Abstract
Our objective was to evaluate the feasibility, safety, and efficacy of intravenous (IV) infusion of allogenic mesenchymal stem cells (MSCs) in ankylosing spondylitis (AS) patients who are refractory to or cannot tolerate the side effects of nonsteroidal anti-inflammatory drugs (NSAIDs). AS patients enrolled in this study received four IV infusions of MSCs on days 0, 7, 14, and 21. The percentage of ASAS20 responders (the primary endpoint) at the fourth week and the mean ASAS20 response duration (the secondary endpoint) were used to assess treatment response to MSC infusion and duration of the therapeutic effects. Ankylosing Spondylitis Disease Activity Score Containing C-reactive Protein (ASDAS-CRP) and other preestablished evaluation indices were also adopted to evaluate the clinical effects. Magnetic resonance imaging (MRI) was performed to detect changes of bone marrow edema in the spine. The safety of this treatment was also evaluated. Thirty-one patients were included, and the percentage of ASAS20 responders reached 77.4% at the fourth week, and the mean ASAS20 response duration was 7.1 weeks. The mean ASDAS-CRP score decreased from 3.6 ± 0.6 to 2.4 ± 0.5 at the fourth week and then increased to 3.2 ± 0.8 at the 20th week. The average total inflammation extent (TIE) detected by MRI decreased from 533,482.5 at baseline to 480,692.3 at the fourth week (p > 0.05) and 400,547.2 at the 20th week (p < 0.05). No adverse effects were noted. IV infusion of MSCs is a feasible, safe, and promising treatment for patients with AS.
Keywords
Introduction
Ankylosing spondylitis (AS) is a chronic, immunemediated inflammatory disease that is associated with inflammation in the axial skeleton, peripheral joints, and nonarticular structures (28). Nonsteroidal anti-inflammatory drugs (NSAIDs) incontrovertibly relieve symptoms of AS (25,34), and their constant use may reduce the rate of ankylosis (26,35). Unfortunately, some patients have a poor response to NSAIDs (22), and they are associated with gastrointestinal and cardiovascular toxicity. Results from two large multicenter clinical trials demonstrated that administration of sulfasalazine had no significant beneficial effects on AS patients unless they also had peripheral joint disease (5,13). Conventional disease-modifying antirheumatoid drugs (DMARDs), however, exert neither symptomatic nor disease-modifying effects on the spine, and although used for the treatment of peripheral joint disease, evidence of efficacy is limited (1,34). The introduction of tumor necrosis factor (TNF) blockers for the treatment of AS has been considered the greatest breakthrough over the past 50 years (1,12,18,29). However, their use can increase the risk of fungal and bacterial infections (15,23,24), and the personal and societal financial burden has limited the availability of effective anti-TNF treatments (1). Moreover, some patients are refractory to treatment with TNF blockers (1,6).
Many studies have demonstrated that AS is an autoimmune disease (1,12). Mesenchymal stem cells (MSCs) are adult stem cells with desirable properties for cell-based clinical therapies (16). Recent studies have proven the immunosuppressive and anti-inflammatory properties of MSCs (9,10,20,39). This has opened the way for novel therapeutic applications for the treatment of autoimmune and inflammatory diseases. Research has shown the safety and efficacy of MSC infusion or transplantation in various autoimmune and inflammatory diseases including graft-versus-host disease (GVHD) (21,37), Crohn's disease (4,8), multiple sclerosis (8), systemic lupus erythematosus (SLE) (30,32,33), and systemic sclerosis (19). In 2011, our research showed that, compared to normal donors, the immunomodulatory ability of MSCs was reduced in AS patients. Accordingly, the dysfunctional MSCs of AS patients induced an abnormal ratio of T helper 17/regulatory T (Th17/Treg) cells, manifested by increased Th17 cells and reduced Treg cells in cluster of differentiation 4-positive (CD4+) T-cell subgroups after a mixed lymphocyte reaction (MLR). Hence, the immunoregulatory dysfunction of MSCs may play a novel role in AS pathogenesis by inducing an imbalanced ratio of Th17/Treg cells (38).
