Abstract
Transplantation of allogeneic adrenal chromaffin cells demonstrated the promise of favorable outcomes for pain relief in patients. However, there is a very limited availability of suitable human adrenal gland tissues, genetically well-matched donors in particular, to serve as grafts. Xenogeneic materials, such as porcine and bovine adrenal chromaffin cells, present problems; for instance, immune rejection and possible pathogenic contamination are potential issues. To overcome these challenges, we have tested the novel approach of cell reprogramming to reprogram human bone marrow (BM)-derived mesenchymal stem cells (hMSCs) using cellular extracts of porcine chromaffin cells. We produced a new type of cell, chromaffin-like cells, generated from the reprogrammed hMSCs, which displayed a significant increase in expression of human preproenkephalin (hPPE), a precursor for enkephalin opioid peptides, compared to the inherent expression of hPPE in naive hMSCs. The resultant chromaffin-like cells not only expressed the key molecular markers of adrenal chromaffin cells, such as tyrosine hydroxylase (TH) and methionine enkephalin (Met-enkephalin), but also secreted opioid peptide Met-enkephalin in culture. In addition, intrathecal injection of chromaffin-like cells in rats produced significant analgesic effects without using immunosuppressants. These results suggest that analgesic chromaffin-like cells can be produced from an individual's own tissue-derived stem cells by targeted cell reprogramming and also that these chromaffin-like cells may serve as potential autografts for chronic pain management.
Keywords
Introduction
Transplantation of allogeneic adrenal chromaffin cells provides significant pain relief in patients with intractable cancer pain and in patients experiencing allodynia, a hallmark of neuropathic pain (1,11,13,25). There is often long-term pain relief without analgesic tolerance (1 year in humans) (24). Following adrenal chromaffin cell transplantation, application of exogenous opiates further alleviate pain without dosage escalation (25), that is, there was a stabilization of exogenous analgesic intake in these patients, strongly indicating that chromaffin cell grafts ameliorated the problem of opioid tolerance (12,13). These adrenal chromaffin cells release a “cocktail” of endogenous analgesic substances, including enkephalins, catecholamines, γ-amino butyric acid, indolalkylamines, and other neuropeptides (15,42). The analgesic effects of chromaffin cells can be partially reversed by intrathecal injection of either the opioid antagonist naloxone or the adrenergic antagonist phentolamine (9,32,33), suggesting that these analgesic effects are mediated by the opioids and catecholamines released by these cells. The antitolerance effects produced by the transplantation of adrenal chromaffin cells may be attributed to the synergistic action of the endogenous molecules released by these transplanted cells.
Although transplantation of allogeneic chromaffin cells demonstrates a viable modality of effective treatment for relieving pain and suffering in patients with the promise of ameliorating opioid tolerance, clinical practice has been hindered due to the limited availability of suitable human adrenal gland tissues. Mature chromaffin cells are postmitotic when they produce enkephalins and catecholamines. Expansion of these cells in culture is not possible. Xenogeneic materials, such as bovine and porcine chromaffin cells, have been extensively studied as potential alternative materials to human chromaffin cells. Transplantation of these xenogeneic cells into the spinal subarachnoid space produces antinociceptive effects on both Ad and C fiber-mediated responses in rodents and in nonhuman primates (16,23,32–34,40) with a gradual decline in analgesic efficacy that can be prolonged by administration of immunosuppressants (23). Although chromaffin cells themselves are not very immunogeneic and highly purified chromaffin cells may minimize immunorejection (4,20), these experimental results suggest that, in transplants, xenogeneic chromaffin cells elicit immunological host rejection and that immunosuppressive therapy is necessary for enhancing long-term graft survival to extend the analgesic effect of these transplants. In addition, there has been concern regarding pathogen contamination by these xenogeneic materials, such as bovine spongiform encephalopathy in bovine chromaffin cell transplantation. Thus, the use of xenogeneic chromaffin cells presents serious problems.
