Abstract
Critical limb ischemia (CLI) is the end stage of peripheral vascular disease (PVD). One third of CLI patients progresses to leg amputation with high associated morbidity and mortality. In no-option patients with end-stage critical limb ischemia, bone marrow cell transplantation has shown promising results, improving leg perfusion to the level of reducing major amputations and allowing limb salvage. We recently reported the successful application of an innovative protocol based on repeated autologous bone marrow cell transplantation, which resulted in an effective and feasible strategy for achieving long-term revascularization in patients with severe CLI. In an effort to understand the clinical benefit provided by stem cells therapy in patients with CLI, we characterized the marrow-derived stromal cells of CLI patients and we provided a correlation between the in vitro features of these cells and the clinical follow up at 12 months. We showed that cells derived from CLI patients had a reduced capacity to proliferate, adhere, and migrate, but that they stimulated proliferation and migration of endothelial cells through the release of VEGF-A, supporting the idea that the paracrine mechanisms underpinned the biological effects of long-term angiogenesis in CLI patients.
Keywords
Introduction
Peripheral vascular disease (PVD) is a growing medical problem in Western societies, affecting 3—10% of the population (16). Based on the severity of symptoms, usually two clinical presentations are distinguished: intermittent claudication (IC) as early moderate manifestation, and critical limb ischemia (CLI) as a more severe form, with poor prognosis and high risk for limb amputation. Currently available treatment options for PVD patients are limited, because a significant proportion of PVD patients are not eligible for surgical vascularization procedures. Cell-based revascularization therapies have been tested in clinical trials investigating the therapeutic benefits in patients suffering from CLI (4). Many, if not all, of these studies used autologous bone marrow cell transplantation, given the concern of immune system reactions, and showed promising results. However, the precise mechanism of action is still unknown.
Although the cellular and molecular mechanisms involved in neovascularization have not been fully elucidated, three possible mechanisms have been proposed through which grafted bone marrow cells might rescue from ischemic damage.
The first mechanism is transdifferentiation of the grafted cells to the phenotype of the host tissue. The evidence of such a mechanism is highly controversial; some studies showed an apparent phenotypic switch after delivery of cells, while other studies could not confirm such a phenomenon (3).
The second possible mechanism is cell fusion. Grafted cells fuse with existing damaged cells and, in response to the genetic information of the recipient cell, they start to express tissue-specific genes. However, the resulting cells are often aneuploid and their survival rate is low (13,20,21).
The third mechanism underlying beneficial effects is based on the secretion by the grafted cell of trophic factors, which can act by: 1) preventing death of resident endothelial cells; 2) stimulating the proliferation of small collateral vessels, leading to connections with branches of the major axial arteries; and 3) stimulating the differentiation of resident stem cell progenitors, suggesting that angiogenesis is not the prerogative of a single cell population, but may result from the contribute of multiple cell types that interact each other by cell—cell interaction and paracrine signaling (14).
Recent studies have evidenced that bone marrow derived-endothelial progenitor cells (EPC) contribute to ischemic tissue repair by the secretion of paracrine factors. The authors employed intramuscular injection of conditioned medium (CM) of EPC in a rat model of chronic hind limb ischemia, demonstrating that the functional recovery of ischemia can be obtained by a cell-free strategy (8).
Here, we aimed to investigate whether the therapeutic benefit of cell therapy depends on the injection of cells themselves or to the release of cytokines necessary to mediate arteriogenesis, looking at the possible correlation between the in vitro activity of marrow-derived stem cells derived from CLI patients and the clinical features of a small set of patients at 12 months of follow up.
Patients and Methods
Inclusion Criteria
Six patients with end-stage CLI (III or IV stage of Leriche-Fontaine classification) were treated with autologous bone marrow cell transplantation between February 2008 and July 2008. The protocol was approved by the Ethics Committee of the Seconda Universitaà of Naples and was registered at the Trial Registration, NCT00306085.
All patients gave written informed consent before evaluation for inclusion. The diagnosis of limb ischemia was confirmed by ultrasound analysis and computed tomography angiography (CTA). All patients underwent measurements of resting ankle-brachial index (ABI), and pain-free walking for a standardized distance to classify the severity of CLI. Routine laboratory screening was performed for all patients. They underwent the same examinations after the treatment at the follow up.
