Abstract
This study aimed to evaluate a 50:50 mix of perfluorohexyloctane/polydimethylsiloxane 5 (F6H8S5) preservation of pancreases in a clinical setting compared with standard solutions for 1) cold ischemia time (CIT) <10 h and 2) an extended CIT >20 h. Procured clinical-grade pancreases were shipped in either F6H8S5 or in standard preservation solutions, that is, University of Wisconsin (UW) or Custodiol. F6H5S5 was preoxygenated for at least 15 min. Included clinical-grade pancreases were procured in UW or Custodiol. Upon arrival at the islet isolation laboratory, the duodenum was removed followed by rough trimming while F6H8S5 was oxygenated for 15-20 min. Trimmed pancreases were immersed into oxygenated F6H8S5 and stored at 4°C overnight followed by subsequent islet isolation. Pancreas preservation using F6H8S5 proved as effective as UW and Custadiol when used within CIT up to 10 h, in terms of both isolation outcome and islet functionality. Preservation in F6H8S5 of pancreases with extended CIT gave results similar to controls with CIT <10 h for both isolated islet functionality and isolation outcome. This study of clinically obtained pancreases indicates a clear benefit of using F6H8S5 on pancreases with extended CIT as it seems to allow extended cold ischemic time without affecting islet function and islet numbers.
Keywords
Introduction
Islet transplantation is an established therapy for type 1 diabetic patients suffering from life-threatening hypoglycemic episodes (10,14,20). In comparison with whole-organ pancreas transplantation, one of the benefits of islet transplantation is a low frequency of serious complications (3). However, this procedure is limited with respect to the number of organ donors required to achieve insulin independence and with regard to long-term islet graft function (1,20,21). Analysis of the Immune Tolerance Network (ITN) trial indicated that the efficiency of the production process can potentially be increased by centralization to experienced islet-processing facilities (19).
Nevertheless, pancreas transportation between geographically distant centers is associated with ischemia and hypoxia, two of the most decisive factors negatively influencing outcomes associated with human islet isolation and islet transplant engraftment (8,24). The approach of supplying explanted human pancreases with oxygen, utilizing the so-called two-layer method (TLM), raised hope to increase the percentage of islet isolations suitable for clinical islet transplantation after extended cold ischemia time (CIT) and to improve the effectiveness of utilizing marginal donors (17,18,24). However, two independent, large-scale studies involving more than 350 human islet isolations could not find a difference between pancreas storage in University of Wisconsin (UW) solution alone or using the TLM consisting of UW solution and the preoxygenated hyperoxygen carrier perfluorodecalin (PFD) (6,15). In agreement with these observations, findings in the porcine and human pancreas indicate that only a marginal proportion of the pancreas is efficiently penetrated by oxygen if PFD is used as the oxygen carrier (2,16). In a pilot study, non-clinical-grade pancreases were roughly trimmed and immersed for approximately 24 h in 400 ml of oxygen-charged PFD alone or perfluorohexyloctane (F6H8), a semifluorinated alkane blended with 50% (v/v) of polydimethylsiloxane with a viscosity of 5 mPas (F6H8S5: I-LET Protect®; Novaliq GmbH, Heidelberg, Germany), until initiation of the isolation procedure. Storage of human pancreases in oxygenated F6H8S5 not only increased islet yield from marginal donors compared with storage in PFD but also improved islet quality in terms of viability, in vitro function, and posttransplant outcome in diabetic nude mice (5). The present organ preservation study for islet isolation and transplantation aimed to (i) clarify if F6H8S5 is as suitable as standard preservation solutions for donor pancreases processed for islet transplantation into type 1 diabetes patients and, as a follow-up study, (ii) evaluate if F6H8S5 could be used for extended CIT with overnight oxygenation of donor pancreases followed by subsequent islet isolation and still maintain transplantable yields and islet quality.
