Abstract
The aim of this study was to evaluate the effects of intraperitoneal transplantation of encapsulated human hepatocytes on liver metabolism and regeneration of mice with acute liver failure. Primary human hepatocytes were immortalized using lentiviral vectors coding for antiapoptotic genes and microencapsulated using alginate-polylysine polymers. In vitro, immortalized human hepatocytes showed low, but stable, synthetic and catabolitic functions over time, when compared to primary hepatocytes. In vivo, mice with acute liver failure and transplanted with encapsulated immortalized human hepatocytes had a significantly improved survival and biochemical profile, compared to mice transplanted with empty capsules. Serum levels of cytokines implicated in liver regeneration were lower in mice transplanted with hepatocytes compared to mice receiving empty capsules. This decrease was significant for IL-6 and HGF at 3 h. Measurement of liver regeneration showed no significant difference between mice transplanted with hepatocytes compared to control groups. Intraperitoneal transplantation of encapsulated immortalized hepatocytes significantly improved survival of mice with acute liver failure by providing metabolic support and without modifying liver regeneration. The lower levels of cytokines implicated in liver regeneration suggest that the metabolic support provided by the encapsulated hepatocytes reduced the inflammatory stress on the liver and herein decreased the regenerative trigger on residual hepatocytes. These data emphasize that metabolic function and regeneration of hepatocytes are two distinct aspects that need to be studied and approached separately during acute liver failure.
Keywords
Introduction
Acute liver failure (ALF) is a devastating clinical syndrome occurring in approximately 2,000 cases/year in the US and is associated with a mortality reaching 80% (26). Treatment of acute liver failure generally remains supportive. Currently, orthotopic liver transplantation (OLT) is the only available therapy showing survival rates from 70% to 85% (37). However, OLT is not always possible due to the shortage of organ donors, and development of alternative methods is crucial.
A cell-based therapy could be a potential solution, as cellular transplantation using primary hepatocytes has been used successfully in rodent (24,29,40) and primate (3,42) models and in humans (13,14,25,38). Studies have shown that allogeneic hepatocyte transplantation positively influenced regeneration of native liver (2,4). The aim of this alternative therapy is to bridge patients with ALF until either a suitable liver allograft is obtained for transplantation or to promote the patient's own liver regeneration sufficiently to resume normal function.
Although human primary hepatocytes are an ideal source for hepatocyte transplantation, shortage of donor organs limits the availability of human livers for the use of this modality. It is therefore essential to find new sources of cells for transplantation. Transplantation of xenogeneic primary or immortalized hepatocytes (32) instead of whole liver or primary human hepatocytes has been considered as a possibility, allowing unlimited availability of liver cells to treat liver diseases (8).
Immune responses are still major barriers for successful cell and organ allo- and xenotransplantation. To protect allo- or xenogeneic cells, semipermeable capsules using polymers have been developed to protect hepatocytes from immune attacks (8). The small pores of the capsules prevent cells and antibodies to cross, while oxygen and glucose can pass through the membranes and maintain metabolic functions. It is known that inflammatory responses to the capsules induce long-term fibrotic changes around the grafts (31). As a result, oxygen and nutrient exchanges progressively stop inducing death of encapsulated transplanted cells (8). During ALF, short-term metabolic support is sufficient to allow the native liver to regenerate.
The aims of the present study were to evaluate in vitro functions of isolated hepatocytes after immortalization and encapsulation, and in vivo the impact of their transplantation on the regeneration of native liver cells in a rodent model of ALF.
Materials and Methods
Animals
Eight- to 10-week-old male C57BL/6 mice (Charles River Laboratories, Les Oncins, France), weighing 25–30 g, were used as recipients. For some experimental groups, Sprague-Dawley rats (Charles River Laboratories), approximately 300 g of body weight, were used as hepatocyte donors. Animals were maintained in conventional housing facilities and experimental protocols were approved by the ethical committee of the Geneva University Medical School and by Geneva veterinary authorities.
