Abstract
We recently found that rat hepatocyte transplantation was efficient (liver repopulation: 2.4%) in a sublethal nude mouse model (less than 33% mortality) of repeated liver injury generated using Jo2, a mouse-specific anti-Fas antibody, at sublethal dose of 250 μg/kg for 3 weeks. Genomic analysis of the livers revealed cell cycle blockade and an antiproliferative status of circadian genes, suggesting a selective advantage. By contrast, in the present study, freshly isolated human hepatocyte transplantation performed in the same mouse model resulted in implantation of less than 6,000 cells per liver (about 0.006% repopulation) in all animals. Genomic analysis of nude mouse livers revealed a lack of P21 upregulation, while a signature of stimulation of liver regeneration was observed, including upregulation of early response genes and upregulation of circadian genes. When we translated this sublethal model to a lethal model (65% mortality) by increasing the Jo2 repeated doses to 375 μg/kg, human hepatocyte engraftment was still very low; however, animal mortality was corrected by transplantation (only 20% mortality). Genomic findings in livers from the mice of the lethal Jo2 transplanted group were similar to those of the sublethal Jo2 transplanted group, that is, no selective advantage genomic signature and signature of mouse liver regeneration. In conclusion, transplanted human hepatocytes acted as if they modified nude mouse liver responses to Jo2 by stimulating liver regeneration, leading to an increased survival rate.
Introduction
In the current context of lack of organ donors, hepatocyte transplantation could be an alternative to liver transplantation (9,11,36,37). Efficient hepatocyte transplantation could allow a transient mainstay of the diseased or partial resected liver to detoxify the organism during the time course of liver regeneration or, in case no liver regeneration occurs, to wait for liver donation for transplantation (18,41).
Animal models of liver deficiency represent an important stage before performing hepatocyte transplantation in humans with the aim of treatment of clinical pathologies. Difficulties reside in developing animal models that not only present liver injury mimicking clinical pathologies, but also predict the response of human liver to human allogeneic hepatocyte transplantation.
Recently, we developed a nude mouse model with repeated acute liver deficiency, by weekly intraperitoneal injection of Jo2, an anti-Fas antibody agonist of Fas ligand (22). This antibody was found to be specific to mouse hepatocytes, since rat hepatocytes were shown to be naturally resistant to the Jo2 effect (14,34). Jo2 has been reported to induce transitory fulminant hepatic failure in various mouse models after a single injection (17,22,26), characterized by an elevation of serological alanine transaminase (ALT) for the first 3 days. The survival rates correlated with histological observations of liver necrosis. We and others have found that liver deficiency injury was a dose-dependent effect and was reversible within 10 days (14). The regenerative capacity of the liver was thus retained after Jo2 injection, making its use attractive for studying the efficacy of hepatocyte transplantation. We also found that when Jo2 was administrated on repeated challenges, each Jo2 injection induced an episode of fulminant hepatic failure.
In our recent study, we found an increased liver engraftment of rat hepatocytes into nude mouse livers when transplantation was followed by a weekly Jo2 treatment with a sublethal (mortality inferior to 33%) dose of 250 μg/kg weight, compared to that obtained when transplanted into control mice (14). Genomic analysis of livers of these sublethal Jo2 weekly treated mice revealed a transcriptome profile with upregulation of cellular cycle blocking transcripts (especially an elevated level of gene p21, cyclindependent kinase inhibitor 1, controlling cell cycle G1 phase arrest), activation of liver injury-inducing interferon (IFN)-γ /signal transducer and activator of transcription 1 (STAT1) pathway, circadian transcript signature of antiproliferative cell status, and minor inflammatory responses. All of these modifications indicated an important cell cycle blockade, following the last Jo2 injection. The overall transcriptome profile suggested that Jo2-treated nude mice might correspond to a model of selective advantage where cell cycle blockade occurred in mouse hepatocytes submitted to weekly Jo2 treatment, while natural resistance to Jo2 of rat hepatocytes allowed them to proliferate.
The first aim of the present study was to assess human hepatocyte engraftment into the Jo2-treated nude mouse model of acute liver failure. For this study, freshly isolated human hepatocytes (FHCs) were transplanted into nude mice subjected to repeated Jo2 injections of 250 μg /kg weight, and genomic analysis of nude mouse hepatic parenchyma was performed.
The second aim of the present study was to evaluate the therapeutic efficacy of hepatocyte transplantation in fulminant hepatitis by using lethal Jo2 doses of 350 μg/kg weight and assessing survival of transplanted and non-transplanted mice.
Materials and Methods
Chemicals and Reagents
All chemical compounds were obtained from Sigma-Aldrich (St. Quentin-Fallavier, France). Culture media, fetal calf serum, and antibiotics were purchased from Invitrogen (Cergy Pontoise, France). Cell culture plates were obtained from Dutscher (Brumath, France).
Animals
Male nude mice 8 to 10 weeks old used for cell transplantation were housed individually, placed in a ventilated cabinet under controlled air pressure and temperature conditions, and under daily cycles of alternating 12-h light/darkness. They had sterile water and sterile standard pelleted rodent diet, ad libidum. Male Sprague–Dawley rats (180–200 g) and mice (CD1) were used for hepatocyte isolation. All animals were purchased from Charles River Laboratories (L'Arbresle, France). All experiments were conducted according to local institution guidelines for the care and the use of laboratory animals.
All operations were performed between 10 am and 12 pm, under general anesthesia, using a sterile surgical technique.
