Abstract
Spinal cord injuries (SCI) cause sensory loss and motor paralysis. They are normally treated with physical therapy, but most patients fail to recover due to limited neural regeneration. Here we describe a strategy in which treatment with autologous adherent bone marrow cells is combined with physical therapy to improve motor and sensory functions in early stage chronic SCI patients. In a phase I/II controlled single-blind clinical trial (clinicaltrials.gov identifier: NCT00816803), 70 chronic cervical and thoracic SCI patients with injury durations of at least 12 months were treated with either intrathecal injection(s) of autologous adherent bone marrow cells combined with physical therapy or with physical therapy alone. Patients were evaluated with clinical and neurological examinations using the American Spinal Injury Association (ASIA) Impairment Scale (AIS), electrophysiological somatosensory-evoked potential, magnetic resonance imaging (MRI), and functional independence measurements. Chronic cervical and thoracic SCI patients (15 AIS A and 35 AIS B) treated with autologous adherent bone marrow cells combined with physical therapy showed functional improvements over patients in the control group (10 AIS A and 10 AIS B) treated with physical therapy alone, and there were no long-term cell therapy-related side effects. At 18 months posttreatment, 23 of the 50 cell therapy-treated cases (46%) showed sustained functional improvement. Compared to those patients with cervical injuries, a higher rate of functional improvement was achieved in thoracic SCI patients with shorter durations of injury and smaller cord lesions. Therefore, when combined with physical therapy, autologous adherent bone marrow cell therapy appears to be a safe and promising therapy for patients with chronic SCI of traumatic origin. Randomized controlled multicenter trials are warranted.
Keywords
Introduction
Spinal cord injury (SCI) predominantly results from motor vehicle accidents, falls, violence, or sports-related injuries. At the cellular level, mild contusion to the spinal cord causes massive neuronal and glial cell loss, demyelination, cavitation, and glial scarring (11), resulting in loss of sensory perception, distal motor paralysis, and severe functional impairment. There are no fully restorative therapies for SCI; however, functional improvement depends on the success of a combination of molecular, cellular, and rehabilitative physiotherapy (36). Safe and effective therapies are clearly needed to enhance the quality of life for SCI patients.
Autologous cellular therapy for SCI patients is attractive for a number of reasons. Methods for the delivery of autologous cell populations back into the patient are being widely used. Additionally, the risk of transplantation rejection is limited, and the ethical concerns of using derivatives of embryonic stem cells are eliminated. Autologous cellular therapies currently being investigated in chronic complete SCI patients include the transplantation of olfactory nervous system cells, neural, or adult stem cells (36). Numerous preclinical studies have shown that the fraction of bone marrow cells (BMCs) that is adherent to plastic and contains the unexpanded mesenchymal stromal cells (MSCs) (9) among other cell types, have stem cell-like characteristics and multipotency (29) and therefore have been used (in the form of MSCs) as a feasible therapeutic option for the treatment of chronic SCI [reviewed in Tetzlaff et al. (35)]. BMCs home to sites of injury and secrete an array of growth (32) and neurotrophic factors (3) that deliver immunomodulatory and neuroprotective functions (25) to induce the survival and regeneration of neurons (7). Additionally, BMCs were induced into synaptically excitable neurons (6) and promoted SCI repair in vivo (12).
Studies by our group (18) and others (8,24,26,33) have demonstrated the long-term safety of autologous BM-derived cell therapy in traumatic SCI patients. We have now developed a strategy in which autologous BMCs are allowed to adhere for 72 h to form “minimally manipulated” adherent BMCs (ABMCs) (see Materials and Methods) that are transplanted into SCI patients via intrathecal injections. This permits delivery of a sufficient number of cells close to the injury site in order to augment neural and astrocytic differentiation (20,31). Our preclinical studies in the zebrafish SCI model (28) suggested that these cells enhance SCI repair. To translate these findings to the clinic, we initially examined the safety and efficacy of this cell therapy strategy in a large mammalian SCI model. Studies in a canine SCI model revealed that ABMCs homed to the injury site and resulted in spared white and gray matter, with some remyelination, neural regeneration, and functional improvement in locomotor scores, and with no side effects observed (Gabr et al., submitted). This led us to initiate a randomized single-observer-blind controlled phase I/II trial in humans, and here we report on the safety and efficacy of intrathecal autologous ABMC therapy in 70 chronic traumatic SCI patients.
Materials and Methods
Isolation and Culture of Human ABMCs
Human ABMCs were isolated from BM aspirates from SCI patients. After signing an informed consent, aspirates were obtained under local anesthesia according to standard procedures approved by the ethics committee of Cairo University. One hundred-milliliter volumes were taken from the iliac crest and placed immediately into a sterile container in cold culture medium [Roswell Park Memorial Institute (RPMI) 1640; Invitrogen, Cairo, Egypt]. All subsequent processing was done under complete clinical aseptic conditions at the Stem Cells Unit of Cairo University Hospital. Cells were diluted 1:1 with RPMI 1640 and layered on top of 15 ml Ficoll-Plaque Plus (GE Healthcare, Cairo, Egypt) and centrifuged for 30 min at 800 × g at room temperature. The cell interface was diluted with media and centrifuged for 10 min at 400 × g. The supernatant was discarded, and the nucleated cells were suspended at a density of 2 × 105 cells/cm2 on poly-l-lysine-coated dishes (BD Biosciences, Dubai, United Arab Emirates) in standard minimum essential medium with α modification (α-MEM; Invitrogen). The cells were incubated for 3 days, and nonadherent cells were removed using three washing steps. The cells were lifted by incubation with Accutase (Innovative Cell Technologies, Inc., San Diego, CA, USA). Prior to transplantation, samples were checked for phenotype, viability, and sterility. All 50 patients in the cell therapy group were treated with autologous ABMC therapy by intrathecal transplantation through lumbar puncture, and each received a cumulative target cell dose of 2 × 106 cells/kg. The procedure was repeated monthly until this target dose was achieved for each patient (median was four injections, range was one to eight injections).