Thus, the purpose of this study was to determine the safety and efficacy of MSC IV infusion for the treatment of AS.
Materials and Methods
Overall Study Design
This was a 20-week open-label clinical study of MSC intravenous (IV) infusion in active AS patients who had a suboptimal response to, or could not tolerate, the side effects of NSAIDs. Patients were strictly screened according to the inclusion and exclusion criteria as defined below, and all discontinued their current treatment for AS for at least 72 h prior to MSC treatment to minimize the impact of their prior therapy. All patients received 1 × 106 MSCs/kg body weight in 10 ml normal saline at baseline (day 0), the first week (day 7), the second week (day 14), and the third week (day 21). MSC infusion is a mature treatment for autoimmune diseases, and the infusion dose of approximately 1 × 106 MSCs/kg body weight has been used previously in GVHD and Crohn's disease (14,36).
The study protocol was approved by the research ethics board of Sun Yat-sen Memorial Hospital and is presented in Figure 1. All patients provided written informed consent for participation in the study.

Flow chart of study. Abbreviations: AS, ankylosing spondylitis; MSC, mesenchymal stem cell; NSAIDs, nonsteroidal anti-inflammatory drugs; bw, body weight; NS, normal saline; W, week(s); IV, intravenous.
Patient Population
Patients between 18 and 40 years old were eligible if they fulfilled the following inclusion criteria. (1) A diagnosis of AS according to the modified New York Diagnostic Criteria. (2) Ability to provide informed consent. (3) Active disease after adequate therapeutic trials. (4) Drug withdrawal because of intolerance to the side effects of NSAIDs such as peptic ulcer, hepatorenal damage, and cardiovascular events. Active disease was defined according to the presence of at least two of the following three criteria: Ankylosing Spondylitis Disease Activity Score Containing C-reactive Protein (ASDAS-CRP) score of ≥3.5 (31), total back pain score ≥4 [on a 0–10 cm visual analog scale (VAS)] and/or morning stiffness ≥1 h, and adequate therapeutic trial of NSAIDS defined as at least two NSAIDs over a 4-week period in total at the maximum recommended or tolerated anti-inflammatory dose unless contraindicated.
Exclusion criteria were active infections or high risk for infections (e.g., diabetes, AIDS, and severe pulmonary heart disease); malignancy or premalignancy condition; intra-articular corticosteroid injections or IV methylprednisolone within the 3 months prior to study entry; abnormal serum creatinine; major surgery within the previous 3 months or planned in the ensuing 6 months; severe comorbidities including cardiovascular, cerebrovascular, liver, renal, and hematological system diseases or mental disease; or pregnant or breastfeeding female patients.
Preparation of MSCs
Thirty-five young male donors, 20 to 30 years of age, were enrolled all from Sun Yat-sen University. After acquiring informed consent of possible risks, 30 ml of bone marrow was obtained by puncture of the posterior superior iliac bone by skilled allied health professionals.
Bone marrow MSCs were isolated from the bone marrow aspirates by density gradient centrifugation as previously described (7,38). The cultured MSCs were identified by the minimal phenotypic and functional criteria proposed by the International Society for Cellular Therapy (ISCT) (11). In brief, human MSCs were harvested and analyzed by flow cytometry with a FACSCalibur flow cytometer (BD Biosciences, San Diego, CA, USA). The primary antibodies used were as follows: isotype-control fluorescein isothiocyanate (FITC) mouse IgG1, CD29_FITC, CD44_ FITC, CD105_FITC (all from BD Pharmingen, Bedford, MA, USA); CD34_FITC (Southern Biotech, Birmingham, AL, USA); CD45_FITC (Caltag, Burlingame, CA, USA); and human leukocyte antigen (HLA)-DR_FITC (Santa Cruz, Santa Cruz, CA, USA).
