Abstract
In cartilage tissue engineering using stem cells, it is important to stimulate proliferation and control the differentiation of stem cells to specific lineages. Here we reported a combined technique for articular cartilage repair, consisting of bone marrow mesenchymal stem cells (BMMSCs) transfected with connective tissue growth factor (CTGF) gene and NaOH-treated poly(lactic-
Keywords
Introduction
Due to the avascular nature and limitation in cell movement, articular cartilage has a limited capacity for self-repair after injury. Therefore, if left untreated, damage to articular joint surfaces progressively causes pain, joint dysfunction, effusions, and, in some instances, joint degeneration and osteoarthritis. Although many methods have been developed to treat cartilage lesions (6,12,22), the results are unsatisfactory because regenerated cartilage has limited durability and shows degenerative changes after long-term follow-up.
Tissue engineering principles that combined living cells with scaffolds are promising techniques for cartilage defect repair (4,10,14,17). The scaffold is one of the key components in tissue engineering strategies. Synthetic biodegradable materials such as poly(lactic-
Live cell implantation is another key component in tissue engineering strategies. Recently, the attempts to employ bone marrow-derived mesenchymal stem cells (BMMSCs) for cartilage tissue engineering have been widely explored (1,13,29). To regenerate hyaline cartilage in situ, the differentiation of these pluripotent progenitor cells toward a chondrogenic phenotype is highly desirable. There have been several reports about the local delivery of various growth factors, including basic fibroblast growth factor (bFGF) (4), transforming growth factor (9), bone morphogenic protein, and vascular endothelial growth factor, to achieve desirable specific organized tissues. Connective tissue growth factor (CTGF) is a 38-kDa cysteine-rich matricellular protein belonging to the CCN family [named after the original three members, cysteinerich 61 (CYR61/CCN1), connective tissue growth factor (CTGF/CCN2), and nephroblastoma overexpressed (NOV/CCN3)] of secreted proteins (27). It is strongly expressed in growth cartilage, especially in hypertrophic chondrocytes, and promotes the differentiation, matrix synthesis, and growth while inhibiting mineralization of articular chondrocytes (3,19). CTGF was also locally used to accelerate the regeneration of articular cartilage in rat knee joints (18). However, free CTGF is currently limited with regard to volume maintenance, and the direct application of CTGF alone in vivo always leads to its rapid diffuseness, denature, and degradation. This means that maintaining an adequate local concentration during the period of cartilage regeneration is markedly important.
In the current study, we transfected recombinant adenoviruses that expressed sense RNA [messenger RNA (mRNA)] of CTGF continuously in BMMSCs and tested the effect of CTGF on the proliferation and differentiation of the cells. Meanwhile, the effect of NaOH-treated PLGA on proliferation and differentiation of BMMSCs was also investigated in culture. Next, we loaded CTGF-modified BMMSCs onto NaOH-treated PLGA scaffolds and tested their effects on repair of full-thickness cartilage defect in rabbit knee joints.
Materials and Methods
Preparation of PLGA Scaffold
Conventional PLGA scaffolds were fabricated as previously described (20). PLGA (50:50wt% PLA/PGA; 11,000–13,500 kDa; Polysciences Inc., Taipei, Taiwan) copolymer scaffolds were fabricated using chloroform (Shanghai Experiment Reagent Co., Ltd., Shanghai, China) and heat treatment. Polymer pellets (0.5 g) were dissolved in 6 ml of chloroform in heat (below 60°C) for 30 min, and a salt-leaching method was applied to produce pores. For this process, 4.5 g of salt (NaCl, up to 250 mm diameter; Sigma-Aldrich, Shanghai, China) was added to the polymer solution, mixed with a stirring rod, and collectively poured onto a Teflon-coated Petri dish (Shanghai Experiment Reagent Co., Ltd.). The polymer solution was vacuum dried at room temperature for 48 h. Then the polymer was soaked in water for 3 days to remove the salt. Some of PLGA scaffolds were soaked in 1 N NaOH (Shanghai Experiment Reagent Co., Ltd.) for 10 min at room temperature. Then the NaOH-treated PLGA scaffolds were rinsed with distilled water several times until the supernatant pH stabilized at 7.4. Both non-treated and NaOH-treated PLGA scaffolds were prepared in a cylinder (Shanghai Experiment Reagent Co., Ltd.; 3 mm in thickness and 5 mm in diameter) and soaked in 70% ethanol for 15 min for sterilization. Samples were coated with gold via a sputter-coater (Beijing HTCY Co., Ltd., Bejing, China) at ambient temperature, and then the samples were examined by scanning electron microscopy (Carl Zeiss, Oberkochen, Germany) (Fig. 1A–D).