For in vitro studies of BMC expansion to form MSCs and for neural induction, ABMCs were left to grow for 12–16 days before they were passaged and replated at a ratio of 1:4. After six passages, the differentiation potentials of passage 6 cells (P6) (labeled as MSCs) were compared to those of corresponding to passage 0 (P0) cells (minimally manipulated ABMCs) from the same BM sample. Osteogenic, adipogenic, and chondrogenic differentiation assays were performed as previously described (29). ABMCs were analyzed by flow cytometry (FACSCalibur; BD Biosciences) after exclusion of dead cells with 7-aminoactinomycin D (7-AAD; BD Biosciences) and staining with labeled antibodies against the following cell surface markers: cluster of differentiation 13-phycoerythrin-cyanine 7 (CD13-PC7), CD29-PC7, CD34-phycoerythrin (PE), CD44-fluorescein isothiocyanate (FITC), CD45-PC7, CD73-PE, CD90-PE, CD105-PE, CD166-PE, CD271-PE, and c-kit-PE (all from BD Biosciences).
Neuronal Induction
Neuronal differentiation was performed as described (3) with the following modifications. Neurosphere induction was done by culturing ABMCs in serum-free Dulbecco's modified Eagle's medium (DMEM) supplemented with 2% (v/v) B27 (both from Invitrogen) and the growth factors epidermal growth factor (EGF; 20 ng/ml; R&D Systems, Minneapolis, MN, USA), basic fibroblast growth factor 2 (bFGF; 20 ng/ml; Sigma, Cairo, Egypt), and heparin (5 μg/ml; Sigma). Neural induction was performed by obtaining a single-cell suspension using Accutase and then plating the cells at a density of 2,000 cells/cm2 in serum-free DMEM/F12 (Invitrogen) with 2% dimethyl sulfoxide (DMSO) and 1 μM forskolin (both from Sigma). Cells were kept under these conditions for 4 days and were then analyzed using an AxioObserver Z1 immunofluorescence microscope and AxioVision software (Carl Zeiss Microimaging GmbH, Jena, Germany).
Immunostaining
Cells were fixed in 4% paraformaldehyde (Sigma) and stored in phosphate-buffered saline (PBS; Invitrogen) at 4°C until stained. 4′,6-Diamidino-2-phenylindole (DAPI; BD Biosciences) was used to stain nuclei. Primary antibodies were anti-type III β-tubulin epitope J1 (antibody name TuJ1), anti-platelet-derived growth factor receptor-α (PDGFR-α) (both from Chemicon, Temecula, CA, USA), anti-nestin and anti-70 kDa neurofilament (NF70) (both from LifeSpan BioSciences, Inc., Seattle, WA, USA), and anti-microtubule-associated protein 2 (MAP2) (Sigma). Secondary antibodies labeled with Alexa Fluor 488, 535, and 610 dyes (Invitrogen) were used. To calculate nestin, PDGFR, and TuJ1-positive cells, 100 squares with a surface area of 0.01 mm2 each were counted per marker. Values are presented as mean ± SEM.
Human Phase I/II Trial
The design of this prospective randomized controlled clinical trial conformed to international SCI clinical trial standards in regard to patient selection criteria, clinical outcome measures, and time of interference (19).
Inclusion Criteria
Traumatic chronic SCI of 10–36 months in duration.
Completion of at least 6 months of physiotherapy postinjury with no spontaneous recovery or neurological improvement (10).
No concomitant systemic disease.
Baseline assessment of American Spinal Injury Association (ASIA) Impairment Scale (AIS) A and B.
Exclusion Criteria
Nontraumatic SCI, including patients with either transverse myelitis or demyelination, or lumbar SCI.
Acute SCI or duration of injury less than 10 months or more than 36 months.
Evidence for clinical and/or electrophysiological improvement in spinal cord functions over the previous 6 months.
Baseline assessment of AIS C or D.
Major impairment of respiratory, cardiac, hepatic, or other systemic functions.
Unfitness for physical therapy, such as patients with muscle fibrosis.
Patients were recruited from the outpatient clinics of Physical Medicine at Al-Azhar, Cairo, and Alexandria University Hospitals and provided therapies (treatment and follow-up) free of charge. During the enrollment period, 159 patients were evaluated, and 70 patients were enrolled after signing an informed consent under a protocol approved by the clinical trial review committees at Cairo University and Al-Azhar University. All of the 70 patients completed the follow-up that included monthly neurological assessments and magnetic resonance imaging (MRI) every 6 months for 18 months. Patients in the trial included 9 females and 61 males, aged 16–45 years. The period since their injury ranged from 12 to 36 months with an average duration of injury of 18.25 ± 5 months and a median of 17.25 month. Neurological levels of enrolled patients were between C3 and T12. Patients were assigned randomly based on coding numbers into two groups: 50 patients who received autologous ABMC transplants in combination with standard physiotherapy, while 20 patients received standard physiotherapy only. To minimize the number of control patients, sample size analysis for controls was based on historical response of SCI patients treated with physiotherapy only at our institute. We made the assumption that 30% of patients treated with cell therapy will have neurological improvement as defined in the neurological outcome criteria below. Analysis calculated for a power of 80% and α (type I error) of 5% and powered to show a statistically significant difference of ≥10 points on the ASIA motor score, a reliable therapeutic outcome measure (10,22), indicated that the neurological outcome of 50 cell therapy-treated patients can be statistically compared with 20 controls. The risks associated with lumbar puncture and cell injections raised significant ethical concerns that limited the inclusion of a sham-injected control group. Observers at Armed Forces Center for Physical Medicine, Rehabilitation, and Rheumatology (Cairo, Egypt) who were blinded to treatment type performed AIS and all other measurements on all patients during the trial.
