Abstract
Adult mesenchymal stem cells (MSCs) are an attractive cell source for new treatment strategies in regenerative medicine. This study investigated the potential effect of matrix assisted MSC transplantation for articular cartilage regeneration in a large-animal model 8 weeks postoperatively. MSCs from bone marrow aspirates of eight Goettingen minipigs were isolated and expanded prior to surgery. Articular cartilage defects of 5.4 mm were created bilaterally in the medial patellar groove without penetrating the subchondral bone plate. Defects were either left empty (n = 4), covered with a collagen type I/III membrane (n = 6) or additionally treated with autologous MSC transplantation (2 × 106; n = 6). After 8 weeks animals were euthanized and the defect area was assessed for its gross appearance. Histomorphological analysis of the repair tissue included semiquantitative scoring (O'Driscoll score) and quantitative histomorphometric analysis for its glycosaminoglycan (GAG) and collagen type II content. All membranes were found to cover the defect area 8 weeks postoperatively. Median defect filling was 115.8% (membrane), 117.8% (empty), and 100.4% (MSC), respectively (not significant). Histomorphological scoring revealed significantly higher values in MSC-treated defects (median 16.5) when compared to membrane treatment (median 9.5) or empty defects (median 11.5; p = 0.015 and p = 0.038). Histomorphometric analysis showed larger GAG/collagen type II-positive areas in the MSC-treated group (median 24.6%/29.5% of regeneration tissue) compared to 13.6%/33.1% (empty defects) and 1.7%/6.2% (membrane group; p = 0.066). Cell distribution was more homogeneous in MSC compared to membrane-only group, where cells were found mainly near the subchondral zone. In conclusion, autologous matrix-assisted MSC transplantation significantly increased the histomorphological repair tissue quality during early articular cartilage defect repair and resulted in higher GAG/collagen type II-positive cross-sectional areas of the regenerated tissue.
Introduction
Cell-based strategies for the treatment of chondral defects have been introduced into clinical routine since the inauguration of autologous chondrocyte transplantation (ACT) (2). Despite 15 years of clinical application, this technique is still not widely accepted as the “gold standard” procedure for localized chondral defect treatment because prospective randomized clinical trials did not confirm clear benefits in the clinical outcome when compared to microfracture treatment (12,19), even though a superior histological outcome was found in ACT (30). In the past years substantial effort has been made to improve the originally proposed technique, towards a tissue-engineering approach involving two- and three-dimensional biomaterials to replace the originally used periosteum for covering the defect area. By such means cell retention has been supported and cell differentiation and tissue formation are thought to be facilitated (23). Thus, second-generation transplantation techniques with a collagenous membrane for coverage of the target defect area have become more and more popular in clinical routine use (11,32).
As donor site morbidity is still considered to play a crucial role in ACT and the chondrocyte expansion is limited during in vitro culture, other cell sources have been evaluated for articular cartilage repair. Bone marrow-derived mesenchymal stem cells (MSCs) are considered as an attractive alternative cell source, based on their capacity to differentiate into various types of connective tissues, including the chondrogenic cell lineage (1,16,22,38). Initial experimental studies in small (4,5, 34) and larger animal models (10,24,37) have already shown a positive effect of MSCs on osteochondral and full-thickness articular cartilage defect repair. Furthermore, first clinical application of autologous MSC transplantation in human osteoarthritic knees resulted in a superior arthroscopic and histomorphological grading when compared to untreated controls (35) or showed to improve clinical symptoms in a full-thickness cartilage defect in the human patella (36). Wakitani and coworkers (35,36) used in their pioneering work a modification of the cell transplantation technique used for autologous chondrocyte implantation (ACT) in clinical routine (2). The authors transplanted autologous bone marrow-derived mesenchymal stem cells into the articular cartilage defect after embedding the cells in a collagen gel while covering the defect area with periosteum. Periosteum, however, has been identified as a major drawback during ACT as it may be responsible for repair tissue hypertrophy and calcification (11,20). In order to avoid this debilitating effect, second-generation ACT technique implies the use of a collagenous membrane to cover the targeted defect area (11,32).