The emerging approach of cell extract-based cell reprogramming developed by Håkelien and Collas et al. (3,5,8) represents a novel technology to furnish the phenotypic characteristics of the target cells to the reprogrammed cells for therapeutic purposes. We previously (41) reported that bone marrow (BM)-derived human mesenchymal stem cells (hMSCs) hold the inherent gene expression of preproenkephalin (PPE), a precursor for enkephalin opioid peptides, such as methionine- (Met-) and leucine- (Leu-) enkephalins, and in culture are able to release a low basal level of Met-enkephalin, a major neurotransmitter that plays an important role in analgesia by activating opioid receptors (15,33). To address the clinical needs for new and safe alternatives to adrenal chromaffin cells, recently, we further reprogrammed these hMSCs with nuclear and cytoplasmic extracts of porcine chromaffin cells and examined the phenotypic development and functional changes of the reprogrammed cells. RT-PCR assays revealed that the expression of human PPE (hPPE) in the reprogrammed hMSC population was greatly enhanced compared to that of naive hMSCs (unprogrammed). Immunoblot analysis confirmed that secretion of Met-enkephalin in serum-free culture of reprogrammed hMSCs was significantly augmented compared to secretions released by naive hMSCs for the same number of cells under identical conditions. Immunocytochemical examination demonstrated strong immunoreactivity in the reprogrammed hMSCs for Metenkephalin and tyrosine hydroxylase (TH), specific markers for adrenal chromaffin cells. By targeted cell reprogramming, we have successfully developed a new type of cells from the reprogrammed hMSCs, which demonstrates the key phenotypic and functional features of adrenal chromaffin cells and thus termed these cells “chromaffin-like cells” (37,38). Transplantation of these chromaffin-like cells into the spinal intrathecal space of animals produced significant analgesic effects on both Ad nociceptor- and C nociceptor-mediated responses in a rat thermal pain model. The analgesic effects lasted for 3 weeks without immunosuppression. Preliminary data from some of this work have been reported previously in abstracts (37,38).
Materials and Methods
Chromaffin Cell Isolation, Purification, and Culture
Sixteen male and female porcine (1 year old) adrenal glands were obtained from a local slaughterhouse and immediately placed in ice-cold Locke's buffer (154 mM NaCl, 5.6 mM KCl, 3.6 mM NaHCO3, 5.6 mM glucose, 5 mM HEPES, pH 7.4; all components are from Sigma, St. Louis, MO, USA) supplemented with 100 UI/ml penicillin, 100 mg/ml streptomycin, and 0.125 ml/ml fungizone (PSF; Sigma-Aldrich) for transport. Preparation of chromaffin cells was carried out using a method reported by us previously (39) with modifications. In brief, adrenal glands were manually perfused three times with warmed (37°C) Locke's buffer followed by digestion of connective tissue with 0.125% collagenase A (Boehringer Mannheim, Mannheim, Germany) in Locke's buffer for 3 × 5 min at 37°C. At the end of the digestion period, the chromaffin cells were isolated from the dissected medulla by mechanical dissociation. The harvested cells were filtered through a 70-μm nylon mesh (BD Falcon, Bedford, MA, USA) and then centrifuged at 150 × g for 10 min in Locke's buffer. The resulting cells were purified on 39.47% Percoll gradients (Pharmacia Biotech, Uppsala, Sweden) by centrifugation at 22,500 × g for 20 min. The portion of the gradients containing purified chromaffin cells was harvested by aspiration and washed three times by centrifugation at 150 × g in Locke's buffer. Freshly isolated chromaffin cells were suspended and plated in 75-cm2 culture flasks (Corning, Cambridge, MA, USA) containing Dulbecco's modified Eagle's medium/F-12 (DMEM/F12, 1:1; Gibco, Grand Island, NY, USA) supplemented with 10% (v/v) fetal bovine serum (FBS, Sigma) and antibiotics (PSF). These chromaffin cells were maintained at 37°C in a 5% CO2 humidified incubation chamber (Fisher, Pittsburgh, PA, USA) fed by replacing culture media twice per week and used for experiments within 4 weeks after culturing because mature chromaffin cells are postmitotic and do not survive longer in cultured conditions.
hMSC Culture
In total, four samples of hMSCs (three males, ages 22, 25, and 32; one female, age 50), which are negative for surface markers associated with hematopoietic cells [e.g., cluster of differentiation 11b (CD11b), CD33, CD34 and CD133 antigens], were obtained from Cambrex (Walkersville, MD, USA). hMSCs were cultured and expanded using the protocol previously developed in our laboratory (30,39,41). In brief, hMSCs were plated in 75-cm2 culture flasks at a concentration of 1 × 105 cells/cm2 and cultured in 20 ml of serum-supplemented growth medium consisting of DMEM, an antibiotic–antimycotic mixture (1:100, Invitrogen, Carlsbad, CA, USA), and FBS (Stem Cell Technologies, Vancouver, BC, Canada), incubated at 37°C in a 5% CO2 humidified incubation chamber, and fed by replacing half of the culture media twice a week. Cells were passaged by incubating with 0.05% trypsin–EDTA (Gibco) for 5 min at room temperature to gently release the cells from the surface of the culture flask after reaching about 80% confluency. Culture medium was added to stop trypsinization, and the cells were centrifuged at 350 × g for 5 min at room temperature, resuspended, and transferred into new culture flasks at a concentration of 1 × 105 cells/cm2 for continuous culture and expansion to reach a sufficient number of cells. hMSCs that underwent less than 10 passages were used for the experiments.