Cell stemness was assessed by testing the ability of MSCs to differentiate into adipocytes, osteoblasts, and chondrocytes. To induce osteogenic differentiation, MSCs were allowed to grow in osteogenic medium [high-glucose Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum (FBS), 50 mg/l ascorbic acid, 10 mM β-glycerol phosphate, and 10 nM dexamethasone; all from Sigma-Aldrich, St. Louis, MO, USA]. To induce adipogenic differentiation, the MSCs were cultured in adipogenic medium (low-glucose DMEM supplemented with 10% FBS, 1 μM dexamethasone, 10 μg/ml insulin, 0.5 mM 3-isobutyl-1- methylxanthine, and 0.2 mM indomethacin; all from Sigma). To induce chondrogenic differentiation, the MSCs were seeded in high-glucose DMEM supplemented with 1% ITS-Premix (Becton-Dickinson, Mountain View, CA, USA), 50 mg/ml ascorbic acid (Sigma), 10−3 M sodium pyruvate (Sigma), 10−7 M dexamethasone (Sigma), and 10 ng/ml transforming growth factor-β3 (R&D Systems, Minneapolis, MN, USA). Finally, Oil red O, alizarin red, and Alcian blue dyes (all from Sigma) were used to identify adipocytes, osteoblasts, and chondrocytes, respectively (Fig. 2). Cell viability was determined at each passage, and morphology was monitored twice a week throughout the culture period. Absence of contamination by pathogens (bacteria, fungi, mycoplasmas, and viruses) and negative endotoxin test were completed at culture initiation and before harvest of MSCs. For the pathogen test, a minimum of 3 ml of culture supernatant was introduced into a Hi media blood culture bottle (C.D.RICH, Chengdu Rich Science Industry Co., Sichuan, China) containing 50 ml of glucose broth (Hopebio, Qingdao Hope Biol-Technology Co., Qingdao, China). All blood culture bottles were incubated at 37°C for an initial period of 24 h and subcultured on MacConkey agar (Oxiod, Basingstoke, UK) for another 72 h. Pathogens were identified on the basis of standard cultural, microscopic, and biochemical characterization. For the endotoxin test, the culture supernatant was analyzed by the limulus amebocyte lysate (LAL) gel-clot assay (Gulangyu Pai, Chinese Horseshoe Crab Reagent Manufactory Co., Xiamen, China). The test was carried out according to manufacturer's instructions. A positive reaction takes place in the presence of endotoxin. In addition, a standard G-banding procedure was used to perform karyotype analysis for propagated MSCs. Briefly, the metaphase chromosomes of MSCs are treated with trypsin (to partially digest the chromosome) and stained with Giemsa stain (both from Sigma). Then banding was used to identify chromosomal abnormalities, such as translocations, because there is a unique pattern of light and dark bands for each chromosome. Passage three to five were used, and 31 AS patients received cells derived from two or more donors.
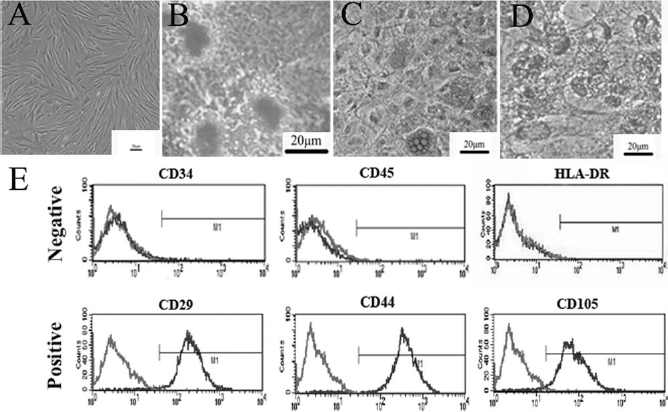
Immunophenotype and differentiation potential of MSCs. (A) Under the 40× microscope, spindle-shaped MSCs grow like intersecting bundles. (B) After osteogenic-inducing culture, MSCs were stained for the formation of mineralized matrix by Alizarin red stain (×400). (C) After adipogenic-inducing culture, MSCs were stained for lipid vacuoles with Oil-red O (×200). (D) After chondrogenic-inducing culture, MSCs were stained for chondrocyte matrix with Alcian blue (×200). (E) MSCs were harvested, labeled with isotype control (light gray) or antibodies against immunophenotype (dark gray), and analyzed by flow cytometry. MSCs expressed CD29, CD44, and CD105, but not of CD34, CD45, and HLA-DR.
