Abstract
Diazoxide (DZ) is a pharmacological opener of ATP-sensitive K+ channels that has been used for mimicking ischemic preconditioning and shows protection against ischemic damage. Here we investigated whether diazoxide supplementation to University of Wisconsin (UW) solution has cellular protection during islet isolation and improves in vivo islet transplant outcomes in a rodent ischemia model. C57/B6 mice pancreata were flushed with UW or UW + DZ solution and cold preserved for 6 or 10 h prior to islet isolation. Islet yield, in vitro and in vivo function, mitochondrial morphology, and apoptosis were evaluated. Significantly higher islet yields were observed in the UW + DZ group than in the UW group (237.5 ± 25.6 vs. 108.7 ± 49.3, p < 0.01). The islets from the UW + DZ group displayed a significantly higher glucose-induced insulin secretion (0.97 ng/ml ± 0.15 vs. 0.758 ng/ml ± 0.21, p = 0.009) and insulin content (60.96 ng/islet ± 13.94 vs. 42.09 ng/islet ± 8.15, p = 0.002). The DZ-treated islets had well-preserved mitochondrial morphology with superior responses of mitochondrial potentials, and calcium influx responded to glucose. A higher number of living cells and less late apoptotic cells were observed in the UW + DZ group (p < 0.05). Additionally, the islets from the UW + DZ group had a significantly higher cure rate and improved glucose tolerance. This study is the first to report mitoprotective effects of DZ for pancreas preservation and islet isolation. In the future, it will be necessary to further understand the underlying mechanism for the mitoprotection and to test this promising approach for pancreas preservation and the islet isolation process in nonhuman primates and ultimately humans.
Keywords
Introduction
Islet transplantation has emerged as an alternative treatment for type 1 diabetes to restore euglycemia (36,39) but remains characterized by variable outcomes. Factors that affect transplant outcomes are largely dependent on the islet isolation procedure and donor variables, such as ischemia time. While donor factors, for the most part, are not subject to change, improvements in organ preservation and the islet manufacturing process offer opportunities to enhance transplant outcomes.
Ischemia and/or ischemia–reperfusion (IR) injuries have been shown to significantly decrease the quantity and quality of isolated islets (15,23,32). Current preservation solutions, such as the University of Wisconsin solution (UW) and histidine–tryptophan–ketoglutarate (HTK), are designed to protect organs from those injuries. However, these solutions do not prevent the deleterious effects of ischemia per se (7,10,33). Other attempts have been made to reduce ischemia-related injuries through the oxygenation of preservation solutions, both by physical and chemical means (5,6,13,28,46), with controversial results. In 2006, our group demonstrated that polyheme (polymerized human hemoglobin) improves islet isolation outcomes through the preservation of mitochondrial integrity (4). Another study has shown that adding nicotinamide supplements to the isolation medium ameliorates the injuries caused by mitochondrial oxidative stress (18). While the specific underlying mechanisms responsible for these observed benefits have not been identified, it is evident that isolation outcomes can be improved through preserving β-cell mitochondrial integrity and reducing oxidative stress.
Numerous studies have shown the involvement of mitochondrial adenosine triphosphate (ATP)-sensitive potassium (mitoKATP) channels in ischemic preconditioning (IPC) (22,37,41). While the role of mitoKATP channels is not as well defined as sarcolemmal ATP-sensitive potassium (sarcKATP) channels, it is known that they are a second subtype of KATP channels that regulate metabolism and membrane excitability and are specifically critical in β-cells for insulin secretion regulation (24,26,27). Recent evidence has suggested the involvement of mitoKATP in mitochondrial membrane potentials maintenance, ion homeostasis, and free radical (reactive oxygen) species (ROS) regulation (2,3,45). Diazoxide (DZ), a mitoKATP channel opener, has been successfully used to mimic IPC and displays cytoprotective effects in various ischemia models (9,11,17,38). Specific to pancreatic tissue, however, the impact of DZ has not yet been examined.