Materials and Methods
Organ Procurement, Preservation, and Solution Oxygenation
Standard organ procurement procedures for the retrieval of pancreases for islet isolation were used for all participating centers in the Nordic Network for Clinical Islet Transplantation (NNCIT) (18). The centers include the Surgical Hospital, Helsinki University (Helsinki, Finland); Oslo University Hospital, Rikshospitalet (Oslo, Norway); Rigshospitalet (Copenhagen, Denmark); Sahlgrenska University Hospital Gothenburg (Gothenburg, Sweden); University Hospital (Malmö, Sweden); Karolinska Institute (Stockholm, Sweden); and Uppsala University Hospital (Uppsala, Sweden). All work involving human organs and tissues was conducted according to the principles expressed in the Declaration of Helsinki and in the European Council's Convention on Human Rights and Biomedicine. Consent for organ donation (for clinical transplantation and for use in research) was obtained from the relatives of the deceased donors by the donor's physicians and documented in the medical records of the deceased subject. The study was approved by the Regional Ethics Committee in Uppsala, Sweden (http://www.epn.se), according to the Act concerning the Ethical Review of Research Involving Humans (2003:460), Permit No. Dnr 2009/371 (from April 10, 2013). This ethical approval covers all organs handled within the NNCIT. All work with human pancreases was conducted as evaluation of a clinically approved product.
In part 1 of the study, each experimental group included 12 fully processed clinical-grade pancreases. Pancreases were either transported in 500 ml of F6H8S5 (Novaliq GmbH) or standard preservation solutions used within the NNCIT (i.e., UW or Custodiol). All solutions were preoxygenated for at least 5-min charge at a flow rate of 2,000 ml/min oxygen.
For part 2 of the study, at the time of pancreas arrival in the evening, the duodenum was removed followed by fine trimming of the pancreas while the F6H8S5 was oxygenated for 15–20 min. The trimmed pancreas was then immersed into the F6H8S5 and placed in +4°C, ending up with a cold ischemic time of 19:17–23:25 h. This was followed by islet isolation as described below. In this second part of the study, additional enzyme, clostripain (Vitacyte, Indianapolis, IN, USA), was added to the digestion enzyme blend (22). Pancreases accepted for clinical islet isolation immediately following each study pancreas were chosen as controls. Control pancreases were isolated immediately after arrival to the islet processing center and had a markedly and highly significant shorter CIT when compared with the pancreases stored in F6H8S5 (Table 3).
Donors, Storage, and Islet Isolation
Donor criteria for clinical islet transplantation were the same as applied for clinical kidney donation aged from 25 to 69 years. Exclusion criteria included glycosylated hemoglobin A1c (HbA1c) higher than 48 mmol/mol according to the International Federation of Clinical Chemistry and Laboratory Medicine (25). Donor and organ transport parameters for comparison included donor age, donor gender, CIT, and HbA1c. Organ and isolation parameters for comparison included pancreas weight, dissection time, weight removed during dissection, digestion time, harvest time, total packed tissue pellet volume prior to separation, digested pancreas (%), total volume of tissue after purification, islet equivalents (IE; IE/g pancreas), islet purity (%), and islet recovery (%) from day of isolation (day 0) to day 1 (changes in IE and islet purity). Islet isolation procedures have previously been described in detail (9,11,13). Briefly, islets were isolated by means of intraductal pancreas perfusion with proteolytic enzymes (Vitacyte), automated digestion at 37°C, and continuous Ficoll (Biochrom, Berlin, Germany)/UW (Viaspan; Apoteket, Uppsala, Sweden) density gradients for islet separation from exocrine tissue. Separated tissue was then maintained in Connaught Medical Research Labs 1066 media (Mediatech Inc., Manassas, VA, USA) with about 10% human serum and standard supplements [10 mM HEPES (Invitrogen AB, Stockholm, Sweden), 10 mM nicotinamide (Swedish Pharmacy, Gothenburg, Sweden), 2 mM L-glutamine (Invitrogen AB), 50 μg/ml gentamicin (Invitrogen AB), 5 mM sodium pyruvate (Swedish Pharmacy), 20 μg/ml ciprofloxacin (Swedish Pharmacy)] in a humidified atmosphere with 5% CO2 at 37°C overnight (minimum 12 h) and at 25°C thereafter (11). Islets were maintained for 1 to 5 days before eventual transplant with medium changes on day 1 and every second day thereafter.