All 50 patients in the cell therapy group were treated with autologous ABMCs as described above. Patients were assessed by clinical examination, MRIs, ASIA scores (including both upper and lower extremities motor scores obtained by summing a six-grade evaluation of 10 key muscles from both upper and lower limbs and a sensory score obtained by using light-touch and pin-prick tests at 28 dermatomes from C2 to S5 in each side of the body), functional independence measurements using a functional rating score (FRS) (a detailed description of the FRS is provided within the neurological assessment tables) (13), bladder and bowel control questionnaire, and electrophysiological somatosensory-evoked potential (SSEP) measurements using a Neuropack II (Nihon-Kohden, Tokyo, Japan) that were taken at baseline and during the 18-month trial. The trial data were regularly reviewed by the Egyptian Ministry of Health review committee and were finally reviewed by an independent oversight committee from Germany.
Physical Therapy
Both control and cell therapy patients were provided with physiotherapy programs for at least 1–2 h three times per week. At times of lumbar punctures, all control and cell therapy patients stopped physiotherapy for 2–3 days. Physiotherapy included mat and transfer activities, self-range of motion, strengthening exercises, ambulation training for paraplegic patients, upright posture on the tilt table, and cardiopulmonary training.
Data and Statistical Analysis
All data were expressed as mean ± SD. In vitro data and quantitative variables for clinical outcome among patients and controls were compared using a Student's t test when applicable. Group outcome statistics among subgroups of patients and controls were compared using two-sample Wilcoxon rank-sum (Mann–Whitney) test or Fisher's exact test among all groups using Microsoft Excel (Redmond, WA, USA), Sigmaplot (Systat, San Jose, CA, USA), or Stata software (College Station, TX, USA), unless otherwise specified. A value of p < 0.05 was considered statistically significant.
Results
In Vitro Differentiation of ABMCs
We evaluated the differentiation potential of minimally manipulated ABMCs isolated from 10 normal human BM samples from healthy volunteers or patients undergoing splenectomy. Flow cytometry of the cells revealed that human ABMCs that are positive for CD44 are also positive for CD29, CD73, CD90, CD105, CD166, and CD271, but lack expression of CD45, CD34, CD13, and c-kit (Fig. 1). Compared to MSCs that are traditionally expanded initially for 2–3 weeks before use (29), human ABMCs were more potently inducible to mesodermal trilineage differentiation into adipocytes, osteocytes, and chondrocytes, with differentiation occurring within 1 week (Fig. 2A–C). Neural induction (3) resulted in the appearance of typical neural morphology in cells displaying multiple primary dendrites (Fig. 2G–I) and atypical neurosphere formation when cells were induced in partial serum conditions (not shown). Compared to culture-expanded MSCs, ABMCs were more potently inducible to cells with upregulated immunoreactivity to the neuronal marker MAP2 (Fig. 2G–I), the neurofilament marker NF70 (Fig. 2J–K), the oligodendrocyte precursor marker PDGFR-α (inset in Fig. 2L), and the neuronal precursor marker type III β-tubulin (TuJ1; Fig. 2L). Quantitative analyses of immunoreactive cells from the same patient (n = 6, performed in six-well plates in triplicates) that were either induced for neural differentiation immediately or after culture expansion (passage 6) (i.e., ABMCs vs. MSCs) revealed no significant difference in nestin expression (not shown), while the levels of both PDGFR-α and type III β-tubulin (TuJ1) were significantly higher in ABMCs than in culture-expanded MSCs (Fig. 2M). Although these studies were performed in vitro, the results suggest that when cells are used for cell therapy in vivo, there might be an advantage for using ABMCs versus culture-expanded MSCs.

Flow cytometric analysis of primary human adherent bone marrow cells (ABMCs) that were utilized for cell therapy. Representative flow cytometry histograms of primary ABMCs that were isolated and cultured for 72 h on poly-l-lysine, removed from the plastic with Accutase, and stained with antibodies against the cell markers as indicated. 7-Aminoactinomycin D (7-AAD) was used to exclude dead cells. FSC, forward scatter; Control, untreated cells; CD44, cluster of differentiation 44.

Multilineage potential of human ABMCs isolated after 72 h. Trilineage differentiation into adipocytes, osteocytes, and chondrocytes, as well as neurosphere induction, and neural differentiation of ABMCs are shown. (A) Oil red staining of ABMCs induced for adipocyte differentiation. (B) Osteocyte differentiation visualized using alkaline phosphatase staining. (C) Alcian blue staining of ABMCs induced for chondrocyte differentiation. (D–F) Uninduced ABMCs stained with 4′,6-diamidino-2-phenylindole (DAPI) and microtubule-associated protein 2 (MAP2) and used as control. (G–I) Induction of neural cells with retinoic acid in serum-free conditions to form neural-like cells (arrow) that were stained with anti-MAP2 antibody. (J–L) Induced cells express 70-kDa neurofilament (NF70; K), platelet-derived growth factor receptor (PDGFR)-α in cells with astrocyte-like morphology (inset in L), and type III β-tubulin (antibody name: TuJ1) (L). In induced ABMCs, inset shows higher magnification TuJ1-positive cell with long dendrites. Scale bars: 10 μM (A and J–L) and 20 μM (B–I). (M) Percentage of nestin, PDGFR, and TuJ1 cells in human minimally manipulated ABMCs at passage 0 (P0) compared to induced mesenchymal stromal cells (MSCs) from the same patients after passage 6 (P6 MSCs; *p < 0.001, t test).