In this present study, our objective was to determine whether mitochondrial integrity preservation through the use of DZ could prevent β-cell ischemia-related injury and, consequently, improve islet function in vitro and in vivo. This study investigated the effects of DZ supplementation of common organ preservation solution and islet isolation process on islet isolation and transplant outcomes in a rodent ischemia model.
Materials and Methods
Animals
Male C57/B6 mice (Charles River Laboratory, Wilmington, MA, USA; 8–10 weeks of age) were used for this study. All animals were housed at the Biologic Resources Laboratory (BRL) at the University of Illinois at Chicago (UIC), and procedures involving animals were performed in accordance with the guidelines of the National Institute of Health and the Animal Care Committee (ACC) at UIC.
Pancreatic Cold Ischemic Model
The mice were divided into five groups: 1) control group (nonischemic control); 2) UW group (cold ischemic control): animal pancreata preserved with UW solution (ViaSpan, Duramed Pharmaceuticals, Pomana, NY, USA); 3) UW + DZ group: animal pancreata preserved with UW solution supplemented with 150 μM DZ (Sigma, St. Louis, MO, USA); 4) UW + DZ + 5-HD group: animal pancreata preserved with UW supplemented with both 150 μM DZ and 100 μM 5-hydroxydecanoate (5-HD; Sigma), a specific blocker of mitoKATP channels; 5) UW + 5-HD group. All animals were anesthetized by isoflurane inhalation using a vaporizer and masks (Viking Medical, Medford Lakes, NJ, USA). The mouse abdomen was opened along the midline, and small clips (Roboz Surgical Instrument, model RS-5420, Gaithersburg, MD, USA) were placed on the lower abdominal aorta and the thoracic aorta. Either a 25-gauge or 27-gauge catheter (Becton Dickinson, Copell, UT, USA) was inserted into the lower abdominal aorta, just above the small clips, and the pancreata were flushed with 8 ml of UW solution, with or without supplemental drugs (DZ and/or 5-HD), at a speed of 1 ml/min controlled by a syringe pump (model 22; Harvard Apparatus, Cambridge, MA, USA). The whole bodies of the mice from all four ischemic groups were then preserved at 4°C for either 6 or 10 h.
Islet Isolation
Following the cold ischemic preservation, islets were isolated from the pancreata by collagenase digestion and discontinuous Ficoll gradient separation, a modification of the original method of Lacy et al. for rat islet isolation (20). In brief, 0.224 mg/ml Collagenase P (Roche, Applied Science, Indianapolis, IN, USA) was dissolved in Hank's balanced salt solution (HBSS; Mediatech, Inc., Manassas, VA, USA), with or without drugs (DZ and/or 5-HD) and injected into pancreatic tissue via bile duct. The enzyme-infused pancreata were then excised and digested in 15 ml conical tubes at 37°C for 11 min. Postdigested tissues were washed twice with HBSS (Mediatech, Inc.) supplemented with or without drugs (DZ and/or 5-HD), and discontinuous Ficoll density gradients (1.108, 1.096, 1.069, and 1.037 kg/m3; Mediatech, Inc.) were used for islet purification. Purified islets were then cultured in Roswell Park Memorial Institute (RPMI) 1640 medium (Mediatech, Inc.) containing 10% fetal bovine serum (FBS; Sigma) at 37°C overnight prior to experimental procedures.
Islet Yield and Static Glucose Incubation
Islets bigger than 50 μm were manually counted for the islet yield. Static glucose incubation was used to compare glucose-stimulated insulin secretion. Briefly, five islets of approximately the same size were handpicked from each group, placed in Krebs–Ringer buffer (KRB; in mM: 119 NaCl, 4.7 KCl, 2.5 CaCl2, 1.2 MgSO4, 1.2 KH2PO4, 25 NaHCO3; all components were from Sigma) containing 2 mM glucose (Sigma) and 20 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES; Sigma) (five replicates) and incubated for 30 min. The islets were then stimulated with 14 mM glucose for 60 min at 37°C and 5% CO2. Following stimulation, 200 μl of supernatant was collected and frozen at −20°C for insulin assessment by ELISA (Mercodia, Uppsala, Sweden), according to the manufacturer's protocol. All samples were tested in duplicate.
