Abstract
Induced pluripotent stem (iPS) cells have potential for multilineage differentiation and provide a resource for stem cell-based treatment. However, the therapeutic effect of iPS cells on acute kidney injury (AKI) remains uncertain. Given that the oncogene c-Myc may contribute to tumorigenesis by causing genomic instability, herein we evaluated the therapeutic effect of iPS cells without exogenously introduced c-Myc on ischemia–reperfusion (I/R)-induced AKI. As compared with phosphate-buffered saline (PBS)-treated group, administration of iPS cells via intrarenal arterial route into kidneys improved the renal function and attenuated tubular injury score at 48 h after ischemia particularly at the dose of 5 × 105 iPS cells. However, a larger number of iPS cells (5 × 107 per rat) diminished the therapeutic effects for AKI and profoundly reduced renal perfusion detected by laser Doppler imaging in the reperfusion phase. In addition, the green fluorescence protein-positive iPS cells mobilized to the peritubular area at 48 h following ischemia, accompanied by a significant reduction in infiltration of macrophages and apoptosis of tubular cells, and a remarkable enhancement in endogenous tubular cell proliferation. Importantly, transplantation of iPS cells reduced the expression of oxidative substances, proinflammatory cytokines, and apoptotic factors in I/R kidney tissues and eventually improved survival in rats of ischemic AKI. Six months after transplantation in I/R rats, engrafted iPS cells did not result in tumor formation in kidney and other organs. In summary, considering the antioxidant, anti-inflammatory, and antiapoptotic properties of iPS cells without c-Myc, transplantation of such cells may be a treatment option for ischemic AKI.
Keywords
Introduction
Acute kidney injury (AKI) is characterized by a rapid decline of renal function due to kidney damage. Important causes of AKI comprise renal ischemia, sepsis, or exposure to nephrotoxins (2, 42). Renal ischemia is the most common cause of AKI, particularly in the perioperative phase of cardiopulmonary bypass surgery (27) and kidney transplantation (2). AKI associates with remarkable increases in morbidity and mortality (23). Despite the proven benefits of pharmacological therapy in a number of preclinical studies (3), the therapeutic approaches to prevent or treat AKI are very limited. Therefore, the search for novel therapeutic modalities for ischemic AKI is an area of intense investigation.
Stem cells have the potential of self-renewal and the ability to differentiate into many types of cells (6, 33, 46). Bone marrow-derived stem cells can mobilize into the ischemic injured kidneys and contribute to renal parenchymal turnover and regeneration (25, 28, 34). Nevertheless, these bone marrow-derived cells only account for a small percentage of cells of regenerative capacity in the injured kidney. Surviving tubular epithelial cells are the predominant cellular components involved in repopulation after ischemic injury (15). Administration of bone marrow-derived mesenchymal stem cells (MSCs) has been demonstrated to markedly accelerate tubular proliferation, repair the kidney, and improve renal function (13, 20, 29, 40, 44). Treatment with bone marrow-derived MSCs seems to favor functional and morphological recovery after AKI. However, the traditional source of MSCs, namely bone marrow, is relatively inaccessible and limited in the provision of a large volume of these cells. Therefore, the prospect of cell replacement therapy utilizing other stem cells for this disease has attracted substantial interest.
Recently, Dr. Yamanaka and colleagues (38, 39) demonstrated that induced pluripotent stem (iPS) cells could be generated from mouse embryonic fibroblasts (MEFs) and from adult human fibroblasts using retrovirus-mediated transfection of four transcription factors, octamer-binding transcription factor 4 (Oct-4), sex-determining region Y box 2 (Sox2), myelocytomatosis viral oncogene homolog (c-Myc), and Krüppel-like factor 4 (Klf4). The iPS cells have been shown to undergo multilineage differentiation (18), be less immunorejective, and lack the ethical concerns associated with embryonic stem cells (ESCs). Tsuji et al. (41) demonstrated that local injection of iPS-derived neural stem cells promoted locomotor function recovery in a mouse spinal cord injury model. Our previous study showed that the direct injection of iPS cells into damaged areas of rat cortex significantly decreased the infarct size, attenuated inflammatory cytokine production, and improved motor function in rats with middle cerebral artery occlusion (9). Our further study in vivo showed that iPS-derived dopaminergic neurons functionally increased dopamine release in transplanted graft and improved the abnormal behavior in Parkinson's diseaselike rats (8). These studies were all rooted in the regenerative capabilities of iPS cells. An emerging interest as well is in the parenteral administration of iPS cells for tissue repair of acutely damaged organs. Renal inflammation is the leading mechanism of ischemic kidney tissue damage. Inflammatory indicators during renal ischemia include increased monocyte infiltration, elevated levels of proinflammatory cytokines, and also increased reactive oxygen species (ROS) (4, 36). Given that iPS cells are able to suppress intracellular ROS at low levels (1) and reduce inflammatory cytokines (5, 9), these findings suggest that the use of iPS cells administered parenterally may be a safe and beneficial future for cell transplantation in tissue repair.
In the present study, we examined whether iPS cells administered via an intrarenal arterial route could protect against AKI in ischemia–reperfusion (I/R) rat model. The oncogene c-Myc may contribute to tumorigenesis by overstimulating cell growth and metabolism and/or by causing genomic instability (32). Therefore, preventing teratoma formation in vivo has become an emergent issue in cell transplantation, especially in cases of ES or iPS cell transplantation. A recent study has successfully generated iPS cells from mouse and human fibroblasts using three of these transcription factors: Oct-4, Sox2, and Klf4 (without c-Myc) (30). Our recent study further showed that hepatic engrafted iPS cells without c-Myc did not result in tumor formation 6 months after transplantation into thioacetamide-treated mice (24). In the present study, we therefore evaluated the therapeutic effect of transplantation of three-gene iPS cells without c-Myc on ischemic AKI. Administration of such iPS cells improves renal function and attenuates renal tubular injury in I/R-induced AKI. Specifically, the iPS cell transplantation exhibits the antioxidant, anti-inflammatory, and antiapoptotic properties in vivo.