Phase I/II Clinical Trial of Autologous ABMCs in Chronic SCI Patients
All 70 SCI patients (AIS A and B) who met the inclusion criteria completed the follow-up (53 thoracic and 17 cervical) in the control and cell therapy groups (see Fig. 3 for study design, and Tables 1–4 for outcome analyses, Tables 5–7 for complete neurological assessment scores, and Tables 8–11 for correlative studies). A total of 50 patients were in the cell therapy group (Table 1), and 20 patients were in the control group in a study designed to evaluate the safety of ABMC therapy in multiple subgroups of SCI patients with the minimum number of controls required to gain an initial emphasis on efficacy (Table 1). Both groups received scheduled standard physical therapy performed by observers who were strictly blinded to treatment.
Number of Patients in Study Groups of SCI Patients Treated With Either Physiotherapy Alone or Physiotherapy Combined With ABMC Therapy at 18 Months After Treatment

SCI, spinal cord injury; ABMC, adherent bone marrow cell; AIS, American Spinal Injury Association (ASIA) Impairment Scale.
Assessment of ASIA and Functional Rating Mean Scores at Enrollment (Before) and at 18 Months After Therapy (After) Comparing AIS A SCI Patients Treated With Physiotherapy Combined With ABMC Therapy Versus AIS A Control Patients Treated With Physiotherapy Alone

Thor, thoracic; cerv, cervical; ND, not determined.
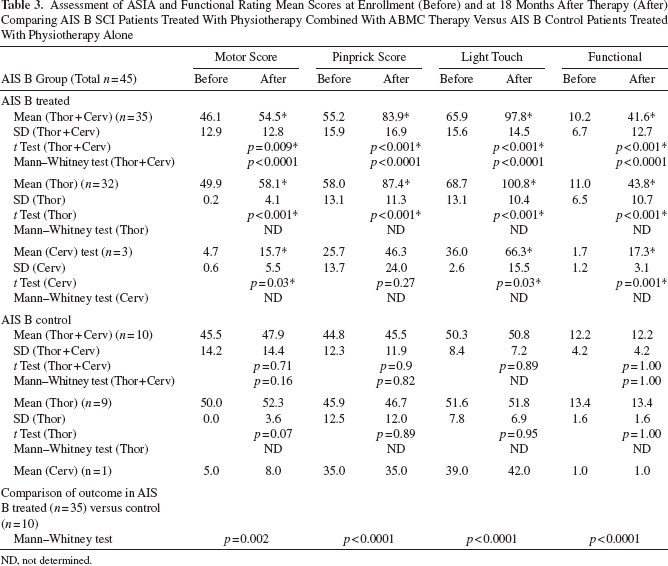
Assessment of ASIA and Functional Rating Mean Scores at Enrollment (Before) and at 18 Months After Therapy (After) Comparing AIS B SCI Patients Treated With Physiotherapy Combined With ABMC Therapy Versus AIS B Control Patients Treated With Physiotherapy Alone
ND, not determined.
Assessment of AIS Conversion Among SCI Patients Treated With Either Physiotherapy Alone or Physiotherapy Combined With ABMC Therapy at 18 Months After Treatment

Comparisons stratified by AIS status among thoracic or cervical AIS A and AIS B SCI patients were underpowered but were suggestive of a nonsignificant trend.
p = 0.05 when compared to 13 thoracic AIS A and AIS B controls, Fisher's exact test.
p = 0.009 when compared to 20 AIS A and AIS B controls, Fisher's exact test.
Nested study analyses 1 and 2 were performed utilizing Stata version 12.1 since the treated group had a higher percentage of AIS B cases as compared to the control group (70% of the treated and 50% of the control groups were AIS B). Treated subjects were randomly excluded such that 50% were AIS B to match the percentage within the control group.
p = 0.02 when compared to 20 AIS A and AIS B controls, Fisher's exact test.
p = 0.001 when compared to 20 AIS A and AIS B controls, Fisher's exact test.
p = 0.08 when compared to 20 AIS A and AIS B controls, Fisher's exact test.
p = 0.003 when compared to 20 AIS A and AIS B controls, Fisher's exact test.
Characteristics of 40 Thoracic SCI Patients Treated With ABMC Therapy

Functional rating score (FRS) (max 61 points) (see Table 6).
PP, pinprick; LT, light touch; shaded, AIS A; clear, AIS B.
Characteristics of 10 Cervical SCI Patients Treated With ABMC Therapy

Functional rating score (FRS) (max 61 points) was utilized to assess motor functional improvement at enrollment, during follow-up, and after 18 months. The scoring system is made of 0–61 points and is comprised of a 0–3 score for each of 17 functions including upper limb, lower limb, sphincter control, reflexes, and skin functions, and 0–10 points for trunk movement. The distribution of scoring is as follows: eating (0–3 points), grooming (0–3 points) and writing (0–3 points) for upper limbs, standing (0–3 points) or walking with brace (0–3 points) for lower limbs, trunk movement (0–10 points), sitting (0–3 points) and turning body over (0–3 points), sphincter control (0–3 points for bladder control and 0–3 points for bowel control), muscle tension (0–3 points), sweating (0–3 points), reflexes (0–3 points), skin condition (0–3 points) and general movement including transfer to bed (0–3 points), bathing (0–3 points), toileting (0–3 points) and dressing (0–3 points). The sum of the FRS is shown from treated thoracic (Table 5), treated cervical (Table 6), and control SCI patients (Table 7) at enrollment and after 18 months of follow-up.