Tissue Procurement
Human hepatocytes were obtained from surgical liver biopsies of patients undergoing segmental hepatectomies for liver tumors. At the start of the intervention, a wedge of macroscopically normal tissue (15–30 g) located within the part of the liver to be resected was excised, immersed in ice-cold phosphate-buffered saline (PBS), and carried to the laboratory (the protocol for the human studies was approved by the institutional ethics committee of the Department of Surgery and informed consent was obtained from the patients).
Isolation of Hepatocytes
Human hepatocyte isolations were performed using a two-step collagenase perfusion method as previously described (34,38,42). Viability was determined by trypan blue exclusion and cells were cultured in serum-free DMEM/F12 (Invitrogen, Basel, Switzerland) containing 1 × 10−6 M dexamethasone (Sigma-Aldrich GmbH, Basel, Switzerland), 1 × 10−8 M 3,3′-triiodo-l-thyronine, 1 × 10−8 M human insulin (Huminsulin, Lilly France S.A.S, Strasbourg, France), 5 μg/ml apotransferrin (Sigma-Aldrich GmbH), 15 mM HEPES, and penicillin (100 U/ml) and streptomycin (100 μg/ml) (Invitrogen). Primary rat hepatocytes were isolated from adult male Sprague-Dawley rats, as previously described (28). Cells were cultured in Williams E medium (Invitrogen), with 10% fetal bovine serum (Invitrogen), penicillin (100 U/ml), streptomycin (100 μg/ml) (Invitrogen), insulin (10 7 M) (Huminsulin, Lilly France S.A.S), dexamethasone (10−6 M) (Sigma-Aldrich GmbH), and EGF (25 ng/ml) (Sigma-Aldrich GmbH).
Immortalization of Human Primary Hepatocytes
Immortalization of human primary hepatocytes was generated by transduction with lentiviral vectors coding for SV 40 large T antigen, Bmi-1, and telomerase in collaboration with Dr. T. Nguyen and Prof. Trono (EPFL) (33).
Encapsulation of Human Hepatocytes
Primary and immortalized human hepatocytes (IHH) were encapsulated as previously described (28,31) (Fig. 1). Hepatocytes were centrifuged and mixed with sterile MOPS washing buffer solution (10 mM MOPS + 0.85% NaCl, Inotech, Dottikon, Switzerland) and with 1.5% sodium-alginate solution (Inotech). Encapsulation was performed using an encapsulation device (Encapsulator, Inotech), where the hepatocyte–alginate suspension flowed through a 200-μm nozzle and was mixed with a polymerization solution (10 mM MOPS + 100 mM CaCl2, Inotech) to hepatocyte beads. Poly-l-lysine (PLL) hydrobromide solution 0.05% (Sigma-Aldrich GmbH) was added to the hepatocyte beads to form a PLL-alginate membrane. Then the beads were immersed in a 0.03% alginate solution (Inotech) to form the outer alginate membrane. After washing, the beads were put into a depolymerization solution (50 mM Na3 citrate, 10 mM MOPS + 0.45% NaCl, Inotech) to dissolve the core alginate, transforming beads into alginate–PLL–alginate capsules of a mean diameter of 400 μm.

Cultured encapsulated primary (A) and immortalized human (B) hepatocytes at day 3. Scale bar: 400 μm.
Evaluation of Metabolic Function
Human hepatocytes (primary freshly isolated or immortalized both free and encapsulated) were cultured in serum-free DMEM/F12 medium (Invitrogen) and incubated at 37°C for 1 week. Culture medium was collected every day and stored at −20°C for assessment of albumin secretion by enzyme-linked immunosorbent assay (ELISA; Bethyl, Lucerne, Switzerland).
To analyze urea synthesis and drug catabolism, 5 × 104 hepatocytes were seeded in each well of a 24-well-plate and ammonium sulfate (0.56 mM), lidocaine (1 mg/ml), or diazepam (1 μg/ml) was added to culture medium at 1, 3, and 7 days of culture. After 6 h, culture media were collected and concentrations of each reagent and urea, a metabolite of ammonium catabolism, were measured. The measurements were performed by the clinical chemistry unit of Geneva University Hospital using Cobas INTEGRA 400 (Roche Diagnostic, Basel, Switzerland) and DxC 800 system (Beckman Coulter Inc, Brea, CA, USA).