Assessment of Mitochondrial Membrane Potentials and Morphology
Rhodamine 123 (Rh123; Sigma) was used to measure mitochondrial membrane potentials, as previously described (1,25). Briefly, 25–30 isolated islets from each group were incubated in KRB solution with 2 mM glucose and 10 ng/ml of Rh123 for 30 min at 37°C. The islets were then placed into a temperature-controlled microfluidic perifusion chamber, which was mounted on an inverted epifluorescence microscope (TE-2000U; Nikon, Melville, NY, USA). The islets were perifused by a continuous flow of KRB buffer at 37°C (pH 7.4) and then stimulated with 14 mM glucose. Rh123 fluorescence was excited at 490 nm, and the emission was measured at 530 nm. Images were collected with a charged coupled camera (Roper Cascade CCD; Photometrics, Tucson, AZ, USA). Data were normalized to the average basal fluorescence intensity that had been recorded during a 5-min period prior to the glucose stimulation. Changes in fluorescence were measured for 15 min. To examine the mitochondrial morphology, each group of islets was incubated with Rh123 in KRB solution for 15 min at a concentration of 10 ng/ml. The islets were then visualized using a Carl Zeiss LSM 510 confocal microscopy (Thornwood, NY, USA) equipped with 60× water immersion objective. The excitation line at 488 nm from the argon–krypton laser and the emission line were detected through a long-pass 505-nm filter. The intensity and the distribution of fluorescence were used to characterize morphology and integrity.
Intracellular Ca2+ Measurement
Intracellular Ca2+ measurements were performed as previously described (1,25). Briefly, 25–30 islets were incubated with 5 μM Fura-2/AM (a calcium indicator; Invitrogen, Carlsbad, CA, USA) for 30 min in KRB containing 2 mM glucose. The islets were then placed into a temperature-controlled microfluidic perfusion chamber mounted on an inverted epifluorescence microscope (TE-2000U, Nikon) and perifused by a continuous flow of KRB at 37°C (pH 7.4). A 14 mM glucose solution was then administered to the islets for 10 min. After a 15-min rinse with 2 mM glucose KRB, 30 mM potassium chloride (KCl) was then administered to the islets. Multiple islets were simultaneously observed with 10–20× objectives. Dual-wavelength Fura-2 was excited ratiometrically at 340 and 380 nm; the fluorescence changes were expressed as F340/F380. Metafluor/Metamorph (Universal Imaging Corporation, Bedford Hills, NY, USA) was used for image acquisition and analysis, and the images were collected with a high-speed, high-resolution charge-coupled device (Roper Cascade CCD). The percent changes of intracellular Ca2+ between both groups were calculated by subtracting maximum increase after glucose and KCl stimulations from the basal (2 mM glucose) Ca2+ level for each group.
Assessment of Apoptosis by Flow Cytometry
The isolated islets were dissociated using Accutase (Sigma) for 10 min at 37°C and then stained with Annexin V and propidium iodide (PI), using the TACS Annexin V-FITC staining kit (R & D Systems, Minneapolis, MN, USA). Briefly, dissociated islets were washed with phosphate-buffered saline (PBS; Mediatech, Inc.) and then incubated with Annexin V-FITC and PI solution for 15 min at room temperature in the dark. After another wash, the cells were resuspended in binding buffer and analyzed by flow cytometery (CyAn ADP Analyzer; Beckman Coulter, Fullerton, CA, USA). Annexin–- and PI–-labeled cells were classified as living cells. Annexin+- and PI--labeled cells were classified as early apoptotic cells and Annexin+- and PI+-labeled cells were classified as late apoptotic or necrotic cells. Four to five independent experiments were performed, and the data are presented as mean ± SD.
