Abstract
Our objective is to evaluate if there is an association between liver fat accumulation after islet transplantation (ITx) and graft survival. A cohort study was conducted in 34 subjects with type 1 diabetes postallogeneic ITx. Liver fat content was evaluated by magnetic resonance imaging (MRI) (change in liver signal intensity on in-phase and opposed-phase images). Kaplan–Meier curves and Cox regression analysis were performed with islet dysfunction duration as the dependent variable and fat liver content as an independent one. Values of p < 0.05 were significant (SSPS®18.0 and MedCalc®12.5). Patients' mean age was 40 ± 8 years (diabetes duration: 31 ± 12 years; male: 41%). Islet survival did not differ in patients without (51 months, 95% CI 40–62 months) or with steatosis (48 months, 95% CI 38–58 months; p = 0.55) during islet dysfunction period. Nevertheless, survival curves appear to separate late in the follow-up, and after 40 months steatosis was associated with shorter graft survival (p log rank = 0.049). This association remained (RR 23.5, 95% CI 1.1–516.0; p = 0.045) after adjustments for possible confounding factors. In this sample of subjects with type 1 diabetes submitted to ITx, steatosis was not associated with islet failure in the whole cohort. However, in subjects with functional islets after 40 months, a shorter graft survival was observed in those with steatosis during the islet dysfunction period, even after adjustments to variables known to be associated with islet failure.
Introduction
Lipotoxicity has been noted as one of the mechanisms causing β-cell dysfunction and cellular death (10,11). Steatosis refers to an abnormal accumulation of free fatty acids (FFA) within hepatocytes, and it reflects an impairment of the normal processes of synthesis or elimination of fat (4). In islet transplantation (ITx), β-cells are infused through the portal vein and engrafted at the level of the hepatic sinusoids (9). The insulin secreted by islets promotes local deposition of fat, and steatosis has been observed in a significant subset of ITx recipients (2,5,14,15,20). The effects of hepatic lipid deposition on islet grafts was studied in an animal model (12) and have been associated with poorer islet graft function and histological appearance (reduced β-cell mass and increased islet fibrosis). Recently, we have reported that higher baseline serum triglycerides and very low-density lipoprotein (VLDL)-cholesterol predict earlier islet graft failure (13). This association was later confirmed in a larger sample from the Collaborative Islet Transplantation Registry (CITR) (18). However, the causal association between serum triglycerides, liver steatosis, and islet dysfunction/failure remains controversial. In clinical ITx, hepatic steatosis has been described as a marker of good islet function, as well as graft dysfunction (2,14,15,20). The quantification of liver lipid deposition prior to ITx and its modification after transplant is essential to clarify these associations.
Accordingly, this study aimed at determining the liver fat content of type 1 diabetes mellitus (DM) ITx recipients and to evaluate its association with islet graft survival.
Research Design and Methods
The research protocol was approved by the University of Miami health research ethics board (Institutional Review Board), and each subject gave written informed consent. A cohort study was conducted in 34 subjects [28 ITx alone; 6 islet after kidney (IAK)] with type 1 DM, postallogeneic ITx between 2000 and 2007 (see Results for the demographics of subjects). Inclusion/exclusion criteria, pancreatic islet isolation, infusion, and immediate posttransplant management were previously described (6,8). The initial maintenance immunosuppressive regimen consisted of tacrolimus (Astellas Pharma US, Inc., Northbrook, IL, USA) and sirolimus (Wyeth Pharmaceuticals, Inc., Philadelphia, PA, USA). Thirteen subjects were converted from tacrolimus or sirolimus to mycophenolate mofetil (Genentech USA, Inc., South San Francisco, CA, USA) or mychophenolic acid (Novartis Pharmaceuticals Corporation, East Hanover, NJ, USA).
Clinical variables (demography and anthropometry), number of infusions, and islet equivalents (IEQs) infused, exenatide use, and immunosuppressive medication were recorded. Outcomes were graft dysfunction [positive C-peptide, fasting plasma glucose (FPG) > 140 mg/dl and/ or postprandial plasma glucose > 180 mg/dl more than three times in a 1-week period and/or glycated hemoglobin (HbA1c) > 6.5% in two consecutive measurements] and graft failure [fasting C-peptide ≤ 0.10 ng/ml (two consecutive measurements in absence of hypoglycemia) or stimulated C-peptide ≤ 0.3 ng/ml]. Islets were considered functional until the date of islet failure. Capillary fasting plasma glucose and postprandial glucose were measured with a One Touch Ultra meter for measuring capillary glucose (Life Scan, Inc., Milpitas, CA, USA).
