Abstract
We previously reported a small-scale study on the efficacy of histidine-tryptophan-ketoglutarate (HTK) solution versus University of Wisconsin (UW) solution on pancreas preservation for islet isolation. In this large-scale, retrospective analysis (n = 252), we extend our initial description of the impact of HTK on islet isolation outcomes and include pancreatic digestion efficacy, purification outcomes, and islet size distribution. Multivariable linear regression analysis, adjusted for donor age, sex, BMI, cold ischemia time, and enzyme, demonstrated similar results for the HTK group (n = 95) and the UW group (n = 157), including postpurification islet yields (HTK: 289,702 IEQ vs. UW: 283,036 IEQ; p = 0.76), percentage of digested pancreatic tissue (HTK: 66.9% vs. UW: 64.1%; p = 0.18), and islet loss from postdigestion to postpurification (HTK: 24,972 IEQ vs. UW: 39,551 IEQ; p = 0.38). Changes in islet size between the postdigestion and postpurification stages were comparable within each islet size category for HTK and UW (p = 0.14–0.99). Tissue volume distribution across purification fractions and islet purity in the top fractions were similar between the groups; however, the HTK group had significantly higher islet purity in the middle fractions (p = 0.003–0.008). Islet viability and stimulation indices were also similar between the HTK and the UW groups. In addition, we analyzed a small sample of patients transplanted either with HTK (n = 7) or UW (n = 8) preserved islets and found no significant differences in posttransplant HbA1c, β-score, and frequency of insulin independence. This study demonstrates that HTK and UW solutions offer comparable pancreas preservation for islet transplantation. More in vivo islet outcome data are needed for a complete analysis of the effects of HTK on islet transplantation.
Keywords
Introduction
The quality of pancreas flush and preservation is one of the most important determining factors for the successful grafting of both whole pancreata and isolated islets. At present, two preservation solutions are primarily utilized for abdominal organ preservation: University of Wisconsin solution (UW, DuPont Pharma, Mississauga, ON, Canada) and histidine-tryptophan-ketoglutarate (HTK, Essential Pharma, Newtown, PA, USA). Each preservation solution possesses a unique composition, as shown in Table 1. While UW has been the preferred preservation solution for greater than 20 years (5,6), HTK, originally developed by Brestchneider as a cardioplegic solution (8), has been gaining favor as an alternative to UW. Several major studies and clinical trials have demonstrated the clinical equivalence of HTK when compared to UW for abdominal organ preservation and transplant outcomes (12,20,24,29) and specifically for whole pancreas transplantation (4,10,11). Notably, since 1995, a number of studies have assessed the ability of HTK to preserve pancreata intended for islet isolation, with results similar to those obtained with other organ preservation solutions and paralleling those demonstrated for other abdominal organs (7,15,16,30). However, these studies have been limited either by small sample size or incomplete analysis of isolation outcomes.
Organ Preservation Solution Compositions

HTK, histidine-tryptophan-ketoglutarate; UW, University of Wisconsin.
Islet transplantation requires supplementary procedures not necessitated by whole-organ transplantation. It is complicated by an intricate islet isolation process consisting of four primary stages: organ preservation, enzymatic perfusion, pancreas digestion and dissociation, and islet purification. Therefore, success of islet isolations is dictated by the effectiveness of each consecutive step, and the quality of the initial pancreas flush and preservation directly impacts the final islet product constitution that is achieved. It is worth noting that, in addition to UW and HTK solutions used for islet isolation, enormous attempts have been made to reduce ischemia injuries through the oxygenation of UW solution such as two-layer storage method (TLM), a combinatorial UW with perfluorochemicals (PFCs), and show improved islet isolation outcomes (14,18). However, the organs preserved in the TLM are not included in the study since our center has not adopted the TLM as organ preservation method.
In a previous small-scale study, we demonstrated that HTK is equivalent to UW in preserving pancreata for islet isolation, with regard to general isolation outcomes, including islet yield, viability, and in vitro function (23). In this single-center, large-scale study, we reexamined isolation outcomes, focusing on the impact of the preservation solution, either HTK or UW, on pancreatic digestion efficacy, purification outcomes, and isolated islet size distribution.