Scanning electron micrograph (SEM) images of scaffolds. (A, B) Sodium hydroxide (NaOH)-treated poly(lactic-
Cell Culture and Transfection
BMMSCs were derived from bone marrow aspirates of the left tibiae of rabbits. The cells were isolated by Ficoll-Paque density gradient centrifugation (Shanghai Experiment Reagent Co., Ltd.), then suspended in α-minimum essential medium with 10% fetal bovine serum (Gibco BRL, Gaithersburg, MD, USA) and 100 mg/ml penicillin–streptomycin (Gibco BRL), and incubated at 37 °C with 5% humid CO2. When the cells reached 90% confluence, they were resuspended for passage. Cells of passage 3 were used for the gene transfection in vitro and transplantation in vivo.
Adenoviruses containing human rhCTGF (Ad-CTGF) or control β-galactosidase (Ad-LacZ) were constructed by Vector Gene Technology Company Ltd. (Beijing, China). Once the BMMSCs were grown to 80% confluence, they were preincubated with 5 ml Dulbecco's modified Eagle's medium (DMEM; Sigma-Aldrich) and then infected with Ad-CTGF or Ad-LacZ at multiplicities of infection of 800 infectious units (ifu) per cell for 4 h. After that, 10 ml of fresh culture medium was added to the cells and then subjected to a 48-h incubation. On the third day, the infected cells were collected.
Determination of DNA, Glycosaminoglycan, and Collagen Contents
BMMSCs and CTGF-modified BMMSCs were incubated into prepared PLGA scaffolds or NaOH-treated PLGA scaffolds (diameter: 5 mm, depth: 3 mm) at a density of 1 × 107 cells/scaffold using a syringe (Shanghai Experiment Reagent Co., Ltd.) under mild negative pressure and then incubated at 37°C, 5% CO2 for 3 h. A 1.5-ml DMEM containing 10% fetal calf serum (FCS; Sigma-Aldrich) and antibiotics (100 U/ml of penicillin G and 100 ng/ml of streptomycin; Sigma-Aldrich) was then added to each well. The culture medium was changed three times a week. On 1, 4, 7, 14, 21, and 28 days, the scaffolds were collected to assay cell proliferation, glycosaminoglycan (GAG), and collagen contents. The DNA amount of cells in each scaffold was determined by a Hoechst dye. The cultivated cell/scaffold construct was homogenized in cold 10-mm ethylenediaminetetraacetic acid (EDTA; Sigma-Aldrich) solution (pH 12.3) with a probe-type sonicator (Scientz Biotechnology Co., Ltd, Ningbo, Zhejiang, China) over ice. After adjusting to pH 7.0, 200 ml of the sample was mixed with 25 ml of Hoechst 33258 dye solution (0.5 mg/ml; Sigma-Aldrich). The fluorescence intensity of Hoechst 33258 intercalated into DNA was measured at the excitation wavelength of 365 nm and the emission wavelength of 460 nm. Synthesis of sulfated GAG was measured according to a previously published method (2). 1,4-Dimethylene blue solution (16 mg/ml; Sigma-Aldrich) was reacted with 100 ml of digested cell/scaffold construct. Chondrotin sulfate (Sigma-Aldrich) was used to construct a standard curve. In order to measure the amount of total collagen synthesized from cells, the cells were labeled with 0.5 μCi of [3H]proline (Beijing Institute of Atomic Energy, Beijing, China) during the cell culture at 37°C in a humidified 5% CO2 incubator for 12 h. The cell was washed with phosphate-buffered saline (PBS; Sigma-Aldrich) three times and dissolved in 1 M potassium hydroxide (Sigma-Aldrich) at 37°C for 24 h, extracted with ice-cold trichloroacetic acid (Sigma-Aldrich) and centrifugation three times to precipitate any macromolecules. The resulting precipitates were dissolved in 0.3 m NaOH, neutralized, mixed with liquid scintillant, and counted in a scintillation counter. The experiment was performed in triplicate.