Induction of Acute Liver Failure
A model of ALF has been established in our laboratory in C57/BL6 mice by acetaminophen administration (500 mg/kg, IP), followed 15 h later by a 30% hepatectomy. Briefly, under general anesthesia a median laparotomy was performed and the left lateral liver lobe was removed as previously described (12,15,16,43).
Hepatocyte Transplantation and Experimental Groups
Hepatocyte transplantation was performed immediately after hepatectomy. Free or encapsulated IHH and free primary rat hepatocytes were resuspended in 3 ml Hank's solution and injected using an 18-gauge needle into the abdominal cavity (40 × 106 hepatocytes per mouse).
The following experimental groups were defined: Group 1—no treatment (n = 12); group 2—transplantation (Tx) of empty capsules (n = 10); group 3—Tx of primary rat hepatocytes (n = 13); group 4—Tx of free IHH (n = 9); group 5—Tx of encapsulated IHH (n = 9).
Measurement of Total Bilirubin and Aspartate Aminotransferase
Total rodent bilirubin and aspartate amino transferase (AST) were analyzed on peripheral blood of mice by the clinical chemistry unit of University Hospital Geneva using DxC 800 system (Beckman Coulter Inc.), following the manufacturer's instructions.
Measurement of Human Albumin
Human albumin was quantified on peripheral blood of mice using Human Albumin ELISA Quantitation Set (Bethyl Laboratories, Inc., Texas, USA) at 24, 48, and 72 h after transplantation, following the manufacturer's instructions.
Quantification of Peripheral Blood Cytokines Levels During Liver Regeneration
Tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), transforming growth factor-β1 (TGF-β1) (R&D Systems, Minneapolis, MN, USA), and hepatocyte growth factor (HGF; Institute of Immunology, Japan) secretions were analyzed on peripheral blood at 4, 24, 48, and 72 h and 7 days after transplantation, following the manufacturers' instructions.
Native Hepatocyte Proliferation
Immunohistochemistry was used to analyze bromodeoxyuridine (BrdU) incorporation and proliferating cell nuclear antigen (PCNA) expression in order to evaluate the proliferation of hepatocytes at 48 and 72 h after transplantation. Immediately after sacrifice, liver were sampled, fixed in 10% buffered formalin, and paraffin embedded. For BrdU incorporation analysis, BrdU (200 mg/kg) was injected intraperitoneally 2 h before tissue sampling (6) and immunostaining was performed using the Zymed BrdU STAINING Kit (Zymed Laboratories, San Francisco, CA, USA) following the manufacturer's instructions.
Expression of PCNA was detected by indirect immunohistochemistry using peroxidase method (30). Liver sections were fixed in methanol/hydrogen peroxide 5% for 15 min and blocked in PBS containing 5% BSA for 30 min. Mouse monoclonal anti-PCNA primary antibody (clone PC10; Signet Laboratories, Inc., Dedham, MA, USA) was incubated at a 1:100 dilution in the blocking solution for 2 h, followed by three washes with PBS. Biotin-conjugated goat anti-mouse secondary antibody (DakoCytomation, Glostrup, Denmark) was incubated at a 1:250 dilution in blocking solution for 30 min. After three washes with PBS, sections were incubated with a streptavidin-biotinylated peroxidase for 30 min (Invitrogen) and the staining was exposed by 3′3-diaminobenzidine Enhanced Liquid Substrate System (Sigma). Finally, the sections were counterstained with hematoxylin. The proliferation index of PCNA- and BrdU-stained tissue was determined at 200x magnification and labeled nuclei were counted in five randomly chosen fields, which approximate 1,000 cells per section. Data were expressed as the percentage of PCNA- and BrdU-stained hepatocytes per total number of hepatocytes.
