Abstract
Pretreatment culture before islet transplantation represents a window of opportunity to ameliorate the pro-inflammatory profile expressed by human β-cells in duress. Anakinra (IL-1 receptor antagonist) and tocilizumab (monoclonal IL-6 receptor antibody) are two known anti-inflammatory agents successfully used in the treatment of inflammatory states like rheumatoid arthritis. Both compounds have also been shown to reduce blood glucose and glycosylated hemoglobin in diabetic patients. We therefore sought to evaluate the impact of anakinra and tocilizumab on human β-cells. The islets were precultured with or without anakinra or tocilizumab and then transplanted in a marginal mass model using human islets in immunodeficient mice. Islet viability was evaluated in an in vitro model. The pretreatment culture led to a significantly improved engraftment in treated islets compared to the vehicle. Anakinra and tocilizumab are not toxic to human islets and significantly reduce markers of inflammation and cell death. These results strongly support a pretreatment culture with anakinra and tocilizumab prior to human islet transplantation.
Introduction
Islet transplantation is an attractive treatment option for type 1 diabetes, but the islet isolation process imposes a significant burden to the islets (30) and could partly account for the graft loss immediately after transplantation. Most centers in Europe culture the islets for up to 2 days before transplantation, which could further contribute to a decreased islet function (18). Albeit often serving a practical purpose, this culture period could present an opportunity to treat the islets prior to transplantation with the aim of maintaining β-cell mass and function by reducing the proinflammatory and apoptotic state of the islets. Our group and others have previously shown that anti-inflammatory agents are capable of decreasing the detrimental effect of proinflammatory cytokines in vitro (23,45) as well as improving the in vivo function (11,27). Developing strategies to counteract inflammation in culture could therefore be an important contribution to improving graft survival and function.
A key mediator of islet dysfunction and destruction is interleukin-1β (IL-1β). Anakinra, a recombinant human IL-1 receptor antagonist, has had success in the treatment of the inflammatory disease rheumatoid arthritis (RA) (19). Studies on human subjects with diabetes who have been treated with anakinra show an improvement of glycemia and a reduction of inflammatory markers like IL-6 and C-reactive protein. These data have been supported by findings in murine models, both in vivo and in vitro (11,40). Tocilizumab, a humanized IL-6 receptor-specific monoclonal antibody, was originally designed as a therapy for RA, but the drug also decreased glycosylated hemoglobin (HbA1c) in diabetic patients with RA (33). It is suggested that IL-6 is involved in the development of type 2 diabetes, but its action on glucose homeostasis is complex because IL-6 seems to have both detrimental and beneficial effects on pancreatic islets. The prevailing understanding is that IL-6 contributes to obesity-induced insulin resistance in type 2 diabetes (18).
In search of optimizing culturing conditions for human islets prior to transplantation, we here investigate the effects of cytokine receptor blockade by anakinra and tocilizumab on human islets' viability in vitro and in vivo and whether these anti-inflammatory agents could protect the pancreatic islets from cytokine-induced damages.
Materials and Methods
Human Islet Isolation and Culture
Human islets were isolated by the isolation laboratory of the Nordic Network in Uppsala, Sweden, using a modified semiautomated digestion method (16). Pancreases were procured from nine deceased donors (three males and six females), after appropriate consent for multiorgan donation, aged between 42 and 77 years with a body mass index between 20.4 and 30.5 kg/m2. Islet purity ranged between 30% and 95% as judged by dithizone staining (Sigma-Aldrich, Steinheim, Germany). One hundred microliters of dithizone was mixed with 100 μl of cell suspension. The dithizone solution itself was made by mixing 10 mg of dithizone with 100 μl of NaOH, 200 μl of ethanol, and finally adding 19.8 ml of phosphate-buffered saline (PBS; BioWhittaker, Verviers, Belgium). Within 7 days from isolation, aliquots of the islet preparations from nine different donors were placed in 90-mm Petri dishes (Sterilin, Heger AS, Norway) and cultured with or without a recombinant human IL-1 receptor antagonist [10 μg/ml, anakinra (Kineret™, Swedish Orphan Biovitrum, Stockholm, Sweden)] or the IL-6 monoclonal antibody [500 μg/ml, tocilizumab (RoActemra, Roche, Basel, Switzerland)], a mixture of proinflammatory cytokines [PIC; IL-1β (10 ng/ml), interferon-γ (IFN-γ) (50 ng/ml), and tumor necrosis factor-α (TNF-α) (10 ng/ml); all from R&D Systems, Abingdon, Oxon, UK] or a combination thereof for up to 48 h in CMRL 1066 (Mediatech, Manassas, VA, USA) containing 2.5% ABO-compatible human serum, 1% penicillin/streptomycin, 10 mM hydroxyethylpiperazine ethane sulfonic acid (HEPES), and 1% l-glutamine (Invitrogen, Oslo, Norway) at 37°C (5% CO2). Islet pellets and supernatants were collected and stored in −70°C until further analysis.