Materials and Methods
Animals
C57/B6 and nonobese diabetic-severe combined imnmunodeficient (NOD-SCID) mice were purchased from the National Laboratory Animal Center (Taipei, Taiwan), and Sprague–Dawley (SD) rats were obtained from the Laboratory Animal Center of National Yang-Ming University (Taipei, Taiwan); they were raised in a sound-attenuated room with a 12-h light–dark cycle. All experimental procedures and protocols were approved by the Ethical Committee for Use of Laboratory Animals of the National Yang-Ming University and complied with the US NIH guidelines for the Care and Use of Laboratory Animals.
iPS Cell Culture
Murine iPS cells were generated from MEFs derived from 13.5-day-old embryos of C57/B6 mice. The iPS cells were reprogrammed by the transduction of retroviral vectors encoding three transcription factors, Oct-4, Sox2, and Klf4, as described previously with brief modifications (39). Briefly, undifferentiated iPS cells were routinely cultured and expanded on mitotically inactivated MEFs (50,000 cells/cm2) in six-well culture plates (BD Technology) in the presence of 0.3% leukemia inhibitory factor in an iPS medium consisting of Dulbecco's modified Eagle's medium (DMEM; Sigma, St. Louis, MO, USA) supplemented with 15% fetal bovine serum (FBS; Invitrogen), 100 mM minimal essential medium (MEM) nonessential amino acids (Sigma), 0.55 mM 2-mercaptoethanol (Gibco), and antibiotics (Invitrogen, Carlsbad, CA, USA). The mouse iPS cells were transfected with pCX-EGFP to express green fluorescence (enhanced green fluorescent protein) constitutively and were maintained and differentiated in vitro as described previously (18). Reverse transcription-polymerase chain reaction (RT-PCR) was used to examine expression of ESC-related genes. Total RNA was extracted from iPS cells using the TRIzol Reagent (Invitrogen). The messenger RNA of target genes was reversely transcribed to complementary DNA (cDNA) using a reverse transcription system (Promega, Madison, WI). cDNA was amplified using Taq polymerase (Takara, Japan) heated to 95°C for 10 min followed by 30 cycles of denaturation at 95°C for 30 s, annealing at 55°C for 30 s, and extension at 72°C for 30 s. The primers used are shown in Table 1. Amplification of eukaryotic translation initiation factor 4 gamma 2 (Nat1) served as the internal control for sample loading. To evaluate the pluripotent capacity of iPS cells to differentiate into different cell types of all three germ layers, several differentiation protocols were applied as described previously (9). As for hepatocyte differentiation, the protocol using a two-step procedure was applied as described previously (37).
The Sequences for the Primers of Reverse Transcription-Polymerase Chain Reaction

endoOct4, endogenous octamer-binding transcription factor 4; endoSox2, endogenous sex-determining region Y Box 2; endoKlf4, endogenous Krüppel-like factor 4; Fbx15, F box protein 15; Eras, ES cell-expressed Ras; Dppa5a, developmental pluripotency-associated 5A; Rex1, RNA exonuclease 1; Nat1, eukaryotic translation initiation factor 4γ2.
The exogenously transfected genes are required for the induction but not for the maintenance of pluripotency (30). To verify the successful generation of iPS cells in this study, we therefore examined the transcripts of endogenous genes of Oct3/4, Sox2, and Klf4, but not those of exogenous transgenes.
iPS cells were fixed with 100% methanol and air-dried. Then an alkaline phosphatase staining kit (Millipore, Billerica, MA) was used according to manufacturer's instructions. Cells were processed for immunocytochemistry. Primary mouse antibodies for stage specific embryonic antigen-1 (SSEA-1; Cell Signaling, Danvers, MA), α-smooth muscle actin (Biomeda, Burlingame, CA), nestin (Chemicon, Billerica, MA), and α-fetal protein (R&D Systems, Minneapolis, MN) were used. Afterwards, secondary antibody (biotinylated goat anti-mouse IgG) was used. Immunoreactive signals were detected by Fluoresave (Jackson ImmunoResearch, West Grove, PA) and a confocal microscope (Olympus, FV300; Tokyo, Japan). To test pluripotency in vivo, 2 × 107 iPS cells were implanted into the subcutaneous space of NOD-SCID mice (n = 6). After 6 weeks, formation of all three dermal-lineaged structures was assessed by ex vivo biopsy and histologic analysis.