Survival Follow-up and Histology
In each group, mouse survival was followed and five animals were euthanized at regular intervals for histopathology analysis of liver tissue.
Statistical Analysis
Data analyses are presented as means ± SEM. For in vitro studies, statistical differences among three or more groups were examined by one-way ANOVA followed by Scheffé's post hoc test. For in vivo studies, differences between two groups were examined for statistical significance using parametric t-test. Survival curves were calculated by Kaplan-Meier method and analyzed with chi-square test. Quantitative variation was considered significant at value of p < 0.05. Statistical analysis was performed using STATISTICA software (STATISTICA 5.5 Software for Windows, Statsoft Inc., Tulsa, OK, USA).
Results
In Vitro Evaluation of Metabolic Function
Over the first week of culture of free or encapsulated primary human hepatocytes, albumin concentration in the culture medium continuously decreased, suggesting a decreased of albumin synthesis (Fig. 2A). Urea concentration was also decreased (Fig. 2B), and ammonium, lidocaine, and diazepam concentrations in the culture media were increased (Fig. 2C–E), suggesting a decreased drug catabolism of free primary human hepatocytes. Decreased urea concentrations, and increased ammonium, lidocaine, and diazepam concentrations were also observed in culture media of encapsulated primary human hepatocytes (Fig. 2F–I) for the first week of culture. Free or encapsulated IHH produced initially lower quantities of albumin (Fig. 2A) and urea (Fig. 2B, F) and showed higher ammonium, lidocaine, and diazepam concentrations in their culture media, suggesting a decreased of their drug catabolism compared to primary human hepatocytes (Fig. 2C–E, G–I). In contrast to primary human hepatocytes, these concentrations remained unmodified during the first week of culture. These data indicated that human hepatocytes can be isolated, immortalized, and encapsulated and still maintain biological functions in vitro.

Metabolic functions of primary human hepatocytes (HH) and immortalized human hepatocytes (IHH), free or encapsulated over 1 week of culture. (A) Albumin secretion in culture medium of free HH and encapsulated HH (n = 3) and free and encapsulated IHH (n = 3). Primary human hepatocytes showed a continuous decrease of albumin secretion whether IHH (free or encapsulated) showed a lower but stable albumin secretion. Drug catabolism analyzed by urea synthesis (B, F) and ammonium clearance (C, G), and lidocaine (D, H) and diazepam (E, I) clearance was evaluated over the first week of culture of free (B–E) and encapsulated (F–I) HH and IHH. The values reported in (C–I) represent ammonium sulfate, diazepam, and lidocaine clearance of the culture media. Thus, an increase of drug concentration over time represents a decrease of hepatocyte catabolism. Free and encapsulated HH showed a decreased drug catabolism characterized by a decreased urea synthesis and increased concentration levels of ammonium, lidocaine, and diazepam in culture medium due to a decreased clearance. In contrast, free and encapsulated IHH showed lower, but stable, urea production and drug clearance. HH, primary human hepatocytes; IHH, immortalized human hepatocytes; D, day. ∗p < 0.05, HH versus free IHH; &p < 0.05, HH versus encapsulated IHH; §p < 0.05, encapsulated IHH versus HH.
In Vivo Studies
ALF in mice was achieved by combination of an overdose of acetaminophen and 30% hepatectomy, resulting in a survival rate of 20% in untreated animals (group 1) (Fig. 3). In mice transplanted with empty capsules (group 2) or free rat hepatocytes (group 3), survival rate remained unmodified (23% for both groups) (Fig. 3), compared to untreated mice. In group 4, Tx of free IHH increased survival rate to 37%, but did not reach statistical significance (Fig. 3). In group 5, Tx of encapsulated IHH significantly increased survival rate to 48% (p < 0.05) (Fig. 3).