The aim of this study was to investigate the impact of mesenchymal stem cell transplantation on the repair tissue formation of articular cartilage defects when a “second-generation” transplantation technique with a collagen type I/III membrane (CHONDROGIDE®, Geistlich, Switzerland) is used.
Materials and Methods
Animals
All animal care, housing, and treatment was performed according to the German animal welfare act of May 25, 1998, after receiving written allowance for the experiment by the animal rights protection authorities in Baden Württemberg, Germany (Regierungspraesidium Karlsruhe, AZ: 35-9185.81/G-117/03). Eight mature female Goettingen minipigs (Elegaard Soro Landevej 302, DK-4261 Dalmose, Denmark) were used for this study. The average age and weight of the minipigs was 19 months (18–24 months) and 32.5 kg (27–35 kg), respectively, at the beginning of the study. Animals were held in big indoor runs with unrestricted movement with food and water ad libitum. Animals were randomized before surgery to the three treatment groups.
Mesenchymal Stem Cell Isolation and Cultivation
Bone marrow aspirates were obtained 6 weeks prior to transplantation by a 0.5-cm anteromedial skin incision at both proximal tibiae, 2 cm below the joint line. A Jamshidi bone marrow needle (8 gauge x 10 cm; Cardinal Health Inc., Waukagen, IL, USA) was used to penetrate the cortical bone and aspirate blood from the bone marrow cavity (6–14 ml) into 10-ml syringes containing 5000 IU of Natrium heparin (25000 IU/5 ml, Ratiopharm GmbH, Ulm, Germany). The anticoagulated blood was resuspended in 15 ml DMEM-LG culture medium (Gibco GmbH, Karlsruhe, Germany) and washed twice with PBS (pH 7.4). Mononucleated cells were isolated by Ficoll-Paque Plus (GE Healthcare AG, Uppsala, Sweden) gradient centrifugation as described previously (38). Cells were expanded up to passage 3 for 6 weeks in expansion medium [high-glucose Dulbecco's modified Eagle's medium (DMEM), 40% MCDB201, 2% fetal calf serum (FCS), 2 × 10−8 M dexamethasone, 10−4 M ascorbic acid-2-phosphate, 10 μg/ml insulin, 10 μg/ml tranferrin, 10 ng/ml selenous acid, 100 U/ml penicillin/streptomycin, 10 ng/ml recombinant human platelet-derived growth factor BB (all from Sigma-Aldrich, Deisenhofen, Germany), and 10 ng/ml recombinant human epidermal growth factor (Strathmann Biotech, Hamburg, Germany)]. Nonadherent cells were removed after 24–48 h. Colonies of adherent MSCs were expanded and replated two to three times at a density of 5 × 103 cells/cm2 (28). To demonstrate the multilineage potential of the obtained MSC population they were successful differentiated in vitro along the chondrogenic, osteogenic, and adipogenic pathway (data not shown).
For MSC transplantation 2 × 106 autologous MSCs were immersed directly before transplantation in 7 μl 1: 500 diluted thrombin and mixed with 13 μl 1:2 diluted fibrinogen available as commercial fibrin glue (TISSUCOL DUO S 0.5 ml immuno, Baxter Deutschland, Unterschleissheim, Germany). Cells and fibrin glue were thereby aspirated in an insulin syringe with no dead space [BD Micro-Fine, 30G (0.30 × 8 mm, 1 ml U-40), Becton Dickinson, Heidelberg, Germany]. We were able to inject 17–18 μl of the 20 μl cell/fibrin glue suspension, resulting in approximately 1.7 × 106 transplanted cells.
Defect Localization and Surgery
All animals were operated under aseptic conditions in general anesthesia on both medial patellar grooves (trochleae) simultaneously as previously described (9). A total of 16 defects were created. A median skin incision was placed between the tibial tuberositas and the distal patellar pole while extending the knee. After soft tissue preparation the patellar tendon and the inferior patellar fat pad were split longitudinally, exposing the patellar groove. Ten millimeters above the intercondylar notch, the articular cartilage on the medial trochlear facet was cut with a 5.4-mm circular cutter down to the calcified zone. Chondral defects were created by miscellaneous removal of the articular cartilage down to the subchondral bone plate under visual control with 3.8x magnifying glasses, without penetrating the subchondral bone plate, confirmed by the absence of bleeding from the subchondral marrow space (Fig. 1A).