Glucose-Stimulated Insulin Secretion Assay
Twenty islets were handpicked, transferred into 12 Transwell trays (Costar, Cambridge, MA, USA), and preincubated in Krebs–Ringer bicarbonate buffer (11.5 mM NaCl, 0.5 mM KCl, 2.4 mM NaHCO3, 2.2 mM CaCl2, 1 mM MgCl2, 20 mM HEPES, and 2 mg/L albumin; all Sigma-Aldrich) containing 1.67 mmol/L glucose (Fresenius Kabi, Halden, Norway) at 37°C (5% CO2) for 30 min before the islets were incubated for 40 min in fresh Krebs–Ringer bicarbonate buffer containing 1.67 mmol/L glucose (basal). Finally, the islets were incubated for 40 min in fresh Krebs–Ringer bicarbonate buffer containing 20.0 mmol/L glucose (stimulated). The supernatants were subsequently collected, and insulin secretion was measured using a human insulin enzyme immunoassay (EIA) (Mercodia AB, Uppsala, Sweden). The capacity for insulin release was expressed as a stimulation index (SI), calculated as the ratio of stimulated to basal insulin secretion.
Viability and Apoptosis Assays
Intracellular adenosine triphosphate (ATP) content was quantified using the CellTiter-Glo® Luminescent Cell Viability Assay (Promega, Madison, WI, USA). Activity of caspase 3/7 was assessed using Caspase-Glo® 3/7 Assay (Promega). Induced cell death was determined by the detection of DNA–histone complexes present in the cytoplasmic fraction of the cells using Cell Death Detection ELISAPLUS (Roche). The assays were performed as described by the manufacturers.
RNA Extraction and Real-Time Quantitative Polymerase Chain Reaction (PCR)
Total RNA was isolated from frozen islet pellets using the RNeasy Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer's description and stored at −70°C until analysis. The concentrations of all RNA samples were quantified using a NanoDrop ND-1000 UV/Vis spectrophotometer (Saveen Werner AB, Malmö, Sweden). One microgram total RNA was reverse-transcribed using the High-Capacity cDNA Archive Kit following the manufacturer's protocol (Applied Biosystems, Foster City, CA, USA). Real-time quantitative reverse transcriptase-PCR using sequence-specific primers was performed on an ABI Prism 7900HT (Applied Biosystems) sequence detector using 2× qPCR Master Mix for SYBR Green (Applied Biosystems), 300 nM sense and antisense primers, cDNA, and water up to 25 μl. The specificities of all SYBR Green assays were confirmed by melting point analysis. Levels of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) messenger RNA (mRNA) were used for normalization of the target gene expression. All samples were run in duplicates using primers for IL-1β, IL-6, IL-8, and monocyte chemotactic protein-1 (MCP-1) (primer sequences available upon request). Standard curves were run on the same plates, and the standard curve method was used to calculate the relative gene expression.
Cytokine Content in Supernatants
IL-1β, IL-6, IL-8, and MCP-1 were measured in cell-free supernatants with EIA (R&D Systems) as described by the manufacturer.