Microcirculatory disturbances were evaluated by laser Doppler measurements (Periflex PF2B, Perimed AB, Stockholm, Sweden) and videocapillaroscopy (Videocap DS Medica srl, Milan, Italy, optical x200) before and after the treatment at two different stages: before infusion (T0) and 12 months after the infusion (T12). The laser Doppler (LD) measurement procedure was performed as follows: LDBO (laser Doppler—-clinostatic position) recorded the blood flow for 5 min with the patient in clinostatic position; LDBP (laser Doppler with lowered leg) recorded blood flow after positioning the limb in supine position activating venoarteriolar response; LD44°C (laser Doppler—-clinostatic position, probe temperature 44°C) recorded blood flow after increasing the temperature of the probe to 44°C to remove the myogenic vessel tone; LDPF (laser Doppler peak flux after induced ischemia)-LDPT (laser Doppler peak time after induced ischemia) recorded blood flow after a mixed hyperemia test by increasing the probe temperature to 44°C and inducing limb ischemia by arm cuff positioned below the knee and inflated to a pressure of 240 mmHg for 3 min.
Bone Marrow Harvest
Bone marrow (200 ml) was extracted from the iliac crest under anesthesia in a sterile manner and anticoagulated with heparin (1:50). The mixture was passed through filtering devices to remove large particulate matter such as fat, bone chips, and/or clots (Fenwal Bone Marrow kit collection 4R2104, Fenwal Inc Blood Technologies, Lake Zurich, IL, USA) and transplanted immediately into femoral artery through a peristaltic pump at 120 ml/h. Forty-five days after the first infusion, the treatment was repeated following the same procedure.
Cell Culture Preparation
A fraction of 10 ml of bone marrow aspirates was collected in tubes with Na Citrate (129 mM) and then diluted with PBS (1:3) and mononuclear cells isolated by density gradient centrifugation with Ficoll. Mononuclear cells were washed with PBS (3x) before plating in Mesencult Basal Medium (StemCell Technologies) + 10% FBS medium or EGM-2 medium (Lonza). Human mesenchymal stem cells (MSC) and human umbilical vein endothelial cells (HUVEC) were purchased from Provitro (Stemcells) and were used as healthy controls. Furthermore, bone marrow-derived cells were also obtained from two healthy donors of 60 and 41 years old.
Cellular Uptake of Acetylated LDL
Cells (4 × 104) were plated in 24-well plates and incubated in EGM-2 medium containing 15 mg/ml DiI-labeled Ac-LDL (Molecular Probes) for 1 h. After fixation with 2% PFA cells were incubated with FITC-labeled Ulex europaeus agglutinin I (ulex lectin, Sigma) for 1 h and visualized with an inverted fluorescent microscope (Leica). Nuclei were stained with DAPI.
Senescence Associated β-Galactosidase (SA-β-Gal) Staining
Cells were cultured on chamber slides and fixed with 2% (v/v) formaldehyde/0.2% (v/v) glutaraldehyde for 10 min. The cells were then washed twice with PBS and incubated with staining solution [30 mM citric acid/phosphate buffer (pH 6), 5 mM K4Fe(CN)6, 5 mM K3Fe(CN)6, 150 mM NaCl, 2 mM MgCl2, 1 mg/ml X-Gal solution (all reagents were purchased from Sigma)] at 37°C for 2 h. The cells were photographed with an inverted microscope (Leica).
RT-PCR and qPCR
Total RNA was isolated from cells by RNAspin Mini kit (Amersham, Bioscience); 600 ng was converted to cDNA using the Quantitect Reverse Transcription kit (Qiagen). PCR products were electrophoresed on 1.5% agarose gels.
The qPCR assays were performed using an iCycler (BIORAD) and the Sybergreen Super mix (BIORAD). The primer sequences and PCR conditions are available on request.
Capillary Tube Formation Assay in Matrigel
For analysis of capillary tube formation, 150 μl Matrigel (Becton Dickinson) was laid into a 24-well plate (Falcon, Germany) and incubated at 37°C for 30 min. CLI-derived MSC, bone marrow cells, or HUVEC were trypsinized and 3 × 104 cells were suspended in 150 μl of EGM-2 medium (Lonza) and plated onto Matrigel. Cells were incubated at 37°C and capillary tube formation in Matrigel was observed under an inverted microscope (Leika) both after 6 and 24 h of incubation.