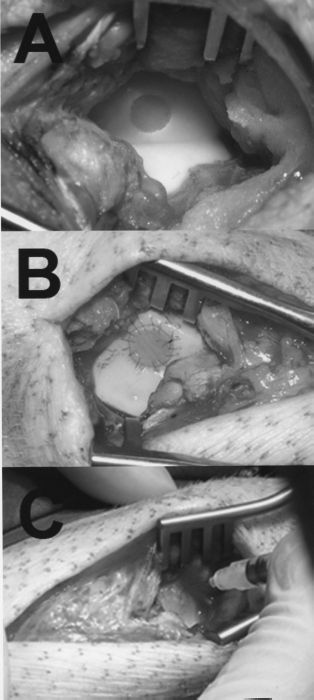
Operating technique. (A) Experimental chondral defect (5.4 mm in diameter) in the medial facet of the patellar groove. (B) Chondral defect covered with a collagen type I/III membrane (8–0 Ethilon® running suture). (C) Mesenchymal stem cells immersed in fibrin glue, being injected under the collagen type I/III membrane.
As controls, defects were either left empty (n = 4) or covered with the commercially available collagen type I/III membrane (n = 6) (Chondrogide®, Geistlich Biomaterials, Wolhusen, Switzerland) fixed with running sutures (8–0 Ethilon®; Ethicon, Norderstedt, Germany) and sealed with fibrin glue (Fig. 1B). In the verum group (n = 6), approximately 1.7 × 106 autologous bone marrow-derived mesenchymal stem cells were injected under the collagen membrane. Fibrin glue was allowed to solidify for 10 min before the wound was closed in layers. All animals were allowed immediate, full weight bearing postoperatively and free exercise.
Animals were euthanized by an intravenously applied overdose of barbiturate (200 mg/kg body weight, EUNARCON®, Pharmacia & Upjohn GmbH, Erlangen, Germany) 8 weeks postoperatively and the hind legs were dissected obtaining both knees for further preparation.
Macroscopic Evaluation
After dissection of the knees, the defect area was digitally photographed and examined for surface irregularities, color, and the amount of defect filling.
Histological Evaluation
Following soft tissue preparation, a cube of approximately 2 × 2 × 2 cm was cut from the medial trochlea, including the repair zone in the center. The defect area was cut longitudinally, dividing it into two equal parts. The lateral half of the defect was fixed in 4% phosphate-buffered formalin for 5 days, decalcified in 0.5 M EDTA (pH 7.4) for 4–6 weeks, being followed by dehydration and paraffin embedding. Serially sections (5 μm) were stained with Safranin O/Fast green (SOFG) according to standard protocols to indicate the presence of sulphated glycosaminoglycans (orange to red), which are commonly found in articular cartilage (29). Immunohistological staining for collagen type II was performed as described elsewhere (17), using primary mouse anti-type II collagen monoclonal antibodies (1:1000 in 1% BSA, ICN ImmunoBiologicals, Lisle, IL, USA) and biotin-SP-conjugated goat anti-mouse IgG (1:500, Dianova, Hamburg, Germany).
Quantitative histomorphometric analysis of the repair tissue was carried out. After digital image acquisition of a representative center cut cross section at 5x magnification, the initial cartilage defect area, the area of total regeneration tissue filling the defect, the area of cartilage flow at the defect edges, and the area of safranin O (SO)-positive stained repair tissue was determined with ImageJ software (ImageJ 1.38x, National Institutes of Health, USA). Values for the total regeneration tissue were expressed as percentage of the original cartilage defect area (=100%). Values for SO-stained areas were expressed as percentage of the total regeneration tissue (without the cartilage flow areas) and medians of each group were compared for differences. The same protocol was used to quantify the collagen type II-positive area within the total regeneration tissue.
Further, semiquantitative histomorphological evaluation of the repair tissue quality was carried out according to O'Driscoll et al. (27), reaching a maximum of 24 points (Table 1. The evaluation was carried out blinded by two of the authors (T.G., M.J.).