Animal Experiments
Seventy-two adult male New Zealand White rabbits (20 weeks old, 3–3.5 kg) were used. All procedures were performed according to the protocols approved by the animal research committee of the university. The cells were loaded on nontreated PLGA or NaOH-treated PLGA scaffold (thickness: 3 mm; diameter: 5 mm) at a density of 1 × 107 cells/scaffold. Under general anesthesia, the knee joint was opened with a medial parapatellar approach. A full-thickness cylindrical cartilage defect of 5 mm diameter and 3 mm depth was created in the patellar groove of the femoral articular cartilage using a hand drill (Stryker Co., Kalamazoo, MI, USA) equipped with a 5-mm diameter drill-bit. Rabbit knees were divided into four groups: group I implanted with CTGF-modified BMMSCs/NaOH-treated PLGA scaffold (
Gross and Histological Examination
The harvested samples were examined and photographed for evaluation. After macroscopic examination, samples were fixed in 4% formalin (Shanghai Experiment Reagent Co., Ltd.), decalcified in 10% EDTA, then embedded in paraffin, and cut to 8-μm-thick sections. Sections were stained with hematoxylin and eosin (H&E; Shanghai Experiment Reagent Co., Ltd.) for the study of morphological details, with toluidine blue (Shanghai Experiment Reagent Co., Ltd.) for cartilaginous matrix distribution. In order to evaluate the histological repair of articular cartilage defects quantitatively, we used a modified version of the grading scale (Table 1), which has previously been described by Wakitani et al. (29).
Mechanical Evaluation
Mechanical properties of the regenerated articular cartilage were compared with that of normal appearing cartilage from the patella groove using an indentation test. Indentation tests were performed on whole intact joint surface using a nondestructive in situ method by Instron 5848 microtester machine (Instron, Canton, MA, USA). The distal femur of 4 cm was harvested from the experimental rabbits, and specimens were placed with the articular surface facing up into a metal sample holder and bathed in PBS containing protease inhibitors (Sigma-Aldrich) to prevent any digestion of the cartilage tissue during testing.
A 0.5-mm diameter conically shaped indenter was fixed to the load cell assembly and used to probe one or two sites at the edge and center of each defect. Five consecutive displacements of 30 μm were performed on test samples. Data were recorded over a time increment of 200 s; for each step, the criterion for a complete relaxation has a relaxation rate of <100 Pa over the final 60 s. After acquisition of data, the Young's modulus,
In Vivo Tracing of Implanted Cells
To investigate the fate of the implanted mesenchymal cells in the defect, Ad-LacZ-labeled BMMSCs were seeded in NaOH-treated scaffolds and implanted in two additional rabbits. The samples were harvested at 6 weeks postoperatively and fixed in 4% formalin overnight. Following that, the samples were stained in 1 mg/ml X-gal (5-bromo-4-chloro-3-indolyl-β-d-galactopyranoside) substrate, 1 mmol/L MgCl2, 10 mmol/L K4Fe(CN)6, and 10 mmol/L K3Fe(CN)6 (all Sigma-Aldrich) in PBS for 24 h. After X-gal staining, the samples were placed in 4% formalin again for complete fixation, and then the samples were decalcified and embedded in paraffin. The sections were counterstained with Safranin O (Shanghai Experiment Reagent Co., Ltd.).