Phosphorylation Pathway Analysis
Islets were treated with a mixture of PIC [IL-1β (10 ng/ ml), IFN-γ (50 ng/ml), and TNF-α (10 ng/ml)] with or without anakinra (10 μg/ml) and tocilizumab (500 μg/ml) for 1 h. Islets (200–400) were handpicked in columns, centrifuged for 1 min at 220 × g, and washed twice in ice-cold PBS. Phosphorylated proteins [p-extracellular signal-regulated kinase 1/2 (ERK1/2; Thr185/Tyr18), p-c-Jun N-terminal kinase (JNK; Thr183/Tyr185), p-protein 38 mitogen-activated protein kinase (p38 MAPK; Thr180/Tyr182), p-signal transducers and activators of transcription (STAT3)] were detected utilizing Luminex® technology (Luminex, Austin, TX, USA) and a dual-laser, flow-based microplate reader system (Bio-Plex® cytokine assay from Bio-Rad, Hercules, CA, USA) following the manufacturer's instructions. Each sample was correlated to the protein content of the islet pellet measured by bicinchoninic acid assay (BCA; Pierce, Rockford, IL, USA).
In Vivo Islet Transplantation and Graft Functionality
Six- to 8-week-old male severe combined immunodeficient (SCID; Cb-17) mice purchased from Taconic M&B (Ry, Denmark) were used as recipients after being rendered diabetic by a single intravenous injection of streptozotocin (200 mg/kg, Sigma-Aldrich). The mice were transplanted with pretreated islets from six different donors (five females, one male). Each donor was distributed across the three experimental conditions (control, anakinra, and tocilizumab).
The animals were housed under standard conditions in an approved unit and given free access to food and water except when fasting. The treatment of the animals was according to the Norwegian Animal Welfare Act and approved by the Norwegian Animal Research Authority. Before transplantation, all recipients had nonfasting blood glucose levels ≥20 mmol/L for 2 consecutive days as measured by a glucometer (Accu-Chek Aviva Nano, Roche Diagnostics, Indiana, USA). The mice were anesthetized by 1.5% isoflurane (Baxter, Kista, Sweden) mixed with oxygen, and a minimal mass graft of 600 islet equivalents (IEQs) was loaded into a PE 50 tubing and then transplanted under the left kidney capsule (42). Subcapsular islet transplantation in murine models has been shown to be significantly more efficient compared to intraportal transplantation and requires fewer islets (37). For these reasons, we chose to deposit the grafts under the kidney capsules. Graft function was assessed by monitoring blood glucose levels daily for the first 7 days, then twice weekly, with rejection defined as blood glucose levels of >11 mmol/L on two consecutive measurements. Glucose tolerance tests were performed 21 days after transplantation after a minimum 6-h fast. The mice were injected intraperitoneally with d-glucose (2 g/kg body weight; Sigma-Aldrich), and blood glucose levels were measured at the indicated time points. Islet graft function was proven by removal of the graft-bearing kidney to confirm the recurrence of diabetes.
Statistical Analysis
Data are presented as means ± SEM, and the GraphPad Prism 4.0 statistical software (La Jolla, CA, USA) was used for data analysis. Differences between groups were evaluated by one-way ANOVA followed by Bonferroni correction. Significance was set at p < 0.05.
Results
Anakinra and Tocilizumab Protect Insulin Secretory Function in Human Islets
To examine the effect of anakinra and tocilizumab on islet function in vitro, we performed a static glucose challenge test in the presence or absence of a PIC cocktail. Human islets cultured with or without anakinra (10 μg/ ml) or tocilizumab (500 μg/ml) for 48 h showed preserved glucose responsiveness under both basal (1.67 mM) and stimulated (20 mM) glucose conditions (Fig. 1A). As previously reported (40), PIC-treated islets showed a significantly reduced capacity for insulin secretion compared to nontreated controls (Fig. 1B). Notably, there was a shift in both basal and high glucose values resulting in a burst of insulin secretion in low-glucose concentration and a lack of compensatory increase in insulin secretion in high-glucose concentration (Fig. 1A). The treatment with anakinra increased the insulin secretion compared to the nontreated control, whereas in the presence of PIC, both anakinra and tocilizumab were able to restore the insulin secretory capacity to a significant level (Fig. 1B).