Materials and Methods
Pancreas Procurement and Isolation Activities
Organ procurement organizations (OPOs) provided pancreata, with consent from donors. The organs were flushed with either HTK (n = 95) or UW (n = 157), depending upon the protocols incorporated by individual OPOs, and transported to the University of Illinois at Chicago (UIC). The islet isolation procedure, including digestion, purification, and culture, was performed for all pancreata according to the previously described protocol (13,21,25). Upon arrival, the pancreas was surface-decontaminated and trimmed of excess fat. The pancreas was then perfused, via the pancreatic duct, with the digestive enzyme, collagenase from several sources (Serva Premium and Serva GMP from Serva Electrophoresis, Heidelberg, Germany; Sigma V from Sigma-Aldrich, St. Louis, MO, USA; Roche Liberase from Roche, Indianapolis, IN, USA). Tissue digestion and islet dissociation were achieved using a modified Ricordi semiautomated method (22).
After digestion was complete, the collected tissue was washed to remove traces of enzyme and incubated in UW, on ice, for 30 min. The refined UIC-UW/Biocoll (UIC-UB) continuous density gradient (3), consisting of a mixture of a high-density solution [1.078 g/ml: 40% Biocoll (Cedarlane, Burlington, NC, USA) and 51% UW] and a low-density solution (1.068 g/ml: 30% Biocoll and 70% UW), was used for the purification procedure. Up to 45 ml of tissue were purified in a single operation of the COBE 2991 Cell Separator (CARIDIAN BCT, Lakewood, CO, USA). Following the centrifugation process, the tissue was collected in 12 fractions. The first two fractions were discarded due to minimal tissue volume (often less than 0.01 ml) and being primarily composed of ductal and adipose cells. In each of the remaining 10 fractions, corresponding to the aforementioned continuous gradient from 1.068 to 1.078 g/ml, a fluid and tissue volume of 30 ml was collected and then recombined based on the percentage of islet purity. Recovered tissue with an islet purity of >69%, 40–69%, and < 40% were defined as the top, middle, and bottom fractions, respectively. A small percentage of isolations required multiple sequential purifications due to a postdigestion tissue volume of greater than 45 ml.
Assessment of Islet Yield, Size Distribution, Purity, and Tissue Volume
Islet yield, size, and purity assessments were manually performed, using dithizone (a zinc chelating agent; Sigma) staining under light microscopy (Leica MZ95; Buffalo Grove, IL, USA) at two time points: postdigestion and postpurification. Islet yield was measured both in actual islet number and islet equivalent (IEQ), a volumetric quantification of islet mass, in which large islets contribute more to the total IEQ count than small islets. Eight discrete categories were designated for islet size quantification: 50–100, 100–150, 150–200, 200–250, 250–300, 300–350, 350–400, and >400 μm.
The islet purity of each postpurification fraction was specified as the estimated percentage of islet tissue present within the total tissue composition. The absolute tissue volume of each fraction was visually approximated, and the relative tissue volume was calculated as the percentage of tissue volume of each fraction versus the total collected tissue volume.
Assessment of Islet Quality and Function
Postpurification islet viability was determined using inclusive and exclusive fluorescent staining with Syto-Green (Invitrogen, Carlsbad, CA, USA) for live cells and ethidium bromide (Sigma) for dead cells (21,31). A static glucose-stimulated insulin release (GSIR) assay was used to evaluate islet function, as previously described (2). Briefly, 10 purified islets were individually selected and preincubated with Krebs–Ringer bicarbonate buffer (KRBB; in mM: 119 NaCl, 4.7 KCl, 2.5 CaCl2, 1.2 MgSO4, 1.2 KH2PO4, 25 NaHCO3, 2 glucose) solution containing 1.67 mM (low) glucose (all from Sigma) and 20 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES; Life Technologies, Grand Island, NY, USA) for 1 h. The islets were then transferred into KRBB solution containing 16.7 mM (high) glucose for another 1 h. The secreted insulin levels of each incubation phase were measured using conventional enzyme-linked immunosorbent assay (ELISA, Mercodia, Uppsala, Sweden); a stimulation index (SI) was calculated by dividing insulin release during the high-glucose stimulation (16.7 mM) by insulin release during the basal glucose (1.67 mM) stimulation.
Exclusion Criteria for Analysis
Isolations that were not completed due to technical errors or equipment malfunction, as well as isolations in which complete data for all primary variables were unavailable, were not included in the analysis (approximately 28% and 25% of the isolation data from the HTK-preserved pancreata and the UW-preserved pancreata, respectively).