Islet Quality Controls
Islet functional viability assays were performed according to standard protocols. Quality control assays (performed the day after islet isolation) included glucose-stimulated insulin secretion (GSIS) from a dynamic glucose perifusion system (Brandel, London, UK) where 20 equally distributed islets were placed into reaction chambers of the perifusion apparatus. The islets were then perifused first with low-glucose solution (1.67 mM) for 36 min followed by high-glucose solution (20 mM) for 42 min and finally with low-glucose solution (1.67 mM) for 48 min. Stimulation index (average of the high-glucose phase divided by average of the low-glucose phase) was calculated, as well as increment of insulin secretion (IIS) (8) and intracellular insulin content per DNA. Insulin was measured utilizing an immunoassay specific for human insulin (Mercodia, Uppsala, Sweden) while DNA was determined using picogreen assay (Thermo Fisher Scientific, Gothenburg, Sweden) according to kit protocols. Islet characterization also evaluated cytokine expression using homogenized islets using a Gyrolab™ workstation (Gyros AB, Uppsala, Sweden) including interleukin-6 (IL-6) and IL-8 (Quantikine; R&D Systems, Abingdon, UK), monocyte chemoattractant protein-1 (MCP-1; R&D Systems) (4) and tissue factor (TF) (Imubind; American Diagnostica, Stamford, CT, USA) (4,14). For each chemokine, two monoclonal antibodies were loaded into a Gyrolab Bioaffy® 200 CD microlaboratory disk to bind and detect the cytokines TF [capture: TFE (Enzyme Research Laboratories, South Bend, IN, USA); detection: CD142 (BD Pharmingen, Stockholm, Sweden)], MCP-1 (capture and detection: anti-CCL2/MCP-1; R&D Systems, Minneapolis, MN, USA), IL-6 [capture: rat anti-human IL-6 (BD Pharmingen); detection: anti-human IL-6 (R&D Systems)], or IL-8 (capture and detection: anti-CXCL8/IL-8; R&D Systems).
Study Endpoints
The primary endpoint was purified islet yield determined as IE. Secondary endpoints included the number of islet isolations fulfilling the standard transplantation criteria of >250,000 IE, a GSIS >2.0, and packed tissue volume <15 ml at day 1 after isolation. Quality control endpoints were GSIS stimulation index (8), GSIS IIS (8), adenosine diphosphate/adenosine triphosphate (ADP/ATP) ratio (12), and cytokine expression (TF, MCP-1, IL-6, and IL-8).
Statistical Analysis
Statistical analysis was performed utilizing Prism 6.0c (GraphPad, La Jolla, CA, USA). Comparison between experimental groups was carried out by the Mann–Whitney test for unpaired groups. Unless specified otherwise, data are presented as median with range. Differences were considered significant at p < 0.05. Values of p > 0.05 were termed nonsignificant (NS).
Results
Donor, Transport, and Islet Isolation Variables
In part 1 of the study, there was no difference between the two groups regarding donor and organ procurement factors (Table 1) when evaluating donor age, donor gender, HbA1c, body mass index (kg/m 2 ), and CIT from cross-clamp to start of dissection at the islet laboratory.
Donor and Transport Parameters

n = 11; BMI, body mass index; CIT, cold ischemia time.
No differences were observed in trimmed pancreas weight, amount of removed fat, dissection time, digestion time, total harvest time, total packed tissue pellet volume, percentage of pancreas digested, total purity at isolation, and after media change (Table 2).