Statistical Analysis
Statistical differences were examined using a oneway factorial ANOVA followed by Scheffe's multiple comparison tests (SPSS Inc., Chicago, IL, USA). A
Results
Determination of DNA, GAG, and Collagen Contents
After 7 days of culture, the DNA content from the cells seeded in NaOH-treated PLGA scaffolds, no matter whether they were CTGF-modified BMMSCs or unmodified BMMSCs, was significantly higher than that in PLGA scaffolds, and it was two times greater after a 28-day culture (

Changes of DNA contents (A), sulfated glycosaminoglycan (GAG) (B), and total collagen (C). Error bars mean standard deviations. *
Gross Examination
At 6 weeks postoperatively (Fig. 3A–D), the defects in the CTGF-modified BMMSCs/NaOH-treated PLGA group (Fig. 3A) were covered with smooth, white, and semitransparent tissue, but the margins of the defects could be recognized and the color tones of their locations were slightly different from the surrounding host cartilage. In the BMMSCs/NaOH-treated PLGA (Fig. 3B) and BMMSCs/ PLGA (Fig. 3C) groups, the defects still had not been totally filled in by the repair tissue with sharply defined margins and clearly discernible edges, but the resurfacings in the BMMSCs/PLGA group were not as smooth as that in the BMMSCs/NaOH-treated PLGA. In the defect control group, the defects were filled with opaque tissue with a concavity toward the center (Fig. 3D).
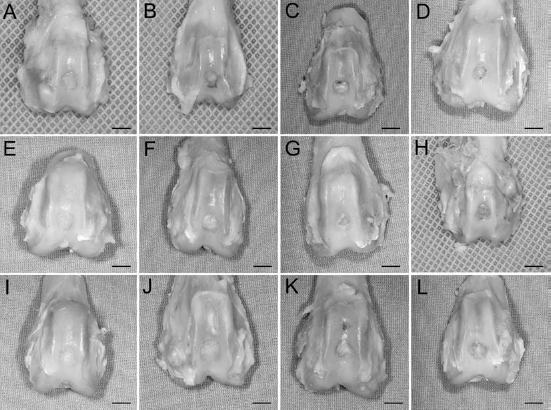
Gross appearance of the specimens. In the CTGF-modified/NaOH-treated PLGA group (A, E, I), the defects were covered with smooth, white, and semitransparent tissue, but the margins of the defects could be recognized at 6 weeks (A), but at 12 weeks (E) and 24 weeks (I), it exhibited continuity with the surrounding intact host cartilage. In the BMMSCs/NaOH-treated PLGA group (B, F, J), the defects still had not been totally filled with the repair tissue at 6 weeks (B). At 12 weeks (F) and 24 weeks (J), the defect was substituted with smooth and semitransparent tissue just like host cartilage, although the junction with surrounding host cartilage was still visible. In the BMMSCs/PLGA group (C, G, K), less than 50% of the defect had been replaced with cartilage-like tissue at 6 weeks (C). More than 70% area of the defects showed regenerated cartilage-like tissue at 12 weeks (G). Although the defects showed a total cartilaginous tissue filling the defects at 24 weeks, the surface of defects was slightly concave and the margins still could be recognized. In the defect control group (D, H, L), the defects were rough and filled with fibrous-like tissue, and a depression was visible at all time points. Scale bar: 5 mm.
At 12 weeks postoperatively (Fig. 3E–H), the defects in the CTGF-modified BMMSCs/NaOH-treated PLGA group (Fig. 3E) were completely filled in with smooth white tissue that resembled the surrounding normal hyaline cartilage, and the margins were barely recognizable. In the BMMSCs/NaOH-treated PLGA (Fig. 3F) and BMMSCs/PLGA (Fig. 3G) groups, the defects were almost entirely filled with cartilage-like tissue of similar color and texture to the surrounding cartilage. There was a discernable circular line marking the junction between the normal and new tissue. However, the proportion of regeneration in the repair area was significantly larger in the BMMSCs/NaOH-treated PLGA group than in the BMMSCs/PLGA group. In the defect control group, there was still a larger vacant position in the center and a thin layer of white and rough fibrous tissue in the defect (Fig. 3H).