Hepatocyte Isolation
All experimental procedures were performed in compliance with respective French laws and regulations after approval by the National Ethics Committee. Informed consent was obtained from the patients for the use of liver tissue for research purposes. Hepatocytes were isolated by a two-step collagenase method according to Alexandre et al. (1) and Richert et al. (38). Donor characteristics are summarized in Table 1. Briefly, on arrival at the laboratory, the liver was removed from the transportation bag, placed on ice-cold washing buffer [0.8 mM Na2HPO4, 0.5 mM ethylene glycol tetraacetic acid (EGTA), 2.7 mM KCl, and 137 mM NaCl, pH 7.4], two to four cannulae (Vasculon 14–20 gauze, vein size dependent, Becton Dickinson, Le Pont de Claix, France) were inserted into the major vessels, and the cut surface of the liver was coated with a collar glue (Super Glue 3, Loctite, Henckel Loctite France, Marne-la-Vallée, France) to avoid leakage of the perfusate. A washing step was initiated by using the prewarmed (37°C) washing buffer without recirculation [20–40 ml/min/cannula (depending on the size of the specimen)] for 10 min at 37°C. This perfusion medium was discarded, and the digestion step was started by recirculation of the digestion buffer at 37°C [0.8 mM Na2HPO4, 2.7 mM KCl, and 137 mM NaCl, 0.05% w/v collagenase (Gibco powdered collagenase for hepatocyte isolation; Invitrogen, Cergy Pontoise, France), pH 7.4] for 20–30 min at a flow rate of 20–40 ml/min/cannula (depending on the size of the specimen). After the digestion step, the cell suspension, obtained by smooth mechanic dissociation and filtered through three stainless steel strainers (850, 425, and 106 mm diameter, respectively, Dominique Dutscher, Brumath, France), was centrifuged at 75 × g for 5 min at 37°C. The cell pellet was washed three times with Hank's balanced salt solution (HBSS). Cell yield and viability were estimated by trypan blue dye exclusion and counting the cells using a Malassez hemocytometer (WWR, Strasbourg, France).
Donor Information for Human Hepatocyte Preparations

M, male; F, female; C, caucasian.
MTT Cytotoxicity Test
Hepatocytes from different species (human, mouse, and rat) were seeded in Biocoat 96-well plates at a density of 50,000 cells/well in Dulbecco's modified Eagle's medium (DMEM) supplemented with 5% fetal calf serum, containing insulin (4 mg/ml), dexamethasone (1 μM), and antibiotics (gentamicine). After overnight plating, cells were treated for 24 h with increasing concentrations of Jo2 (0, 0.1, 1, 10, 100, 500, 1,000 ng/ml). After 24 h of treatment, a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) test was performed, according to literature data (5), to assess cell viability.
Liver Injury Induction
The purified hamster anti-mouse Fas monoclonal antibody, Jo2 (PharMingen, Becton Dickinson, France), was used to induce acute liver failure in nude mice, by intraperitoneal injections. Animals received either a repeated 250 μg/kg body weight dose of Jo2 (n = 21) weekly during 21 days (Group 250), or at day 0, a single 250 μg/kg body weight followed at day 7 by a 375 μg/kg body weight dose of Jo2 (n = 21) repeated weekly until day 21 (Group 375). Three animals in each group were kept alive until day 57 (for 8 weeks). A control group of animals was not submitted to Jo2 treatment.
Hepatocyte Transplantation
Intrasplenic hepatocyte transplantation was performed on 8- to 10-week-old male nude mice under anesthesia [by breathing 2 L/min O2 containing 3% enflurane (Forene®, Abbott, Rungis, France)] 24 h after the first liver injury induction. After an abdominal midline incision, the spleen was mobilized, and 1 × 106 human hepatocytes suspended in 100 μl HBSS were slowly injected into the spleen parenchyma using a 30-gauge needle. Hemostasis was realized at the point of injection by dabbing with Surgicell® gauze (Johnson & Johnson, New Brunswick, NJ, USA). Hepatocytes migrate to the liver, and successful injection was approved by a short-term paleness of the liver lobes. The animals were sacrificed 21 days after hepatocyte transplantation, except for three animals per group, for which the experiment kept running for 8 weeks. These three animals were subjected to weekly Jo2 injections and sacrificed on day 57 (Table 2). One part of the liver was snap frozen in liquid nitrogen and stored at −80°C until genomic analysis could be performed, and one part was embedded in optimal cutting temperature (OCT) compound (Tissue-Tek, Elkhart, IN, USA) and snap frozen in isopentane, for cryosection and immunostaining with an anti-human cytokeratin 18 (CK18) antibody (M 7010, DAKO, Les Ulis, France) (see below).
Different Experimental Groups of Nude Mice, Treated or Not by Jo2 (Anti-Fas Antibody) Weekly Injection and Transplanted or Not With Freshly Isolated Human Hepatocytes (FHCs)

Experimental Groups for Hepatocyte Transplantation (Table 2)
The control group of animals did not undergo liver injury and were transplanted with freshly isolated human hepatocytes (Group Transpl, n = 5).
Two groups underwent liver injury either with weekly 250 μg/kg of Jo2 (Group 250) or with weekly 375 μg/kg of Jo2 (Group 375). They were either submitted or not to transplantation:
Without hepatocyte transplantation (Group 250Jo2, n = 6 and Group 375Jo2, n = 11)
Transplanted with freshly isolated human hepatocytes in nude mouse treated by repeated body weight dose of Jo2 (Group Transpl_250, n = 15)
Transplanted with freshly isolated human hepatocytes in nude mouse treated by repeated 375 μg/kg body weight dose of Jo2 (Group Transpl_375, n = 10)
Seric Parameters
Blood samples were taken each day following Jo2 injection from day 7 to day 22, on anesthetized animals (as above) via the caudal vein. Serum samples were obtained by centrifugation of collected blood 10 min at 3,000 × g. Catalytic activity of ALT (U/L) was determined by means of reflectometry using an automatic Kodak Ektachem 500 analyzer (Vincennes, France).