Islet Transplantation and In Vivo Evaluation
The isolated islets were assessed for in vivo function through allogenic transplantation under the kidney capsule of isogeneic diabetic C57/B6 mice. Induction of diabetes was achieved by intraperitoneal injection of streptozotocin (Sigma) at a dosage of 200 mg/kg of body weight. Mice with blood glucose levels higher than 20 mM or 360 mg/dl were considered recipients for islet transplantation. After overnight incubation, 350 islets were transplanted under the left kidney capsule in each mouse under isoflurane (HenrySchein, Columbus, OH, USA) anesthetization; the animal was placed on a surgically clean field below a temperature-controlled heat lamp. A left flank incision (~1 cm) was performed until the kidney was found. The kidney capsule was then carefully opened with the tip of a sterile 21-gauge needle. Islets, previously pelleted into polyethylene microtubing (PE50, Becton Dickinson), were then carefully delivered under the kidney capsule. The incision was closed with 5–0 monofilament nylon surgical sutures (Johnson & Johnson, New Brunswick, NJ, USA). Pain management was through a single postsurgery subcutaneous administration of buprenorphine (Zoopharm Pharmacy, Windsor, CO, USA) at a dose of 0.1 mg/kg. Blood glucose levels were monitored from the tail vein using a glucose meter (Alphatrak, Abbott Laboratories, Abbott Park, IL, USA) every day during the first week and every other day thereafter. Transplantation was considered successful if the blood glucose levels were lower than 210 mg/dl for 2 consecutive days. On day 14, an intraperitoneal glucose tolerance test (IPGTT, glucose 2 g/kg of body weight) was performed to assess graft function. All animals were fasted overnight, and blood glucose levels were measured at several time points (0, 15, 30, 60, 90, and 120 min). Area under the curve (AUC) was used to compare each group. Six weeks posttransplantation, recipients underwent a graft-bearing nephrectomy. Returning to hyperglycemia was interpreted as indirect evidence of islet graft function rather than spontaneous recovery of the native pancreas.
Statistical Methods
Statistical analysis was carried out by Student's t test and Pearson chi-square test. For multiple comparisons between each group, one-way ANOVA was used and then the significant differences determined using the Bonferroni post hoc test (Graphpad Software, La Jolla, CA, USA). Values of p < 0.05 were regarded as statistically significant.
Results
DZ-Supplemented UW Solution Increases Islet Yield
In the 6-h cold ischemia (CI) model, the UW + DZ group had a significantly higher islet yield compared to the UW group (237.5 ± 25.6 vs. 108.7 ± 49.3 islets/mouse, respectively; p < 0.01) (Fig. 1A). The difference between the UW + DZ and the control group was not significant (p = 0.156). To test whether the protective effect of DZ was specifically due to the opening of the mitoKATP channels, 100 μM of 5-HD (a specific mitoKATP closer) was added to the UW + DZ solution during pancreas preservation and islet isolation. Pancreata from the UW + DZ + 5-HD group had significantly lower islet yields compared to the group without 5-HD (p < 0.01); however, the islet yields were similar to the UW group (117 ± 33.1 vs. 108.7 ± 49.3 islets/mouse, respectively; p > 0.05) (Fig. 1A). The UW + 5-HD as a negative control of DZ also had low islet yield, even lower than the UW group (65.4 ± 12.3).
Isolated islet yield in the rodent pancreatic CI preservation model. (A) Islet yields from the nonischemia control (n = 3) and four 6-h cold ischemia (CI) groups: the University of Wisconsin (UW) (n = 7), the UW + diazoxide (DZ) (n = 8), the UW + DZ + 5-hydroxydecanoate (5-HD) (n = 7), and UW + 5-HD (n = 5). **p < 0.01. (B) Islet yields from two 10-h CI groups: the UW (n = 3), the UW + DZ (n = 4), the UW + DZ + 5-HD (n = 5), and the UW + 5-HD (n = 4). *p = 0.02.
In the 10-h CI model, both the UW and the UW + DZ groups had much lower islet yields than in the 6-h CI model. Yet, the UW + DZ group still had a significantly higher islet yield than the UW group (108.2 ± 22.1 vs. 53.2 ± 12.3 islets/mouse, respectively; p = 0.02) (Fig. 1B). Similar to the 6-h model, both the UW + DZ + 5-HD and UW + 5-HD groups had much lower yields (24 ± 12.4 and 21 ± 5.1, respectively).