PP, pinprick; LT, light touch; shaded, AIS A; clear, AIS B.
Characteristics of 20 Control Cervical and Thoracic SCI Patients Treated With Physical Therapy Only
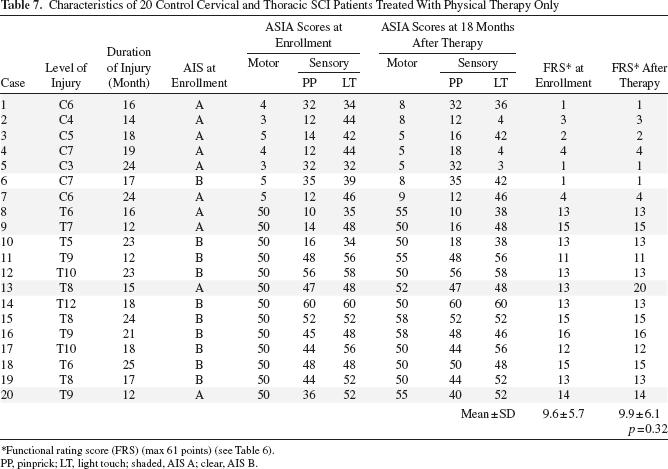
Functional rating score (FRS) (max 61 points) (see Table 6).
PP, pinprick; LT, light touch; shaded, AIS A; clear, AIS B.
Clinical Improvement Among Thoracic SCI Patients Who Were Treated With ABMC Therapy Combined With Physical Therapy and Showed ASIA Motor Score Improvement of ≥10 Points

Cervical SCI Patients Who Were Treated With ABMC Therapy Combined With Physical Therapy and Showed ASIA Motor Score Improvement of ≥10 Points

Correlation Between Duration of Injury and Clinical Outcome Among 40 Thoracic SCI Patients Treated With ABMC Therapy Combined With Physical Therapy

Correlation Between Level of Injury and Clinical Outcome Among 40 Thoracic SCI Patients Treated With ABMC Therapy Combined With Physical Therapy


Study design of autologous intrathecal transplantation of ABMCs combined with physiotherapy in chronic SCI patients. Diagram shows enrollment, inclusion criteria, and follow-up criteria. SCI, spinal cord injury; AIS, American Spinal Injury Association (ASIA) Impairment Scale; MEP, motor evoked potential; SSEP, somatosensory evoked potential; MRI, magnetic resonance imaging; FIM, functional independence measures utilizing motor function rating scores (FRS).
Most of the patients receiving cell therapy experienced mild side effects common with lumbar punctures including headache and mild pain at puncture site immediately after the procedure. However, they were temporary and were completely resolved by symptomatic treatment. Neuropathic pain was reported as both at and below neurological level pain starting at 12 h and for up to 12 weeks postinfusion in 15 of 50 patients (30%) treated with cell therapy. Five of these 15 patients had chronic pain at enrollment that increased from 4–5/10 to 7/10. Four of the control patients reported chronic neuropathic pain from previous injuries at enrollment that remained the same at follow-up. Pain episodes were usually resolved with gabapentin or oxycarbamazepine within 2 weeks. For up to 18 months posttransplantation, no long-term side effects were detected in ABMC-treated patients.
Clinical neurological assessment and grading of the injury according to the AIS were performed for all patients at presentation and during each follow-up. At the final follow-up at 18 months, long-term safety, functional improvement, and improvement of both motor score of ≥10 points and dermatomes were considered as three primary outcomes, and all three were detected in 23 of the 50 patients in the autologous ABMC therapy group (46%). In contrast, the 20 patients in the control group only improved their motor scores by 3 ± 2.4 points for thoracic SCI and 2.7 ± 1.9 points among cervical SCI patients in the AIS A group (Table 2) and nonsignificantly by 2.3 ± 3.6 points among thoracic SCI control patients and 3 points in one cervical SCI control patient in the AIS B group, respectively (Table 3), compared to their enrollment scores. Thus, among the control group of 20 patients, mean motor scores increased from enrollment scores by a few points, an effect that is attributed to physiotherapy, but none reached ≥10 points on the motor score. Among the two groups of 10 patients with cervical SCI and 40 patients with thoracic SCI who were treated with the combination of ABMCs and physical therapy, 5 cervical SCI patients (50%) and 18 thoracic SCI patients (45%) showed signs of clinical neurological improvement.
Analyses of neurological ASIA scores among AIS A and AIS B subgroups were much more supportive of a therapeutic benefit for SCI patients who were treated with the combination of physiotherapy and ABMC therapy (Tables 2–3). An increase in the ASIA motor score of ≥10 points was considered a reliable measure of therapeutic benefit among chronic SCI patients whose spontaneous repair plateaued at >1 year after injury (22), it was considered a primary therapeutic outcome for cell therapy and was seen in 26 of 50 patients treated with cell therapy, but not in any of the 20 controls (Fisher's exact test; p = 0.001). Among AIS A patients, 12 of 15 patients treated with cell therapy but none of 10 controls increased their ASIA motor score by ≥10 points. This difference was statistically significant (Fisher exact test; p = 0.01). Similarly, among AIS B patients, 14 of 35 patients treated with cell therapy but none of 10 controls increased their ASIA motor score by ≥10 points, though this trend was not significant (Fisher's exact test; p = 0.09).