Gel Electrophoresis and Western Blotting Analysis
Human albumin levels in nude mice serum, in suspension of 106 hepatocytes and in liver samples 22 days after the beginning of the experiment, were estimated qualitatively and semiquantitatively. Proteins (10–50 μg) were separated by 7% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) according to Laemmli (27) and then electrophoretically transferred onto nitrocellulose membrane of 0.22 μm (Schleicher and Schuell, Dassel, Germany) for Western blot analysis (4). The blots were incubated overnight at 4°C with the primary antibody: an affinity-purified human albumin antibody produced in goat (1:8,000, Bethyl Laboratories, Inc., Interchim, France). Detection of the primary antibody was performed using a commercial Vectastain Elite ABC Kit (Vector Laboratories, Biovalley, France) as described by the manufacturer. Positive signals were detected by the enhanced chemiluminescence method (Amersham Life Science, Inc., Velizy-Villacoublay, France). Relative amounts of albumin were determined by densitometry using a scanner and quantified using a computer program (Quantity One Basic 1-D Analysis Software, Biorad Laboratories, Marnes-la-Coquette, France).
Liver Ki-67 Immunostaining
Cryofrozen liver sections (4 μm) were fixed in cold methanol for 10 min and rinsed in phosphate-buffered saline (PBS) for 3 min before being immersed in a 10-mM citrate sodium bath (pH 6) exposed to microwaving during 10 min. After 1 h at room temperature, slides were rinsed in PBS (5 min, three times), specific endogenous peroxidase activity was inhibited by H2O2 (0.5% in methanol) for 30 min, followed by another rinsing step in PBS (5 min, three times) and then incubated overnight at 4°C with Ab833 rabbit polyclonal antibody to human Ki-67 antigen (Abcam, Interchim, France) diluted (1:50) in PBS. After rinsing in PBS (5 min, three times), a 1:200 dilution in PBS of biotinylated goat anti-rabbit IgG (H+L) antibody (Jackson ImmunoResearch Laboratories, Interchim, France) was added for 1 h. A commercial kit (Vectastain® Elite® ABC kit; Vector Laboratories) was used for amplification of the signal before 0.3 mg/ml diaminobenzidine (supplemented with 0.03% H2O2) was added to each slide (10 min, in the dark). After two successive sterile water washes (3 min each), sections were dried for 1 h before being mounted in Eukitt® medium (CML, Nemours, France). Percentage of Ki-67-positive cell staining was determined between the different samples. Negative control was mouse IgG (X 0931; Dako; 1:50).
Ki-67 immunostaining was performed in all animals of Group Transpl_250 and Group Transpl_375, on the liver removed at the time of autopsy, and on animals that died during the experiment, if the death was noticed rapidly after its occurrence.
Liver CK18 Immunostaining
This was performed the same as for Ki-67 except as outlined below.
Antigen retrieval was by microwaving for 20 min in 10 mmol/L Tris buffer, 1 mmol/L EDTA, pH 9. The slides were incubated 30 min at room temperature with monoclonal mouse anti-human cytokeratin 18 antibody (Dako) diluted (1:50) in PBS. After rinsing in PBS (5 min, three times), a 1:200 dilution in PBS of biotinylated goat antimouse IgG (H+L) antibody (Jackson ImmunoResearch Laboratories, Interchim, France) was added for 30 min at room temperature. Visualization was performed using the Vectastain® Elite® ABC kit and diaminobenzidine as described above before the sections were dried for 1 h before being mounted in Eukitt® medium.
Hepatocyte Engraftment
In order to evaluate the number of engrafted cells, albumin content of transplanted livers, as determined by Western blotting, was divided by the albumin content of 106 hepatocytes of the given batch of transplanted hepatocytes.
Statistics
Results were expressed as mean ± SEM. Differences among means were assessed using computer-aided unpaired t test (Statview® 4.5 software, Abacus Concept, Berkeley, CA, USA). When multiple comparisons were done (comparing different groups to a control group), Bonferroni correction was used. Survival curves were analyzed using log rank test. Values of p < 0.05 were considered significant.
Genomic Analysis
RNA Preparation
One part (left lobe) of mouse liver samples was frozen in liquid nitrogen immediately after removal and was used for RNA preparation and DNA microarray experiments. Total RNA from liver tissue samples was extracted after homogenization with a rotor stator homogenizer (Ultra Turrax; IKA, Staufen, Germany) with TRIzol Reagent (Invitrogen) and purified using RNeasy Mini Kit (Qiagen, Courtaboeuf, France) according to the manufacturers' instructions. Total RNA was quantified by absorbance at 260 nm (A260 nm), and purity was checked by the A260 nm/A280 nm ratio (Gene Quant pro photometer, Amersham). The RNA samples were stored at −80°C before analysis.