Survival rate of mice with ALF untreated (open square; n = 13) and transplanted with empty capsules (open triangle; n = 10), free primary rat hepatocytes (open circle; n = 13), free IHH (filled diamond; n = 8), and encapsulated IHH (filled square; n = 9) were followed. After transplantation with empty capsule or free rat hepatocytes, mice with ALF showed a survival rate similar to untreated mice. In contrast, mice transplanted with encapsulated IHH showed a survival rate improved to 48% after 100 days (p < 0.05). ALF, acute liver failure.
To evaluate the effect of hepatocyte transplantation on metabolism of mice with ALF, we measured aspartate aminotransferase (AST) and total bilirubin serum levels of mice transplanted with empty capsules or encapsulated IHH after ALF induction. Serum levels of AST were measured at 3, 24, 48, and 72 h and 7 days after ALF induction and transplantation. Our results showed that the serum levels increased 3 h after induction of ALF and then decreased over 1 week to reach baseline levels of naive mice (Fig. 4). The serum levels of AST were not statistically different at any time point when comparing mice transplanted with encapsulated IHH, mice receiving empty capsules, or untreated mice. This result indicates that the liver injury was similar in all groups of mice. Total bilirubin levels were measured at 3, 24, 48, and 72 h after ALF induction and transplantation (Fig. 5). Our results showed an increase of total bilirubin in both groups from 3 h after induction of ALF. This increase tended to be less pronounced 3 h after ALF induction and was significantly lower 24 h after ALF induction in mice transplanted with encapsulated IHH compared to mice receiving empty capsules (Fig. 5).

Serum levels of aspartate aminotransferase (AST) of mice with acute liver failure (ALF) either untreated (hatched bars; n = 5), transplanted with empty capsules (black bars; n = 5), or transplanted with encapsulated IHH (white bars; n = 5) at 0, 3, 24, 48, and 72 h and 7 days after ALF induction. The serum levels of AST increased in animals 3 h after induction of ALF. This increase was similar at all time points in all mice. IHH, immortalized human hepatocytes.

Serum levels of total bilirubin of mice with ALF transplanted with empty capsules (black bars; n = 5) and encapsulated IHH (white bars; n = 5) at 0, 3, 24, 48, and 72 h after ALF induction. The serum concentration of total bilirubin increased in animals 3 h after induction of ALF. This increase was less pronounced at various time points in mice transplanted with encapsulated IHH than in mice receiving empty capsules. This difference was statistically significant at 24 h after ALF induction (3.5 ± 0.71 μmol/L vs. 6.3 ± 2.5 μmol/L in mice transplanted with encapsulated IHH vs. mice transplanted with empty capsules, respectively). ALF, acute liver failure; IHH, immortalized human hepatocytes.
Transplanted hepatocyte function was also evaluated by detection of human albumin in peripheral blood of mice transplanted with encapsulated IHH (Fig. 6). We observed no human albumin detection in mice transplanted with empty capsules. In contrast, human albumin was detected in mice transplanted with encapsulated IHH with decreasing secretion between 24 and 48 h but then the secretion remained stable between 48 and 72 h.
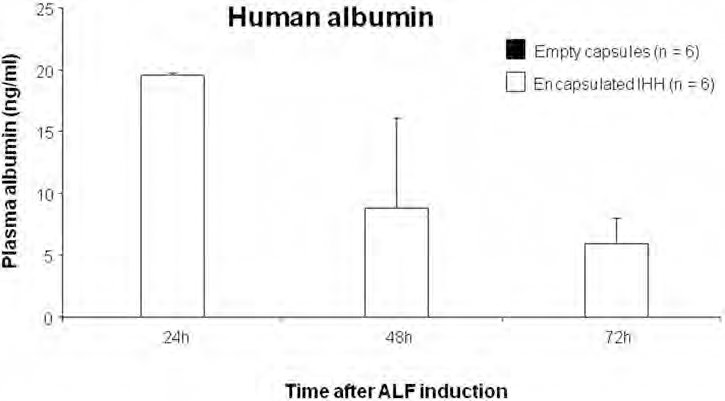
Serum levels of human albumin in mice with ALF transplanted with empty capsules (black bars; n = 5) and encapsulated IHH (white bars; n = 5) at 24, 48, and 72 h after ALF induction. Human albumin could not be detected on peripheral blood of mice with ALF transplanted with empty capsules any times after ALF induction. After transplantation with encapsulated IHH, human albumin was detected on peripheral blood of mice with ALF from 24 h (19.5 ± 0.37 ng/ml). Human albumin secretion from encapsulated IHH decreased progressively from 24 h until 72 h after transplantation with (8.84 ± 4.2 ng/ml) and (5.96 ± 2.01 ng/ml) at 48 and 72 h after transplantation, respectively.