Analyses of all ASIA neurological motor and sensory score means as well as functional independence scores among treated AIS A patients (n = 15) compared to AIS A controls (n = 10) (Table 2) demonstrated a statistically significant improvement in motor, pinprick, and light touch sensory scores and functional independence scores after cell therapy among the 15 AIS A cell therapy-treated patients (Table 2), and a nonsignificant trend for light touch scores among cervical patients (p = 0.09) (Table 2). In contrast to cell therapy-treated patients, the 10 AIS A controls had only 2.4 (for thoracic SCI) and 1.9 (for cervical SCI) point increases in their ASIA motor score after physiotherapy compared to enrollment scores, but none of the changes in pinprick and light touch sensory scores were statistically significant (Table 2). Moreover, neurological outcome comparisons of the 15 AIS A-treated patients compared to the 10 AIS A controls were highly statistically significant (p = 0.002 for motor, p < 0.0001 for pinprick, p = 0.0002 for light touch, and p < 0.0001 for functional scores) (Mann–Whitney test) (Table 2).
Similarly, analyses of ASIA motor and sensory score means and functional independence scores among treated AIS B patients (n = 35) compared to AIS B controls (n = 10) (Table 3) demonstrated a statistically significant improvement in motor, pinprick, and light touch sensory scores and functional independence scores after cell therapy among the 15 AIS A cell therapy-treated patients (Table 3) (with nonsignificant trend for pinprick scores among the three cervical patients; p = 0.27) (Table 3). In contrast, the 10 AIS B controls had only 2.3 ± 3.6 (for thoracic SCI) and a 3.0 (for a cervical SCI patient) point increases in their ASIA motor score after physiotherapy compared to enrollment scores, but none of the changes in motor, pinprick, and light touch sensory scores were statistically significant (Table 3). Collectively, we detected a statistically significant improvement of neurological and functional measures in both AIS A and AIS B SCI patients treated with the combination of physiotherapy and ABMCs compared to controls treated with physiotherapy only (Tables 2 and 3).
We next analyzed the potential for AIS conversion in ABMC-treated patients (Table 4). A total of 17 (14 thoracic and 3 cervical) of 50 patients treated with cell therapy had AIS conversion from AIS A to AIS B or C and from AIS B to AIC C, but none of the 20 controls had any AIS conversion. This difference was statistically significant (p = 0.009, Fisher's exact test) (Table 4). Moreover, AIS conversion was detected in 14 of 40 thoracic SCI patients (including AIS A and AIS B), but none of the 13 thoracic SCI controls (p = 0.05) (Table 4). However, because the treated group had a higher percentage of AIS B cases as compared to the control group (70% of the treated and 50% of the control groups were AIS B), we assessed if this could account for the results. To test this, we performed two independent nested study analyses in which treated subjects were randomly excluded (utilizing Stata version 12.1), such that 50% were AIS B, to match the percentage within the control group. In effect, 20 treated subjects who were AIS B were randomly excluded from analysis. In the first nested analysis, 12 of 30 treated subjects underwent AIS conversion as compared to none of 20 controls (p = 0.001; Fisher's exact test). Similarly in the second nested analysis, 10 of 30 treated subjects underwent AIS conversion as compared to none of the 20 controls (p = 0.003; Fisher's exact test) (Table 4). From these data in which initial AIS status was frequency matched, we conclude that the cell therapy-treated group underwent a statistically significantly higher rate of AIS conversion as compared to the control group. Next, we stratified the patients by AIS status. Among the cell therapy subgroup of 15 patients with a baseline AIS A (8 with thoracic SCI and 7 with cervical SCI), 2 patients converted to AIS C, 6 patients converted to AIS B, and the other 7 cases remained in the same AIS A (Table 4). Of these 7 AIS A nonconverters, 5 were cervical SCI patients, and the remaining 2 were thoracic SCI patients. Thus, the overall AIS A to B or A to C conversions among AIS A patients treated with cell therapy were 8 of 15 patients but none in the controls (Table 4).
In the cell therapy subgroup of 35 patients with a baseline AIS B, 9 patients converted to AIS C, 14 patients remained in AIS B despite improving their score by ≥10 points, and the other 12 cases remained in AIS B. Collectively, AIS conversion among AIS A and AIS B patients treated with cell therapy were 14 of 40 patients with thoracic SCI and 3 of 10 patients with cervical SCI (Table 4).
We then performed “within group” statistical analyses of the AIS conversion comparing treated AIS A patients versus control AIS A patients and treated AIS B patients versus control AIS B patients (Table 4). Although the analysis was underpowered, there was a nonsignificant trend in AIS conversion for treated AIS A or B versus the corresponding AIS A or B control SCI patients. Moreover, the responders who experienced an improvement of more than one AIS (i.e., AIS conversion from A to C) were two patients with thoracic SCI but none with cervical SCI (Table 4). These data suggest that patients with thoracic SCI might potentially respond better to the combination of ABMCs and physical therapy; however, these differences and group outcome analysis must be challenged in dose escalation studies and with a larger cohort of SCI patients.