Surgery and iPS Cell Transplantation
Adult SD rats (male, 8 weeks old, approximately 300 g) were anesthetized with sodium pentobarbital (50 mg/kg, intraperitoneally). Each rat was placed on their right side, and left kidney was exposed via a flank incision and dissection of the surrounding tissue. The left renal artery was cannulated by a length of stretched PE10 tubing via the left femoral artery. For induction of total ischemia in the kidney, the left renal artery was clamped with a small vascular clamp for 45 min. Reperfusion was initiated by removal of the clamp. The right kidney was removed simultaneously. Phosphate-buffered saline (PBS) (200 ml) or various iPS cell doses (5 × 104, 1 × 105, 5 × 105, 5 × 106, and 5 × 107 cells per rat) in 200 ml PBS were then administered via the left intrarenal arterial route for 60 s. The sham-operated animals underwent similar operative procedures but without clamping of left renal artery and right kidney nephrectomy. Rats were sacrificed 2 days later to collect blood and kidney tissue samples. The kidney was divided into two parts. One part was stored for histological analysis, and the other part was quickly frozen and stored in liquid nitrogen for protein analysis. At the time of sacrifice, blood was collected to measure blood urea nitrogen (BUN) and creatinine. The blood samples were allowed to clot and then centrifuged at 2,500 rpm for 30 min. Serum was collected for measurement of BUN and creatinine using an Olympus AU-2700 autoanalyzer (Olympus Ltd., Tokyo, Japan). To determine kidney blood perfusion in rats treated with different cell doses of iPS cells, 20 rats were assigned to four groups: sham operation, I/R + PBS, I/R + iPS 5 × 105 and 5 × 107 cells per rat, respectively. Kidney blood perfusion was measured by laser Doppler perfusion imager system (Moor Instruments Limited, Devon, UK) at the baseline, 45 min following ischemia, and at 5, 15, 30, and 45 min of reperfusion phase.
Histology and Tubular Injury Score (TIS)
The kidney tissues were fixed with 4% phosphate-buffered formalin solution, embedded in paraffin block, and cut into 3-μm sections. Sections were stained with hematoxylin and eosin (H&E) and periodic acid–Schiff reagent. To quantify tubular injury, sections were evaluated by assessing 20 randomly selected high power fields (40x objective). A score for each tubular cross-section per intersection was assigned as follows: 0, normal histology; 1, tubular cell swelling, loss of brush border, nuclear condensation, up to 25% of the tubular cross section showing nuclear loss; 2, same as for score 1, except for changes affecting 25–50% of the section; 3, changes affecting 50–75% of the section; and 4, changes affecting 75–100% of the section.
Immunohistochemistry
Immunohistochemical staining was performed with mouse anti-GFP antibody (Cell Signaling), mouse anti-CD68 antibody (ED1; AbD Serotec, Oxford, UK), mouse antiproliferating cell nuclear antigen (PCNA) (Santa Cruz, Santa Cruz, CA), and mouse anti-4-hydroxynonenal (4-HNE) (Abcam, Cambridge, UK). Briefly, paraffin sections of kidneys were deparaffinized with xylene and rehydrated in an alcohol series and water. Kidney sections were subjected to antigen retrieval and were blocked with a peroxidase-blocking reagent. Sections were incubated with the primary antibody overnight at 4°C. After washing, the kidney sections were incubated with Envision system horseradish peroxidase-labeled polymer (Dako, Glostrup, Denmark) for 1 h at room temperature. The sections were visualized with 3,3′-diaminobenzidine tetrahydrochloride (Dako) and counterstained with hematoxylin. The number of positive cells was evaluated by counting stained cells per high power field (HPF) in at least 15 randomly selected fields. The value for the brown deposits in the 4-HNE assay was analyzed by Image Pro Plus software. The 4-HNE staining percentage of the total tubular area was determined, and the mean value of 15 randomly selected fields was calculated. Apoptotic cells in the kidney were identified by terminal deoxynucleotidyl transferase (TdT)-mediated digoxigenin-deoxyuridine nick-end labeling (TUNEL) staining. The TUNEL method for the in situ apoptotic assay was performed according to the method of Gavrieli et al. (12) with minor modifications (14). TUNEL-negative controls were stained without TdT enzyme.
Western Blotting
Renal tissue proteins were separated on a 12% sodium dodecyl sulfate polyacrylamide gel by electrophoresis (SDS-PAGE) and then transferred to polyvinylidene fluoride membranes (Millipore). The membranes were blocked with blocking buffer (5% nonfat dry milk in PBS containing 0.1% Tween 20) and incubated overnight with monoclonal rabbit anti-nitric oxide (NO) synthase 2 (Santa Cruz), rabbit anti-monocyte chemoattractant protein-1 (MCP-1; Abcam), mouse anti-intracellular adhesion molecule-1 (ICAM; Abcam), rabbit anti-interleukin-10 (IL-10; AbD Serotec), rabbit anti-B-cell lymphoma 2 (Bcl2; Cell Signaling), rabbit anti-Bcl-2-associated X protein (Bax; Santa Cruz, Heidelberg, Germany), rabbit anti-caspase-3 (Sigma), or rabbit anti-GAPDH (Sigma) antibodies diluted in blocking buffer at 4°C. After being washed, the membranes were incubated with horseradish peroxidase-conjugated horse anti-mouse (Sigma) antibodies or goat anti-rabbit (Abcam) for 1 h at room temperature. Finally, the membranes were exposed to a Western Lighting Chemiluminescence Reagent (PerkinElmer LAS, Boston, MA). The immunoblot density was analyzed using Image Lab 3 software (Bio-Rad Laboratories, Hercules, CA).
Enzyme-Linked Immunosorbent Assay (ELISA)
The kidneys were homogenized in 1 ml of PBS containing antiproteases [0.1 mmol/L phenylmethylsulfonyl fluoride, 0.1 mmol/L benzethonium chloride, 10 mmol/L EDTA, and 20 kallikrein inhibitor unit (KI) aprotinin A] and 0.05% Tween 20. The samples were then centrifuged at 3,000 × g for 10 min, and the supernatants were immediately assayed using the tumor necrosis factor (TNF)-α or IL-6 ELISA kit (R&D Systems).