Anakinra and tocilizumab protect insulin secretion capacity in human islets. Enhanced insulin secretion in human islets treated with anakinra (ANA) or tocilizumab (TCZ) in the presence of proinflammatory cytokines (PIC) [interleukin-1β (IL-1β; 10 ng/ ml), interferon-γ (IFN-γ; 50 ng/ml), and tumor necrosis factor-α (TNF-α; 10 ng/ml)]. The effects of the treatment on low (1.67 mM) and high (20 mM) glucose-stimulated insulin secretion (A), expressed as the stimulation index calculated by dividing the concentration of insulin in high glucose on its corresponding value in low glucose concentration (B) assessed by insulin enzyme immunoassay (EIA). Data are representative of six independent experiments, and values are mean ± SEM. *p < 0.05 versus untreated controls, #p < 0.05 versus PIC-treated islets.
Anakinra and Tocilizumab Are Not Detrimental to Human Islet In Vitro
To evaluate whether anakinra and tocilizumab are toxic to human islets, we analyzed the levels of caspase 3/7 activity, DNA fragmentation, and ATP content in the islets. Treatment with anakinra (10 μg/ml) or tocilizumab (500 μg/ml) for 48 h resulted in no significant differences in apoptosis as measured by caspase 3/7 activity (Fig. 2A). Nor was the viability (cell death EIA) affected (Fig. 2B), suggesting that anakinra and tocilizumab are not toxic to human islets with the doses used in this study. To assess whether cytokine-induced apoptosis in human islets could be repressed by anakinra or tocilizumab, human islets treated or not were exposed to a mixture of PIC. As shown in Figure 2, proinflammatory cytokines significantly increased apoptosis (Fig. 2A) and cell death (Fig. 2B), whereas ATP content (Fig. 2C) was decreased in human islets. In contrast, both anakinra and tocilizumab abolished the PIC-induced apoptosis (Fig. 2A) and cell death (Fig. 2B) in human islets, suggesting that anakinra and tocilizumab protect the islets from apoptosis caused by proinflammatory cytokines. Notably, anakinra restored the ATP content in PIC-exposed islets, while tocilizumab did not (Fig. 2C).

Anakinra or tocilizumab are not toxic to human islets. After a 48-h culture of human islets with or without anakinra (ANA) or tocilizumab (TCZ), the caspase 3 activity (A), DNA fragments (B), and ATP content (C) were measured as indicated in Materials and Methods. In parallel experiments, human islets exposed to proinflammatory cytokines [PIC: IL-1β (10 ng/ml), IFN-γ (50 ng/ml), and TNF-α (10 ng/ml)] were treated with or without ANA or TCZ, and caspase 3 activity (A), cell death (B), and ATP content (C) were measured as indicated in Materials and Methods. Data are representative of six independent experiments, and values are mean ± SEM. *p < 0.05 versus untreated controls, #p < 0.05 versus PIC-treated islets.
Anakinra Protects Human Islets Against Proinflammatory Cytokines
Anakinra significantly reduced the mRNA expression of the proinflammatory mediators IL-8 (Fig. 3A), MCP-1 (Fig. 3B), IL-1β (Fig. 3C), and IL-6 (Fig. 3D) in human islets at basal levels, whereas tocilizumab did not. However, the reduced mRNA expression was not reflected in a decreased protein accumulation of IL-8 (Fig. 4A), MCP-1 (Fig. 4B), IL-1β (Fig. 4C), or IL-6 (Fig. 4D) in the media from human islets treated with anakinra or tocilizumab for 48 h compared to nontreated controls. When human islets were exposed to PIC, we found an approximately fourfold increase in the mRNA expression of IL-8, MCP-1, and IL-6 compared to nontreated controls (Fig. 3A, B, and D). Anakinra totally abolished this cytokine-induced inflammation, whereas tocilizumab did not (Fig. 3A, B, and D). To evaluate whether the cytokine-induced expression of IL-8, MCP-1, and IL-6 was associated with protein synthesis and release, protein levels in supernatants from human islets treated with anakinra in the presence of cytokines were measured by EIA. In agreement with the mRNA data, incubation with anakinra led to reduced protein levels of IL-8 (Fig. 4A), MCP-1 (Fig. 4B), and IL-6 (Fig. 4D) in cytokine-treated human islets. Conversely, anakinra seems to have no effect on the protein levels of IL-1β in cytokine-induced human islets (Fig. 4C).