Fasting lipids [total cholesterol, high-density lipoprotein (HDL)-cholesterol, VLDL-cholesterol, and triglycerides] were measured by the enzymatic method in a Roche Cobas 6000 analyzer (Roche Diagnostics, Indianapolis, IN, USA) using the manufacturer's reagents and procedures, and low-density lipoprotein (LDL)-cholesterol was determined by the Friedewald equation (7). FPG (hexokinase-method in a Roche Cobas 6000 analyzer), and HbA1c [high-performance liquid chromatography (HPLC); BioRad, Richmond, CA, USA] were measured. C-peptide (double antibody radioimmunoassay in a Roche Cobas 6000 analyzer using the manufacturer's reagents and procedures) was measured at fasting and during mixed meal test (Boost high protein; Novartis/Sandoz–Nestle Nutrition, Vevey, Switzerland).
Patients had abdominal magnetic resonance imaging (MRI) scans (1.5-T machine; Siemens, Iselin, NJ, USA), and 95 MRIs from 34 patients were available. Table 1 describes the number of MRIs/patient in each period (baseline, off insulin, islet dysfunction, and islet failure), the time when the MRI was performed (from first islet infusion), and the MRI fat content (expressed in % of signal change—see description below). When more than one exam was available per patient in each period, the median value was used for analysis. Thus, six patients had at least one MRI available at baseline, 6 during off-insulin period, 25 during islet dysfunction, and 20 after islet failure. A radiologist (A.C.W, with 10 years of experience that included subspecialty training in abdominal imaging) reviewed the MRIs on a picture archiving and communication system workstation (Impax; Agfa, Mortsel, Belgium). The radiologist was unaware of the patients' clinical data. Magnetic resonance images were obtained on a 1.5-T magnet, using the body coil for excitation and surface coil for reception. The signal intensity from regions of interest in the liver and spleen was recorded for T1-weighted in-phase gradient-echo and opposed-phase gradient-echo sequences. These regions of interest were drawn to include representative areas of parenchyma that did not contain blood vessels or an artifact and were in the exact same location at the paired in-phase and out-of-phase sequences. Liver signal intensity was recorded as the mean of multiple readings from regions of interest placed in the anterior and posterior segments of the right lobe and in the medial and lateral segments of the left lobe. The signal intensity was measured at the level of and below the porta hepatis whenever possible (12 regions of interest). Relative liver signal intensity loss was quantified on in-phase and opposed-phase T1-weighted gradient-echo images as the percentage relative signal intensity change in the liver by using the following formula (17): (SIin - SIopp)/SIin. 100, where SI is the average liver signal intensity divided by the average spleen signal intensity, SIin is the signal intensity on in-phase images, and SIopp is the signal intensity on opposed-phase images. The signal intensity of the spleen was used as a denominator in the formula to adjust for the lack of an objective signal intensity scale on the MRI (17). The signal intensity of the spleen was obtained at levels that corresponded to the three levels at which the signal intensity of the liver was obtained; however, in some patients, the size of the spleen was too small for placement of regions of interest at three levels. The regions of interest were placed on the spleen to include representative areas of parenchyma that did not contain blood vessels or an artifact, and we avoided any regions of obvious visual homogeneity in signal intensity. Estimating liver steatosis based on this technique allows the numerical quantification of fat deposition, while most of the other techniques only describe the presence or the absence of liver fat deposition. A positive result is associated with the presence of steatosis, and a signal change ≥3% corresponds to at least grade 1 steatosis in histology (1,16,19). The radiologist also described the presence of steatosis based on subjective visual assessment of MRIs, which is how this diagnosis is usually made in the clinical setting, as well as how it has been reported in the previous islet transplant series (2,8,14,15,20).
Number of MRIs, Time When the MRI Was Performed (in Months From First Islet Infusion), and MRI Fat Content (in % of Signal Change) Before (Baseline) and After Islet Transplant (Off-Insulin, Islet Dysfunction, and Islet Failure Periods)

MRI, magnetic resonance imaging. If more than one MRI/patient was available in each period, the median (interquartile interval) was calculated and used for analysis for score change and time from first infusion.
not significant for differences among groups.
Statistical Analysis
Variables were described as mean (SD), median (interquartile range), and number of cases (%). Variables with nonnormal distribution were log transformed before analysis. The variations in liver fat content according to islet function were evaluated by t test with Bonferroni correction. Patients were also stratified based on the absolute quantity of liver fat during islet dysfunction. This period was chosen because a higher number of patients had a MRI available during islet dysfunction. Baseline characteristics were analyzed by t test or chi-squared test. Kaplan–Meier curves and Cox regression analysis were performed with islet dysfunction duration as the dependent variable and fat liver content (signal change ≥3% during islet dysfunction) as the independent one. Values of p < 0.05 (two-tailed) were significant. SSPS®18.0 (IBM, Armonk, NY, USA) and MedCalc® 12.5 (Ostend, Belgium) were used for analysis.