DNA Microarray Experiments
DNA microarray experiments were conducted as recommended by the manufacturer of the GeneChip system (Affymetrix, Santa Clara, CA, http://www.affymetrix.com). Samples were hybridized to MOE430A GeneChip arrays. The resultant image files (.dat files) were processed using the Microarray Analysis Suite 5 (MAS5; Affymetrix) software package from which numerical signal intensity (Signal) data and categorical expression level measurements (Absolute Call) were obtained. All data were quality checked and were transferred to other software packages for analysis. The mouse genome-specific MOE430A GeneChip expression probe arrays containing more than 45,000 probe sets interrogating more than 39,000 transcripts included wellsubstantiated genes and also expressed sequence tag (EST) clusters. One GeneChip was used per animal.
Statistical Data Analysis.
Data Pretreatment
The MAS 5 processed raw expression intensities were subjected to a filtering. Only probe sets with a median intensity of larger than 50 and 3 (or more) “present” or “marginal” calls in at least one of the treatment groups were used in the subsequent statistical analyses. A total of 18,156 probe sets satisfied the two filter criteria. The intensity data were then log2-transformed with an offset of 1.
Statistical Analysis
A one-way analysis of variance (ANOVA) using group as the independent variable was fit to the filtered and transformed data of all samples. The transplanted Transpl_250 and Transpl_375 groups were compared to the transplanted control group. These comparisons were made using linear contrasts within the framework of the ANOVA models (2). The calculated fold changes are based upon geometric means. The principal component analysis (PCA) and ANOVA calculations were performed with Partek Pro 6.2 (Partek, Inc., St. Louis, MO, USA). The Affymetrix probe sets were annotated using the Database for Annotation, Visualization and Integrated Discovery (DAVID) 2.0 (8). Expressed sequence tags (ESTs) were not considered for biological interpretation.
Results
Mouse Selectivity of the Cytotoxicity of Anti-Fas Jo2 Antibody
Human hepatocytes were cultured overnight and treated with increasing concentrations of Jo2 (0, 0.1, 1, 10, 100, 500, 1,000 ng/ml). Assays with rat and mouse hepatocytes were performed in parallel. After 24 h in culture, a MTT test was performed, and viability of cells treated with increasing concentrations of Jo2 was expressed as percentage of the control value (0 ng/ml).
As shown in Figure 1, no cytotoxic activity of Jo2 against human or rat hepatocytes was found up to 1,000 ng/ml, while the sensitivity of mouse hepatocytes to more than 1 ng/ml was confirmed.

Cytotoxicity of Jo2 at increasing concentrations, on hepatocyte cultures from different species. Viability was assessed by MTT assay; 24-h incubation with Jo2.
Induction of Liver Injury Following Jo2 Administration to Nude Mice
For all groups of nude mice, at each time point during the experiment, ALT levels increased significantly (p < 0.05, compared to nontreated control group) 24 h after 250 μg/kg or 375 μg/kg Jo2 treatment (Fig. 2), confirming that liver injury was induced.
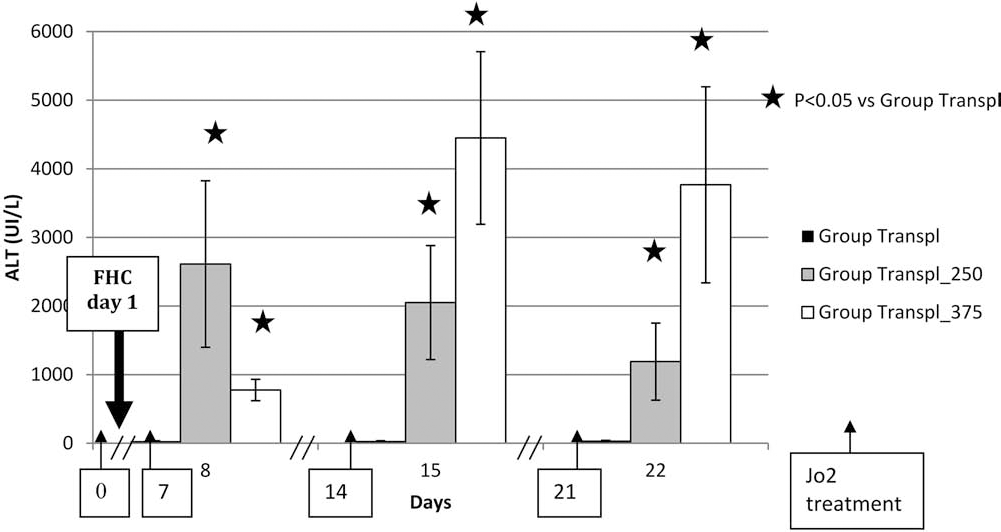
Alanine transaminase (ALT) content in the serum of nude mice. ALT content in the serum of nude mice after transplantation with human freshly isolated hepatocytes (FHCs), and treated without (Group Transpl) or with antibody anti-Fas (Jo2) at a weekly dose of 250 μg/kg (Group Transpl_250) or at a weekly dose of 375 μg/kg (Group Transpl_375). Results are expressed in UI/L as the mean ± SEM in mice serum. Stars indicate a significant increase compared to the control Group Transpl. Arrows indicate each Jo2 injection.
Engraftment Rate of Human Hepatocytes and Levels of Human Albumin in the Serum of Transplanted Nude Mice
Due to the important interindividual fluctuation within each experimental group, all individual results of hepatocyte engraftment calculations and serum human albumin are shown and summarized in Table 3.
Individual Characteristic of Human Hepatocyte Transplantation Results: Hepatocyte Viability Before Transplantation (%), Number of Engrafted Hepatocytes at Autopsy, Human Albumin in Nude Mouse Serum (ng/ml) at Autopsy

FHCs, fresh human hepatocytes; Nd, not determined.