Cytokine Levels During Liver Regeneration After Hepatocyte Transplantation
To evaluate whether transplanted human hepatocytes have an impact on mice liver regeneration, serum levels of the most important cytokines implicated in liver regeneration (i.e.; TNF-α, IL-6, HGF, and TGF-β1) were measured, by ELISA 3, 24, 48, and 72 h after ALF induction (Fig. 7). In control mice transplanted with empty capsules, there was an increase of TNF-α, IL-6, and HGF at 3 h after induction of ALF; in contrast, TGF-β1 began to increase at the end of liver regeneration between 72 h and 7 days. In mice transplanted with encapsulated IHH, serum level of TNF-α tended to be lower than that of mice receiving empty capsules, 3 h after ALF induction. Significant lower serum levels of IL-6 and HGF were measured in mice transplanted with encapsulated IHH compared to control mice transplanted with empty capsules 3 h after ALF induction (p < 0.05) (Fig. 7). Serum levels of TGF-β1 were similar between mice transplanted with empty capsules and encapsulated IHH at any times after ALF induction.

Serum levels of tumor necrosis factor-α (TNF-α) (A), interleukin-6 (IL-6) (B), hepatocyte growth factor (HGF) (C), and transforming growth factor-β1 (TGF-β1) (D) of mice with ALF transplanted with empty capsules (black bars; n = 5) and encapsulated IHH (white bars; n = 5) at 0, 3, 24, 48, and 72 h after ALF induction. Serum levels of TNF-α, IL-6, and HGF were lower in mice transplanted with encapsulated immortalized human hepatocytes (IHH) compared to mice Tx with empty capsules 3 h after ALF induction with a significant difference only measured for IL-6 (B) and HGF (C) levels (p < 0.05). No significant difference was detected for TNF-α and TGF-β 1.
Liver Regeneration
To complete analysis of liver regeneration of transplanted mice, BrdU incorporation and PCNA expression in native liver tissue were carried out 48 and 72 h after ALF induction (Figs. 8 and 9). PCNA is a cell cycle nuclear protein expressed throughout cell cycle and BrdU is a thymidine analogue, which is incorporated into DNA during DNA synthesis. The incorporation of BrdU and the expression of PCNA have been described to correlate with the degree of cell proliferation (30). BrdU incorporation (Fig. 8A–E) was similar in mice transplanted with encapsulated IHH and with empty capsules (n = 5) at 48 h (20.1% vs. 19.5%, respectively) and 72 h (8.4% vs. 8.9%, respectively) after ALF induction. In the same way, PCNA expression levels (Fig. 9A–D) were similar in mice transplanted with encapsulated IHH and with empty capsules at 48 h (22% vs. 19.6%, respectively) and 72 h (7.3% vs. 7.9%, respectively) after ALF induction. The proliferation index is expressed as the percentage of PCNA- and BrdU-stained hepatocytes per total number of hepatocytes.

Bromodeoxyuridine (BrDU) incorporation in native liver cells of mice with ALF transplanted with empty capsules (black bars, n = 5) or encapsulated IHH (white bars, n = 6) at 48 and 72 h after ALF induction. (A–D) Representative micrographs of immunohistochemistry detection of BrDU+ cells within tissue sections of native liver from mice with ALF transplanted with empty capsules (left panels, A, C) or encapsulated IHH (right panels, B, D) 48 h (A, B) or 72 h (C, D) after ALF induction (original magnification 200x). (E) Proliferation index expressed as the percentage of BrdU-stained hepatocytes per total number of hepatocytes. Our data showed no significant difference between mice transplanted with encapsulated IHH compared to mice receiving empty capsules (20.1 ± 3.1% vs. 19.6 ± 6.1% at 48 h and 8.4 ± 1.6% vs. 8.9 ± 3.4% at 72 h, respectively). Scale bar: 200 μm.