Human Islet Transplantation
After the portal vein was accessed percutaneously under fluoroscopic and ultrasound guidance, the islets were resuspended into 60-ml syringes and slowly injected into the intraportal catheter. During infusion, syringes were turned constantly to avoid sedimentation or clumping of islets. Heparin was administered throughout the procedure for a total dose of 5,000 units pretransplant followed by enoxaparin (Lovenox®) 30 mg subcutaneously twice daily for 1-week posttransplant.
The clinical outcomes were determined at 6 months posttransplant (the UW n = 5, three from male donor and two from female donor; the HTK n = 3, one from male donor and two from female donor). The recipients who required a second transplant prior to the sixth month after first transplant were excluded from the analysis because the islet graft function and survival may be affected by combined and cumulative effects from other factors such as immunosuppressant toxicity. The beta-score was calculated as an indicator of islet graft function by measuring fasting plasma glucose values based on glucose oxidase methodology (Beckman Coulter Unicel Synchron DXC 800, Brea, CA, USA), glycated hemoglobin (HbA1c) using high-performance liquid chromatography (HPLC; Variant II Turbo Hemoglobin A1C Recorder pack 270-2415, Bio-Rad, Hercules, CA, USA), stimulated C-peptide levels using a quantitative chemiluminescent immunoassay (CLIA kit for human, KA2802, Abnova, Walnut, CA, USA), and insulin independence with or without insulin requirement.
Statistical Analysis
For the analysis of donor, pancreas, and isolation characteristics, as well as postpurification viability, and clinical outcomes, the results were expressed as mean ± standard deviation (SD) and percentage. Differences between HTK and UW groups were analyzed by using unpaired Student's t tests and chi-square (Fisher's exact, as appropriate) tests. Level of statistical significance for these comparisons was set at p < 0.05.
For the statistical analysis of digestion efficacy and purification outcomes, SAS version 9.2 (Cary, NC, USA) was used. Multivariable linear regression was used to compare HTK and UW, adjusting for age, sex, body mass index (BMI), cold ischemia time (CIT), and enzyme used, for the following outcomes: digestion time and efficacy; postdigestion and postpurification IEQ and the difference between postdigestion and postpurification IEQ; percentage of trapped islets; postdigestion and postpurification IEQ per gram of pancreas; percentage change between postdigestion and postpurification IEQ in each size group; absolute and relative tissue volume; and islet purity of each density range (fraction). Whether the effect of solution (HTK vs. UW) on absolute and relative tissue volume was modified by CIT was tested for each density layer, but no interactions were found to be significant (i.e., any differences in the effect of HTK vs. UW on volume did not differ across CIT strata). The effect modification by CIT was similarly explored for digestion time and efficacy, percentage of trapped islets, and IEQ per gram of pancreas, and no interactions were found to be significant.
Multivariable mixed linear regression models were used to compare distributions between HTK and UW for the percentage change between postdigestion and postpurification IEQ across islet size groups and absolute and relative volumes and purity across density ranges, adjusting for age, sex, BMI, CIT, and enzyme. These mixed models incorporated the correlation due to clustering by specifying the unique isolation identification number as the unit of cluster and specified the autoregressive covariance structure with empirical standard errors. The level of statistical significance for these multivariable models was set at p < 0.01 to minimize type 1 error with the multiple comparisons.
Results
Donor, Pancreas, and Isolation Characteristics
The comparison of donor, pancreas, and isolation characteristics between the HTK and the UW groups is summarized in Table 2. No significant differences were found between the two groups with regard to donor age (p = 0.68), donor BMI (p = 0.36), or donor sex (p = 0.86). A significant difference was observed for CIT, for which the HTK group had a longer ischemia time than the UW group (p = 0.02). Additionally, pancreas mass was significantly heavier in the UW group than in the HTK group (p = 0.02). The frequency of use of each enzyme type was similar between groups (p = 0.86).
Digestion Efficacy and Islet Yield
The average digestion time was not found to be significantly different between the HTK and the UW groups (15.5 min vs. 14.8 min, respectively; p = 0.24), when adjusted for age, sex, BMI, CIT, and enzyme. Enzyme digestion efficacy, calculated by dividing the mass of the digested tissue by total pancreas mass, was similar between the HTK and the UW groups (66.9% vs. 64.1%, respectively; p = 0.18).