Malondialdehyde Assay and Nitrate/Nitrite Concentration
Malondialdehyde (MDA), a biomarker for assessing lipid peroxidation, in the kidney tissue was assayed using a commercial kit for thiobarbituric acid reactive substances (TBARS; Cayman Chemical, Ann Arbor, MI). Optical density at 530 nm (OD530) was determined using an ELISA reader (Bio-Rad Laboratories). Nitrite and nitrate are the primary oxidation products of NO subsequent to reaction with oxygen, and therefore, the nitrite/nitrate concentration was used as an indicator of NO synthesis. Nitrite/nitrate levels in kidney tissues were measured after enzymatic conversion of nitrate to nitrite using nitrate reductase (Cayman Chemical). Subsequently, the total nitrite was assayed by adding 100 μl Griess reagent (0.05% naphthalethylenediamine dihydrochloride and 0.5% sulphanilamide in 2.5% phosphoric acid) to each sample. Optical density at 550 nm was measured, and the total nitrite/nitrate concentration of each sample was calculated by comparison against the OD550 of a standard solution of sodium nitrate prepared in saline.
Ex vivo Green Fluorescence Protein (GFP) Imaging
To assess teratoma or tumor formation, I/R rats were sacrificed 1, 2, and 6 months after iPS cell transplantation (5 × 105 cells per rat), respectively. The organs including kidney, liver, spleen, stomach, intestine, muscle, lung, and brain were examined by histological analysis. In addition, organs of whole abdomen and kidneys were further examined by ex vivo GFP imaging as previously described (24). The excitation filter of 470 nm with a lamp supply of optical lighting of 150 W (Southern California Services, USA) was used as an excited light source to project on the foci of ex vivo transplanted tissues or organs. The GFP imaging capture and photography was based on the recordings of a digital camera (Olympus) through the optical configuration of a dissected microscope (SZ60, Olympus) with a 515-nm viewing (emission) filter.
Statistical Analysis
All data are shown as means ± standard errors of means. Statistical analysis was performed by the one-way ANOVA followed by the Tukey's post hoc analysis for multiple comparisons using the computer software Statistical Package of Social Science (SPSS 15.0, 2006; SPSS Inc., Chicago, IL). Differences in the means between two groups were assessed with unpaired, two-tailed t test. Difference in the survival between two groups was analyzed by the log-rank test. A value of p < 0.05 was considered statistically significant.
Results
Characterization of MEF-Derived iPS Cells
In the present study, we generated mouse iPS cells by introducing three genes (Oct4/Sox2/Klf4) but without c-Myc. Undifferentiated iPS cells were cultured on inactivated MEFs (Fig. 1A) and formed colonies very similar to ESCs. The iPS cell clones were positive for alkaline phosphatase (ALP) and stage-specific embryonic antigen-1 (SSEA-1) immunofluorescent staining (Fig. 1A). Compared to MEFs, RT-PCR showed that the iPS cells expressed a gene signature similar to ESC marker genes, including Oct4, Sox2, Nanog, Klf-4, F box protein 15 (Fbx5), embryonic stem cell-expressed Ras (Eras), developmental pluripotency-associated 5A (Dppa5a), and RNA exonuclease 1 (Rex1) (Fig. 1B). We investigated the pluripotency of iPS cells using embryoid body formation (Fig. 1A) and various differentiation protocols. The iPS cells were able to differentiate into muscle-like cells (mesodermal lineage, positive for α-smooth muscle actin), astrocyte-like cells (neuroectodermal lineage, positive for nestin), and hepatocyte-like cells (endodermal linage, positive for α-fetal protein) (Fig. 1C). These iPS cells expressed various stemness genes and possessed pluripotency, namely they were able to differentiate into the various different cell types from all three germ layers. To test pluripotency in vivo, iPS cells were implanted into the subcutaneous space of NOD-SCID mice. After 6 weeks, ex vivo biopsies and histological analysis revealed the formation of structures from all three dermal lineages (Fig. 1D).

Characterization of mouse induced pluripotent stem (iPS) cells without c-Myc. (A) Top left: Morphology of the iPS cell colonies. Bottom left: iPS cell colonies were positive for alkaline phosphatase (ALP) stain (blue). Top right: positive signals for stage-specific embryonic antigen-1 (SSEA-1) in the iPS cells detected by immunofluorecent staining. Right lower: In vitro embryoid body (EB) formation. (B) As compared to mouse embryonic fibroblasts (MEFs), the RT-PCR results showed that the iPS cells express a gene signature similar to that of mouse embryonic stem (mES) cells marker genes. (Nat1, internal control) (see Table 1 for gene definitions). (C) iPS cells differentiated into all three germ layers in vitro, including smooth muscle-like cells [positive for α-smooth muscle actin (α-SMA)] (left), astrocyte-like cells (positive for nestin) (middle), hepatocyte-like cells [positive for α-fetal protein (AFP)] (right). (D) 2 × 107 iPS cells were implanted into the subcutaneous space of nonobese diabetic-severe combined immunodeficient (NOD-SCID) mice (n = 6). After 6 weeks, ex vivo biopsies and histological analysis revealed three dermal lineage formations (striated muscle-like tissue, columnar epithelium-like tissues, and neuroepithelium-like tissue) in the subcutaneous grafts. Scale bar: 100 mm in iPS (A, top left), EB (A, bottom right), and cartilage (D, left). Other scale bars: 200 μm.