Summary of the Histomorphological Grading According to O'Driscoll et al.'s (27) Score System

Values are given as absolute numbers of samples and percentages of total group size. SOFG, Safranin O Fast Green.
Statistical Analysis
As variance of the evaluated groups was significantly different, the nonparametric Kruskal-Wallis test was used to test for differences in the histopathological score, the histomorphometric data, and for SO or collagen type II-positive regeneration areas of the three treatment groups. In case of significance the nonparametric Mann-Whitney U-test for independent samples was used for evaluating differences between the single treatment groups under closed testing procedures. Significance was reported if p < 0.05. Data analysis was performed with SPSS for Windows 10.0 (SPSS Inc. Chicago, IL, USA).
Results
All animals recovered well from surgery within 1–2 days. Observation of gait did not reveal any gross limping after 2–4 days. There were no complications.
Macroscopic Results
Worst and best macroscopic results of each group are shown in Figure 2. The worst results were characterized by irregular white and reddish repair tissue and a rough surface (Fig. 2A–C). In contrast, the best macroscopic results did show a more differentiated, opaque white repair tissue with a smooth surface similar to the adjacent cartilage (Fig. 2D–F). The most favorable macroscopic result was found in the membrane + MSC-treated group (Fig. 2F).

Macroscopic results after 8 weeks. Worst (A–C) and best (D–F) macroscopic results of the three treatment groups. Empty defects (A, D); membrane (B, E); membrane + MSC (C, F). The best macroscopic overall result was seen in the membrane + MSC group with a smooth surface and a white, opaque color, similar to the adjacent cartilage (F).
Overall macroscopic results displayed a considerable interindividual variability. In 8 out of 12 samples of the membrane and membrane + MSC group we found partial loosening of the running suture (Fig. 2B, C, F), but like in all other membrane covered samples, no loss or delamination of the collagenous membrane was found histologically (Fig. 3).

Representative histomorphological section of a membrane-covered defect with autologous MSC. After 8 weeks the collagen membrane is still covering the defect area demonstrated by homogeneous collagen bundles (M: membrane, S: sutures, CF: small area of cartilage flow, C: original cartilage).
Microscopic Results
In all 12 defects covered with the collagenous membrane, remnants of the scaffold in terms of homogeneous, dense collagen fibers were seen covering the defect area (Fig. 3, M = membrane), indicating that no membrane detachment or delamination occurred within 8 weeks of full weight bearing.
The median dimension of the total repair tissue filling the defect area compared to the original defect size (=100%) was slightly elevated in the empty defect (n = 4) and the membrane group (n = 6) with 115.8% (interquartile range: 17.8) and 117.8% (interquartile range: 74.2), respectively, whereas the median defect dimension of the membrane + MSC group was 100.4% (interquartile range: 68.4). These differences were not statistically significant. The increment of total defect area filled with repair tissue in both control groups was found to base on an extension of the reparative tissue towards the subchondral bone.
Quantitative histomorphometry revealed that the largest percentage of SO-positive stained regeneration tissue was found in the membrane + MSC group with a median of 24.6% (interquartile range: 25.1) compared to 13.6% (interquartile range: 15.9) in the empty and 1.7% (interquartile range: 12.3) in the membrane group (Fig. 4, empty boxes). This tendency towards a larger area of SO-positive tissue in the membrane + MSC group was not statistically significant (Kruskal Wallis test: p = 0.066). When assessing the percentage of collagen type II-positive stained area, membrane-only-treated defects again displayed the lowest values (median: 6.2%, interquartile range: 24.8) whereas the membrane + MSC-treated defects displayed higher values (median 29.5%, interquartile range: 43.7). Empty control defects showed a similar amount of collagen type II accumulation as found in membrane + MSC-treated defects (median: 33.1%, interquartile range: 23.4). Differences did not reach significance in the Kruskal-Wallis test (Fig. 4, filled boxes).

Box plot showing the Safranin O (empty boxes) and collagen type 2 (filled boxes)-positive regeneration tissue areas in percent of the total regeneration area (=100%; without the cartilage flow areas). The horizontal bar represents the median, the box the 25–75 quartiles. No significance was reached for the higher values between the membrane and membrane + MSC group.