DZ-Supplemented UW Solution Improves Glucose-Stimulated Insulin Secretion and Total Insulin Content
Insulin secretions were performed at both basal (2 mM) and stimulated (14 mM) glucose concentrations. For comparison between the UW + DZ group and the UW group, basal secretions were similar (0.264 ng/ml ± 0.05 vs. 0.332 ng/ml ± 0.12, p = 0.29); however, the UW + DZ group had a higher insulin secretion when stimulated with 14 mM glucose with 0.97 ng/ml ± 0.15 versus the UW group with 0.758 ng/ml ± 0.21 (p = 0.009) as shown in Figure 2A. The addition of 5-HD in UW + DZ did not have a negative impact on the 14 mM glucosestimulated insulin secretion but even secreted more insulin (1.30 ng/ml ± 0.67), but this was not significantly different from the UW + DZ group (p = 0.55). For the UW + 5-HD group, the stimulated insulin level was significantly lower than both the UW and UW + DZ groups (0.262 ng/ml ± 0.14, p = 0.031). With the addition of 150 μM of DZ to control islets during the static incubation, both basal and glucose-stimulated insulin secretions were almost completely inhibited (Fig. 2A).
Glucose-stimulated insulin secretion and total insulin content of islets. (A) 2 mM (first column) and 14 mM (second column) glucose-stimulated insulin secretion (mean ± SD) for nonischemia control islets (n = 3), four 6-h CI groups: the UW, the UW + DZ, the UW + DZ + 5-HD, and the UW + 5-HD (n of each group = 5). **p = 0.009, *p = 0.031. (B) Total insulin content measurement for the UW control islets (n = 7), the UW + DZ (n = 7), UW + DZ + 5-HD (n = 7), and UW + 5 = DH (n = 5). **p = 0.002.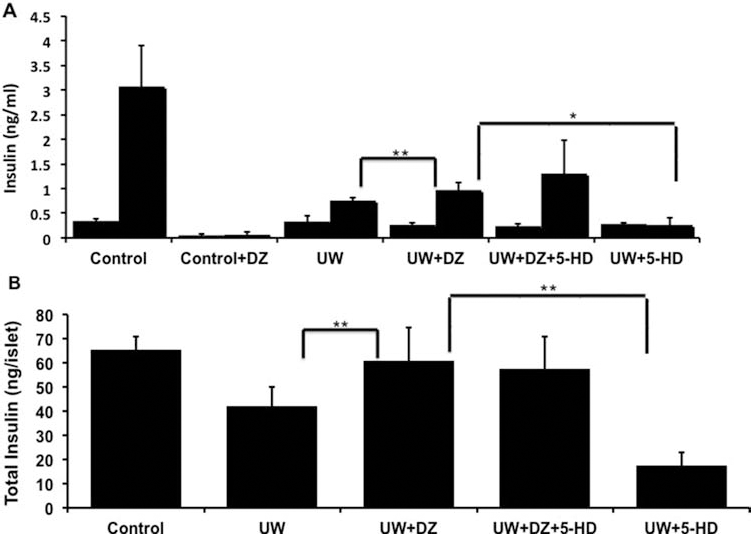
Additionally, the islets from the UW + DZ group had significantly higher insulin contents (60.96 ng/islet ± 13.94) than the islets from the UW group (42.09 ng/islet ± 8.15; p = 0.002). Similar to insulin secretion data, the UW + DZ + 5-HD had comparable total insulin content (57.65 ng/islet ± 13.22) to the UW + DZ group. The UW + 5-HD group had significantly lower insulin content (17.61 ng/islet ± 5.14, p < 0.01) (Fig. 2B).
DZ-Supplemented UW Solution Preserves Mitochondrial Integrity
Observation of mitochondrial morphology, using Rh123, indicated that the mitochondrial structure in the UW + DZ group had less mitochondrial swelling, fragmentation, and aggregation than the UW group (Fig. 3). Additionally, the islets from the UW + DZ group had significantly improved mitochondrial function, as measured by glucose-stimulated mitochondrial potential changes when stimulated with 14 mM glucose, than islets from the UW group (28.73% ± 9.87 vs. 4.59% ± 1.23 for the UW + DZ group, respectively; p < 0.05) (Fig. 4A, B).