iPS Cells Improved Renal Function and Attenuated Tubular Injury in I/R-Induced AKI
Rats that developed AKI displayed impaired renal function and severe tubular injury at 48 h after I/R (17). Initially, 5 × 105 iPS cells were administered via intraarterial, intraperitoneal, and intravenous routes, respectively. BUN and serum creatinine levels at 48 h after I/R were significantly reduced only in the I/R + iPS group via intra-arterial route compared with the PBS-treated (I/R + PBS) group (Fig. 2A). Then, 5 × 105 iPS cells in 200 ml PBS were administered via intra-arterial route for 60 s (low speed) and 10 s (high speed), respectively. BUN and serum creatinine levels at 48 h after I/R were significantly reduced in the low-speed group, but not in the highspeed group (Fig. 2B). Accordingly, intra-arterial route for iPS cell injection with a low speed was preferential in the present study. To further assess the dose effects of iPS cells on I/R-induced AKI, rats were administered with iPS cells of 5 × 104, 1 × 105, and 5 × 105 cells per rat, respectively. I/R + iPS groups had a significant reduction in BUN and serum creatinine levels in a cell dose-dependent manner compared with the I/R + PBS group, and the maximal effect was observed at 5 × 105 iPS cells (Fig. 2C). Histological examination indicated that there were no tubular lesions in the kidneys from the sham-operated group. In I/R rats receiving PBS, the kidneys had severe tubular injury lesions at 48 h, including loss of the brush border, flattening and loss of the tubular epithelium, nuclear fragmentation, and hyaline casts (Fig. 2D). The tubular injury score (TIS) was significantly increased in I/R + PBS group compared with the sham-operated group. In contrast, iPS cell transplantation markedly attenuated the tubular injury in I/R kidneys, as evidenced by fewer casts and less tubular debris (Fig. 2D).

Induced pluripotent stem (iPS) cell transplantation improved renal function and attenuated tubular injury in rats with ischemia–reperfusion (I/R)-induced acute kidney injury. (A) Blood urea nitrogen (BUN) and serum creatinine levels at 48 h after reperfusion in the sham-operated group, I/R + phosphate-buffered saline (PBS) group, and I/R + iPS groups treated with 5 × 105 cells per rat via intra-arterial (IA), intraperitoneal (IP), and intravenous (IV) routes, respectively (n = 5 in each group). (B) BUN and serum creatinine levels at 48 h after reperfusion in the sham-operated group, I/R + PBS group and I/R + iPS groups treated with 5 × 105 cells in 200 ml PBS via intra-arterial route for 10 s (high speed) and 60 s (low speed), respectively (n = 5 in each group). (C) BUN and serum creatinine levels at 48 h after reperfusion in the sham-operated group, I/R + PBS group, and I/R + iPS groups treated with 5 × 104, 1 × 105, and 5 × 105 cells per rat, respectively (n = 9 in each group). (D) Histological evaluation of kidney samples was taken from sham (top left) or I/R injury rats receiving PBS (top right), iPS 5 × 105 cells (bottom left), iPS 5 × 107 cells (bottom right) at 48 h after reperfusion. Tubular injury score in the sham-operated group, I/R + PBS group, and I/R + iPS groups, respectively. Scale bar: 50 μm. Data were expressed as mean ± SEM. *p < 0.05 versus sham group. #p < 0.05 versus I/R + PBS group.
Safety of iPS Cell Number Administration
To define the safety window for intrarenal arterial injection of iPS cells, I/R rats were treated with 5 × 105, 5 × 106, and 5 × 107 iPS cells, respectively. TIS and BUN levels were significantly reduced in the I/R + iPS 5 × 105 group, and the reduction was modest in the I/R + iPS 5 × 106 group (Fig. 3A, B). In contrast, TIS and BUN were significantly higher in I/R + iPS 5 × 107 group than those in I/R + iPS 5 × 105 group. The renoprotective effect for AKI was not found by iPS cell transplantation at 5 × 107 cells per rat. To further elucidate why transplantation with a high cell dose failed to attenuate AKI, renal blood perfusion was monitored for 45 min during reperfusion by laser Doppler imaging (26). In the I/R + PBS and I/R + iPS 5 × 105 groups, blood perfusion of the kidney was intact at baseline before ischemia and was totally blocked during ischemic induction. Kidney perfusion returned to the baselines by reperfusion for 45 min (Fig. 3C). However, the recovery of renal perfusion was significantly impaired at all time points of reperfusion phase in the I/R + iPS 5 × 107 group as compared with the I/R + PBS group. Microscopic examination was performed on the kidneys of I/R + iPS 5 × 107 group, showing evidence of cellular emboli in the small arteries affecting the kidneys (data not shown). Since the optimal renoprotective effects have been observed by transplantation of 5 × 105 iPS cells (Figs. 2 and 3), we therefore used this cell dose in the following experiments.

Changes of tubular injury score, blood urea nitrogen (BUN), and blood perfusion ratio of the kidneys in the recovery phase of reperfusion. (A) Tubular injury score and (B) BUN levels in the sham-operated group, I/R + phosphate-buffered saline (PBS) group, I/R + iPS groups treated with 5 × 105, 5 × 106, and 5 × 107 iPS cells per rat, respectively. (C) The images and quantification of blood perfusion in the rat kidneys were detected by laser Doppler perfusion imager system. Blood perfusion recovery was determined at 5, 15, 30, and 45 min of reperfusion (n = 5 in each group). *p < 0.05 versus sham group. #p < 0.05 versus I/R + PBS group. +p < 0.05 versus I/R + iPS 5 × 105 group.
Mobilization of iPS Cells Into the Peritubular Area
To localize the iPS cells transfected with pCX-EGFP within the I/R kidneys, immunohistochemical staining with anti-GFP antibody was used to trace the distribution of the iPS cells administered via the intraarterial route. Forty-eight hours after iPS cell transplantation in the I/R rats, GFP-positive cells were detected in kidney tissues. The iPS cells were mainly located in the peritubular area, rather than within the renal tubules (Fig. 4A). Results also showed that the engraftment of GFP-positive cells was highest in the kidneys, followed by the spleen and by the liver (Fig. 4B).