Results
Patients' mean age at baseline was 40 ± 8 years, and the diabetes duration was 31 ± 12 years. Fourteen patients (41%) were male, and all were white. The mean body mass index (BMI) was 23.5 ± 2.3 kg/m2, HbA1c was 7.1 ± 1.1%, basal C-peptide was 0.15 ± 0.2 mg/dl, and insulin dose/kg was 0.51 ± 0.11 UI/kg/day. Subjects received a mean of 1.85 ± 0.78 islets infusions. In a mean follow-up from first islet infusion of 45 ± 23 months, all patients have achieved the goal of glucose stability and avoidance of hypoglycemia, and insulin independence was observed in 26 (77%).
Steatosis, based on subjective visual assessment of MRIs by a radiologist, was identified only in 6 (18%) out of the 34 patients (no cases at baseline or during off-insulin period, 5 during islet dysfunction, and 1 during both islet dysfunction and after islet failure). Twenty-one patients (62%) had a MRI signal change corresponding to grade 1 steatosis (signal change ≥3%) during the follow-up (Table 1), and these proportions appeared to be higher during off-insulin and islet dysfunction periods (baseline: 0%; off-insulin period: 67%; islet dysfunction: 48%; and islet failure: 25%), but the changes were not statistically significant (p > 0.05). The fat liver content did not vary significantly after islet transplant [median (interquartile interval) - signal change at baseline: −7.30% (-11.55% to −4.88%); off-insulin period: 5.4% (-3.59% to 12.11%); islet dysfunction: 2.41% (-3.35% to 10.11%); and islet failure: −3.43% (-7.44% to −0.54%); p > 0.05] (Table 1) in the whole sample.
Clinical and laboratory baseline characteristics of patients based on the presence of MRI signal change corresponding to grade 1 steatosis during the islet dysfunction period are presented in Table 2 (n = 25). The groups were similar for all clinical and laboratory characteristics, with the exception for a higher prevalence of males (23% vs. 67%; p = 0.03), a tendency for higher insulin dose at baseline (0.48 ± 0.09 vs. 0.56 ± 0.13 UI/kg/day; p = 0.08), and for lower frequency of exenatide use (69% vs. 33%; p = 0.07) among those with steatosis during islet dysfunction period.
Subject Characteristics According to the Presence or Absence of Grade 1 Steatosis (MRI Signal Change ≥3%) During Islet Dysfunction
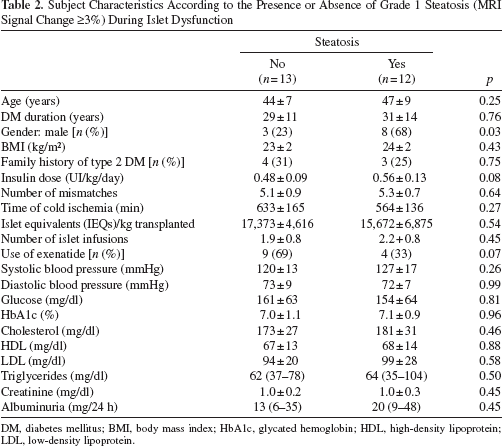
DM, diabetes mellitus; BMI, body mass index; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
The islet graft survival did not differ in patients without (51 months, 95% CI 40–62 months) or with MRI signal change corresponding to grade 1 steatosis during islet dysfunction period (48 months, 95% CI 38–58; p = 0.55; Figure 1). However, the survival curves appear to separate late in the follow-up, and in the subset of patients with functional islets after 40 months (n = 15 subjects), steatosis was associated with shorter graft survival (p log rank = 0.049). When the presence of steatosis was based on subjective visual assessment of MRIs, and not on objective quantification of the signal change, no differences on islet survival was observed between patients with or without steatosis (data not shown).
A Cox regression analysis was performed in order to adjust for possible confounders. The presence of MRI signal change corresponding to grade 1 steatosis remained associated with shorter graft survival (RR 23.5, CI 95% 1.1–516.0; p = 0.045) after adjustments for gender, number of human leukocyte antigen (HLA) mismatches, IEQs/kg, islet infusions, and use of exenatide.
Conclusions
In this sample of subjects with type 1 DM submitted to ITx, the fat liver content did not change significantly over time, as well as the presence of steatosis grade 1 during islet dysfunction period was not associated with earlier islet failure when data from all patients were analyzed together. However, in subjects with functional islets after 40 months, a shorter graft survival was observed in those with steatosis during islet dysfunction period, even after adjustments to variables known to be associated with islet failure.