Reprogramming hMSCs with the Extracts of Porcine Chromaffin Cells
The technology for cellular extract-based cell reprogramming, originally reported by Håkelien and Collas et al. (3,5,8), was applied by us with some modifications (37,38).
Preparation of Porcine Chromaffin Cell Extracts. Cultured porcine chromaffin cells were counted and washed twice in PBS (Sigma) and in cell lysis buffer (20 mM HEPES, pH 8.2, 50 mM NaCl, 5 mM MgCl2, 1 mM dithiothreitol, and protease inhibitors; all from Sigma), sedimented at 400 × g, resuspended in 1 volume of cell lysis buffer, and incubated for 30 min on ice. Cell samples were then sonicated in 200 μl of aliquot on ice with a pulse sonicator (PowerGen 125, Fisher Scientific) in short pulses until all cells and nuclei were lysed and confirmed by microscopic observation. The lysate was centrifuged at 15,000 × g for 15 min at 4°C. The supernatant was aliquoted and stored in liquid nitrogen for later use. Before applying cell reprogramming, the protein concentrations of the cell extracts were determined (~30 mg/ml).
Permeabilization of hMSCs With SLO. hMSCs were resuspended from the culture, washed in Ca2+- and Mg2+-free PBS (Mediatech, Inc., Manassas, VA, USA), and centrifuged at 120 × g for 5 min at 4°C. The collected hMSCs were resuspended in aliquots of 1 × 105 hMSCs/100 μl of Ca2+- and Mg2+-free PBS in 1.5-ml tubes. Cell samples were permeabilized with streptolysin O (SLO; Sigma-Aldrich) at a final concentration of 200 ng/ml and incubated in an H2O bath at 37°C for 50 min with occasional agitation to mix the cells. The cell samples were then placed on ice, diluted with 200 μl of cold PBS, and sedimented at 150 × g for 5 min at 4°C.
Reprogramming hMSC With the Extracts of Porcine Chromaffin Cells. The reversibly permeabilized hMSCs (1 × 105) were resuspended in 100 μl of the extracts of porcine chromaffin cells in a 1.5-ml tube containing an ATP-regenerating system (1 mM ATP, 100 μM GTP, and 1 mM of each NTP, 10 mM creatine phosphate, 25 μg/ml creatine kinase) (Sigma, St. Louis, MO, USA). Cell samples were incubated in an H2O bath at 37°C for 1 h with occasional agitation. To reseal the membranes of hMSCs, 1 ml of DMEM containing 2 mM of CaCl2 (Sigma) and antibiotics were added to the tube and incubated at 37°C for an additional 1 h. Finally, CaCl2-containing DMEM was replaced by fresh DMEM with 10% FBS, and reprogrammed hMSCs were transferred to a 24-well plate (BD, Franklin Lakes, NJ, USA) at a concentration of 1 × 104 cells per well or into a 75-cm2 culture flask at a concentration of 1 × 105 cells/cm2 and incubated at 37°C in a 5% CO2 humidified incubation chamber (Fisher). Twenty-four hours later, the floating cells were removed. The reprogrammed hMSCs grew well in culture and were expanded continuously, fed by replacing half of the culture media twice per week, and underwent regular cell passages when confluence was reached. Cells between passage 2 (1 week after cell reprogramming) and passage 4 (2 weeks after cell reprogramming) were used for experiments, and the viability of chromaffin-like cells was examined by trypan blue (Lonza, Walkersville, MD, USA), exclusion to be ≥95% at the time point before each experiment.