Human albumin was undetectable in sera from nontransplanted nude mice (receiving Jo2 repeat treatment). In mice transplanted with human hepatocytes, human serum albumin could be measured at low levels at day 22, in all groups: group not treated with Jo2 (1.004 ± 0.293 ng/ml serum), group receiving 250 μg/kg of Jo2 (1.719 ± 0.683 ng/ml serum), and group receiving 375 μg/kg of Jo2 (3.569 ± 1.302 ng/ml serum).
Results of liver repopulation are listed in Table 3. The human hepatocyte engraftment rate was calculated by measuring the percentage of human albumin in transplanted mouse livers. In the three groups, half of the animals showed strictly no liver engraftment of human hepatocytes. In the remaining animals, the human hepatocyte engraftment remained very low, whatever the dose of Jo2. Even if taking the best engraftment rate of human hepatocytes (6,000 cells by liver), and hypothesizing that a nude mouse liver contains approximately 108 hepatocytes (19), it would correspond to 0.006% of repopulation, which can be considered as a failure of human hepatocyte engraftment into the recipient liver. These results were confirmed by immunostaining with an anti-human CK18 antibody and evaluation of engraftment by counting the number of positive cells within randomly chosen microscope fields (data not shown).
There was no evidence for ectopic engraftment since no human hepatocyte presence could be found in the spleen, heart, bowel, or lung tissue (data not shown).
Taken together, the results observed with human hepatocytes were opposite to those previously reported with rat hepatocytes (42). Indeed, engraftment of transplanted rat hepatocytes was observed in all animals, and the repopulation rate was estimated at 2.4%, which was sevenfold higher than that of rat hepatocyte repopulation in the nude mouse not treated with Jo2 (42).
Effect of Transplantation on the Survival Rate of Jo2-Treated Nude Mice
We first confirmed that repeated 250 μg/kg Jo2 injections was a sublethal model, with a survival rate of 67% at 3 weeks, mortality occurring during the 24 h following each Jo2 injection, as found in our previous experiments (42). When nude mice were challenged with higher weekly doses of 375 μg/kg Jo2 from day 7 until day 21, survival rate decreased to 33% at 3 weeks. As we aimed at comparing the benefits of hepatocyte transplantation on the survival rate of Jo2-treated nude mice, the survival rate at day 1 (i.e., the day of hepatocyte transplantation) was considered 100% (Fig. 3).

Nude mouse survival after treatments. Survival (% of day 1) of nude mice treated with antibody anti-Fas (Jo2) at a weekly dose of 250 μg/kg (Group 250Jo2 and Group Transpl_250) or at a weekly dose of 375 μg/kg (Group 375Jo2 and Group Transpl_375). Arrows indicate each Jo2 injection.
In the group submitted to 250 μg/kg Jo2 doses, the survival rate after transplantation of human hepatocytes was 64.3%, equivalent to the nontransplanted corresponding controls (Fig. 3), also confirming our previous data (42).
In the lethal model, the survival rate increased to 80% when animals were transplanted with human hepatocytes (Fig. 3), and this gain of survival was found to be significant (p = 0.046).
Genomic Analysis of Transplanted Mice
Genomic analysis was performed in mice submitted to transplantation only (Transpl_group) and in mice submitted to subsequent repeated 250 μg/kg Jo2 treatments (Transpl_250 group) or to repeated 375 μg/kg Jo2 treatments (Transpl_375 group).
When comparing genomic analysis results from Transpl_250 group and Transpl_group, 41 different genes were upregulated, and 16 genes were downregulated (Table 4).
Genes Up- and Downregulated in Group of Nude Mouse Treated by Weekly 250 μg/kg Jo2 Dose and Transplanted With Freshly Isolated Human Hepatocytes (Transpl_250 group) Versus Control Transplanted Group of Nude Mice (Transpl_group)