Clinical Assessment
The primary efficacy endpoint was the percentage of assessment in Ankylosing Spondylitis Response Criteria (ASAS)20 responders at the fourth week. ASAS20 response is defined as improvement of >20% and >1 unit on a 0–10 cm VAS compared with baseline in at least three of the four domains (31) [patient global, back pain, Bath Ankylosing Spondylitis Functional Index (BAFSI), inflammation of the spine] as well as no worsening of >20% and >1 unit on a 0–10 cm VAS in the remaining.
The secondary efficacy endpoint was the mean duration of ASAS20 response. The duration of ASAS20 response of every patient was expressed as the difference of the time point (weeks) when ASAS20 response ended and the time point (weeks) when ASAS20 response emerged. The mean duration of ASAS20 response was defined as the ratio of the sum of the duration of ASAS20 response of every patient and the patient number of the final cohort.
Additional efficacy assessments included the ASAS40 Improvement Criteria and ASAS Partial Remission Criteria. ASAS40 response is defined as improvement of at least 40% and absolute improvement of at least 2 units on a 0–10 cm VAS compared with baseline in three or more of the four domains with no worsening at all in the remaining domain. ASAS Partial Remission Criteria is defined as a value not above 2 units on a 0–10 cm VAS in each of the domains. ASDAS-CRP, BASFI, Bath Ankylosing Spondylitis Metrology Index (BASMI), Maastricht Ankylosing Spondylitis Enthesis Score (MASES), Total Back Pain (TBP), Patient Global (PG), Erythrocyte Sedimentation Rate (ESR), and CRP were used to assess the clinical effects of MSC infusion. Professionally trained researchers were responsible for assessing spinal mobility by BASMI.
Bone marrow edema, caused by local inflammation of bone marrow, can be displayed clearly after a magnetic resonance imaging (MRI) scan. MRI of the spine from C2 to S1 was performed with a 3.0-Tesla MR scanner (Philips Medical Systems, Best, The Netherlands) at baseline and the fourth and 20th week for 12 of the patients. The MRI sequence included a T1-weighted turbo spin-echo (TSE) sequence and a fat-saturated short tau inversion recovery (STIR) sequence.
MR images were first analyzed using the ASspiMRI-a spine-specific (spi) scoring system for measuring AS by MRI (2), which is based on grading disease activity on a scale of 0 to 6. In addition to the ASspiMRI-a scoring system, the inflammation area (Fig. 3B, underlined in red) and average intensity (Fig. 3B, underlined in green) of each inflammatory site were calculated using the Philips Extending MRI Workspace 2.6. The background value (BV) was obtained by taking 10 normal sites of the vertebral body of one layer and calculating the average. The inflammation extent of each inflammatory site was calculated by the formula: value of inflammation area (VIA) × [value of average intensity (VAI) - BV].