Both the adjusted mean postdigestion and postpurification IEQ were marginally higher in the HTK group than in the UW group but were not significantly different (postdigestion IEQ: 329,253 for HTK vs. 308,008 for UW; p = 0.34; postpurification IEQ: 289,702 for HTK vs. 283,036 for UW; p = 0.76), as shown in Figure 1. The difference between mean postdigestion and postpurification IEQ was similar for both groups (39,551 for HTK vs. 24,972 for UW; p = 0.38). The adjusted percentage of trapped islets in the postdigestion sample was similar between the groups (HTK: 18.1% vs. UW: 16.9%; p = 0.71). Additionally, the adjusted postdigestion and postpurification IEQ per gram of pancreas were not significantly different between the groups (postdigestion IEQ: 3,379.2 for HTK and 3,377.2 for UW; p = 0.99; postpurification IEQ: 2,810.8 for HTK and 2,951.0 for UW; p = 0.53).

Islet quantification (IEQ): postdigestion, postpurification, and the difference between postdigestion and postpurification (islet loss via purification). The number of isolations analyzed was 95 and 157 for histidine-tryptophan-ketoglutarate (HTK) and University of Wisconsin (UW) solution, respectively. Data were adjusted for age, sex, body mass index (BMI), cold ischemia time (CIT), and enzyme. Statistical significance was set at p < 0.01. IEQ, islet equivalent.
Donor, Pancreas, and Isolation Characteristics
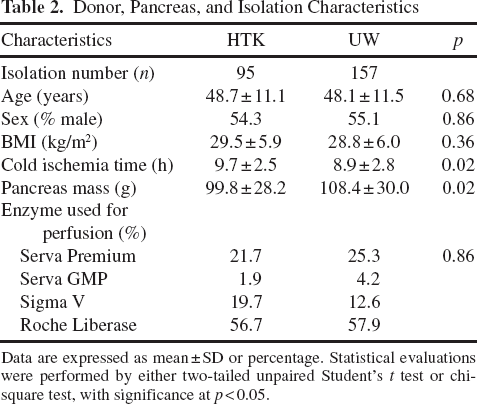
Data are expressed as mean ± SD or percentage. Statistical evaluations were performed by either two-tailed unpaired Student's t test or chi-square test, with significance at p < 0.05.
Islet Size Distribution
The islet size distribution change between the postdigestion and postpurification stages, as expressed by the adjusted percentage change of islets within each size category, did not differ between the HTK and the UW groups for any of the eight size categories (Fig. 2). Using mixed model analysis, the results indicated that the overall distribution across size groups also did not differ between the groups (p = 0.55).

IEQ change (%) between the postdigestion and postpurification stages, across the eight islet size categories. The number of isolations analyzed was 95 and 157 for HTK and UW, respectively. Data were adjusted for age, sex, BMI, CIT, and enzyme. Statistical significance was set at p < 0.01.
Total Tissue Volume and Islet Purity Following Purification
The distribution of tissue volume across purification fractions, compared between the HTK and the UW groups, is summarized in Figure 3. There were no significant differences between the two groups with regard to the tissue volume distribution within the purification fractions, expressed as either absolute volumes (p = 0.07–0.84) or as proportions to the overall tissue volume (p = 0.016–0.94). The overall volume distribution across purification fractions did not differ between the HTK and the UW groups (absolute volume: p = 0.12; relative volume: p = 0.52) in the mixed model analysis. The isolated islet purity within each purification fraction was also compared between the two groups (Fig. 4), with p values ranging from 0.003 to 0.47. All fractions demonstrated similar purity values, except for fractions 6 and 7, of which the HTK group had a significantly higher purity (p values of 0.008 and 0.003, respectively). In the mixed model analyses, the overall purity distribution across purification fractions did not differ between the HTK and the UW groups (p = 0.61).

Tissue volume distributions across purification fractions. (a) Absolute tissue volume distribution (ml). (b) Relative tissue volume distribution (%). The number of isolations analyzed was 95 and 157 for HTK and UW, respectively. Data were adjusted for age, sex, BMI, CIT, and enzyme. Statistical significance was set at p < 0.01.

Islet purity distribution across purification fractions. The number of isolations analyzed was 95 and 157 for HTK and UW, respectively. Data were expressed as percentages and adjusted for age, sex, BMI, CIT, and enzyme. Sample size ranged from 167 to 249 across fractions. Statistical significance was set at p < 0.01.
Isolated Islet Quality
Islet viability after purification was similar between the two groups (HTK: 90.6% vs. UW: 90.2%; p = 0.63), as were the GSIR stimulation indices (HTK: 3.81 vs. UW: 3.34; p = 0.14), when adjusted for age, sex, BMI, CIT, and enzyme.