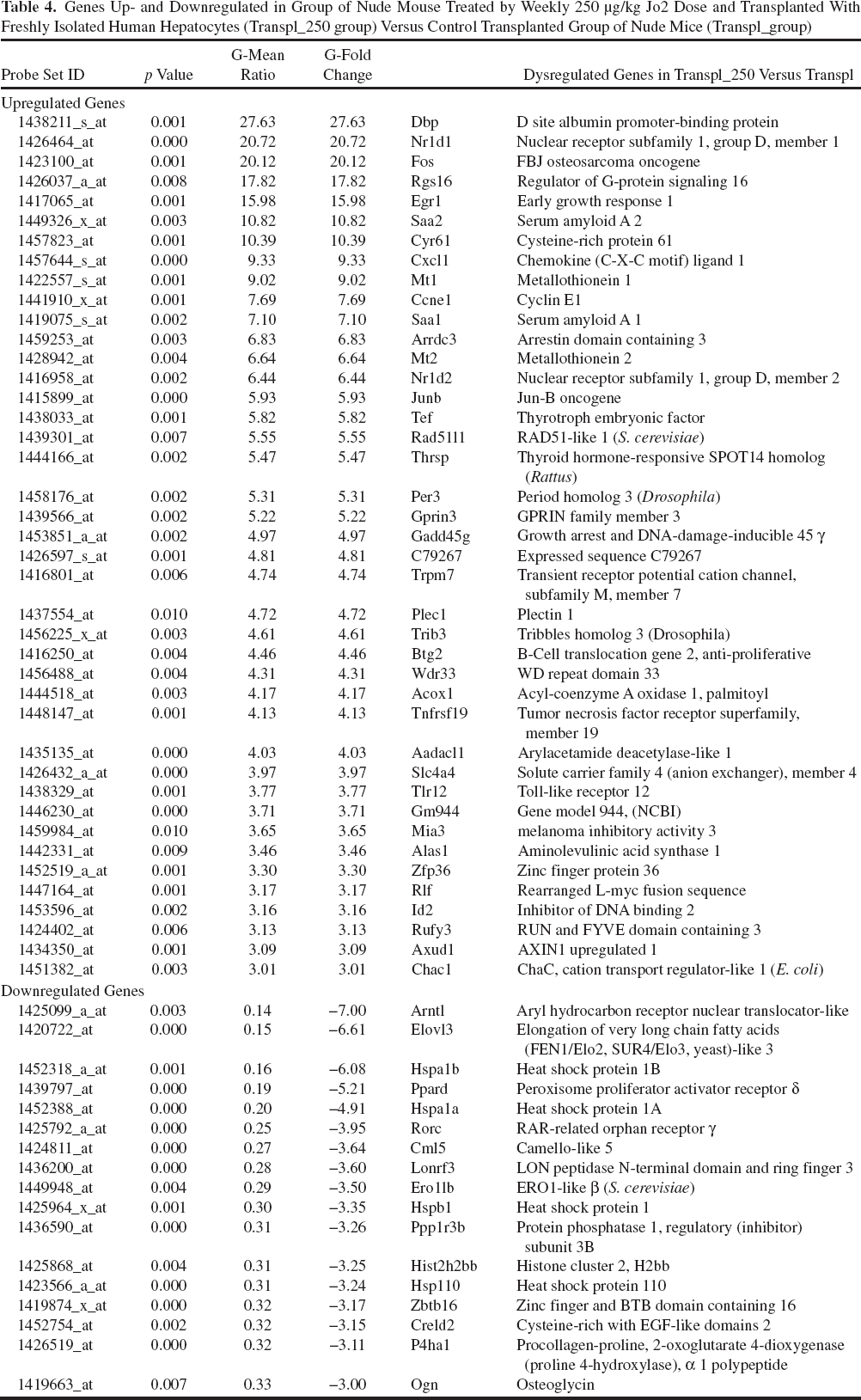
Among the upregulated genes, we could isolate genes involved in cell proliferation such as Fos, a proto-oncogene, Egr1 (early growth response 1), cyclin 1, and methallothioneins. These genes are known to appear early after liver injury, and they signal a liver regeneration response. Some inflammation marker genes were upregulated such as serum amyloid (Saa) 1 and 2. A few genes that can be seen with macrophage activation were also found such as chemokines or Toll-like receptor (Tlr).
Interestingly, the most upregulated genes were clock genes: Dbp (D-site of albumin promoter) (27-fold) and Nr1D1 (nuclear receptor subfamily 1, group D, number 1). Other circadian cycle regulator genes were also upregulated like Nr1D2 (nuclear receptor subfamily 1, group D, number 2) also called Rev-Erb β, Per3 (Period 3), and Tef (thyrotrophic embryonic factor). This upregulation of circadian genes was interpreted as linked with increased cellular proliferative status.
Among the downregulated genes were some heat shock proteins and Arntl (aryl hydrocarbon receptor nuclear translocator-like), also called Bmal1 (brain and muscle aryl hydrocarbon receptor nuclear translocator-like 1), another circadian gene.
Taken altogether, the results were different from those obtained when comparing rat hepatocyte transplantation into the repeated 250 μg/kg Jo2 injections nude mouse model versus rat hepatocyte transplantation alone (42). While all the circadian genes were downregulated in the livers of nude mice transplanted with rat hepatocytes, they were strongly upregulated in livers of nude mice transplanted with human hepatocytes (Fig. 4).

Comparative mRNA expression of circadian-related transcripts in nude mice transplanted in rat hepatocytes (white) or fresh human hepatocytes (black) after weekly injection of 250 μg/kg Jo2. mRNA measured by Affymetrix mouse genome-specific MOE430A GeneChip array.
In our previous rat hepatocyte transplantation study, we found a “signature” for blockade of cell proliferation (with P21, a cell cycle arrest gene, as the most upregulated gene associated with a downregulation of circadian genes), of interferon γ pathway activation, and of moderate inflammation, and we could not isolate early genes. In the present study, where we compared nude mice receiving a weekly 250 μg/kg Jo2 dose after transplantation with human hepatocytes (Transpl_250 group) with human hepatocyte transplantation alone (Transpl_group), we found an inverted “signature” of stimulation of liver regeneration: lack of P21 upregulation, upregulation of early response genes, and upregulation of circadian genes.
Genomic analysis comparing human hepatocyte transplantation in the 375 μg/kg Jo2 model (Transpl_375 group) to human hepatocyte transplantation alone (Transpl_group) revealed 26 upregulated genes and six downregulated genes (Table 5).
Genes Up- and Downregulated in Group of Nude Mouse Treated by Weekly 375 μg/kg Jo2 Dose and Transplanted With FHCs (Transpl_375 group) Versus Control Transplanted Group of Nude Mice (Transpl group)

The upregulated genes were similar to those found when comparing Transpl_250 group to Transpl_group: they belong to the family of regulators of cell proliferation, such as metallothionein, Egr, cyclin E1, and to inflammation genes such as Saa1 and Saa2, or matrix metalloprotease (Mmp). Thus, the signature of Transpl_375 group very closely resembled that of Transpl_250 group. The major difference was the absence of neither up- nor downregulation of circadian genes. The PCA of the three groups showed two distinct, clearly separated groups: Transpl_375 and Transpl, while Transpl_250 and Transpl_375 were close (Fig. 5).