Proliferating cell nuclear antigen (PCNA) expression in native liver cells of mice with ALF transplanted with empty capsules (black bars, n = 5) or encapsulated IHH (white bars, n = 6) at 48 and 72 h after ALF induction. (A–D) Representative micrographs of immunohistochemistry detection of PCNA+ cells within tissue sections of native liver from mice with ALF transplanted with empty capsules (left panels, A, C) or encapsulated IHH (right panels, B, D) 48 h (A, B) or 72 h (C, D) after ALF induction (original magnification 200x). (E) Proliferation index expressed as the percentage of PCNA-stained hepatocytes per total number of hepatocytes. Our data showed no significant difference between mice transplanted with encapsulated IHH compared to mice receiving empty capsules (22 ± 4.9% vs. 19.6 ± 6.2% at 48 h and 7.3 ± 2.1 vs. 7.9 ± 2.4 at 72 h, respectively). Scale bar: 200 μm.
Discussion
Temporary support of terminal liver diseases by hepatocyte transplantation is a promising alternative to whole liver transplantation. However, major limiting factors of this therapy are scarcity of donor livers for hepatocyte isolation and the inability to isolate an adequate number of transplantable hepatocytes. A possible alternative for primary human hepatocyte transplantation is to use immortalized hepatocytes that can be grown in culture, stored by cryopreservation, and exhibit differentiated liver functions (36). These cells allow to perform cell transplantation within hours with a sufficient quantity and adequate quality.
In our study, we tested the in vitro function of primary human hepatocytes after isolation, immortalization, and encapsulation. Over 1 week in culture, a decreasing albumin and urea synthesis, as well as drug catabolism (ammonium, diazepam, and lidocaine), was observed for primary human hepatocytes (Fig. 2). These results are in accordance with several studies showing that free primary hepatocytes are difficult to maintain in culture (7,35). Interestingly, fresh and encapsulated IHH showed low albumin secretion and drug catabolism, as well as urea synthesis, indicating that immortalization of human hepatocytes modified their metabolic functions, but these remained stable over 1 week in culture compared to primary hepatocytes. Our data confirm that human hepatocytes can be isolated, immortalized, and encapsulated and still maintain biological functions in vitro for at least 1 week as previously described (9,20–23,27).
As expected, intraperitoneal Tx of empty capsules did not modify the survival rate of mice with ALF compared to untreated mice. Intraperitoneal Tx of free xenogeneic hepatocytes (primary rat hepatocytes or IHH) did not significantly increase survival rate of mice with ALF compared to untreated mice, suggesting destruction of these free cells by immune responses.
In contrast, Tx of encapsulated IHH significantly increased survival rate of mice with ALF. This effect was associated with improved metabolic functions such as albumin synthesis and bilirubin conjugation estimated by a reduction of total bilirubin level in blood. Our results indicate that encapsulated IHH provided life-supporting liver-specific metabolic functions until native liver regeneration was achieved. Our data are in accordance with previous studies showing improvement of survival rate and biochemical parameters (hyperammonemia) of animals (rat, pig) with ALF after intrasplenic or intraportal transplantation of free IHH (17–19,41). However, in these studies, the authors used immunosuppressive regimens to protect donor cells from immune responses. The negative or positive effects of various immunosuppressive regimens on liver regeneration have to be considered and it is likely that patients with ALF would be at high risk to develop infectious complications or other serious side effects when exposed to these drugs. Thus, we preferred to use encapsulation instead of immunosuppression to protect xenogeneic hepatocytes.