Migration Assay
A total of 3 × 104 cells were resuspended in 250 μl of endothelial basal medium (EBM) and pipetted in the upper chamber of a modified Boyden chamber (Costar Transwell assay, 8 μm pore size, Corning, NY). The chamber was placed in a 24-well culture dish containing 750 μl complete endothelial growth medium (EGM-2) and as negative control 750 μl EBM with 0.1% BSA. After 24-h incubation at 37°C, transmigrated cells were counted by independent investigators at the inverted microscope.
Adhesion Assay
Hypoxic culture conditions were achieved in a BD GasPak EZ Anaerobe Gas Generating Pouch System (BD Biosciences, San Diego, CA). As certified by the manufacturer, the Anaerobe Gas Generating Pouch System produces an atmosphere containing 10% carbon dioxide and 1% oxygen.
Confluent HUVEC monolayers were subjected to normoxic and hypoxic culture for 16 h. Cells were serum starved in EBM plus 0.5% fetal bovine serum (FBS) at least 8 h before hypoxic culture to minimize the effects of growth factors in the expansion media. Carboxyfluorescein succinimidyl ester (CFSE) (Invitrogen)-labeled cells (5 × 104) were added to each preconditioned monolayer and preadhesion fluorescence was measured using a microscope (Leica). After 3 h, nonadherent cells were washed away and postadhesion fluorescence was measured. We calculated the percentage of adherent cells by the following formula: percentage of cells bound = (postadhesion fluorescence-mono-layer only)/(preadhesion fluorescence-monolayer only) × 100. Results are representative of three independent experiments and p-values are provided.
Conditioned Medium Preparation
Marrow-derived stromal cells were grown in basal medium and incubated at 37°C in hypoxic conditions for 48 h. After that, the supernatants were collected, filtered using Amicon Ultra-15 membranes (Millipore), and stored at −80°C until used.
Enzyme-Linked Immunosorbent Assay (ELISA)
The concentrations of SDF-1, VEGF, and MMP-9 in conditioned media were measured using commercial ELISA kits (R&D Systems) according to the manufacturer's instructions. Optical density was measured at 450 nm using an ELISA reader (TECAN).
Cell Proliferation and Survival
Patient MSC were grown in low-serum medium Mesencult Basal Medium (MBM) + 0.2% FBS. HUVEC were plated (1 × 104 cells/well in 96-well plates) in EGM-2 media (Cambrex) and allowed to attach overnight. The day after, they were starved for 24 h in EBM + 0.1% BSA at 37°C. Conditioned medium was added to starved cells in normoxic and hypoxic conditions. After 24 h, the number of living cells was measured by determination of ATP cellular levels using ViaLight® Plus Kit (Lonza). The number of apoptotic cells was determined by cytofluorometer using propidium iodide (PI) and annexin V staining. All experiments were performed in triplicate with patient CM generated from six different donors.
Statistical Analysis
All data are represented as mean ± SD. Statistical significance was evaluated by performing the Student's t-test and significance was accepted if the value was p < 0.05.
Results
Patient Characteristics and Clinical Follow-up
Six patients were diagnosed of severe CLI (III or IV stage of Leriche-Fontaine classification), showing an extensive arterial occlusion, reduced ankle-brachial index (ABI), invalidant claudication (IC), and pain at rest (Table. 1). With regard to the cardiovascular risk profile, three of six patients had hypertension, two of them had hypercholesterolemia, two were current smokers, and two were diabetic. Two patients already suffered an acute myocardial infarct (AMI). One patient had amputation of right leg (patient 4), while patient 6 had advanced necrosis of the first finger of the left foot. For the advanced disease stage, all patients were at high risk of amputation and were subjected to repeated autologous bone marrow transplantation as ultimate option.
Clinical Characteristics of CLI Patients Enrolled in the Study

Twelve months after transplantation improvement in perfusion by quantitative analysis of Laser Doppler imaging was assessed with subsequent assessment of ABI index and pain-free walking distance (Tables 1 and 2). According to the results, we classified patients 2, 3, 4, and 5 as responders, and patients 1 and 6 as nonresponders, even if the number of injected cells in the femoral artery was comparable.
Blood Flow Measurements at T0 and T12

Total cells infused in each leg.