Anakinra decreases the mRNA expression of IL-1β, IL-6, IL-8, and MCP-1 following PIC treatment of human islets. mRNA expression for interleukin-8 (IL-8; A), monocyte chemotactic protein-1 (MCP-1; B), IL-1β (C), and IL-6 (D) was examined by real-time PCR in human islets cultured for 48 h with or without anakinra (ANA) or tocilizumab (TCZ) and in the presence or absence of proinflammatory cytokines (PIC). The housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used for normalization of the target mRNA expression. Each column represents mean ± SEM of six different donors.*p < 0.05 versus untreated controls, #p < 0.05 versus PIC-treated islets.
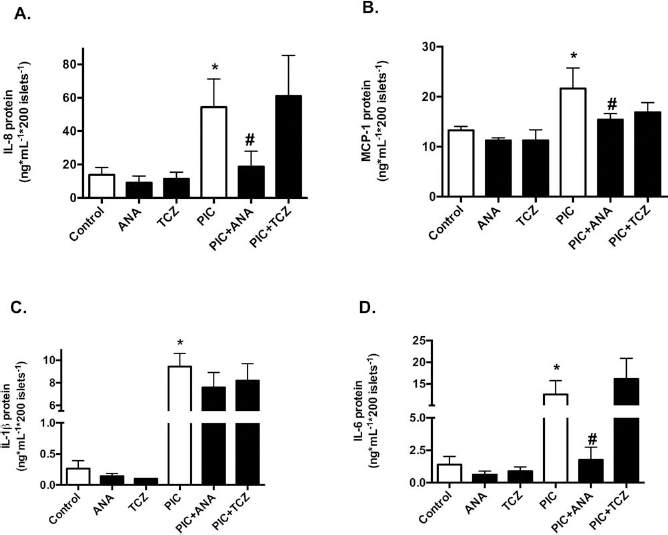
Anakinra reduces the secretion of inflammatory cytokines in human islets exposed to PIC. Protein levels of IL-8 (A), MCP-1 (B), IL-1β (C), and IL-6 (D) in supernatants from human islets cultured for 48 h with or without anakinra (ANA) or tocilizumab (TCZ) and in the presence or absence of proinflammatory cytokines (PIC). Chemokines and cytokines secreted to the culture media were measured by EIA. Data are representative of six independent experiments, and values are mean ± SEM. *p < 0.05 versus untreated controls, #p < 0.05 versus PIC-treated islets.
Cytokine Receptor Blockers Interfere with Stress-Associated Pathways in Human Islets Treated with Proinflammatory Cytokines
The molecular pathway involved in cytokine-induced islet dysfunction after the isolation process is multifactorial (1,7). Here we show that PIC stimulation of human islets for 1 h results in an increased phosphorylation of p38 (Fig. 5A), ERK 1/2 (Fig. 5B), JNK (Fig. 5C), and STAT3 (Fig. 5D). When the islets were treated with anakinra, the levels of p-p38 (Fig. 5A) and p-JNK (Fig. 5C) were significantly reduced compared to islets exposed to PIC only, whereas treatment with tocilizumab significantly reduced the levels of p-ERK 1/2 (Fig. 5B) and p-STAT3 (Fig. 5D).