In Vivo Islet Graft Function
The clinical transplant outcomes 6 months posttransplant from the isolated islet preparations either preserved in HTK (n = 7) or UW (n = 8) were analyzed (Table 3). Results indicated that the HTK and UW groups had no statistically significant differences in the frequency of insulin independence (HTK: 3/7 = 43% vs. UW: 6/8 = 75%; p = 0.31), glycated hemoglobin (HbA1c; HTK: 5.9 vs. UW: 6.1; p = 0.30), or beta-score for islet graft function (HTK: 4.3 vs. UW: 5.5; p = 0.23).
Discussion
The results from this large-scale study demonstrate that both HTK and UW organ preservation solutions are comparable in their ability to preserve pancreata intended for islet isolation. This finding further supports our previous results.
Although considerable effort has been devoted to expanding the clinical application of islet transplantation for the treatment of patients with type 1 diabetes mellitus, many variables continue to pose challenges for clinicians. Among these, the quality of pancreas flush and preservation is a key factor contributing to the outcome of the islet isolation procedure. For example, poor pancreatic flush and long cold ischemia duration often result in a lower digestion efficacy due to cellular edema and structural changes of collagen (9). Such endocrine and exocrine tissue injury primarily occurs during pancreas cold ischemic preservation but can be further influenced by isolation factors, such as organ reperfusion and associated reperfusion injuries, enzymatic digestion, centrifugation, and purification. Due to the multiphase processes inherent in the islet isolation procedure, which can exacerbate the progression, the selection of a suitable preservation solution is even more critical for islet transplantation than for whole pancreas transplantation. HTK and UW each have a distinct composition and differing pathways of action to prevent cellular injury; UW prevents cellular edema primarily via the osmotically active impermeants raffinose, lactobionate, and hydroxyethyl starch, while HTK achieves this via histidine, mannitol, tryptophan, and ketoglutarate.
In the present study, we compared the impact of preservation solution during the islet isolation process through the analysis of pancreatic digestion efficacy, purification outcomes, and islet size distribution. In terms of digestion efficacy, no significant difference was found between the HTK and the UW groups, with adjustment for age, sex, BMI, CIT, and enzyme. The extent of enzymatic cleavage, based on the percentage of trapped islets (islets encased in exocrine tissue) observed after the digestion phase, also did not differ between the groups. These combined data suggest that both preservation solutions similarly impact pancreatic digestion efficacy.
The analysis of purification outcomes is often used as an indirect measurement of pancreatic cellular edema. For single cells, the extent of edema is often measured by means of a cell resistance assay. However, the same technique cannot be applied to whole islets, which possess a three-dimensional cytostructure composed of 1,000–2,000 individual cells. Thus, the analysis of islet and exocrine tissue distribution and islet purity in a continuous gradient of 1.068–1.078 g/ml served as an indirect approach to assess the impact of preservation solutions on edema. The islet isolation procedure relies on a density-based purification, following the digestion phase, to separate islets and exocrine tissue; the intrinsic density difference between islet tissue (~1.059 g/ml) and exocrine tissue (1.059–1.074 g/ml) (19) is so minimal that even a slight change in the density of either tissue would have a detrimental effect on recovered tissue mass and purity. For instance, trapped islets, a product of incomplete digestion and dissociation, have a relatively high density that is similar to that of exocrine tissue. An elevated percentage of trapped islets within a digested tissue population will result in a rightward shift in islet purity and, consequently, a lower overall islet purity. On the other hand, exocrine cellular edema will result in a leftward shift, and consequently, incomplete separation of these cell types during purification will lower overall islet purity. Our results revealed no significant difference between the HTK and the UW groups in tissue volume distribution, both in terms of absolute and relative volume, within purification fractions (Fig. 3). A slightly higher tissue volume was observed in the lowest density fractions (fractions 1 and 2) within the UW group, whereas a slightly higher tissue volume was observed in the highest density fractions (fractions 8-10) within the HTK group. Due to the low islet purity within these fractions, exocrine tissue comprised the majority of the volume. In order to eliminate other influencing factors, the relative tissue volume, as a percentage, was analyzed, adjusting for age, sex, BMI, CIT, and enzyme. These results paralleled those observed for the absolute tissue volume.
