effect (assessed by TcPO2) (12). Only one study observed a significant correlation between clinical response and number of CD34+ cells; however, this study was done in a small number of patients (n = 8) (20).
The main limitation of our study was that we measured serum levels of angiogenic cytokines and not their tissue concentrations, and this might have affected the results in that we did not find a significant correlation between the proangiogenic cytokines and a clinical effect of SC therapy, but we confirmed that there was no significant systemic vasculogenesis and side effects. On the other hand, serum levels of angiogenic inhibitor endostatin were quantitatively many times higher than the proangiogenic cytokines, and therefore we feel that the serum levels of endostatin are more conclusive. The levels correspond with endostatin's real production and can be a reflection of regulation of angiogenesis. The advantage of our study was the ability to assess adverse events and risk of systemic vasculogenesis over a prolonged period, which is clinically very important. The other limitation was that we could not verify the endostatin increase theory by comparing it with appropriate imaging techniques of local vasculogenesis after SC therapy, which is currently not available or requires special techniques (19).
Conclusion
Our study showed a significant increase in serum levels of angiogenic inhibitor endostatin after SC therapy in diabetic patients with NO-CLI. This increase correlated with the number of injected CD34+ cells; this finding can possibly reflect a feedback regulation of angiogenesis. No significant increase in serum levels of proangiogenic cytokines during 6 months of follow-up, and no changes in the retina indicate safety and low risk of autologous stem cell treatment in terms of induction of systemic angiogenesis.
Footnotes
Acknowledgments
Supported by grants from Grant Agency of Charles University (GAUK 362311) and the project (Ministry of Health, Czech Republic) for development of research organization 00023001 (IKEM, Prague, Czech Republic) – Institutional support. The authors declare no conflicts of interest.