The integrity of mitochondrial morphology. Two representative confocal images of rhodamine 123 (Rh123)-stained islets from the UW group (A, B) and the UW + DZ group (C, D). Contrast has been balanced to reveal the details of mitochondrial morphology. N = 3–4 for each experimental condition, 10–20 islets/group. Scale bar: 5 μm. Alteration in mitochondrial membrane potential changes induced by glucose. Islets were loaded with Rh123, and the intensity of Rh123 fluorescence was monitored in response to 14 mM glucose stimulation. The confocal images were digitally sharpened and contrast balanced to reveal the details of mitochondrial morphology. (A) A representative figure of observed mitochondrial potentials changes. (B) A summary of mitochondrial potential changes, represented as percentage over basal level of the UW + DZ- and the UW-treated islets. Data represent mean ± SD of four to five experiments (islets n = 15–20) performed. *p < 0.05.

DZ-Supplemented UW Solution Improves the β-Cell Calcium Signaling in Response to Glucose
To determine if preserved mitochondrial morphology and function using DZ was associated with alterations in calcium signaling, the calcium influx responses were examined, using the fluorescence imaging of Fura-2. The islets from UW + DZ group showed improved responsiveness to glucose and potassium challenges (Fig. 5A). The Ca2+glu index of the islets from the UW + DZ group was 122.3% ± 20.87 vs. 56.1% ± 13.45 for islets from the UW group (p < 0.05). The Ca2+KCl index of islets from the UW + DZ was 132.1% ± 22.2 vs. 98.8% ± 10.9 for islets from the UW group (p < 0.05) (Fig. 5B).
Intracellular calcium concentration responses to glucose and KCl. (A) Representative record of islet responses from the UW + DZ and the UW groups. Islets were loaded with 5 μM Fura 2-AM and then stimulated with 14 mM glucose and 30 mM KCl. Calcium changes are reflected by the ratio changes of 340/380 nm. The responses of 15–30 islets tested in both groups are recorded. (B) The delta [Ca2+]glu and [Ca2+]KCl of the UW + DZ and the UW groups. *p < 0.05.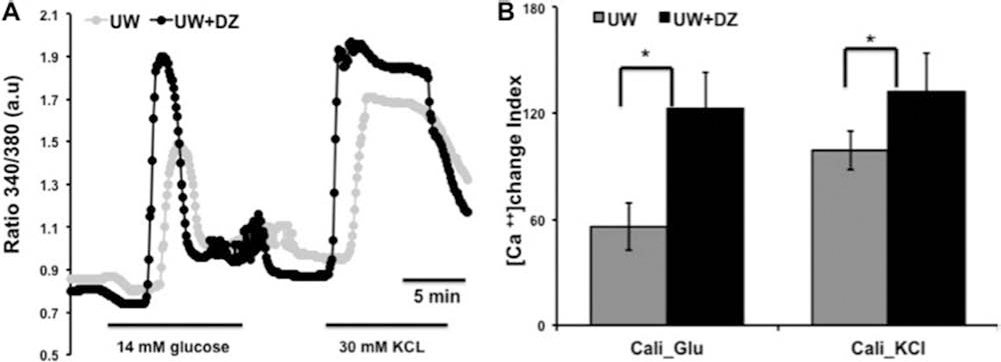
DZ-Supplemented UW Solution Decreases Apoptosis
Annexin V Flow Cytometric Analysis

Annexin V and propidium iodide (PI) staining for the percentage of live, apoptotic, and necrotic cells present in the dissociated cell population of ischemia groups and nonischemia control group. The p values were determined between University of Wisconsin (UW) (n = 3) and UW + diazoxide (DZ) (n = 3) groups. Data from three separate experiments are presented, each with five independent samples (mean % ± SD). 5-HD, 5-hydroxydecanoate.