RT-PCR
The expression of hPPE genes in the population of chromaffin-like cells was analyzed by RT-PCR 1 week (passage 2) after cell reprogramming. Naive hMSCs with the same number of passages served as controls. RNAs from the cells were isolated using TRIzol reagents (Invitrogen) according to the manufacturer's protocol and treated with RNAase-free DNAase (Promega, Madison, WI, USA). The concentration of RNA was quantified by absorbance at 260 nm. RT-PCR was performed using a SuperScript One-Step RT-PCR with Platinum Taq (Invitrogen) by means of specific primer sets for gene hPPE (forward: 5′-AC ATCAACTTCCTGGCTTGCGT-3′ and reverse: 5′-GCT CACTTCTTCCTCATTATCA-3′) and β-actin (forward: 5′-GACAGGATGCAGAAGGAGAT-3′ and reverse: 5′-TT GCTGATCCACATCTGCTG-3′). RT-PCR products were quantified using the Qgel 1D program (Stratagene, Cambridge, UK) and expressed as hPPE/β-actin (an internal control) ratio. In total, three independent experiments were performed for this experiment. Statistical analysis was performed using Student's t tests at a significance of p < 0.05.
Immunoblot
The production and secretion of Met-enkephalin opioid peptides in chromaffin-like cell culture was examined at 1 week (passage 2) following cell reprogramming using an established protocol of immunoblot assay as we performed previously (41). Cultured hMSCs with the same number of passages served as controls. In brief, cultured cells were resuspended from cell culture flasks and transferred onto a 12-well culture plate (BD) containing serum-supplemented growth medium at a concentration of 1 × 105 cells/well. After recovering overnight, the serum-supplemented culture medium was replaced by serum-free culture medium. Twenty-four hours later, the medium was collected and purified with YM-30 microcon (Millipore Corp., Bedford, MA, USA). Purified medium samples (200 μl for each) obtained from chromaffin-like cells and naive hMSCs were applied to Hybond ECL nitrocellulose membranes (Amersham Life Science, Piscataway, NJ, USA) using the Slot Blot Hybridization Manifold (GENEMate, Kaysville, UT, USA). The membranes were blocked with 3% normal donkey serum (Jackson ImmunoResearch, West Grove, PA, USA) in PBS containing 0.5% Tween 20 (PBST; Sigma) for 2 h at room temperature and then incubated with a specific Met-enkephalin rabbit antibody (1:600, ImmunoStar Inc., Hudson, WI, USA) overnight at 4°C. After washing three times with PBST, the membranes were incubated with anti-rabbit IgG peroxidase-linked species-specific whole donkey antibody (1:3,000, Amersham Biosciences, Piscataway, NJ, USA) for 90 min at room temperature. The membranes were washed with PBS, incubated with an ECL Plus detection reagent for 5 min, and then exposed to Hyperfilm ECL (Amersham Biosciences). The films were developed and scanned by a computer. The quantification of immunoblot band density was assessed by densitometric analysis using the NIH image program (ImageJ, NIH, Bethesda, MD, USA). Data were expressed as the mean±SEM of four independent experiments. Student's t tests were performed to compare the levels of Met-enkephalin released by chromaffin-like cells with the levels released by naive hMSCs for the same number of the cells under the same condition of the serum-free cultures. Significance was set at p < 0.05.
Immunocytochemistry
Fluorescence immunocytochemical staining was performed on cultured chromaffin-like cells 2 weeks (passage 4) post-cell reprogramming. Naive hMSCs served as controls. Cell samples in eight-well culture chambers (Thermo Scientific, Swedesboro, NJ, USA) were washed with PBS and fixed in 4% paraformaldehyde (Sigma) in PBS (pH 7.4) for 20 min at room temperature. To examine the proliferative potential of chromaffin-like cells, cell samples were incubated with 1 μM of bromodeoxyuridine (BrdU; Sigma) for 24 h and then treated with 2 N HCL (Fisher Scientific, Fair Lawn, NJ, USA) for 30 min. Following washing in PBS, cell samples were blocked in PBS buffer containing 0.1% Triton X-100 (Sigma-Aldrich) and 3% donkey serum (Jackson ImmunoResearch) for 30 min, followed by incubation with sheep anti-BrdU (1:500; Fitzgerald, Concord, MA), mouse anti-TH (1:300; Sigma-Aldrich), or rabbit anti-Met-enkephalin (1:600; ImmunoStar Inc., Hudson, WI, USA) antibodies overnight at 4°C. Then the cells were washed in PBS and incubated with their corresponding secondary antibodies, including rhodamine (TRITC)-conjugated donkey anti-sheep IgG (1:200; Jackson ImmunoResearch), fluorescein isothiocyanate (FITC)-conjugated donkey anti-mouse IgG, and rhodamine (TRITC)-conjugated donkey anti-rabbit IgG antibodies for 2 h at room temperature in the dark. Finally, the cells were washed with PBS, counterstained with DAPI (Vector Labs, Burlingame, CA, USA), and viewed by immunofluorescence microscopy (Zeiss, Jena, Germany). The number of cells fluorescently immunostained by a specific antibody and the number of cell nuclei stained by DAPI were counted in five randomly selected microscopic fields at 200× magnification. In total, three independent experiments were performed, and about 50–100 cells were counted for each experiment. The ratio of the cells fluorescently immunostained by specific antibodies to the total number of counted cells was recorded.