Patients treated with intrathecal injections of autologous ABMCs noticed modest improvement in neurological functions as early as 4–6 weeks posttransplant. Patients experienced enhanced response to tactile and sensory stimuli first. This was associated with a variable increase in pain between 8 and 12 weeks posttreatment in 15 of 50 patients. After 12 weeks, neuropathic pain had significantly subsided beyond the pretreatment levels in most patients, and those patients who showed clinical improvements started to gain increased muscle strength. Patients with AIS conversion showed improved trunk movements that allowed them to sit and turn in bed. Additionally, improved muscle strength was associated with enhanced sexual potency and with stronger bowel and bladder sphincter controls that allowed these patients to live catheter free. No urological changes were reported in the patients who did not have improvement in motor or sensory scores.
All patients showed no cortical SSEP before therapy, while 65% of patients treated with ABMCs, but none of the control patients, showed reappearance of cortical impulse at 6–12 months posttransplant, with improved nerve conduction (for data from representative patients, see Fig. 4A–C). In the group treated with cell therapy, there was a significant correlation between the patient's clinical improvement and shorter duration of injury (< 2 years) (Table 10). Data analysis demonstrates that among patients with thoracic SCI who were treated with cell therapy and have shown improvement were five patients, patient 8, patient 12, patient 19, patient 38, and patient 40, who had injury durations of 20–24 month. On the contrary, three patients who were treated with cell therapy, patient 7, patient 30, and patient 37, had injury durations of 36, 32, and 36 months, respectively, and all three were among the nonresponders. These data suggest that cell therapy might be beneficial during the window of opportunity of the earlier phases of chronic SCI. Certainly, the optimum timing of interference with cell therapy might be different for each SCI patient, depending on the extent of injury and the initial response, and this timing must be further investigated in a larger cohort of SCI patients and defined based on personalized medicine approaches.

Electrophysiological and MRI features of ABMC-treated chronic SCI patients at 18 months posttransplantation. (A) Motorevoked potential responses recorded using a Neuropack II in the posterior tibial muscle in a patient treated with ABMCs and a second patient from the control group (representative recordings from one patient from each group). Recording from the control patient with flat activity (top), while the recording from the ABMC-treated patient (bottom) at 12 months posttreatment demonstrates an electrically evoked response with a latency of 20–30 ms that was recorded for the posterior tibialis muscle. (B) SSEP obtained using a Neuropack II of patient 21 showing an improvement in the latency from 51.2 to 45 ms and from 49 to 47.2 ms 12 months posttreatment. (C) Nerve conduction study of patient 20 showing improvement in the motor response amplitude of deep peroneal nerve conduction from 1.17 to 2.40 mV on right side at 12 months posttreatment. (D) Representative MR images before and after autologous ABMC transplantation from patient 6 (cervical SCI) showing compression and edema of the spinal cord at C6–C7, while the image at 12 months posttreatment demonstrates a healed area with minimal gliosis. No abnormal masses or opacities were detected using MRI for any of the treated patients.
Further data analyses demonstrated that there was a significant correlation between the patients' clinical improvement and higher thoracic injuries (T3–T9) (Table 11) and/ or shorter craniocaudal length of the cord lesion in the baseline MRI. Clinical improvements in patients with smaller lesions might be predictable when utilizing similar cell doses to repair variable injuries, while it is unclear at this time why patients with higher thoracic injuries responded better than those with lower lesions. One possibility that remains to be investigated is that the dynamics of cerebrospinal fluid (CSF) flow and the distribution of cells after injections with lumbar puncture might favor homing of cells to the higher thoracic region. In contrast, there were no correlations between the patients' clinical improvement and age, level of injury, or degree of injury. Electrophysiological studies demonstrated that nerve conduction velocities and SSEP showed variable degrees of neurological impairment that correlated with the degree of injury. Three patients who demonstrated clinical improvement after cell therapy showed electrophysiological signs of reinnervation at 1 year posttreatment.
Recovery of spinal cord compression and edema was observed in the patients treated with cell therapy, as shown in representative MR images at 12 months posttransplant from patient 6, who had a C6–C7 cervical SCI (Fig. 4D). Moreover, these changes were associated with a significant improvement in quality of life. This was most evident in two particular patients: patient 6 (with a C7 cervical SCI), who was quadriplegic prior to beginning the study, and patient 10 (with a T4 thoracic SCI), who was paraplegic before cell therapy. Both patients regained mobility and were able to walk 9–12 months posttreatment.
Discussion
Our studies demonstrate that intrathecal transplantation of autologous minimally manipulated ABMCs, when combined with physiotherapy, induces functional motor and sensory improvement in chronic SCI patients. These repair features are likely to be unique to BM-derived cells (5,36), since the outcome after transplantation of olfactory ensheathing cells (OECs) in subjects with chronic SCI was modest (21). Mononuclear BMCs with limited culture expansion have been used in human trials to improve neurological function in one of two chronic SCI patients when delivered locally (33). Importantly, similar to other autologous transplant trials, our study demonstrates a notable absence of long-term side effects related to cell therapy. In contrast, human neural progenitor cells derived from embryos were inconclusively linked to brain tumor development in an ataxia telangiectasia patient (1). Still, BM-derived cell therapy with intrathecal injections might not be completely free of side effects (18). Fifteen cell therapy-treated patients reported a short-term increase in pain during the first 12 weeks after initiation of therapy. Since pain episodes started hours after cell injections and coincided with the enhanced response to tactile and sensory stimuli in 10 patients but was also reported from five treated patients and four controls who had chronic pain from previous trauma and were with no sensory enhancement, it was difficult to ascertain whether these pain episodes were side effects of cell therapy or in some cases due to the restoration of normal musculoskeletal and visceral pain sensation. Accordingly, additional direct measures for changes in the central pain threshold should be implemented to further discern the origin(s) of pain in cell therapy-treated subjects. One important exclusion criteria in this trial was nontraumatic SCI, including patients with either transverse myelitis or demyelination. Since BM-derived cells might have immunomodulatory functions (25), their utilization in SCI patients with inflammatory disorders, especially when used without immunosuppression, requires further validation in preclinical animal models due to the potential for neuroinflammatory exacerbations as recently reported (17) in one patient treated with cell therapy.