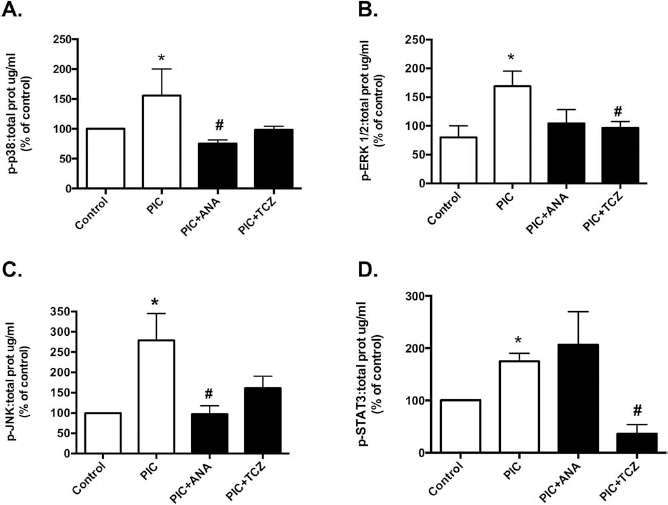
Anakinra and tocilizumab affect the phosphorylation pathways induced by PIC in human islets. Protein extracts from human islets exposed to proinflammatory cytokines (PIC) and treated with or without anakinra (ANA) or tocilizumab (TCZ) for 1 h were subjected to analysis of phosphorylated (p) proteins [38 kDa protein mitogen-activated protein kinase (p38; A), extracellular signal-regulated kinase 1/2 (ERK1/2; B), c-Jun N-terminal kinase (JNK; C), and signal transducers and activators of transcription 3 (STAT3; D)] by the Bio-Plex Cytokine Assay and normalized to total protein as described in Materials and Methods. Data are representative of six independent experiments, and values are mean ± SEM. *p < 0.05 versus untreated controls, #p < 0.05 versus PIC-treated islets.
Islets Cultured with Anakinra and Tocilizumab Engraft Better In Vivo
Finally, we wanted to evaluate the in vivo function of human islets cultured prior to transplantation with anakinra or tocilizumab. Diabetic SCID mice were transplanted with 600 IEQs handpicked human islets treated or not with anakinra (10 μg/ml) or tocilizumab (500 μg/ml) for 48 h. Engraftment was defined as time to achieve normoglycemia (sustained blood glucose less than 11 mM in each animal) posttransplantation. Every animal in all three groups was cured after the islet transplantation; however, the time to reach normoglycemia was significantly shorter in the groups receiving anakinra- or tocilizumab-treated human islets compared to untreated controls (median 2 vs. 7 days, p < 0.05). The nonfasting glucose levels were significantly lower in the treated groups versus the control for 3 weeks (p < 0.05). At posttransplantation day 28, four out of six animals in the control group were diabetic again, while the corresponding number for the anakinra and tocilizumab group was two out of six animals. Mice having received human islets cultured with anakinra or tocilizumab prior to transplantation exhibited lower glucose levels on the intraperitoneal glucose tolerance test (IPGTT) compared to control mice as shown in Figure 6B, with area under the curve analysis for the IPGTT shown in Figure 6C.

In vivo monitoring of human islets treated with or without anakinra (ANA) or tocilizumab (TCZ) for 48 h in vitro. After culture of human islets with or without anakinra (black triangle) or tocilizumab (circle), aliquots of 600 IEQs were transplanted under the kidney capsule of diabetic immunodeficient mice (n = 6 in each group). Graft function was evaluated by measuring nonfasting blood glucose levels for 28 days posttransplantation (A), and an intraperitoneal glucose tolerance test was performed at 21 days posttransplantation (B), with area under the curve analysis displayed in (C). Loss of graft function was defined by two consecutive blood glucose measurements >11 mM. Values are mean ± SEM. *p < 0.05 versus untreated controls.
Discussion
In the present study, we show that by blocking IL-1 signaling with the IL-1β receptor antagonist anakinra, and the IL-6/IL-6R signal by tocilizumab, in human islets in culture, viability and function are improved both in vitro and in vivo.