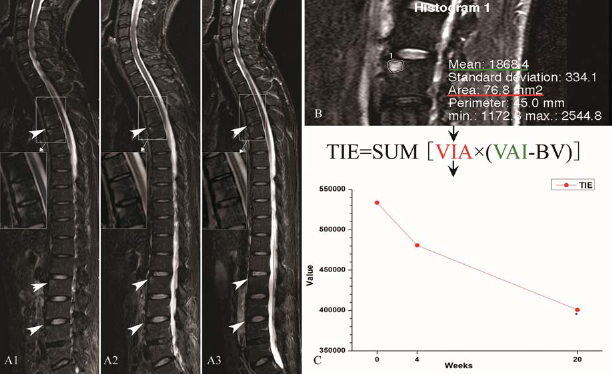
MRI measurements and the corresponding values for spinal bone marrow edema of AS patients. (A1–A3) The full spine magnetic resonance imaging (MRI) scan sagittal images of an AS patient who received MSC infusions at the baseline (A1), fourth (A2), and 20th week (A3) by the fat-saturated short tau inversion recovery (STIR) sequence. The sites with high signal (marked by white big arrow) show the place where bone marrow edema occurred. More detail of edema can be appreciated at higher resolution (amplified by white small arrow). (A) After the MSC infusion, bone marrow edema parts of the AS patients shrunk and got remitted gradually. (B) The method to measure bone marrow edema area (red line) and average intensity (green line) with Philips Extending MRI Workspace 2.6, and to calculate the inflammatory extent of spinal vertebral bone marrow edema in AS patients, according to the displayed formula. (C) The declined trends of all AS patients with total spine vertebral bone marrow edema value at three time points. Abbreviations: TIE, total inflammation extent, VIA, value of inflammation area, VAI, value of average intensity, B V, background value of noninflammatory vertebral body.
The summation of the inflammation extent of all inflammatory sites in all scanning layers was defined as the total inflammation extent (TIE) of each patient. We compared TIE of the 12 patients who were evaluated by MRI at baseline, the fourth week, and the 20th week. All the evaluations above for each patient, before and after treatment, were conducted by three experienced radiologists independently who were blind to the trial.
Other parameters monitored throughout the trial included body temperature, pulse rate, respiration rate, blood pressure, complete blood count (CBC), routine urine and stool testing, blood creatinine, alanine transaminase, and aspartate transaminase levels, antinuclear antibody testing, electrocardiogram, and chest radiographs. These data were obtained by skilled allied health professionals strictly according to international standardized procedures when the patients were enrolled in this study.
Statistical Analysis
Data were expressed as mean ± standard deviation (SD) and analyzed using SPSS13.0 (IBM, Armonk, NY, USA). Single factor analysis of variance was performed by ANOVA, followed by Dunnett's test, with an α value of p < 0.05 being regarded as statistically significant.
Results
Demographic and Baseline Disease Characteristics
A total of 37 AS patients who met the inclusion and exclusion criteria who were seen at our hospital between April 2011 and July 2011 were selected for this study. Four of the 37 patients refused the protocol and did not participate in this clinical trial, and two patients dropped out. Therefore, the final cohort included 31 patients. Of the 31 patients, 22 patients had a suboptimal response to NSAIDs, and 9 could not tolerate the side effects.
The mean age of the patients was 27.8 years (range, 22–33), the mean disease duration from diagnosis was 33.9 months (range, 28–39.6), and the mean ASDAS-CRP score at entry was 3.6 ± 0.6. Other demographic data and baseline disease characteristics for all patients are presented in Tables 1 and 2. Twelve of the 31 patients were evaluated by MRI, and their demographic data and disease characteristics before and after treatment are shown in Tables 2 and 3.
Disease Characteristics Before and After MSC IV Infusion (Data Are Expressed as Mean ± SD).

Abbreviations: MSC, mesenchymal stem cell; I V, intravenous; ASDAS, Ankylosing Spondylitis Disease Activity Score; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; MASES, Maastricht Ankylosing Spondylitis Enthesis Score; TBP, Total Back Pain; PG, Patient Global; ESR, Erythrocyte Sedimentation Rate; CRP, C-reactive protein; MRI, magnetic resonance imaging; B, baseline; W, week(s).