In Vitro Characterization of Marrow-Derived Stem Cells
To investigate the marrow-derived cells status in patients suffering from severe CLI, mononuclear cells (MNC) were isolated from marrow aspirates derived from patients, previously described, and two healthy donors (Table. 1). Two different protocols were used for cell culture in order to assess the identity of their clonal progeny. MNC (2 × 106 cells) were seeded onto fibronectin-coated tissue culture plates and cultured in the presence of MBM + 10% FBS medium, to promote mesenchymal stem cells outgrowth. After 48 h, the nonadherent cells in MBM medium were removed to prevent hematopoietic cell contamination and further growing in MBM medium. Colonies appeared after 14 days in culture and CFUs were determined. The total number of CFUs was lower in marrow-derived cells of CLI patients compared with healthy donors (3.2 ± 1.5 CLI patients vs. 9.7 ± 0.8 healthy donors). In terms of morphology, both cell populations presented spindle morphology characteristic of mesenchymal cells in culture (Fig. 1A).
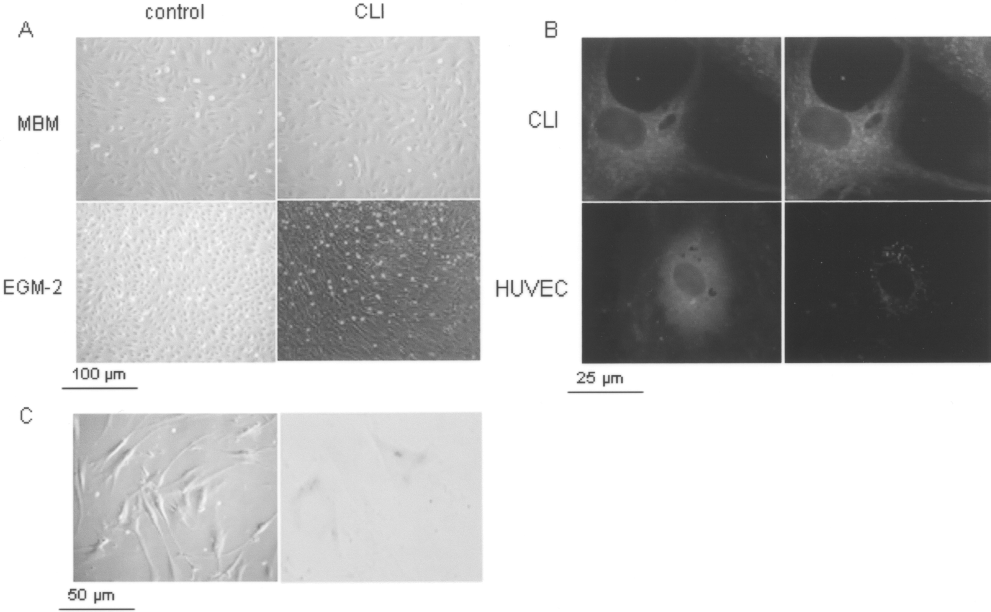
(A) Morphology of CLI-derived cells. Representative photographs of marrow-derived stromal cells seeded in MBM (10x magnification, upper panel) and in EGM-2 (lower panel). Mononuclear cells isolated from bone marrow of adult healthy young donor (left upper panel) and HUVEC CELLS cultured in EGM-2 (10x magnification) were used as controls (left lower panel). (B) Fluorescence microscopy images of lectin binding using FITC-Ulex europaeus agglutinin I and uptake of Ac-LDL. CLI BM-MNC (upper) and HUVEC (lower) showed lectin binding (green fluorescence), whereas only HUVEC cells showed uptake of acLDL-DiI (red fluorescence) (40x magnification). (C) Representative photographs of in vitro vacuoles formation (left panel) and blue-stained cells for SA-β-Gal activity are shown (20x magnification, right panel).
In order to assess whether the marrow-derived cells have the capacity to acquire an endothelial cell-like phenotype in vitro, we investigated this possibility by culturing MNC in endothelial induction medium. MNC (2 × 106 cells) were seeded onto fibronectin-coated tissue culture plates in EGM-2. After 48 h, the nonadherent cells were collected and replated in EGM-2 medium. Colonies appeared after 7 days in culture. The cells derived from the healthy donors appeared as well-circumscribed monolayer of cobblestone-appearing cells, while those derived from CLI patients showed multiple spindle-shaped cells (Fig. 1A) that progressively appeared as tubuloreticular pattern (not shown). To assess the endothelial phenotype, Dil-Ac-LDL uptake and lectin binding of isolated EPCs were determined. Stromal cells derived from healthy donor and CLI patients showed an intense lectin binding activity, but an absent DiL-ac-LDL uptake, compared to the HUVEC cells used as control (Fig. 1B).