The rate of spontaneous functional recovery after SCI is rapid, with motor improvement almost complete by 9 months (10). The majority of AIS A chronic SCI patients (94%) remain with a complete neurological injury at 5 years postinjury (16). We found that 17 of 50 cell therapy-treated patients but none of 20 controls achieved AIS conversion. Among the cell therapy subgroup of 15 patients with a baseline AIS A, 2 patients converted to AIS C and 6 patients converted to AIS B. Similarly, among the cell therapy subgroup of 35 patients with a baseline AIS B, 9 patients converted to AIS C. Moreover, in 50 patients treated with cell therapy (AIS A and AIS B), 23 patients had increased their ASIA motor score with ≥10 points and dermatodes together with a significant improvement in their functional independence measures after 18 months of follow-up. These clinical outcome data of this initial phase I/II study utilizing a single cell dose in a limited number of SCI subjects within each subgroup are encouraging. Still, it remains possible that the cell dosage that we have used was suboptimal for the therapeutic threshold to observe more substantial beneficial effects from ABMC therapy. The data must be challenged in dose escalation studies and with a larger cohort of SCI patients.
When compared with cell transplantation in acute SCI, delayed cell transplantation either in chronic SCI patients or perhaps in the subacute setting is likely to be more successful due to a more permissive injury environment when the inflammatory response has subsided. Additionally, the localized and repeated delivery of BM cells is likely to be more effective than the systemic delivery and homing of BMCs (15,33) for several reasons. First, axonal regeneration likely plays a major role in repair, either directly or mostly indirectly through the recruitment of neural progenitor cells, as we recently demonstrated (28). Second, ABMCs might induce the activation of spinal cord ependymal stem cells in the region around the central canal (4). Third, ABMCs also produce neurotrophic factors and anti-inflammatory mediators (25) that can support the host spinal cord tissue by creating new neuronal pathways in fibrous scar tissues or by propagating short regenerated neuronal fibers. Moreover, ABMCs might contain cells that help form connections between the distal and proximal ends of the spinal cord and facilitate regeneration of transplanted cells (12).
ABMCs injected into the spinal cord that produced injury repair were minimally manipulated BMCs that were allowed to adhere for 72 h but were not expanded in culture to make MSCs. Moreover, as we demonstrated in the in vitro studies, the cell phenotypes utilized in this study are distinct from the widely used BM mononuclear cells or the culture-expanded MSCs. Prolonged culture of BMCs has been shown to alter the biological and phenotypic properties as well as the immunomodulatory and differentiation capacities of BMCs (30). The true identity of the BMC(s) that helped regenerate neural tissues and may have given rise to myelin-forming cells remains illusive. Potential candidates include nonhematopoietic cells that have multipotent activity and share surface marker phenotypes, such as MSCs, adventitial reticular cells, vascular pericytes, bone lining cells (14), and fibroblasts. In light of the induced pluripotency of adult human fibroblasts by genetic factors (34) or altered nuclear transfer (23) and the direct conversion of fibroblasts into functional neurons (27), we cannot exclude the possibility that BM-derived fibroblasts participate in the repair process or become reprogrammed in vivo in response to SCI microenvironment. In conclusion, autologous intrathecal delivery of minimally manipulated ABMCs lacks ethical or immunological concerns and might be an effective therapeutic strategy that significantly enhances the quality of life (2) of SCI patients and is relatively safe at up to 18 months posttransplantation when administered within regulated clinical trials. Long-term safety is being monitored and will be subsequently reported, and future multicenter randomized controlled trials are warranted.
Footnotes
Acknowledgments
We thank Drs. Joseph Bertino (the Stem Cell Institute of New Jersey), Robert DiPaola (the Cancer Institute of New Jersey), Pranela Rameshwar (New Jersey Medical School), George Heinrich (University of Medicine and Dentistry of New Jersey), and Mervat El Ansary (Cairo University School of Medicine) for helpful discussions. We thank Dr. S. R. Pine (The Cancer Institute of New Jersey) for assistance with flow cytometry. We also appreciate the excellent work of all of the Physicians, Physical Therapists, Nurses, Nurse Assistants and Technicians at the Central Hematology Laboratory, Department of Clinical Pathology, Faculty of Medicine, Cairo University; Department of Physical Medicine, Rehabilitation and Neurosurgery at Faculty of Medicine, Al-Azhar University; and Armed Forces Center for Physical Medicine, Rehabilitation, and Rheumatology, Al-Agouza, Cairo, Egypt. This work was generously supported by the Cancer Institute of New Jersey, the Foundation of the University of Medicine and Dentistry of New Jersey, Cairo University, the Military Medical Academy of Egypt, and the Egyptian Academy for Scientific Research and Technology. H.E.S. is a founder and stockholder, and W.A.E. and H.G. are collaborators of Celvive, Inc., a company that has licensed the cell therapy technology from UMDNJ. All of the other authors declare no conflicts of interest. We thank the New Jersey Health Foundation for the funding of the follow-up studies for this trial through Celvive, Inc.