At 24 weeks postoperatively (Fig. 3I–L), the defects in the CTGF-modified BMMSCs/NaOH-treated PLGA group were completely covered with semitransparent tissue, and the color and glistening of the regeneration surface were similar to the adjacent normal cartilage (Fig. 3I). It was difficult to discern the host cartilage from the repaired tissue, and the margins had disappeared. In the BMMSCs/NaOH-treated PLGA group, the defects showed cartilaginous tissue completely filling the defects; however, the margins remained visible (Fig. 3J). In the BMMSCs/PLGA group, the integration of the regenerated cartilage with host cartilage was worse than that in group II, and the surface of the defects was slightly concave (Fig. 3K). The defects in the defect control had a rough surface containing much more fibrous tissue (Fig. 3L).
Histological Examination
At 6 weeks postoperatively (Fig. 4), the defects in the CTGF-modified BMMSCs/NaOH-treated PLGA group were repaired with hyaline-like cartilage, as shown by toluidine blue staining (Fig. 4A, E). The NaOH-treated PLGA scaffolds had been completely absorbed. Extracellular matrix staining in the regenerated articular cartilage was less than that in the normal cartilage. Repaired tissues in the BMMSCs/NaOH-treated PLGA (Fig. 4B, F) and BMMSCs/PLGA group (Fig. 4C, G) showed more fibrochondral-like characteristics, although fibrous tissue still dominated over the defect. Small portions of cartilaginous tissue formed on the site closely adjacent to host cartilage, but the cartilaginous tissue in the BMMSCs/ PLGA group was less and thinner than that in the BMMSCs/ NaOH-treated PLGA group. The defects in the defect control showed noncartilaginous structure and was almost filled with fibrous tissue without staining of toluidine blue (Fig. 4D, H).

Histological appearance of the repaired tissue at 6 weeks postoperatively with hematoxylin and eosin (H&E) staining (A, B, C, D) or toluidine blue staining (E, F, G, H). The arrow indicates the boundary between the newly formed cartilage and the normal adjacent cartilage. Scale bar: 250 μm. CTGF-modified BMMSCs/NaOH-treated PLGA group (A, E): Hyaline-like cartilage was regenerated, but extracellular matrix staining in the regenerated articular cartilage was less than that in the normal cartilage. BMMSCs/NaOH-treated PLGA group (B, F): Fibrous tissue still dominated over the defect, although small portions of cartilaginous tissue formed on the site closely adjacent to host cartilage. BMMSCs/PLGA group (C, G): The cartilaginous tissue in the BMMSCs/ PLGA group was less and thinner than that in the BMMSCs/NaOH-treated PLGA group. Defect control group (D, H): The defect was filled with fibrous tissue without staining of toluidine blue.
At 12 weeks postoperatively (Fig. 5), the defects in the CTGF-modified BMMSCs/NaOH-treated PLGA group (Fig. 5A, E) were filled with regenerated cartilage similar to hyaline cartilage and abundant extracellular matrix, as shown by toluidine blue staining. However, the regenerated cartilage was slightly thicker than the surrounding host cartilage. The defects in the BMMSCs/NaOH-treated PLGA group also contained hyaline cartilage-like regeneration tissue at the surface; however, it was noteworthy that the new cartilage had incomplete integration with adjacent cartilage and was thinner than normal (Fig. 5B, F). Cellular orientation was disorganized. The defects in the BMMSCs/PLGA group were replaced by regenerated cartilage, which was significantly thinner than the normal surrounding cartilage and had incomplete integration with the adjacent cartilage (Fig. 5C, G). In some cases, fissures and cracks were observed at the integration site between neocartilage and adjacent native cartilage. The defects in the defect control still existed with a layer fibrous tissue at its base and had no cartilage-like tissue (Fig. 5D, H).