Organ and Isolation Parameters

MC, media change.
Likewise, for part 2 of the study, there was no difference between control and F6H8S5 extended CIT-preserved pancreas regarding donor and organ procurement factors (Table 3) when evaluating donor age, donor gender HbA1c, and body mass index (kg/m 2 ), but, naturally, there was a difference in terms of CIT. There were differences in trimmed pancreas weight, digestion time, and total packed tissue volume, but not in total harvest time, percentage of pancreas digested, total purity after isolation, or after media change (Table 4).
Donor and Transport Parameters Extended Cold Ischemia

BMI, body mass index; CIT, cold ischemia time.
Organ and Isolation Parameters Extended Cold Ischemia
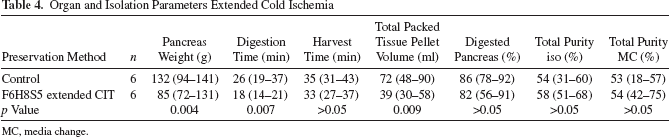
MC, media change.
Outcomes
In part 1 of the study, the primary endpoint of purified islet yield isolated from control pancreases was not different from F6H8S5-preserved pancreases [223,044 (range: 113,804–568,586) vs. 189,109 (range: 68,913–374,000), respectively, p > 0.05] (Fig. 1A). Similarly, there was no difference in IE/g pancreas between controls and F6H8S5-preserved pancreases [2,120 IE/g pancreas (1,026–3,480 IE/g pancreas) vs. 1,646 IE/g pancreas (650–2,473 IE/g pancreas), respectively, p > 0.05] (Fig. 1A), nor was there a difference for the secondary endpoint of islet purity [49% (16–67%) vs. 35% (17–56%), respectively, p > 0.05].

Control versus F6H8S5-preserved pancreas isolation outcomes, quality control outcomes, and islet size distribution. Total IE and IE/g pancreas (A); insulin release from dynamic glucose stimulation, values are presented as mean ± SEM where controls are black circles and F6H8S5 are gray triangles (B); islet size distribution as percentage of total IE number (C); and expression of cytokine tissue factor, MCP-1, IL-6, and IL-8 (D). IE, islet equivalents; SI, stimulation index.
The ability to secrete insulin in response to a glucose challenge in a perifusion assay (Fig. 1B) as measured by the IIS also resulted in no differences between controls and F6H8S5 [151 (44–312) vs. 152 (37–498), p > 0.05]. There was no difference in insulin content between the two groups (data not shown) or stimulation index with a median of 10.15 (4.3–42.6) for controls versus 8.75 (3.8–38.0) for F6H8S5 (p > 0.05).
The concentrations of TF [0.044 pmol/mg DNA (0.002–0.160 pmol/mg DNA) vs. 0.061 pmol/mg DNA (0.050–0.121 pmol/mg DNA), p > 0.05], MCP-1 [0.011 pmol/mg DNA (0.003–0.024 pmol/mg DNA) vs. 0.015 pmol/mg DNA (0.006–0.019 pmol/mg DNA), p > 0.05], and IL-8 [0.042 pmol/mg DNA (0.000–0.128 pmol/mg DNA) vs. 0.063 pmol/mg DNA (0.036–0.161 pmol/mg DNA), p > 0.05] were not different between controls and F6H8S5, respectively. Levels of IL-6 were different but very low in concentration comparing controls [0.002 pmol/mg DNA (0.0005–0.0079 pmol/mg DNA)] with F6H8S5 [0.0042 pmol/mg DNA (0.0025–0.0123 pmol/mg DNA), p = 0.025] (Fig. 1D). There was no difference between the groups regarding size distribution of islets (Fig. 1C). Recovery was similar between the groups with a median of 101% (64%–178%) for controls versus 107% (93%–147%) for F6H8S5 (p > 0.05).