DZ-Supplemented UW Solution Improves Islet In Vivo Graft Function
The percentage of mice cured at week 2 was significantly higher for the UW + DZ group (n = 7) than for the UW group (n = 7) (85.7% vs. 42.5%, respectively; p < 0.05) (Fig. 6A). Mice transplanted with islets from the UW + DZ group also displayed a significantly lower number of days required to reach normoglycemia than the mice transplanted with islets from the UW group (7.2 days ± 3.3 vs. 12.4 days ± 2.5, respectively; p < 0.05) (Fig. 6B). The 28-day glycemic profile of mice transplanted with islets from both groups is depicted in Figure 7A. In order to verify in vivo graft function in responses to glucose challenge, IPGTTs were conducted at day 14. Mice in the UW + DZ group had a significant decrease of glycemic AUC, compared to mice in the UW group (13,825.7 mg/dl ± 2,901.9 vs. 24,351.4 mg/dl ± 6,261.4, respectively; p < 0.01) (Fig. 7B, C).
Cure rate and days to reach normoglycemia after islet transplantation into diabetic mice. (A) The number of mice reaching normoglycemia at day 14 after 350 islets were transplanted from the UW + DZ (n = 6 of 7) and the UW control (n = 3 of 7) mice. *p < 0.05. (B) The number of days to reach normoglycemia in cured mice (UW + DZ: 7.2 days ± 3.3 vs. UW: 12.4 days ± 2.5, *p < 0.05). In vivo graft function. (A) The 28-day glycemic profiles (mean ± SEM) for the UW group (n = 7) and the UW + DZ group (n = 7). (B) The 120-min glycemic profiles of intraperitoneal glucose tolerance tests (IPGTTs; mean ± SEM). (C) Area under the curves (AUCs) of IPGTT were calculated for both groups (mg/dl, mean ± SEM), *p < 0.05.

Discussion
This is the first report to show that DZ supplementation during pancreas preservation and during the islet isolation process improves islet yield and function through preserving mitochondrial integrity. Considerable evidence supports the beneficial effects of DZ in IR (2,16,40, 41,44,47). While the cytoprotection mechanism of DZ has not been completely elucidated, postischemic mitochondrial protection has been shown to occur though sustaining mitochondrial membrane potentials, reducing calcium influx, preventing opening and release of cytochrome C, and sparing ATP during the period of sustained ischemia (16,17,37).
DZ supplementation led to a significantly increased postisolation islet yield in both the 6- and 10-h ischemia models. The effect of DZ on islet yield could be abolished by the addition of 5-HD, which is a specific mitoKATP channel closer. This indicates that the higher islet yield with DZ supplementation was achieved by keeping mitoKATP channels open during prolonged ischemia.
Our results also demonstrated that the islets in the UW + DZ group had a significantly enhanced glucosestimulated insulin secretion and total insulin content, compared to the islets in the UW group. Interestingly, the effect of DZ on insulin secretion was not reversed by the addition of 5-HD. This finding suggests that the effect of DZ on sarcKATP channels may also contribute to this observed preservation in islet function. The effect of DZ on insulin secretion has been extensively studied in pancreatic β-cells and demonstrated to specifically function through sarcKATP channel modulation. In rat islets transplanted into a diabetic environment, DZ causes prolonged improvement of β-cell function, accompanied with increased insulin content and preproinsulin mRNA levels (14). DZ also preserves insulin stores and pulsatile secretion by inducing β-cell rest (34,42) and protects rat islets against the toxic effect of streptozotocin (8,19). We speculated that in addition to preventing ischemia or IR-related mitochondrial injury, the attenuation of ischemic-induced calcium overload via sarcKATP channel opening and resting β-cell membrane potential could prevent insulin granule dumping during pancreas preservation and the islet isolation process; this was supported in part in this study, as the DZ-treated islets had significantly higher total insulin content. Intracellular calcium overload has also been long considered a precursor to IR-related injury and cell death. In pancreatic IPC, DZ may decrease intracellular calcium overload either by sparing ATP production to support ATP-dependent calcium pumps or by stabilizing membrane potentials through activating sarcKATP channels and preventing voltage-dependent calcium influx. However, the affinities of DZ to mitoKATP and sarcKATP channels in pancreatic β-cells are not known. In cardiomyocytes, the affinity of DZ to the mitoKATP channel (0.4 μM) has been shown to be roughly 2,000-fold higher than the affinity for the sarcKATP channel (855 μM) (12,31), but it is not known whether this applies similarly to the situation in pancreatic β-cells. In future studies, the underlying mitoprotective mechanism has to be determined, especially related to which stage: either ischemia or reperfusion or both.