Histological appearance of repaired tissue at 12 weeks postoperatively with H&E staining (A, B, C, D) or toluidine blue staining (E, F, G, H). The arrow indicates the boundary between the newly formed cartilage and the normal adjacent cartilage. Scale bar: 250 μm. CTGF-modified BMMSCs/NaOH-treated PLGA group (A, E): The defects were filled with regenerated cartilage similar to hyaline cartilage and abundant extracellular matrix. However, the regenerated cartilage was slightly thicker than the surrounding host cartilage. BMMSCs/NaOH-treated PLGA group (B, F): The defects also contained hyaline cartilage-like regeneration tissue at the surface of the defect; however, the new cartilage had incomplete integration with adjacent cartilage with disorganized cellular orientation. BMMSCs/PLGA group (C, G): The regenerated cartilage was much thinner than that in the BMMSCs/NaOH-treated PLGA group. Defect control group (D–H): The defects still existed with a layer fibrous tissue at its base.
At 24 weeks postoperatively (Fig. 6), the regenerated cartilage in the CTGF-modified BMMSCs/NaOH-treated PLGA group was homogeneously stained by toluidine blue with a smooth surface and the same thickness as the surrounding host cartilage (Fig. 6A, E). Elongated chondrocytes were arranged parallel to the surface in the superficial zone, and spherical cells were arranged in a columnar form in the deep zone similar to normal articular cartilage. No difference could be observed microscopically between the neoformative cartilage and the surrounding host cartilage. In the BMMSCs/ NaOH-treated PLGA group, the newly formed cartilage in the defect appeared to be bonded to the adjacent host articular cartilage (Fig. 6B, F). The repair tissue showed extensively metachromatic staining but had thinner newly formed cartilage. The cell arrangement in the new cartilage still lacked the typical zonal organization. The defects in the BMMSCs/PLGA group were replaced by bony tissue, which was covered by a layer of thin fibrous tissue (Fig. 6C, G). The defects in the defect control were covered with fibrous tissue, and no hyaline cartilage was detected (Fig. 6D, H). The surface of the repaired areas was still much lower than the surrounding tissue.

Histological appearance of repaired tissue at 24 weeks postoperatively with H&E staining (A, B, C, D) or toluidine blue staining (E, F, G, H). The arrow indicates the boundary between the newly formed cartilage and the normal adjacent cartilage. Scale bar: 250 μm. CTGF-modified BMMSCs/NaOH-treated PLGA group (A, E): The regenerated cartilage was homogeneously stained by toluidine blue with a smooth surface and the same thickness and cell arrangement as the surrounding host cartilage. BMMSCs/NaOH-treated PLGA group (B, F): In most samples, the newly formed cartilage in the defect appeared to be bonded to the adjacent host cartilage, but it was thinner and had no typical cell organization in the normal cartilage. BMMSCs/PLGA group (C, G): The defects were replaced by bony tissue, which was covered by a layer of thin fibrous tissue. Defect control group (D, H): The defects were covered with fibrous tissue, and no hyaline cartilage was observed.
Histological grading scores are presented in Figure 7. Overall, within the corresponding time points, all treated groups had significantly higher scores than the defect control group (

Histological grading of repaired tissue at 6, 12, and 24 weeks postoperatively. *p < 0.05 for significant difference.
Mechanical Evaluation
From the indentation test, the values of Young's modulus of repaired tissues were determined and compared with each other. The indentation test was not performed on the samples from the control defect group because no cartilage was found (Fig. 8). At 6 weeks postoperatively, the value of Young's modulus for regenerated cartilage from all treated groups was significantly higher than that from the normal cartilage (

Young's modulus of regenerated cartilage at 6, 12, and 24 weeks postoperatively. Pa=pascals. *p < 0.05 for significant difference.
Implanted Cell Tracing
LacZ gene transduced BMMSCs were detected by X-gal staining, as indicated by blue color under light microscopy (Fig. 9A, B). Histological sections of repaired tissue with Safranin-O counterstaining also showed the presence of implanted cells in the defect. These cells stayed in the regenerated cartilage (Fig. 9C, arrows) and subchondral bone (Fig. 9D, arrows).