The fulfillment of transplantation release criteria was 5 out of 12 isolations for control and 4 out of 12 for F6H8S5-preserved organs.
In part 2 of the study, even though pancreas weight, digestion time, and total packed tissue volume before separation were significantly lower in the F6H8S5 group (Table 4), the islet yield for control compared to F6H8S5 with extended CIT (19:17–23:25 h) did not differ with 264,891 IE (175,653–589,654 IE) versus 389,021 IE (290,218–645,652 IE), p = 0.065, respectively (Fig. 2A). However, when comparing IE/g pancreas, there was a difference between controls and F6H8S5-preserved pancreases [2,179 IE/g pancreas (1,330–4,181 IE/g pancreas) vs. 5,011 IE/g pancreas (2,215–5,997 IE/g pancreas), respectively, p = 0.038] (Fig. 2A). No difference was found for purity with 54% (31%–60%) versus 58% (51%–68%) for control and F6H8S5 with extended CIT, respectively. As for recovery after 1 day of storage, no difference was found between the groups with a median of 100% (56%–143%) for control versus 91% (78%–116%) for F6H8S5 (p > 0.05).

Control versus F6H8S5 extended CIT-preserved islet isolation outcomes, quality control outcomes, and islet size distribution. Total IE and IE/g pancreas (A); insulin release from dynamic glucose stimulation, values are presented as mean ± SEM where controls are black circles and F6H8S5 prolonged CIT are gray triangles (B); islet size distribution as percentage of total IE number (C); and expression of cytokines tissue factor, MCP-1, IL-6, and IL-8 (D). IE, islet equivalents; SI, stimulation index.
Islets isolated from organs with extended CIT exhibited a preserved capacity to secrete insulin in response to a glucose challenge similar to those in the control group (Fig. 2B). There was no difference in terms of stimulation index with a median of 11.4 (4.7–17.8) for controls and 11.3 (3.7–14.9) for F6H8S5 (p > 0.05).
The concentrations of TF [0.30 pmol/μg DNA (0.041–0.534 pmol/μg DNA) vs. 0.32 pmol/μg DNA (0.234– 0.471 pmol/μg DNA), p > 0.05], MCP-1 [0.13 pmol/μg DNA (0.024–0.398 pmol/μg DNA) vs. 0.065 pmol/μg DNA (0.026–0.170 pmol/μg DNA), p > 0.05], IL-6 [0.015 pmol/μg DNA (0.0017–0.074 pmol/μg DNA) vs. 0.030 pmol/μg DNA (0.011–0.098 pmol/μg DNA), p > 0.05], and IL-8 [0.67 pmol/μg DNA (0.311–1.094 pmol/μg DNA) vs. 0.54 pmol/μg DNA (0.368–1.778 pmol/μg DNA), p > 0.05] were not different between controls and F6H8S5, respectively (Fig. 2D).
No difference was found regarding size distribution between the groups (Fig. 2C). As for isolation outcome in terms of fulfillment of transplant criteria for controls versus F6H8S5 with extended CIT, five out of six versus six out of six preparations met these criteria, respectively.
Discussion
Preservation of pancreases for subsequent islet isolation using F6H8S5, a lipophilic oxygen carrier characterized by improved penetration into deeper tissue layers compared to PFD, proved as effective as UW and Custadiol when used within CIT up to 10 h. The F6H8S5 preservation solution maintained the functional capacity of islets with first- and second-phase insulin release responses to high glucose similar to that of controls, indicating its suitability for use in clinical islet isolation.