Mobilization of induced pluripotent stem (iPS) cells to peritubular area. (A) The renal sections were immunostained with anti-green fluorescence protein (GFP). GFP-positive cells (arrows in the bottom right) were detected in the peritubular area only in the I/R rats treated with 5 × 105 iPS cells. (B) The sections of kidney, liver, and spleen were immunostained with anti-GFP 48 h after 5 × 105 iPS cell transplantation in I/R rats. GFP-positive cells (arrows) were mainly detected in the kidneys. Quantification of GFP-positive cells per high power field (HPF) was shown in the right lower panel. Scale bar: 50 mm. Data were expressed as mean ± SEM (n = 5 in each group). *p < 0.05 versus kidney.
iPS Cell Transplantation Reduced Tissue Oxidative Stress
Previous studies have shown that the production of ROS contribute to I/R kidney injury (31, 43). The expression of inducible NO synthase (iNOS) was significantly upregulated at 48 h in the I/R + PBS group, and its upregulation was significantly reduced in the I/R + iPS group (Fig. 5A, B). I/R + PBS resulted in a significant increase in nitrite/nitrate and MDA levels compared to the sham-operated rats. These increases were significantly reduced by iPS cell transplantation (Fig. 5C, D). Furthermore, using immunohistochemical analysis for 4-HNE adducts in renal tubular cells, the increased stain percentage was substantially reduced by iPS cell transplantation compared to the I/R + PBS group (Fig. 5E).

Induced pluripotent stem (iPS) cell transplantation reduced tissue oxidative stress. (A, B) Inducible nitric oxide synthase (iNOS) protein by Western blotting, (C) nitrate/nitrite levels, (D) tissue malondialdehyde (MDA), and (E) 4-hydroxynonenal (4-HNE) by immunostaining in the kidney tissues of the sham-operated, I/R + phosphate-buffered saline (PBS) group, and I/R + iPS group (n = 9 in each group). Scale bar: 50 mm. *p < 0.05 versus sham group. #p < 0.05 versus I/R + PBS group. GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Effects of iPS Cell Transplantation in Macrophage Infiltration and Tubular Cell Apoptosis and Proliferation
Infiltration of macrophages, in the presence of CD68 (ED-1) positive cells, was significantly increased in the I/R + PBS group compared to the sham-operated group. After iPS cell transplantation, the number of macrophages was substantially reduced (Fig. 6A, B). Injured renal tubules were repaired by the proliferation of surviving renal tubular cells. Immunostaining for PCNA showed that, after iPS cell transplantation, there was a greater increase in the number of PCNA-positive tubular cells compared to the I/R + PBS group (Fig. 6A, C). TUNEL-positive cells ranged from 10 to 70 cells per HPF in the I/R rat studies (10, 35, 40). In the renal cortex, the majority of TUNEL-positive cells was in the tubular compartment. To minimize the false TUNEL-positive cells by negative controls (Fig. 6A), apoptotic cells were significantly increased in the I/R + PBS group compared to the sham-operated group, but the increase was significantly reduced by iPS cell transplantation (Fig. 6D).

Induced pluripotent stem (iPS) cell transplantation decreased macrophage infiltration and tubular cell apoptosis as well as stimulated tubular cell proliferation. The kidney sections of the sham-operated, I/R + phosphate-buffered saline (PBS), and I/R + iPS groups were immunostained with anti-CD68 (ED-1) and anti-proliferating cell nuclear antigen (PCNA) and by terminal deoxynucleotidyl transferase-mediated digoxigenin-deoxyuridine nick-end labeling (TUNEL) assay (n = 9 in each group). TUNEL-negative controls were stained without TdT enzyme. Scale bar: 50 mm. *p < 0.05 versus sham group. #p < 0.05 versus I/R + PBS group.
iPS Cell Transplantation Reduced Renal Inflammation and Apoptosis-Related Protein Expression
The proinflammatory cytokines (TNF-α, IL-6, MCP-1, and ICAM-1) and anti-inflammatory cytokine (IL-10) in the kidney tissues were measured at 48 h after I/R. A marked elevation in TNF-α and IL-6 levels was observed in the I/R + PBS group. In contrast, this increase was suppressed in the I/R + iPS group (Fig. 7A). A significant increase in MCP-1 expression was found in the I/R + PBS group compared to the sham-operated group. The upregulated expression of MCP-1 was reduced in the I/R + iPS group compared to the I/R + PBS group. The changes in ICAM-1 expression were modest and did not reach statistical significance among the three groups. The reverse changes were noted by decreased IL-10 expression in the I/R + PBS group and increased expression in the I/R + iPS group (Fig. 7B). Based on the results of TUNEL assay, we further assessed the Bax/Bcl-2 ratio and caspase-3 (CPP32) expressions in the kidney tissues. A marked increase of the Bax/Bcl-2 ratio and a significant enhancement in CPP32 expression were observed in the I/R + PBS group compared to the sham-operated group. The upregulation of the apoptosis-related proteins was significantly reduced almost to the levels of the sham-operated group by iPS cell transplantation (Fig. 7C).