We then determined the growth rate of these populations. After the appearance of colonies in both media, cumulative population doublings of cells derived from CLI patients were measured and compared to the healthy controls. Cells were split and cultured for approximately 30 days to measure the cell doubling time. Cells derived from patients showed longer doubling time compared to the control cells, both in MBM and EGM-2 media (EGM-2: average doubling time patients 3.7 ± 1.9 days; average doubling time control cells 1.5 ± 0.3 days; MBM: average doubling time patients 5.7 ± 1.8 days; average doubling time control cells 2.1 ± 0.3 days).
In both culture conditions, morphology of cells looked rather normal soon after initial expansion, similar to the controls, whereas in late passages cells started to exhibit vacuoles, debris appeared in the medium, and underwent irreversible growth arrest within 5—7 passages. To see whether this phenotype was related to the appearance of senescence, the cultures were assayed for SA-β-Gal. CLI-derived cells showed an increase of SAP-Gal activity during subcultivation; the appearance of senescence is not related to the age of patient, but to progressive cell divisions, because this phenotype appeared also in wild-type cells (p = 0.0234) (Fig. 1C). Therefore, CLI-derived cells showed a significant decrease in the growth rate and appearance of senescence phenotype, compared to control.
Phenotypic Characterization of Cultured Cells
We determined the phenotype for these adherent cell populations, measuring the expression of a combination of markers by RT-PCR (Table. 3) on RNA purified at early passages.
Primers and PCR Conditions Used in This Study

Marrow-derived cells from healthy donors and CLI patients grown in both conditions uniformly expressed the mesenchymal markers CD90, CD105, CD73, and CD44 and were negative for the endothelial markers von Willebrand factor (vWF), CD31, CD34, and CD144 (VE-cadherin) (Fig. 2). It is critical to consider that marrow-derived cells exhibited a striking similarity to vascular mural cells called pericytes (5,7). Therefore, we measured the expression of CD146, PDGFRβ, NG2, and αSMA, typical markers of pericytes. Cells derived from healthy donors and from CLI patients grown in both media were positive for pericytes markers.

Gene expression profile. RT PCR analysis was performed on (lane 1) HUVEC, (lane 2) BM-MNC from patient 1, (lane 3) patient 2, (lane 4) patient 3, (lane 5) patient 4, (lane 6) BM-EGM-2, (lane 7) BM-MBM, (lane 8) patient 5, and (lane 9) patient 6 to detect expression of different markers.
No cells in these cultures express markers of hematopoietic-derived cells (CD45, CD14, CD115), excluding any contamination (Fig. 2).
These data suggest that long-term culture of marrow-derived cells resulted in a homogenous perivascular cell population, both in medium specific for mesenchymal and endothelial outgrowth, indicating that marrow-derived cells is an obtainable perivascular cell source in vitro.
To examine whether cells exhibited biological features relevant to vessel formation and/or remodeling, a series of assays was performed. First, we asked whether marrow-derived cells from CLI patients were able to form networks of vessel-like structures in vitro. When seeded into extracellular matrix composites in the presence of proangiogenic culture medium, CLI-derived cells were able to form three-dimensional networks of vessel-like structures, as healthy donor cells (Fig. 3), suggesting that these cells had an intrinsic capacity to form tubules-like structures and this ability can be exploited by these cells to create a vascular networks (10,15).

Matrigel assay: in vitro tubule formation promoted by different cell populations (20x magnification).
Several studies have demonstrated that incorporation of marrow-derived cells into new or remodeling vessels is important in augmenting angiogenesis and is related to the adhesiveness toward endothelial cells and migration property (10,11,17). To evaluate the ability of CLI-derived cells to adhere to endothelial cells, we preconditioned HUVEC cells monolayer in hypoxic conditions for 16 h and CFSE-labeled marrow-derived cells were allowed to adhere to hypoxic HUVEC cells for 3 h. A consistent number of marrow-derived cells from healthy donors were found to adhere on hypoxic HUVEC cells, whereas cells derived from CLI patients showed a consistent reduction in the adhesion (Fig. 4A), suggesting that the reduced adherence might be a possible consequence of the disease.