We studied the serum levels of aspartate aminotransferase (AST) after induction of ALF in untreated mice, in mice transplanted with empty capsules and in mice with encapsulated IHH. Our results showed that the AST levels were similar in all groups of mice, meaning that the liver injury was similar. These data suggest that the transplantation of encapsulated IHH or empty capsules have no impact on the liver injury itself.
Primary human hepatocytes are the ideal cellular component for clinical transplantation, but these cells are of limited availability. It is difficult to identify human livers suitable for cell isolations, and development of a standardized and centralized method of cell isolation and conservation has not been performed yet. Up to now, human hepatocytes have been isolated from unused segments of donor livers obtained from split/reduced grafts, and from whole livers considered unsuitable for transplantation for reasons such as excessive fat, fibrosis, trauma, or prolonged ischemia. The quality and the quantity of primary human hepatocytes from explanted livers unacceptable for whole liver transplantation are difficult to purify and tend to have lower viability and functionality and are unlikely to be satisfactory for transplantation (36). Furthermore, primary human hepatocytes are difficult to maintain in culture and loose liver-specific functions within days. Consequently, the use of human hepatocytes for hepatocyte transplantation, at present, is a limited option. Thus, for these reasons, we have decided to concentrate our research on immortalized human hepatocytes.
Serum levels of TNF-α, IL-6, HGF, and TGF-β1 were lower in mice transplanted with encapsulated immortalized hepatocytes, compared to mice receiving empty capsules (p < 0.05 for IL-6 level and HGF level at 3 h after transplantation). The lower levels of these different cytokines have also been described in other studies using hepatocyte transplantation or bioartifical liver treatment (BAL) to treat ALF (5,39). The reduction in the serum levels of cytokines did not reflect a reduced liver injury, as shown by the similar AST levels in mice transplanted or not (Fig. 4). Thus, the lower levels of TNF-α, IL-6, HGF, and TGF-β1 could be explained by an increase of clearance of these cytokines by the transplanted IHH.
Native liver regeneration of mice with ALF transplanted with encapsulated IHH analyzed by BrdU incorporation and PCNA expression was not significantly different than that of mice transplanted with empty capsules. These data indicate that encapsulated hepatocytes transplanted intraperitoneally did not stimulate native liver regeneration. Previous studies have shown that intrasplenic transplantation of syngeneic or allogeneic hepatocytes (free or encapsulated) in rat model of cirrhosis or ALF improved liver regeneration (1,2,4,10). Our different results may be explained by the use of xenogeneic immortalized hepatocytes in our experiments, in contrast to syngeneic or allogeneic hepatocytes. We found that transplantation of encapsulated IHH decreased the secretion of proinflammatory cytokines TNF-α and IL-6 as well as the secretion of HGF in the first phase after ALF induction (3 h). These cytokines have been described to play a role in liver regeneration rendering hepatocytes into a state of replicative competence immediately after liver injury (11).
Therefore, our data suggest that the metabolic support provided by the encapsulated hepatocytes reduced the inflammatory stress on the liver and herein decreased the regenerative trigger on residual hepatocytes. It is also possible that transplanted hepatocytes produce factors that are inhibitory to liver regeneration rather than stimulatory.
In conclusion, intraperitoneal transplantation of encapsulated immortalized hepatocytes significantly improved survival of mice with ALF, provided life-supporting liver-specific metabolic functions, and decreased secretion of cytokines implicated in liver regeneration, without modifying native liver regeneration. These data emphasize that metabolic function and regeneration of hepatocytes are two distinct aspects that need to be studied and approached separately during acute liver failure.
Footnotes
Acknowledgments
This study was funded by M.D.–Ph.D. scholarship No. 323500-115048/1 from the Swiss National Science Foundation to A.S., research Grant No. 3100A0-109888 from the Swiss National Science Foundation to L.B. and C.G.G. We gratefully acknowledge all the staff of the Cell Isolation and Transplantation Center and Nadja Mayer-Perriraz for their technical support. The authors declare no conflicts of interest.