Liver steatosis, based on subjective visual assessment, has been reported in 20% of the patients submitted to ITx (2,8,14,15). The ≥3% change in MRI signal intensity used at the present study corresponds to steatosis grade 1 at histology (1,16,19). This technique is more sensitive in detecting this alteration, which may explain the high steatosis frequency observed in our cohort (62%). Recently, Venturini et al. reported lower C-peptide levels in patients with steatosis detected by ultrasound (20). This method has been used in the clinical practice to diagnose steatosis; however, it is not the most sensitive one (1,16,19). Additionally, the ultrasound only categorizes the patients based on the presence or absence of steatosis and does not quantify the degree of fat accumulation. The gold standard method to describe the quantity of liver fat is the MRI with spectroscopy. However, this technique requires special procedures during image acquisition, and its interpretation is only possible with specific software that is not widely available. Although spectroscopy was not available for this study, an alternative way to determine the quantity of fat deposited in the liver was performed using the change in signal in the opposed-phase MRI (1,16,19,21). This technique has been previously validated and correlates with liver fat content determined by histopathology (3). It is particularly useful for this scenario since the liver signal intensity was recorded as the mean of multiple readings from regions of interest placed in the anterior and posterior segments of the right lobe and in the medial and lateral segments of the left lobe accounting for the heterogeneous pattern of fat distribution created by pancreatic islets. In the present study, the use of MRI made possible the identification of more patients with clinical significant steatosis and the evaluation of even subtle increments in fat accumulation, which could not be detected by conventional methods.

Percentage of subjects with functional islets according to the presence of liver steatosis at magnetic resonance imaging (MRI) corresponding to grade 1 at histology during islet dysfunction. Continuous black line, without steatosis; dashed gray line, with steatosis. p Log rank = 0.55 for whole period and p = 0.049 when only patients with more than 40 months of islet survival were included in the analysis.
An interesting aspect of our results is the lack of association between islet survival and steatosis when the whole follow-up period was taken into consideration, contrasting with results observed in the subset of patients with functional islets after 40 months, as well as after the adjustments for gender, number of HLA mismatches, IEQs/kg and islet infusions, and use of exenatide. Probably, these cofactors play more important roles in the early phases of islet survival, while islet death secondary to lypotoxicity requires a longer exposition period to represent a clinical significant phenomenon.
Another aspect that deserves a commentary is the tendency for higher insulin dose/kg in patients with steatosis during islet dysfunction period. Higher insulin needs to achieve the same HbA1c at baseline could represent increased insulin resistance in this subset of patients, and the association between steatosis and insulin resistance is well defined in the literature (4). However, other classical clinical markers of insulin resistance, such as BMI, blood pressure, HDL-cholesterol, and triglycerides, were not different between groups. In addition, family history of type 2 diabetes, a marker of a genetic predisposition to insulin resistance, was similar between groups.
Owing to the retrospective aspect of this study, not all time points are available for all subjects. Since not all patients had an MRI available at baseline, we cannot be sure if steatosis was not present in some patients before transplant. That is an important limitation that needs to be considered when interpreting the results from this analysis. In addition, another limitation is the small sample size, which can be accountable for some of our negative results, especially when considering the lack of modification in liver fat during the follow-up in comparison with baseline results. Our sample probably is underpowered for this analysis, and a more expanded series would provide more compelling evidence of a potential interaction between steatosis and graft function. However, we believe that it also demonstrates the strength of our findings, since we were able to show a significant association between liver fat and islet failure even with a reduced number of patients. Another possible limitation is the fact that steatosis is not a homogeneous process. It can affect one area more than others and even be focal. Imaging with T1 in and out of phase averages the signal intensity of several areas and may not call small grades of “focal' steatosis.
In conclusion, steatosis was not associated with islet failure in the whole cohort of ITx recipients. However, in subjects with functional islets after 40 months, a shorter graft survival was observed in those with steatosis during islet dysfunction period, even after adjustments to variables known to be associated with islet failure. Prospective clinical trials should address whether it is caused by lipotoxicity and if strategies targeting resolution of steatosis may prolong islet graft survival.
Footnotes
Acknowledgment
This study was supported by NIH/ NCRR (U42 RR016603, M01RR16587); JDRFI (#4-2000-946, 4-2004-361); NIH/NIDDK (5 R01 DK55347, 5 R01 DK056953); State of Florida, and the Diabetes Research Institute Foundation (Hollywood, FL). C.B.L. takes full responsibility for the work as a whole, including the study design, access to data, and the decision to submit and publish the manuscript. C.B.L. researched data, contributed to discussion, wrote manuscript, reviewed/ edited manuscript; E.M.L.P. researched data, contributed to discussion, wrote manuscript, reviewed/edited manuscript; A.C.W. researched data, contributed to discussion, wrote manuscript, reviewed/edited manuscript; L.G.M.Z. contributed to discussion; V.L. researched data, reviewed/edited manuscript; K.B. researched data, reviewed/edited manuscript; A.C. contributed to discussion; C.R. contributed to discussion, reviewed/edited manuscript; R.A. contributed to discussion, wrote manuscript, reviewed/edited manuscript. The authors declare no conflicts of interest.