(A) Adhesion of marrow-derived stromal cells on HUVEC cells preconditioned in hypoxic conditions. The p-values, calculated for each group, were determined by comparison of the average of adherent cells before and after the incubation (HUVEC/ BM p = 0.0059; HUVEC/patient 1 p = 0.0005; HUVEC/patient 2 p = 0.0011; HUVEC/patient 3 p = 0.007; HUVEC/patient 4 p = 0.009; HUVEC/patient 5 p = 0.0080; HUVEC/Patient6 p-value=0.0007. (B) qPCR analysis showed different levels of expression of CXCR4 gene in CLI BM-MNC and normal cells used as a control. (C) Migration toward proangiogenic factors of marrow-derived stromal cells from patient 1, patient 2, patient 3, patient 4, patient 5, patient 6. Normal MNC and HUVEC were used as control.
To explore whether the hypoxia-induced cell adhesion is mediated by CXCR4 signaling, we measured the expression of CXCR4 gene. The surface expression of CXCR4 has been reported to diminish with passages, thus we compared RNA from cells at the same passage. Q-PCR showed that the adherence of healthy donor cells to hypoxic HUVEC occurred by low expression of CXCR4, while increase in CXCR4 expression is related to a decreased adherence in CLI-derived cells, suggesting that adhesion of marrow-derived cells was not mediated by CXCR4-dependent mechanism (Fig. 4B), at variance from HUVEC cells.
Then we examined the migratory ability of marrow-derived cells, derived both from healthy controls and CLI patients in the presence or absence of proangiogenic factors (VEGF, hFGF, IGF-1, hEGF). Cells derived from healthy donors showed twofold increase of migration in presence of growth factors, whereas the response of cells derived from CLI patients was significantly inhibited compared to the control cells, suggesting that the chronic nature of the vascular disease of our patients had a detrimental effect on migration (Fig. 4C).
Cytokines Released From CLI-Derived Cells
We previously demonstrated that marrow-derived cells of CLI patients showed a reduced migratory capacity; therefore, it is difficult to sustain that these cells are able to migrate from bone marrow and to home to injured vessels, promoting the formation of new blood vessels in vivo. Although we cannot exclude this mechanism, we reasoned that bone marrow cells locally injected may be able to promote endothelial cell functions.
According to this hypothesis, we investigated whether marrow-derived cells secreted diffusible factors, supporting the migration of endothelial cells, or the proliferation of these cells, by preventing cell death.
To do this, we first verified whether the conditioned medium (CM) of CLI-derived cells sustained HUVEC cells migration. Low-serum medium conditioned by CLI cells markedly stimulated endothelial cell migration to a much greater extent than control, suggesting that soluble factors released by marrow-derived cells stimulated endothelial cells migration (Fig. 5A).

(A) Migration toward conditioned media of marrow-derived stromal cells from patient 1, patient 2, patient 3, patient 4, patient 5, patient 6, and HUVEC cells. (B, C) HUVEC proliferation and survival. Beneficial effects of the conditioned medium derived from patients on the proliferation of HUVEC cells and on apoptosis of HUVEC cells, evaluated with annexin-V staining.
We then evaluated the proliferation of HUVEC cells in presence of CM of CLI patients. HUVEC cells were incubated with CM derived from normal and CLI-derived cells grown in normoxia and hypoxia for 24 h and the proliferation of endothelial cells measured. As shown in Figure 5B, CM from CLI patients promoted the proliferation of endothelial cells in hypoxic conditions, whereas the medium derived from normal cells were not able to support the viability of HUVEC cells. We then verified whether increased cells viability resulted in reduced apoptosis of cells. To do this, we used the annexin-V staining to quantify the apoptotic cells. HUVEC cells, when grown for 24 h in medium containing low growth factor levels, showed a negligible rate of apoptotic cells in normoxia (EBM: 0.14%), whereas the incubation of HUVEC cells in hypoxic condition resulted in a significant increase of apoptotic cells (hypoxic EBM: 4.25%), a condition that was fully rescued by adding CM of marrow-derived cells (hypoxic CM: 1.25%; p = 0.019) (Fig. 5C).