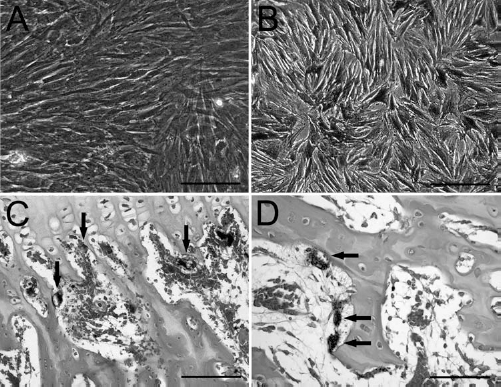
X-gal staining for tracing of implanted cells. Adenovirus containing human β-galactosidase (Ad-LacZ) gene transduced BMMSCs were detected in vitro under light microscopy, as indicated by color (A, B). Histological sections also showed the presence of implanted cells in the regenerated cartilage (C, arrows) and subchondral bone (D, arrows). Safranin-O counterstaining: The scale bar represents 100 μm. X-gal: 5-bromo-4-chloro-3-indolyl-β-d-galactopyranoside.
Discussion
In cartilage tissue engineering using stem cells, it is important to stimulate proliferation and control the differentiation of stem cells to specific lineages. With this point in mind, we seeded CTGF-modified BMMSCs on NaOH-treated PLGA scaffolds and then evaluated the ability to stimulate cell proliferation and chondrogenic differentiation. There are three aspects to the novel technique for articular cartilage repair that we describe in this study: 1) NaOH-treated PLGA scaffolds enhance proliferation of BMMSCs; 2) gene therapy with CTGF stimulates chondrogenic differentiation of BMMSCs and cartilagous matrix production; and 3) implantation of CTGF-modified BMMSCs/NaOH-treated PLGA scaffold promotes biological resurfacing with hyaline-like cartilage in full-thickness articular cartilage defects in rabbits.
Surface properties of a scaffold material play a significant role in cell adhesion, proliferation, and subsequent functions. Previous studies have demonstrated that compared to conventional PLGA, NaOH treatment of PLGA scaffolds greatly promotes the adhesion, proliferation, and subsequent functions of various cell lines like osteoblasts as well as vascular and bladder smooth muscle cells (11,15,20,23,30). Kay et al. observed that when chondrocytes were exposed to two-dimensional NaOH-treated and nontreated PLGA substrates, greater numbers of chondrocytes adhered to the altered surface (11). To determine their efficacy toward articular cartilage applications, Park et al. fabricated three-dimensional NaOH-treated PLGA scaffolds via chemical etching techniques using 1 N NaOH for 10 min and found that NaOH-treated PLGA three-dimensional scaffolds enhanced chondrocyte functions compared to nontreated PLGA scaffold, as indicated by significantly increased chondrocyte numbers, total intracellular protein content, and the amount of extracellular matrix components (20). These effects may be explained by the fact that PLGA scaffolds treated with NaOH have a more hydrophilic surface, increased surface area, altered porosity, and a greater degree of nanometer roughness than the nontreated PLGA (20). To our knowledge, the present study was the first one to investigate the effects of NaOH-treated PLGA scaffold on proliferation and chondrogenic differentiation of BMMSCs. Our results showed that BMMSCs loaded in NaOH-treated PLGA had a higher DNA content than those in PLGA after 14 days; however, no difference in GAG and collagen was observed between these two groups. These results suggested NaOH-treated PLGA had proliferative effects on BMMSCs but had no effects on chondrogenic differentiation. These findings expanded the prior knowledge of NaOH-treated PLGA as a potential scaffold to enhance cell functions and transferred them into BMMSCs.