Demographic Data and Disease Characteristics of All Enrolled Patients and the Subgroup of Patients Evaluated by MRI at Baseline

HLA, human leukocyte antigen; NSAIDs, nonsteroidal anti-inflammatory drugs.
Outcome Measures of the 12 Patients Evaluated by MRI

Abbreviations: ASDAS, Ankylosing Spondylitis Disease Activity Score; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; MASES, Maastricht Ankylosing Spondylitis Enthesis Score; TBP, Total Back Pain; PG, Patient Global; ASAS, Assessment of SpondyloArthritis International Society; ESR, Erythrocyte Sedimentation Rate; CRP, C-reactive protein.
Clinical Effects
After being tested for the phenotype expression and multiple lineage differentiation potential according to the ISCT minimal functional criteria, MSCs were intravenously infused into selected AS patients. The percentage of ASAS20 responders at the fourth week was 77.4% (24/31). In addition, ASAS20 Response Criteria were achieved by 2 patients (6.5%) at the first week, 13 patients (41.9%) at the second week, 19 patients (61.3%) at the third week, 21 patients (66.7%) at the eighth week, 17 patients (54.8%) at the 12th week, 10 patients (32.3%) at the 16th week, and 2 patients (6.5%) at the 20th week (Fig. 4A). The mean duration of ASAS20 response was 7.1 weeks after MSC IV infusion (Fig. 4B). There were two patients who previously achieved and maintained the ASAS20 Criteria to the 20th week, while 10 patients never reached the ASAS20 Criteria. ASAS40 Response Criteria were achieved by eight patients (25.8%) at the fourth week and two patients (6.5%) at the 20th week. No patient achieved ASAS Partial Remission Criteria at any time point during the study (Fig. 4A).
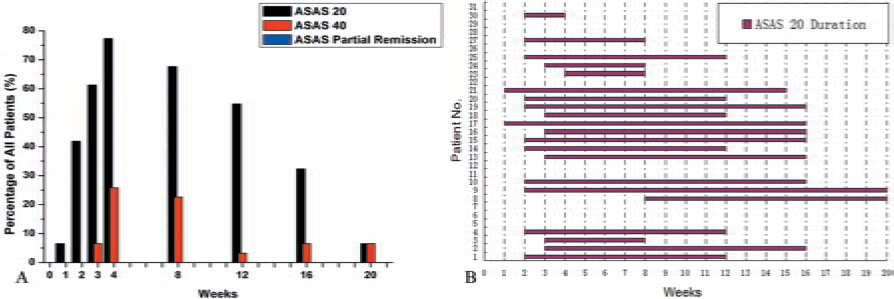
Histogram representation of the treatment response of AS patients to MSC IV infusion assessed by the ASAS20 Improvement Criteria, the ASAS40 Improvement Criteria, the ASAS Partial Remission Criteria, and the ASAS20 duration time. (A) One patient (6.7%) achieved the ASAS20 Improvement Criteria after just one infusion of MSCs, and all the patients (100%) achieved it at the fourth week and maintained the efficacy for at least 8 weeks. Eight patients (53.3%) met the ASAS20 Improvement Criteria at the 16th week and no patient at the 20th week. The ASAS40 Response Criteria were achieved by two patients (13.3%) after three infusions of MSCs, nine patients (60.0%) at the fourth week, six patients (40.0%) at the eighth week, and no patient at the 12th week and thereafter. No patient achieved ASAS Partial Remission Criteria at any time point. (B) The histogram reflects the time points of 31 AS patients to reach ASAS20 Improvement Criteria and the respective duration of maintaining ASAS20 improvement. Data indicate that the average duration time of ASAS20 in the 31 AS patients was 7.1 weeks.