Altogether these data suggested that the marrow-derived cells of CLI patients possibly secreted trophic factors in hypoxic conditions able to promote HUVEC cell proliferation. It is known that hypoxia activates vascular endothelium leading to upregulation of cell adhesion molecules and chemokines, regulating endothelial progenitor cells mobilization, mainly enhanced by VEGF-A, matrix metalloproteinase (MMP)-9, and stromal cell-derived factor-1α (SDF-1α). We hypothesized whether these mediators of endothelial trafficking were upregulated in conditioned medium of CLI cells in normoxic and hypoxic conditions. In hypoxia conditions, a significant increase in the accumulation of VEGF-A in marrow-derived cells from healthy donor was observed, whereas the oxygen level did not significantly influence VEGF-A secretion in HUVEC cells (Table. 4).
VEGF-A, MMP-9, and SDF-1 Concentrations in the Conditioned Media From Culture of Expanded Marrow-Derived Cells Incubated in Hypoxic and Normoxic Conditions for 48 h

Interestingly, the secretion of VEGF-A in CLI patients was responsive to oxygen level in patients 2, 3, 4, and 5, who were classified as responders according to the amelioration of clinical parameters and perfusion, while no variation was seen in nonresponders patients (patients 1 and 6; Table 3), suggesting that the paracrine activity of at least VEGF-A is related to the beneficial role of cell therapy in CLI patients.
Discussion
Cell-based therapy for peripheral vascular disease patients has moved remarkably fast from bench to bedside. Results of the first clinical trial appeared in 2002 (19) and, since then, there has been a steady rise in the number of clinical reports, currently accumulating to at least 40 (including 836 patients) (1,2,4). In spite of the rapid accumulation of data, the Trans-Atlantic Inter-Society consensus document II does not mention cell therapy as a future treatment option for PVD patients for several reasons. Among them, problems about safety, dose and administration route, timing, and frequency are the first cause of concern and need to be addressed in designing a stem cell-based clinical approach. Furthermore, there is accumulating evidence that other issues must also be taken into account; that is, the specific stem cells to be used and their precise mechanism of action, the specific vascular identity of the transplanted cells, and the long-term survival of engrafted cells in absence of a normal supportive tissue environment (the so-called niche).
The question of how stem cells exert their therapeutic effect is certainly not trivial. Recent studies demonstrated that growth factors improve stem cell therapy for ischemic disease, suggesting that the paracrine signaling positively affects angiogenesis (12). It is now clear that marrow-derived stromal cells can support migration, proliferation, and survival of endothelial cells in vitro through release of VEGF-A (18). Overexpression of proangiogenic growth factors (VEGF and PDGF-B) in umbilical cord blood (UCB)-derived progenitor cells improves autologous or allogenic stem cell therapy for ischemic diseases (6). Tissue levels of trophic factors, including VEGF, enhanced ischemia-induced neurogenesis by the intravenous administration of human MSCs. The effects were more pronounced with earlier passage than with later passage human MSCs, which may be related to the differential capacity in trophic support, depending on age (14). Moreover, production of angiogenic growth factors by peritoneal macrophages stimulates neovascularization in the rat ischemic limbs (9).
Altogether, these findings suggest that different cell types through the production of angiogenic growth factors can stimulate neovascularization in stem cell therapy. Recent study by Di Santo et al. demonstrated that conditional medium derived from endothelial progenitor cells stimulated the therapeutic angiogenesis of bone marrow cells transplantation, promoting tissue revascularization and functional recovery (8).
The present study documents for the first time on a small set of CLI patients that the amelioration of clinical and functional parameters in vivo is related to the capability of marrow-derived cells in secreting soluble proangiogenic factors. The absence of this mechanism may correlate with failure of treatment.
Taking into consideration these observations, we propose that the beneficial effect of bone-marrow stem cells transplantation is related to the essential paracrine action of factors, mainly VEGF, released from marrow-derived stromal cells, which support endothelial cells' proliferation, viability, and migration. Our data showed a correlation between the secretion of VEGF and the clinical outcomes in a small set of CLI patients at 12 months of follow up.
In summary, the promising experimental treatment for arterial occlusive disease using growth factors to stimulate neovascularization has to be considered. Targeting the VEGF signals may be a novel, efficient strategy for treating ischemic diseases.
Footnotes
Acknowledgments
We are grateful to Fondazione Luigi Califano and Fondazione Banco Napoli, which gives us the support needed to perform the research. A special thanks to all patients, Dr. Adriano Taddeo and Dr. Silvia Della Bella for technical assistance, and Dr. Paola Mantovano and Giuseppe Sica, who helped us in coordinating the clinical trial.