Of note was that the distribution of SO- and collagen type II-positive areas differed between the treatment groups: SO- and collagen type II-positive areas were found in all three treatment groups allocated at the lower defect edge close to the adjacent cartilage with cells migrating from the subchondral marrow space into the defect area (Fig. 5A). Interestingly, in the membrane + MSC group, SO- and collagen type II-positive areas were also found in the central part of the regeneration tissue in two defects (Fig. 5B).

Safranin O Fast Green staining of two specimens depicting different cell sources for GAG-positive tissue formation. (A) Empty defect specimen with cell influx at the defect rim where the subchondral bone plate was opened during defect preparation. No other zones of GAG-rich tissue are seen within the defect. (B) In this membrane + MSC-treated defect, GAG-rich tissue formation is also found within the middle of the defect.
Repair tissue morphology revealed a noticeable difference in the total number of cells and the cellular distribution within the defect area. Whereas five out of six of MSC-treated defects displayed evenly distributed cells within the reparative tissue, membrane-only-treated defects revealed a high number of cells predominantly allocated at the bottom of the repair tissue adjacent to the subchondral bone. Cells in the empty defects showed a more widespread distribution towards the surface of the defect, but with no apparent SO- or collagen type II-positive staining in the centre of the regenerated tissue.
Repair tissue morphology as semiquantitatively assessed by the O'Driscoll score showed significantly higher score values for defects treated with MSCs (median: 16.5, interquartile range: 5.25) when compared to both other groups (empty defect group: 11.5 and 4.75, p = 0.038; membrane group: 9.5 and 7.25, p = 0.015, Kruskal-Wallis test: p = 0.029) (Fig. 6). The O'Driscoll score values for the subgroups are shown in Table 1. Membrane + MSC-treated defects resulted in a well integrated and fairly thick repair tissue with a smooth surface towards the joint cavity. Safranin O staining indicated a higher content of glycosaminoglycan in the extracellular matrix when compared to the control groups. Additionally, cellular morphology showed areas with round and oval chondrocyte-like cells in the membrane + MSC group as well as in untreated defects partially lying within lacunae and surrounded by a GAG-and collagen type II-rich intracellular matrix.
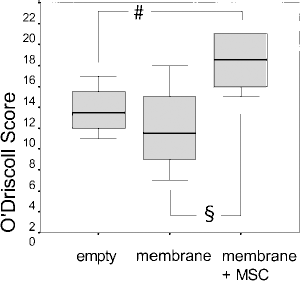
Box plot showing median, 25 and 75 quartiles of the histomorphological scoring results (O'Driscoll score, minimum: 0, maximum: 24 points) after 8 weeks. Differences in score values between empty defect group and membrane + MSC group as well as between membrane and membrane + MSC group were significant with p = 0.038 (§) and p = 0.015 (#), respectively.
Discussion
Mesenchymal stem cells have been identified as possible cell source for articular cartilage regeneration years ago, when Caplan described the function of MSCs for bone and cartilage formation in embryos (3). Since then it was a long way until techniques for isolation and expansion of MSCs found extensive use in experimental cartilage repair studies. Within the last years various studies were conducted to evaluate the potential benefit of using MSCs in full-thickness cartilage defects in small animal models (rabbits) (4,15,18,33,39).
Few studies reported experimental data from a large-animal model like sheep (10) or horses (37) with full-thickness cartilage defects penetrating the subchondral bone plate cartilage layer into the subchondral bone. Lee et al. recently reported the use of MSC in a cartilage defect model similar to ours in the pig, simply injecting the MSCs into the knee (21).
For the presented study we used the well-established Goettingen minipig model (8). To avoid extensive healing response by “intrinsic” stem cells from bone marrow below the defects, we used a chondral defect model ideally not penetrating the subchondral bone plate. We evaluated the effect of matrix-assisted autologous mesenchymal stem cell implantation when an implantation technique similar to second generation ACT was applied. We anticipated that the use of a collagen type I/III membrane for defect coverage limits the defect repair process to the endogenous repair response and the implanted mesenchymal stem cell population: first by avoiding the use of periosteum and secondly by preventing the influx of cells from the synovium by membrane coverage. Periosteum and the synovial membrane both are known as a potential source for chondroprogenitor cells and growth factors stimulating and promoting neochondrogenesis (14,25,26).