The human islet isolation procedure triggers an inflammatory state in the islets causing decreased islet survival in the peri- and posttransplantation phases (9,36). This inflammatory response is distinguished by the elevation of IL-1β, IFN-γ, and TNF-α (26), proinflammatory cytokines activating pathways that induce both apoptosis and necrosis. A significant amount of research has demonstrated the detrimental effects of these cytokines on both human and rodent islets (8,35). For these reasons, islet culture with exogenous IL-1β, IFN-γ, and TNF-α has become an established in vitro model by which pathways of cell death are induced. Pretreatment culture of human islets with anti-inflammatory agents possesses a significant opportunity for recovery and improvement of the islets before transplantation (23). In this study, we sought to evaluate the effect of two clinically approved anti-inflammatory agents, anakinra and tocilizumab, with the potential to dampen the islet inflammation and thereby improve survival and engraftment.
We revealed that a 48-h incubation of human islets with or without anakinra or tocilizumab does not negatively affect the viability and functional potency of the islets. The insulin secretory capacity (SI) is a convenient parameter that is used to assess the islets' function, though some caution must be taken into account when interpreting the data, especially if there is a high insulin release from the islets when they are stimulated with low concentrations of glucose. In our study, we found that the SI is equal to untreated controls (tocilizumab) or significantly better (anakinra). Similar findings have previously been reported in a study performed on rodent islets treated with anakinra, where the observed effect was no deficit in the glucose-stimulated insulin secretion (GSIS) and preserved in vivo function (40). Others have also described no reduction of viability in vitro when human islets are cultured for 48 h with or without anakinra (28). Moreover, when human islets are stressed by a mixture of proinflammatory cytokines (IL-1β, TNF-α, and IFN-γ), we find a significantly suppressed glucose-stimulated insulin secretion, whereas anakinra and tocilizumab abolish this effect. Additionally, we demonstrate that anakinra and tocilizumab prevent cell death, probably through increased ATP levels (3). The enhanced ATP levels may also explain the improvement in insulin secretory capacity of human islets in both basal and high glucose challenge after anakinra treatment. We emphasize that a short-term treatment with anakinra and tocilizumab in vitro is not toxic to human islets. When we block the IL-1β signaling in cultured human islets, we find a reduction of proinflammatory mediators (i.e., IL-1β, IL-6, IL-8, and MCP-1). Human islets produce several cytokines as a response to the stressing nature of the isolation process and cell culture (30), and we found that anakinra attenuates the inflammatory effects of these cytokines. IL-8 and MCP-1 are key factors in selectively recruiting monocytes and T-lymphocytes to sites of inflammation (10,12), and increased levels of MCP-1 and IL-8 in human islets have been linked to impaired graft function (6). On the contrary, we did not find that tocilizumab could counteract the negative effects of the proinflammatory cytokines. We speculate that this could be due to the potency of IL-1β. Pancreatic islets are particularly sensitive to the actions of IL-1β because the selective toxicity is executed at very low concentrations (25).
IL-6 has been shown to have both pro- and anti-inflammatory properties in human islets (5,13,34), as well as in other cell types (38,39). The former study (38) revealed that IL-6 exerts the anti-inflammatory effects through the induction of the (endogenous) IL-1 receptor antagonist, suggesting a crosstalk between these cytokines. This information, taken together with the fact that tocilizumab blocks both types of IL-6 receptors (soluble and membrane bound) (43) leading to increased levels of IL-6, could be beneficial for human islets, but further studies remain to be performed before any conclusions are drawn. Still, this puzzling quality of tocilizumab to improve islet function and viability, but not reduce inflammatory markers, begs the question of whether the principle of function is very different from anakinra. The answer to that probably lies in the understanding of the complex IL-6 biology.