In the second part of the study, islet isolated from pancreases with extended cold ischemic time stored in F6H8S5 showed preserved quality and quantity when compared with islets isolated from pancreases preserved with standard solutions with CIT of less than 10 h. The limitation of not having a control group with similar CIT as the study group in this part of the study is based on the knowledge that long CIT is harmful for the tissue (8,23) and to deliberately prolong the CIT on clinical-grade pancreases stored in standard organ preservation solutions would be unethical. As evidenced by similar IE recovery, expression of inflammatory cytokines and islet function, storage of islets overnight did not seem to have detrimental effects on islets from F6H8S5-preserved organs compared to controls. There was a trend of slightly higher expression of cytokines for the F6H8S5 group in part 1 of the study, reaching significance for IL-6 (Fig. 1D), a trend not observed in part 2. Perhaps F6H8S5 could have some minor effect on islet immunogenicity. However, there was a significant increase in cytokine expression in both groups of part 2 of the study compared to part 1. The only difference between the two parts, besides CIT, was the addition of clostripain to the isolation protocol for part 2 (22) and maybe a confounding component affecting interpretation between parts of the study. F6H8S5 is approved for clinical preservation of pancreases for islet isolation but not for whole-pancreas transplantation. The reason for this is that F6H8S5 is eliminated during the islet isolation process and that only trace amounts of the substance remain in the islet product. F6H8S5 was specifically chosen as an alternative to standard preservation solutions based on its lipophilicity. It is highly biocompatible with a viscosity of 3.5 mPas. The density of 1.13 g/cm 3 also makes it a suitable preservation solution since its density is close to that of human pancreas. As such, the need for custom-made vessels for transport needed for previously utilized PFD-based preservation methods can be avoided.
F6H8S5 was developed for static oxygenation of organs, a standard practice in organ preservation. Other methods of oxygenated preservation make use of complex technical equipment not readily available at all organ-procurement facilities and require training and expertise to connect the organs to these machines. In the case of persufflation, perfusion with gas, there is the issue of expanding and reducing of gases due to change in air pressure during a flight, something that can be avoided using a static liquid-based oxygenation system. A special problem using machine-based liquid perfusion of the pancreas is the rapid development of a severe edema affecting both the endocrine and exocrine tissues.
A practical and cost-effective setup for processing pancreases arriving during late evenings or nighttime is presented. One person can easily, within a short period of time, perform a rough, quick trimming to remove the duodenum and excess fat from the pancreas and then place it into oxygen-charged F6H8S5 solution while awaiting subsequent islet isolation the following day. This system offers several advantages: 1) the intermittent need for night work or extended/irregular work hours, which increases the risk for performance-related errors compared to regular, daytime working hours (7), would be avoided; 2) it would reduce costs associated with personnel working during non-office hours; and 3) it might also give a central isolation facility the possibility to handle several organs arriving at the same time (15) without compromising islet functionality.
In summary, this study of clinically obtained pancreases preserved for islet isolation and transplantation shows that F6H8S5 is a suitable preservation solution for pancreases intended for islet isolation. Furthermore, it seems to allow extended cold ischemic time without inflicting islet function and numbers.
Footnotes
Acknowledgments
The authors would like to thank all personnel within the Nordic Network for Clinical Islet Transplantation (http://nordicislets.com) involved in organ preservation, isolation, and islet quality assessments for their great technical skills. Grants from the Swedish Research Council (K2015-54X-12219-19-4), the Ernfors Family Fund, Barn Diabetes Fonden, the Swedish Diabetes Association, the Diabetes Wellness Foundation, the National Institutes of Health (2U01AI065192-06), EXODIAB (Excellence of Diabetes Research in Sweden), and the Juvenile Diabetes Research Foundation supported this study. Olle Korsgren's position is in part supported by NIH 2U01AI065192-06. Magnus Ståhle, Aksel Foss, Bengt Gustafsson, Marko Lempinen, Torbjörn Lundgren, Ehab Rafael, Gunnar Tufvesson, and Bastian Theisinger performed experiments, data analysis, research design, and wrote the manuscript. Dr. Bastian Theisinger is an employee of Novaliq GmbH. All the other authors declare no conflicts of interest.