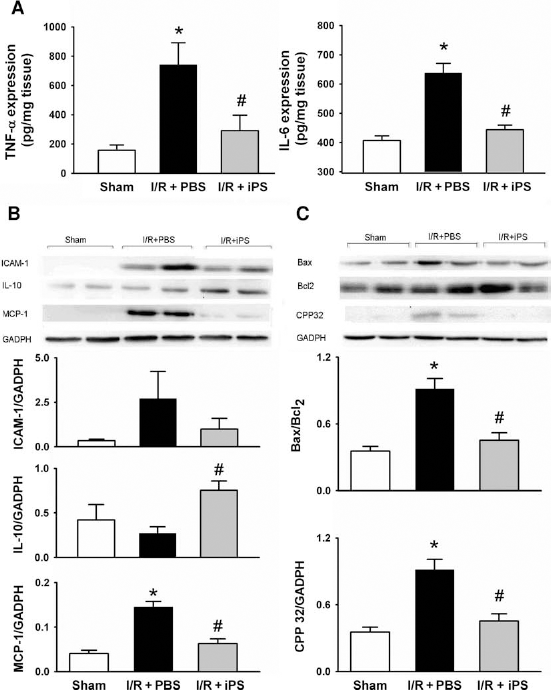
Induced pluripotent stem (iPS) cell transplantation reduced inflammatory cytokines and apoptotic proteins. (A) Tumor necrotic factor (TNF)-α and interleukin (IL)-6 protein levels were measured by enzyme-linked immunosorbent (ELISA) assay in the I/R kidneys. (B) The expressions of IL-10, monocyte chemoattractant protein (MCP)-1, and intracellular adhesion molecule (ICAM)-1, as well as (C) the ratio of B-cell lymphoma 2-associated X protein/B-cell lymphoma 2 (Bax/Bcl-2) and active caspase-3 (CPP32) were measured using immunoblotting assay (n = 9 in each group). *p < 0.05 versus sham group. #p < 0.05 versus I/R + PBS group.
iPS Cell Transplantation Improved Survival of Rats with I/R-Induced AKI
The survival rates in the I/R + iPS, I/R + PBS, and sham-operated groups were analyzed since survival is an outcome index for therapeutic modality of AKI. Seventy-five percent of I/R rats receiving PBS died within 8 days, while the mortality rate of I/R rats 14 days after iPS cell transplantation was only 8.4% (Fig. 8A). The survival rate in I/R + iPS rats was significantly higher than I/R + PBS rats (p < 0.001), indicating that iPS cell transplantation prevents acute lethality of AKI. BUN and serum creatinine levels of the survived rats were measured for 2 weeks in the I/R + PBS and I/R + iPS groups (Fig. 8B, C). The peak of BUN or serum creatinine occurred 2–4 days after ischemic injury and was closely associated with the mortality in I/R + PBS group. The renal function deterioration was significantly attenuated in I/R + iPS group. Then BUN and serum creatinine levels in both groups progressively decreased until day 7.

Induced pluripotent stem (iPS) cell transplantation protected rats from acute kidney injury-induced lethality. The rats were underwent ischemia–reperfusion surgery and intra-arterial injection of phosphate-buffered saline (PBS) or iPS 5 × 105 cells on day 0 and then were followed up for 14 days. (A) Survival curves among the three groups of sham operation (n = 10), I/R + PBS (n = 12), and I/R + iPS (n = 12) were assessed and compared by log-rank test. (B) Blood urea nitrogen (BUN) and (C) serum creatinine levels of survived rats in I/R + PBS and I/R + iPS groups were measured at days 0, 2, 4, 7, and 14. The number in brackets means number of survived rats. Data were expressed as mean ± SEM. *p < 0.05 versus I/R + PBS group.
No Tumor Formation in I/R Rats Receiving iPS Cells Without c-Myc
Teratoma or tumor formation is likely to occur in iPS cell transplantation (23, 31). We used histological examination and ex vivo GFP imaging to assess whether iPS cells lacking exogenous c-Myc could reduce the incidence of tumorigenesis (Fig. 9). The gross necropsy and ex vivo GFP imaging all indicated no formation of GFP-positive tumors in the kidneys and other organs of abdomen. Histological examination further confirmed no formation of tumor-like structures in the grafts or other organs of I/R rats 6 months after transplantation of iPS cell without c-Myc.

No tumor formation in I/R rats 2 months after transplantation of induced pluripotent stem (iPS) cells without c-Myc. (A) Paraffin-embedded sections of kidney, spleen, liver, muscle, lung, stomach, intestine, and brain were stained with hematoxylin and eosin. Scale bar: 50 mm (in top four panels) and 500 mm (bottom panels). (B) Gross necropsy findings and ex vivo green fluorescence protein (GFP) imaging in the whole abdomen and kidneys. (C) Incidence of tumor formation in I/R rats 1, 2, and 6 months after transplantation of iPS cells without c-Myc, respectively.
Discussion
Previous studies have shown the therapeutic potential of MSCs in experimental AKI models (7, 13, 29, 40, 44). Most MSCs were derived from bone marrow, a source that is not available in large volumes. Recently, Yamanaka et al. (38, 39) have demonstrated that iPS cells could be generated from human somatic cells or MEFs, suggesting that iPS cells will be more readily accessible for stem cell-based therapy. To the best of our knowledge, this is the first study to demonstrate the therapeutic effect of iPS cells without exogenously introduced c-Myc in an AKI experimental model. There are three major discoveries in the present study: first, intrarenal arterial administration of iPS cells is able to diminish the pathological severity of I/R-induced AKI; second, iPS cell transplantation improves renal function after AKI and dramatically reduces the AKI-related mortality; and third, iPS cells exhibit antioxidative stress, anti-inflammatory, and antiapoptotic effects on kidney injury. Therefore, iPS cells represent a favorable cell source for stem cell-based transplantation and the use of iPS cells administered parenterally may be a safe and beneficial future for cell transplantation in kidney repair.