Figure 5 shows the mean scores and values of the preestablished evaluation indices at baseline and at 1, 2, 3, 4, 8, 12, 16, and 20 weeks after MSC infusion. The minimum mean of ASDAS-CRP scores (indicating disease activity), which appeared at the fourth week, was 2.4 ± 0.5 and declined significantly compared to the mean ASDAS-CRP score at baseline (p < 0.01). After the fourth week, the mean ASDAS-CRP score gradually increased and reached 3.2 ± 0.8 at the 20th week with an increase of 0.8. Although the mean ASDAS-CRP score had decreased by only 0.4 from baseline at the 20th week, the difference was still statistically significant (p < 0.05) (Table 1 and Fig. 5A). The mean CRP value reached a minimum (18.9 ± 14.7 mg/L) (p < 0.05, compared to baseline) at the fourth week, but the mean ESR value reached a minimum (17.7 ± 14.5 mm/h) (p < 0.01, compared to baseline) at the eighth week (Table 1 and Fig. 5C). Similar variations were found in the mean BASFI score (indicating functional improvement) and the mean BASMI score (indicating spinal mobility) (Table 1 and Fig. 5D).

Graphic representation of variations of the efficacy evaluation indices after MSC IV infusion. (A) The mean ASDAS-CRP score decreased from 3.6 ± 0.6 to 2.4 ± 0.5, which was its minimum after four infusions of MSCs, and then increased slowly (after 12 weeks) to 3.2 ± 0.8. Similar variations were found in the mean total back pain, patient global, stiffness and fatigue score (B), as well as the mean CRP value (C) and BASFI, BASMI (D). As shown in the graph, the mean ESR value had a smaller rangeability compared to the indices above and reached its minimum at the eighth week (C). *p<0.05 and **p<0.01. Abbreviations: ASDAS, Ankylosing Spondylitis Disease Activity Score; CRP, C-reactive protein; ESR, Erythrocyte Sedimentation Rate; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index.
ASspiMRI-a score was 7.2 ± 2.9 at the baseline, 7.0 ± 2.3 at the fourth week, and 6.9 ± 2.0 at the 20th week, respectively. Although there was no statistical significance (F = 0.031, p > 0.05), ASspiMRI-a score was slightly decreased. The average TIE, which takes into account both area of inflammation and intensity, of the 12 patients decreased from 533,482.5 ± 108,106.9 at baseline to 480,692.3 ± 134,234.8 at the fourth week (p > 0.05) and 400,547.2 ± 137,004.4 at the 20th week (p < 0.05) (Fig. 3).
Dropouts and Safety
One patient withdrew at week 4 for personal reasons, and another patient withdrew at week 12 to be treated with traditional Chinese medicine. In the remaining patients, transient fever and injection point pain occurred occasionally after MSC infusion, but no new symptoms or signs of AS appeared, and no clinically significant deteriorations were found in all the preestablished laboratory tests and radiological examinations. Thus, except for the two above-mentioned points, there were almost no adverse effects in this 20-week clinical trial.
Discussion
Although until now there have been no reports of the safety and therapeutic efficacy of MSC infusion for the treatment of AS, our previous study showed that the reduced immunomodulatory potential of MSCs in AS patients may play a novel role in AS pathogenesis (38). To the best of our knowledge, this is the first study assessing the efficacy of MSC infusion for the treatment of AS. The results showed that MSC infusion resulted in an improvement of ASAS scores and symptoms and that the results lasted for up to 7 weeks posttreatment. During the study, no clinically significant deteriorations were found in all of the preestablished laboratory tests and radiological examinations in all patients, indicating that MSC infusion is safe for the treatment of AS treatment over a 20-week period.
Our primary endpoint was the proportion of patients who achieved ASAS20 Criteria at the fourth week. After four infusions of MSCs, the percentage of ASAS20 Criteria responders reached 77.4%. In this trial, however, no patient reached the ASAS Partial Remission Criteria. There are two possible reasons for this finding. First, all of the enrolled patients had high disease activity (ASDAS-CRP > 2.1) (31) and failed treatment with NSAIDs; thus, it was difficult to reach this criteria after only four infusions of MSCs. Second, the MSC infusion protocol in this trial can possibly be improved by increasing the number of MSCs in each infusion and increasing the infusion frequency.