Studying the signaling pathway involved in the activation of inflammation revealed that tocilizumab reduced the activation of STAT3 and ERK1/2, whereas anakinra reduced the p38 and JNK kinase pathways in human islets. The STAT pathway is implicated in growth and apoptosis signaling (29,41), and in β-cells, the apoptotic effects of IL-6 is thought to be exerted through the activation of STAT3 (34). This could indicate an important way for tocilizumab to influence the regulation of lifespan in the islets, seeing that the chief role of ERK1/2 is the regulation of proliferation, cell growth, and survival (32). Chen and colleagues recently described a conserved pathway controlling age-dependent β-cell proliferation involving the ERK pathway (4). ERK activation has previously been described in both cancer cells and murine β-cell expansion in pregnancy (15,17), but it has also been shown to increase cell survival and inhibit glucose-induced apoptosis in both human and murine β-cells (14,44).
Evidence suggests that MAPKs participate in cytokine-induced β-cell damage (24). Both p38 and JNK are known to be activated by IL-1β and play important roles in cytokine-mediated apoptosis in islets (2,22). Our results indicate that anakinra protects the islets from cytotoxicity through reduced p38 and JNK kinase activation. This is supported by the findings of others that demonstrate improved graft survival by inhibition of the p38 and JNK pathways in human islets (26).
We also tested the effect of short-term culture of human islets with anakinra or tocilizumab in vivo by using an islet transplantation model with marginal human islet grafts. Our in vivo data show that recipients having received islets treated with anakinra or tocilizumab prior to transplantation achieved normoglycemia faster than the control group (p < 0.05), and nonfasting blood glucose levels were significantly lower for 3 weeks. We also observed that mice in the untreated group lost their graft function in four out of six instances, compared to two out of six animals in the treated groups. This indicates better engraftment in the groups having received islets treated with anakinra or tocilizumab. The glucose stress test shows that mice transplanted with islets treated with anakinra or tocilizumab had reduced blood glycemia during the glucose challenge, and the overall increase in blood glucose was significantly lower compared to the control, as indicated by the AUC (Fig. 6C). The limitation of this study is that no treatment using the combination of anakinra and tocilizumab was given.
In a recent publication (28), it was described that anakinra had no effect on glucose tolerance and graft survival in mice transplanted with human islets. In contrast to our own study, the recipients were treated posttransplantation with no exposure of the islets to anakinra in culture. Claims of freshly isolated islets being superior to cultured islets (31) in the context of transplantation supports the notion of short-term pretreatment culture with anakinra as an effective strategy to improve islet survival.
The glucose-lowering effect of anakinra is thought to be a result of enhanced β-cell secretory function. In clinical studies, improved glycemia in patients treated with anakinra correlated with improved β-cell secretory capacity (20,21). This could be the reason why recent clinical findings describe better outcome of islet transplantation when the patients had received an anti-inflammatory drug regimen that also included anakinra (27). Taken together with the positive effects on human islets, this could indicate a role for anakinra, not only as an efficient anti-inflammatory treatment after transplantation but also in pretreatment culture.
In conclusion, the current study provides data that demonstrate the efficacy of anakinra and tocilizumab in pretreatment culture of human islets. Anakinra has been well studied in the clinical setting of diabetes, and the positive data are supportive of our own findings. Conversely, we question the use of tocilizumab in spite of our findings because knowledge is sparse about the complex nature of IL-6, both on β-cells and other types of tissue. Therefore, further studies are needed to determine whether IL-6 receptor blockade is justifiable in islet transplantation.
Footnotes
Acknowledgment
Human islets were provided through the JDRF award 31-2008-413 (ECIT Islet for Basic Research program). The authors are grateful to all members at the human islet facility at Uppsala University and the Nordic Network for Islet Transplantation. This work was supported by the Department of Transplantation Medicine at Oslo University Hospital Rikshospitalet and grants from HelseSoer-Oest, The Norwegian Diabetes Association, UNIFOR, the Swedish Medical Research Council (VR K2011-65X-12219-15-6), and the Juvenile Diabetes Foundation International. The positions of Aksel Foss and Olle Korsgren are in part supported by NIH 2U01AI065192-06. Aside from this, the authors declare no conflicts of interest.