Principal component analysis (PCA) of genomic analysis of nude mice after treatment. PCA of genomic analysis of the three groups of nude mice after transplantation with human freshly isolated hepatocytes treated without (“A” Group Transpl) or with antibody anti-Fas (Jo2) at a weekly dose of 250 μg/kg (“B” Group Transpl_250) or at a weekly dose of 375 μg/kg (“C” Group Transpl_375).
Discussion
Nude Mouse Model of Repeated Lethal Liver Deficiency
In the present experiment, repeated liver injury was induced in nude mice by a weekly challenge of Jo2 at 250 or 375 μg/kg for 3 to 8 weeks. The 67% survival observed at 250 μg/kg confirmed our previous report (42) and was therefore considered as a sublethal dose. Survival rate decreased to 36%, when repeated liver injury was induced by 375 μg/kg Jo2, confirming lethality. In both groups, mortality was observed each 24 h following Jo2 injection.
Failure of Human Hepatocyte Liver Engraftment in Repeatedly Jo2-Treated Nude Mice
To assess human hepatocyte engraftment, we used two criteria: serum human albumin rate and human albumin in nude mice liver. In both groups, serum human albumin concentrations were variable and did not correlate with liver repopulation. This was previously reported for the uPA (urokinase plasminogen activator) transgenic mouse model transplanted with human hepatocytes, in which human hepatocyte engraftment raised a rate up to 90%, while the proportion of murine/human albumin in the blood sample did not exceed 10% (29), suggesting that functional human hepatocyte engraftment can occur without improvement of serum albumin. Moreover, the smaller the engraftment, the more difficult it is to detect the human serum albumin (6,31). Thus, human serum albumin might not be the good criteria for evaluation of cell engraftment.
Regarding liver repopulation by human hepatocytes, we obtained a rate of human hepatocyte engraftment that did not exceed 6,000 cells per liver, a much lower rate than what could be expected from our rat hepatocyte engraftment results of 2.4% (42). This corresponds to a repopulation rate evaluated at 0.006% and could be interpreted as a failure of engraftment of human hepatocytes in Jo2 untreated, 250Jo2, and 375Jo2 groups. Such a failure in liver repopulation could be expected in animals not receiving Jo2 since no selective advantage occurred. For groups submitted to Jo2 treatment, the lack of liver repopulation contrasts with the results described when either murine syngenic or human immortalized hepatocytes were transplanted.
In the literature, the engraftment rate of hepatocytes into recipient livers of animals submitted to repeated Jo2 administration in a murine syngenic model is very variable: Fujino et al. (12) obtained a mean repopulation rate of 7%, whereas Gilgenkrantz's group obtained between 4% and 30% of repopulation, depending on the model used (32,33). A 4% mean implantation rate (range from 2% to 6%) was reported in a model of CBA mouse submitted to repeated Jo2 challenges, after transplantation of transduced B-cell CLL/lymphoma 2 (BCl2) overexpressing rat hepatocytes (32), while a mean 30% of repopulation (ranging from 1% to 30%) was obtained in an apoliprotein E knockout (ApoE-/-) mouse submitted to repeated Jo2 challenges after transplantation of transduced BCl2 overexpressing hepatocytes that expressed constitutively ApoE (33). These engraftment rates were, however, low compared to those obtained in urinary protein-urokinasetype plasminogen activator (uPA) transgenic mice, retrorsine models, or fumaryl-acetoacetate hydrolase (FAH) null mouse, where repopulation by mouse hepatocytes raised up to 80–85% (3,16,25,43).
By using the xenogenic human–rodent transplantation model severe combined immunodeficient (SCID) beige mouse with liver injury caused by repeated Jo2 challenge of 0.2 mg/kg in which HepaRG, an immortalized female human hepatocyte cell line, was transplanted, there was a lack of engraftment in 18 over 29 mice despite the multiplication potential of immortalized cells. Nevertheless, engrafted mice survived, whereas the others died within a few days (21).
Our model of rodent xenogenic transplantation into livers submitted to repeated injury by anti-Fas showed a repopulation of nude mouse liver by rat hepatocytes of 2.4%. Thus, our model was less efficient in developing the appropriate stimuli for rat hepatocyte implantation than for syngenic mouse hepatocyte implantation (32,33), even if genomic analysis showed that it corresponded to a model of selective advantage when transplanting rodent (rat) cells into nude mouse hosts (42). The partially efficient model for rat hepatocyte implantation was poorly supportive for human hepatocyte implantation.
Literature reported difficulties to get implantation of human hepatocyte into mouse models, even in transgenic model uPA mouse or FAH null mouse, where selective advantage climate was the strongest. Dandri et al. described a 70% success rate of the transplanted mice in a model of transplantation of human hepatocytes into uPA/RAG2 mouse liver, with an all-or-none response for human hepatocyte engraftment (7). It is noteworthy that in one third of their animals, however, human hepatocytes did not engraft at all. They correlated this 30% failure result with the time of warm ischemia (more than 90 min) and with the viability of the transplanted cells (less than 80%). These results were confirmed in a model of the uPA/SCID mouse where engraftment was successful in a median of 22% (0–87%) of animals, linked with donor age and warm ischemia (24). In our study, viability of suspended hepatocytes was not a criterion for better results in liver repopulation.