The therapeutic mechanisms of transplanted MSCs have been widely investigated in previous studies (13, 20, 29, 40, 44, 45). The engraftment frequency of bone marrow-derived stem cells has been reported to be relatively small (16), and the majority of regenerative cells were found to be derived from resident kidney cells and not from external cells (21). Thus, the protective effect of administered MSCs against AKI is thought to occur via differentiation-independent mechanisms (7, 40, 44, 45). The apparent functional recovery of the injured kidney after MSC treatment is believed to be due to various cytokines secreted by MSCs, which create an appropriate microenvironment in an autocrine/paracrine manner; this promotes kidney repair (7, 16, 20, 45). It remains uncertain whether iPS cells are able to accelerate tubular proliferation in the same way as MSCs. In the present study, the administration of iPS cells substantially reduced the accumulation of nitrate/nitrite, MDA, and 4-HNE; decreased apoptotic events; reduced apoptotic proteins; and suppressed the expression of proinflammatory cytokines. At the same time, the treatment restored the pathological changes occurring in the renal tubules and rescued animals from AKI-associated lethality. ROS, cytokines, and chemokines have been implicated in the pathogenesis of ischemic kidney injury (4, 36). The anti-inflammatory and antioxidative properties of the iPS cells in our study are similar to those reports for MSCs (1, 9, 44, 45), and therefore, the rescuing effect of iPS cells on AKI is expected. Furthermore, it is important to determine the safety dosage of stem cell injection. Levi et al. (22) showed that injection with large number (1 × 1 0 6 per 200 ml) of adipose-derived stromal cells affected the kidneys, liver, and lungs with disseminated intravascular coagulation and resulted in death of all mice. We also found that injection with 5 × 107 iPS cells affected the small arteries of the kidneys with cellular emboli, leading to the reduction of the blood reperfusion after ischemia. The failure of rescuing kidney injury at a high cell dose may be partly due to an impairment of the renal circulation by iPS cell clumping and aggregation. Thus, the cell dose for transplantation and the monitoring of renal blood perfusion are extremely crucial when carrying out cell therapy for treatment of AKI using iPS cells.
It has been reported that MSCs may be mobilized into the circulation in response to injury signals and contribute to tissue repair at injury sites, including bone fracture, cartilage defect, muscular dystrophy (11), liver failure (5, 19), and kidney injury (13, 29). It has been reported that chemokines/cytokines [Chemokine (C-C motif) ligand 25 (CCL25), stromal derived factor-1 (SDF-1), and hepatocyte growth factor (HGF)] and their corresponding receptors [chemokine (C-C motif) receptor 9 (CCR9), CCR4, and MNNG HOS transforming gene (c-MET)] mediated the specific migration of these endogenous MSCs from bone marrow to the injured liver (5). Morigi et al. (29) demonstrated that intravenously infused MSCs are mobilized to injured renal tissue. Herrera et al. (13) also showed that CD44 and hyaluronic acid interactions recruit exogenous MSCs to the injured renal tissue and this enhances renal regeneration after intravenous injection of MSCs. In previous studies by other groups (40) and the present study, MSCs and iPS cells were given via an arterial route directly, and all of these stem cells were then mobilized to the injury site in the kidney. Future investigations are necessary to elucidate the interrelationship between iPS cells and injury signals (chemokines/ cytokines), as well as the paracrine effects of the transplanted iPS cells on the ambient renal tissue in the recipients with AKI for further clarification of therapeutic mechanism and their regulation.
In addition, we previously found that by suppressing the c-Myc expression, teratoma-like formation was effectively blocked in rats transplanted with docosahexaenoic acid-treated iPS cells after 4 months of transplantation (8). Therefore, obviation of c-Myc would be a feasible way to reduce the possibility of genomic instability and the incidence of tumor formation. The present study and our recent investigation (23) further passed on opinion that there is no observable teratoma or tumor formation over 6 months in the recipients of iPS cells without c-Myc (with only three genes, Oct4/ Sox2/Klf4). Accordingly, the nonviral vector delivery and reprogramming without pro-onocogenes address a critical safety concern for the potential utility of iPS cells in future studies.
In conclusion, the current study demonstrated that iPS cell transplantation is able to protect against kidney injury in an experimental I/R rat model. In kidney tissues with I/R-induced injury, iPS cell transplantation provides antioxidative, anti-inflammatory, and antiapoptotic effects. Production of nitrate/nitrite and MDA, as well as other proinflammatory cytokines, was substantially decreased. These findings suggest that iPS cells are a potential resource for stem cell-based therapy against I/R-induced AKI. However, further investigations and long-term monitoring after iPS cell transplantation using the AKI model are still needed. Further optimization of iPS cell-based therapy for AKI and an elucidation of the precise therapeutic mechanisms by which it acts will help the in vivo findings to be extended to clinical practice.
Footnotes
Acknowledgments
This study was supported by the grants from the National Science Council (NSC99-2314-B-010-004-MY3 and NSC97-3111-B-075-001-MY3), Taipei Veterans General Hospital (V97S5-004, V98S5-002, V99S5-002, and V98E1-002), and National Yang-Ming University (Ministry of Education, Aim for the Top University Plan), Taiwan. We are grateful to the Division of Experimental Surgery, Department of Surgery for technical assistance. The kind assistance in the measurement of renal perfusion by Laser Doppler Imaging from Dr. P.-H. Huang (the Division of Cardiology, Taipei Veterans General Hospital) is also appreciated. The authors declare no conflict of interest.