The duration of effectiveness is as important as response rate. Based on the mean ASAS20 duration, our results indicated that the duration of effectiveness of our protocol could last as long as 7.1 weeks after the last infusion of MSCs. It should be noted that the ASAS20 duration calculated by the time between the earliest and the last point (weeks) of achieving ASAS20 criteria may underestimate the duration of effectiveness. Among the 31 patients, there were two patients who maintained achieving the ASAS20 criteria at the 20th week, and 10 patients never reached the ASAS20 criteria at all time points. Thus, patients responded to MSC infusion very differently, and further research to determine the reasons is necessary.
In the present study, ASDAS-CRP was used to evaluate disease activity. From baseline to the fourth week, the mean ASDAS-CRP score decreased by 1.2, which indicated clinical improvement (31). We also observed that the mean ASDAS-CRP score increased 0.8 from the fourth week to the 20th week. From this, we can infer that the anti-inflammatory effects of the MSCs attenuate with time. The mean BASFI score, which indicates functional improvement, had a variation similar to the mean ASDAS-CRP score. This suggests that MSC infusion can simultaneously reduce disease activity and improve function of AS patients. Similar variations were observed in the mean ESR and CRP values, which are objective indictors.
No significant difference in bone marrow edema was observed between baseline and the fourth and 20th weeks after MSC infusion using the common MRI detection method ASspiMRI-a. However, there are limitations of ASspiMRI-a that need to be considered. The system rates according to the percentage of inflammation area over the total vertebral area on MRI sagittal slices; it cannot evaluate changes of inflammation area <25% of the vertebral body properly, and it cannot effectively detect vertebral inflammation intensity changes before and after treatment. Therefore, we proposed a new MRI evaluation method, which can effectively identify the changes of vertebral inflammation area and intensity in AS patients before and after the treatment. We used TIE to evaluate the inflammation extent of the whole spine of 12 patients. Although this evaluation method is complex, it can comprehensively reflect the inflammation extent of the whole spine, and its validity was proven by the small differences between groups. A statistically significant decrease in average TIE at the 20th week compared with the baseline value was observed. However, the time points for MRI detection may not be the most optimal because bone marrow edema changes seen on MRI often occur later than the improvement of clinical symptoms.
The improvement of patient symptoms, decrease in CRP and ESR, and reduction of inflammation by MRI all suggest that MSC infusion is effective in the treatment of patients with AS. The mechanism of this improvement may include migration of MSCs to sites of inflammation (3) and reduction of local inflammation by cell–cell contact and cytokine secretion, which can suppress the proliferation and function of its surrounding inflammatory cells (16,20,27) and suppression of local inflammation by adjusting Th17/Treg proportion (17).
There are some limitations of this study that should be considered. This was not a multicenter randomized controlled clinical trial; therefore, bias may also exist in this study. Only 31 AS patients with high disease activity participated in the study, and thus they are not representative of the whole population of AS patients. MSC infusion frequency and time interval need to be improved to provide better treatment to more AS patients. Additionally, the MRI method evaluating TIE is new and also needs to be validated in a large patient cohort. Though restricted by the above-mentioned shortcomings, this study does not provide a standard treatment program for AS therapy, and we can still draw the following conclusion: MSCs IV infusion is a feasible, safe, and promising treatment for patients with AS.
Footnotes
Acknowledgment
The authors acknowledge Jinglian Zhong, Zhiqiang Bai, and Ziliang Chen for rendering us help and support for MRI scanning. This work is supported by the National Natural Science Foundation of China (81271951), Guangdong Natural Science Foundation (S2012010009654, S2012010008927, 10151008901000076), and Guangzhou Science and Technology Foundation (2014J4100167). The authors declare no conflicts of interest.