To our knowledge, only a few literature reports have described human hepatocyte transplantation into a nude mouse model. Results of human hepatoblasts (and not mature hepatocytes) transplanted into nude mice after 50% partial hepatectomy gave a liver repopulation from 0.05% to 1% after 6 weeks. However, older hepatoblasts, obtained in the last part of pregnancy (later than 20–24 weeks of gestation), that is, mature cells close to hepatocytes, were not able to repopulate this nude mouse liver (29). When human hepatocytes were transplanted into SCID mice, affected by uPA-/- knockout, a well-known model of selective advantage, repopulation of human hepatocytes varied from 10% to 90% (7), altogether less than those obtained in syngenic models or murine–murine transplantation models (20). On the other hand, immune mechanisms, even in immunosuppressed mice could act as a brake for xenotransplantation (23).
Meuleman et al. (31) underlined the ability of transplanted human hepatocytes to develop gap junctions with murine host cells, leading to new biliary canalicules. However, these engrafted human hepatocytes presented abnormal glycogenic accumulation that could be due to inappropriate recognition of murine signals.
Thus, one could assume that transplanted human hepatocytes did not receive adapted signals from the mouse liver to engraft and survive as well as in human environment.
Increased Survival Rate of Nude Mice Transplanted with Human Hepatocytes and Subsequently Challenged with Repeated Lethal Doses of Jo2
The 36% mortality rate induced by repeated 375 μg/kg Jo2 treatment could be partially corrected by human hepatocyte transplantation in the lethal model, with a survival rate of 80% in transplanted mice. Thus, human hepatocytes seemed to be able to protect nude mice from liver deficiency, not only for a few days, but even for long term (up to 57 days). This improvement of survival with human hepatocyte transplantation was observed despite a failure of human hepatocyte engraftment.
The increased survival of mice could be due either to hepatocyte action itself or to the transitory presence of fragments of hepatic cells or of growth factors delivered by hepatocytes. However, it has been noted that after liver injury, mortality cannot be corrected by dead hepatocyte transplantation, contrary to living cell transplantation (45). A recent study showed that human albumin released by nonviable human hepatocytes is not detectable in mouse serum 8 days after transplantation (39). In the present study, human albumin was detectable in the serum of transplanted mice at day 22 or day 57, suggesting that if survival improvement was somehow linked to the presence of hepatocytes, it was due to living cells and not dead hepatocytes.
Genomic expression of nude mouse livers strongly differed when rat or human hepatocytes were transplanted. When human cells were transplanted, cell cycle blockage was not seen, and P21 gene expression, which was the most upregulated gene after rat hepatocyte transplantation, was not modified. Many early genes of proliferation were upregulated in mice transplanted with human hepatocytes, whereas none of them was identified when rat hepatocytes were transplanted (42). Moreover, an inverted (up–down) regulation of circadian genes was observed. The circadian genes are known to be involved in the timing of the regenerating mouse livers (30,35,40). The expression of cell cycle-related genes being under control of a circadian clock induces the key regulators of mitosis directly and unidirectionally in proliferating cells. These clock genes can modulate carcinogenesis in liver (10,28). The strong upregulation of circadian rhythm key regulators Per3, their regulator Nr1d2 (alias Rev-erbβ), and their immediate downstream targets (Dbp and Tef) would indicate a high proliferative status of cells in mouse livers. This could be correlated with the absence of P21 (15). When rat hepatocytes were transplanted, we obtained a signature of mouse cell cycle blockage, whereas when human hepatocytes were transplanted, we obtained a signature of a stimulation of proliferation. Taken together, the genomic results could explain the absence of engraftment–-no more selective advantage—cumulated with this paradoxical increased survival rate—stimulation of host cell proliferation— observed in Jo2-treated nude mice transplanted with human hepatocytes. Results of Ki-67 immunostaining show in the two Jo2-transplanted groups a much higher percentage of Ki-67-positive cells in surviving animals compared to those that died during the time course of the experiment (Table 6). This finding correlates with the results of genomic analysis showing regeneration of recipient liver and also with the increased survival rate. Human hepatocytes, by their presence, act as if they could modify the nude mouse cellular environment and trigger cell proliferation.
Percentage of Ki-67-Positive Liver Cells in Group of Nude Mouse Transplanted With Freshly Isolated Human Hepatocytes and Treated by Weekly 250 μg/kg Jo2 Dose (Transpl_250 group) or 375 μg/kg Jo2 Dose (Transpl_375 group)

We isolated “survivor” animals, but killed for the necessity of the experiment (“sacrifice”), from those that died because of hepatotoxicity of Jo2, during the experiment (“death”).
Literature indicates that cell chronology is species dependent, autonomous, and is not modified by the host nor by the donor cells in a xenogeneic rodent model of transplantation (44).
It could be possible that the differences in chronology of human and murine hepatocyte (6,13) would require a permanent murine signal, as in transgenic uPA or FAH mouse models, to become in-phase for both species and trigger better liver repopulation. It is thus of most importance to assess the outcome of more frequent Jo2 administrations, that is, to shorten the time interval between two challenges where there may be a lack of signal for hepatocyte division and to sustain liver regeneration in recipient livers.
Conclusion
Human hepatocyte transplantation into a nude mouse model of repeated liver injury by Jo2 treatment was a failure in terms of engraftment into recipient liver parenchyma. Genomic analysis showed that the presence of human hepatocytes in nude mouse liver parenchyma modified the changes induced by Jo2 treatment with upregulation of cell proliferation, disappearance of cell cycle blockage, and reversion of circadian antiproliferative status genes. These modifications of the Jo2-induced changes following human hepatocyte transplantation could, at least partly, underline the improved survival rate of nude mice from the group treated with lethal Jo2 when submitted to human hepatocyte transplantation.
Footnotes
Acknowledgments
The authors declare no conflicts of interest.