When BMMSCs were transfected with adenoviruses expressing CTGF gene, these transfected cells continuously produced CTGF protein and stimulated the chondrogenic differentiation of BMMSCs, resulting in an increase in expression of total GAG and collagen contents compared with the control BMMSCs transfected with control adenoviruses. These findings clearly provided direct evidence for stimulatory roles of CTGF in chondrogenic differentiation of BMMSCs. Our results were also consistent with a previous report, in which chondrocytic differentiation of rat MSCs was stimulated when exogenous CTGF was supplemented in culture (18). In addition, no difference in DNA content was observed between the CTGF-modified BMMSC groups and BMMSC groups either on conventional PLGA or NaOH-treated PLGA, suggesting that produced CTGF protein could not stimulate the proliferation of BMMSCs. However, previous reports showed that CTGF promoted the proliferation and differentiation of chondrocytes (3,19) and osteoblasts in vitro (27). One possible reason for this may be the different characteristics between undifferentiated BMMSCs and other differentiated cell lines (16,18,19). Furthermore, when CTGF-modified BMMSCs were loaded in NaOH-treated PLGA, both DNA amount and GAG and collagen content were the greatest, indicating that CTGF and NaOH-treated PLGA act in a complementary fashion in chondrogenic differentiation and proliferation of BMMSCs.
The in vivo study showed that improved histological and gross results were found in the BMMSCs/NaOH-treated PLGA group compared with BMMSCs/PLGA group, supporting the prospect of using NaOH-treated PLGA in vivo as a potential scaffold in cartilage tissue engineering. Moreover, the NaOH-treated PLGA scaffold had been completely absorbed as early as 6 weeks after implantation, and no inflammatory reaction was recognized during the 24-week-long follow-up. These results further confirmed that the NaOH-treated PLGA scaffold was still biodegradable and biocompatible, which was encouraging because it is possible that NaOH treatment could induce potentially harmful chemical changes and alter these properties of PLGA (15,20).
However, the repaired tissue induced by BMMSCs/ NaOH-treated PLGA was still not satisfactory for clinical use because the objective histological and gross results were not equivalent to normal articular cartilage. One possible reason is that the implanted BMMSCs in NaOH-treated PLGA scaffold may differentiate into unnecessary cells, which in turn lead to a relative decrease in the chondrocyte-like cells needed for cartilage regeneration. However, CTGF has a short half-life period in vivo, which affects its bioactivity and longevity. Nashida et al. applied a sustained release strategy by immobilizing CTGF into the gelatin hydrogel to stimulate cartilage repair in both the monoiodoacetic acid (MIA)-induced experimental rat osteoarthritis model and full-thickness defects of rat knee joints (18). Here, we report on the development of another drug delivery system of CTGF. Autogenous BMMSCs served as a vehicle to carry CTGF gene. Compared with BMMSCs/NaOH-treated PLGA scaffold implantation, addition of BMMSCs carrying the CTGF gene showed a significant improvement in both histological and gross results and induced healing of the joint surface with regenerated hyaline cartilage as early as 12 weeks after implantation.
The length of adenovirus-mediated transgene expression in vivo is varied. In this study, the in vivo tracing experiment also confirmed that these implanted BMMSCs can survive for at least 6 weeks after implantation, suggesting that these BMMSCs can continuously secrete CTGF protein in vivo and stimulate cartilage regeneration. This finding was also consistent with the previous studies in knee joint and temporomandibular joint (25,31).
To further investigate the quality and stability of repaired tissue, the indentation test was used for the evaluation of the mechanical property of the cartilage. At 12 weeks after implantation, the newly regenerated tissue in the NaOH-treated PLGA group has comparable strength to the normal cartilage, suggesting that this neocartilage is able to meet the functional demands placed upon it. This is very important in cases with cartilage defects under conditions of load bearing because, from a functional standpoint, mechanically inferior repair tissue may break down with time and loading, becoming fragmented or disintegrating altogether (7,26).
In conclusion, in vitro and in vivo results in the present study revealed that the composite of CTGF-modified BMMSCs/NaOH-treated PLGA scaffold effectively stimulated proliferation and chondrogenic differentiation of BMMSCs, which in turn led to successful hyalinelike cartilage regeneration with similar histological and mechanical properties to normal cartilage. The present findings indicate that CTGF-modified BMMSCs/NaOH-treated PLGA scaffold composites can have significant clinical benefits in cases involving full-thickness articular cartilage defects. Nevertheless, there has been much soul searching and investigation to evaluate whether the attractive basic concepts of gene therapy can be used clinically as a safe and effective therapeutic mode.
Footnotes
Acknowledgments