The distribution of islet purity across fractions also served as an indicator of purification outcome and an indirect measurement of cellular edema. Within the top fraction (> 69%), the islet purity distribution was similar between the HTK and the UW groups. However, within the middle fraction (40-69%), a significantly higher purity was observed in fractions 6 and 7 of the HTK group (Fig. 4).
It is generally accepted that superior islet purification is only possible when the intrinsic density difference between islets and exocrine tissue is preserved during the isolation process. Based on these purification outcomes, our analysis suggests that HTK and UW have equal efficacy in preserving islet and exocrine densities, demonstrating similar top fraction islet purity (in which the majority of islets were recovered) and tissue volume distribution across purification fractions. Although, it appears that HTK may be slightly more effective in preserving exocrine tissue density, as the UW group shows lower islet purity in the middle fraction, which may be caused by exocrine tissue edema.
Recipient, Isolation, and Islet Characteristics and Clinical Islet Transplantation Outcomes

The recipient characteristics, final islet product information, and 6 months posttransplant clinical outcomes from the 15 islet transplants performed at University of Illinois at Chicago (UIC) (HTK = 7 and UW = 8) were summarized (one UW was missing data on glucose stimulation index and one HTK recipient was missing data on beta-score). Differences between HTK and UW groups were analyzed by using unpaired Student's t tests and Fisher's exact test. Recipients who required a second transplant prior to the sixth month following the initial transplant were excluded from the analysis (UW: n = 5 and HTK: n = 3) because the islet graft function and survival may be affected by combined and cumulative effects from other factors such as immunosuppressant toxicity. The beta-score was used as an indicator of islet graft function based on fasting plasma glucose values, glycated hemoglobin (HbA1c), insulin dose, and stimulated C-peptide levels. IEQ, islet equivalent.
In addition to the impact on tissue distribution and islet purity, cellular edema, by definition, also influences islet size. Increased intracellular water retention results in islet enlargement. Consequently, this produces a shift in the islet size distribution of a given islet preparation, with a higher number of islets allocated to the large size categories and a lower number of islets designated to the small size categories. With this in mind, we compared the islet size distribution of the HTK and the UW groups postdigestion (earliest possible dissociation time point) and postpurification (latest possible time point). No discernible difference between islet size was observed at either time point (Fig. 2), supporting the aforementioned findings regarding digested tissue distribution and islet purity. In addition to an increased percentage of large islets, it is also expected that more islets will be lost during purification in the presence of cellular edema. As a volumetric quantification of mass, IEQ can also be used as an indicator of the occurrence of edema. In the presence of cellular edema, the total IEQ from the postdigestion to the postpurification stage will change. In this study, no significant difference was found in total IEQ between these time points. The similarity in size-related variables within this study indicates either the absence of significant intraislet cellular edema, regardless of preservation solution used, or that edema did occur but was not significantly different between the two groups.
This present study is limited mainly by the imprecise assessment techniques that are commonly employed during the islet isolation procedure. Subjectivity is inherent in the established methods for islet yield, size, purity, and volume quantification, and the results may differ depending on the assessor. However, we believe that variation due to user error and subjectivity did not have a great effect on this analysis because all isolations were performed by experienced isolation technicians, according to a standardized islet isolation protocol. Additionally, the large number of isolations analyzed in this study (n = 252) minimizes the impact of user variation on the overall results.
An additional limitation is the potential impact of the 30-min UW incubation, prior to purification, on cellular edema. However, pancreatic tissue from both groups was treated identically in this manner, and the relative duration of this incubation was so brief that we believe any impact would be negligible. Despite the limitations, the results of this study are consistent: HTK and UW solution function similarly to prevent cellular edema during islet isolation.
While there is a consensus that both solutions are acceptable for abdominal organ transplants, new debate has arisen in response to recent studies, as to which solution offers the most comprehensive cold ischemia protection in response to recent studies (17,26–28). These studies indicate that HTK is associated with reduced survival of abdominal organ transplants, especially with increased cold ischemia time. Other studies suggest that, while HTK-flushed pancreata appear more edematous, there is no evidence of impaired early graft function (1,4). However, there have been no studies examining the long-term effects of human pancreas preservation with HTK on in vivo islet grafting. Therefore, although our initial patient data indicate comparable graft function and clinical outcomes, we believe that an in-depth comparison of the effects of HTK and UW on in vivo islet graft outcomes is warranted to garner a complete understanding of the impact of HTK on islet transplantation.