The islets from the UW + DZ group displayed better preserved mitochondria morphology than islets from the UW group, with a more perinuclear distribution, less swelling, and less mitochondrial fragmentation. These results were also in accordance with previous studies in cardiomyocytes, which have demonstrated that DZ treatment preserves mitochondrial morphology (30). Additionally, the islets from the UW + DZ group had superior calcium responses to glucose and potassium challenge, as measured by in vitro calcium influx assays. Because glucose-stimulated calcium influx depends on mitochondrial integrity and energetic status, this measurement can be used as an indirect assay for mitochondrial integrity and β-cell function. The results of our calcium influx assays suggested that DZ-induced mitochondria preservation consequently preserves calcium influx and insulin secretion.
While the percentage of early apoptotic cell populations was similar between the UW and UW + DZ groups, the UW + DZ group had significantly smaller population of necrotic/late apoptotic cells and significantly larger population of living cells when compared to the UW group. These findings suggest that DZ may reduce IR-related injury by suppressing or stopping early apoptotic cells from entering late apoptotic status. As previously discussed, DZ protects mitochondria during ischemia. Ischemia–reperfusion studies in the heart and brain have also shown that DZ reduces ROS formation during reperfusion (21,29). In the presence of ROS, the calcium overload in damaged mitochondria can facilitate the opening of the mitochondrial permeability transition pore (mPTP). The opening of the mPTP further compromises cellular energetics and leads to increased cell necrosis and apoptosis. Ischemia and reperfusion injures cells by a variety of mechanisms, and DZ seems to act at multiple levels (30,43).
Our in vivo data indicated that the islets isolated from the UW + DZ group had a higher percentage cure rate and reduced the number of days needed to reach normoglycemia when compared to the UW group. IPGTT at day 14 showed that the islets treated with the UW + DZ had superior responses to glucose challenge, confirming the in vitro findings of the improved islet function with DZ treatment.
Overall, significant beneficial effects were observed with the addition of DZ to the pancreas preservation and islet isolation processes via a direct involvement of mitoprotection. While the specific mechanism of cellular and mitochondrial protection with DZ is still under investigation, our results suggest a combination of different cytoprotective mechanisms involved and are consistent with findings in other tissues studies. There are many unanswered questions or limitations in this study. Most of the results were derived from direct observation and did not provide direct information on the molecular mechanism of diazoxide mitoprotection on β-cells. For example, an association of mitoKATP-mediated ROS production for β-cell protection was not determined. The exact anti-apoptotic mechanism of diazoxide when supplemented to common organ preservation solutions is still unclear. Several recent studies indicated that heat shock protein 90 (Hsp90) mediates antiapoptotic effect induced by diazoxide preconditioning (35,48). It has been shown that Hsp90 interacts directly with various components of the tightly regulated programmed cell death machinery, upstream and downstream of the mitochondrial events. Diazoxide preconditioning provides cardioprotection by translocation of connexin 43 (Cx43) to the inner mitochondrial membrane through an Hsp90-dependent pathway. Additionally, it is unclear either how much involvement sarcKATP channels have in the β-cell protection. Last, but not the least, a definite long-term outcome on in vivo islet graft function needs to be determined since it is extremely important for clinical application.
In conclusion, this study established a new approach for the application of mitoKATP channel openers to improve islet isolation and transplant outcomes. In the future, it will be necessary to further understand the underlying mechanism for the mitoprotection and test this promising approach for pancreas preservation and the islet isolation process in humans.
Footnotes
Acknowledgments
We thank Dr. Chen ML for her excellent technical assistance. This study was supported by a startup grant by the College of Medicine at the University of Illinois at Chicago (J.O.), The Chicago Diabetes Project, a research grant by the Gift of Hope Foundation in Illinois (J.O. and Y.W.), and NIH R01 DK091526 (J.O.). The authors declare no conflicts of interest.