The collagen type I/III membrane used in our experiment is already used in human matrix-associated chondrocyte transplantation and was extensively tested in vitro (6,7) and in clinical routine (11,32). Our histological evaluation confirmed the possibility to securely fix the collagen type I/III membrane with running sutures and fibrin glue on articular cartilage defect in vivo, even though cartilage thickness in the minipig model was only half the amount found in humans and full weight bearing was allowed directly postoperatively. Even if the running sutures showed partial loosening at 8 weeks follow-up, no membrane detachment was found, indicating that loosening of the suture had taken place after ingrowth of repair tissue that stabilized the membrane.
The main finding of this study was a superior repair tissue quality with significantly higher histomorphologic score values in the MSC-treated group when compared to the controls. This is in accordance with the findings of Lee et al., who reported on the beneficial use of MSCs being suspended in hylan G-F 20 (Synvisc) and being injected into the knee of adult pigs after creation of a cartilage defect (21). In contrast to our study, Lee et al. had used the femoral condyle as defect side and sacrificed the animals after 6 and 12 weeks. Unlike Lee and coworkers, our data indicated only small differences in the total defect filling within the different treatment groups 8 weeks postoperatively, even though the cell number per unit and the cell distribution differed considerably between the three treatment groups. Apparently, in our experiment even empty defects were able to produce a sufficient amount of repair tissue up to 8 weeks. We have already shown in the same in vivo model, that untreated articular cartilage defects potentially heal with fibrocartilagineous repair tissue, which is condemned to degrade over time when followed up for 12 months (8). Our data further indicate that the collagen type I/III membrane forms a functional barrier, reducing synovial cell influx to the defect site from the joint cavity. As a result, lowest score values with a minimal amount of GAG- and collagen type II-positive repair tissue were found in membrane-only-treated defects. Earlier published work has shown that cells from the joint cavity (e.g., synovial membrane) may substantially contribute to repair tissue formation during articular cartilage defect healing (13,14).
Looking at the distinct parameters of the O'Driscoll score, the MSC implantation improved the SO staining, the surface regularity, and structural integrity, and at the same time positively influenced the parameters “hypo-cellularity” and “chondrocyte clustering.” Throughout all treatment groups similar values were reached for the parameter “bonding to adjacent cartilage” indicating good integration of the reparative tissue in all the three treatment groups.
Although meticulous defect preparation was performed, some minor injury to the tidemark was found histologically in about half of the defects at the defect edges, even though no macroscopic bleeding was seen during defect preparation. In these defects we noticed cellular ingrowth into the defect area from the subchondral marrow space (see arrows in Fig. 5A). This was noticed independently of the treatment group. Cell influx from the bone cavity is also known to heal osteochondral defects to a critical size (31). Obviously, the minimal cell influx in our control defects (either empty or membrane alone defects) was not sufficient to regenerate the whole defect area and therefore GAG formation was only found at the defect edges or basis of the defect in these groups. In contrast, the MSC group showed a more homogenous distribution of cells, and a more intense GAG- and collagen type II-positive extracellular matrix throughout the defect area. This effect was confirmed by significant higher histomorphologic score value in the MSC group. Whether this GAG and collagen type II deposition was performed by the implanted MSCs or by other cells being attracted by signaling molecules released from the implanted cell population should be clarified by other studies.
In this study we could show a beneficial effect of autologous mesenchymal stem cells on the regeneration of articular cartilage defects when second-generation cell transplantation technique was used in vivo. Of note was that cell influx from the joint space was reduced in the membrane-alone group compared to the empty defects, which resulted in a reduced amount of GAG and collagen type II accumulation. Further studies have to show the long-term outcome of such a cell-based treatment strategy for articular cartilage repair before transferring this protocol into clinical routine.
Footnotes
Acknowledgments
We would like to thank Dr. vet. K. Kleinschmidt for her expertise in animal anesthesia and animal care and R. Föhr and K. Goetzke for their expertise in preparing the histological sections.