Pain Behavioral Tests
All animal experiments were performed in accordance with federal guidelines for proper animal care and an approved Institutional Animal Care and Use Committee protocol. In total, 12 adult male Sprague–Dawley (SD) rats (250–300 g body weight; Charles River Laboratories, Hollister, CA, USA) were used. Foot withdrawal latencies were measured before cell transplantation using high (Ad nociceptor: 6.5°C/s) and low (C nociceptor: 0.9°C/s) rates of radiant heating on the dorsal surface of the feet according to the methods previously reported by us (16,17). The latencies from the onset of the stimulus to foot withdrawal responses were measured over 1 h at 10-min intervals. Chromaffin-like cells cultured 2 weeks (passage 4) after cell reprogramming were used. After the baseline for foot withdrawal responses was established, a single dose (1 × 105) of chromaffin-like cells in 20 μl of DMEM was injected intrathecally between the L4 and L5 lumbar vertebrae levels into the rats (n = 6) under anesthesia with pentobarbital (50 mg/kg, IP; Abbott Laboratories, North Chicago, IL, USA). We have observed previously that transplantation of this amount of chromaffin-like cells is appropriate to produce optimal analgesic responses in rats (37). Control rats (n = 6) received injections of the same amounts of naive hMSCs. Immunosuppressants were not used for cell transplantation. Foot withdrawal latencies were remeasured 1 week following cell transplantation and then weekly at 1-week intervals until latencies returned to baseline. Foot withdrawal latencies were expressed as the mean±SEM. Data from baseline (before cell transplantation) and response latencies at different time points after cell transplantation were compared and analyzed using t tests followed by Bonferroni post hoc correction. Significance was set at p < 0.05.
Results
hPPE Gene Expression of Chromaffin-Like Cells
One week after cell reprogramming with the cellular extracts of porcine chromaffin cells, RT-PCR was performed to examine the expression of hPPE. The molecular size of the RT-PCR product for the hPPE gene fragment was 425 bp. As expected, naive hMSCs demonstrated a low level of inherent hPPE gene expression. The chromaffin-like cells generated from the reprogrammed hMSCs showed a significantly enhanced expression profile for the gene hPPE compared to that of naive hMSCs (p > 0.01) (Fig. 1), suggesting that cell reprogramming further increases the expression of hPPE genes in the population of reprogrammed hMSCs.

The expression of hPPE gene in the chromaffin-like cells was analyzed by RT-PCR at 1 week (passage 2) after cell reprogramming. Naive human mesenchymal stem cells (hMSCs) with the same number of passages served as controls. RT-PCR products (425 bp) were quantified using the Qgel 1D program (Stratagene, Cambridge, UK) and expressed as human preproenkephalin (hPPE)/β-actin (an internal control) ratio. In total, three independent experiments were performed. Statistical analysis showed that hPPE expression of chromaffin-like cells (A) is significantly upregulated compared to that of naive hMSCs (B) (*p < 0.01, Student's t test).
Met-Enkephalin Secretion of Chromaffin-Like Cells
In parallel to the time point examined for the expression of hPPE genes by RT-PCR (1 week post-cell reprogramming), the serum-supplemented culture medium for chromaffin-like cell and naive hMSC cultures was replaced by a serum-free culture medium. Twenty-four hours later, the medium was collected and purified for Met-enkephalin detection by immunoblot assays. As shown in Figure 2, the level of Met-enkephalin released by chromaffin-like cells was significantly augmented compared to that released by naive hMSCs for the same number of cells (1 × 105 cells/well) in serum-free cultures (p < 0.01). Although naive hMSCs were able to produce and release a low level of Met-enkephalin into the serum-free medium, augmented production and secretion of Met-enkephalin opioid peptides in chromaffin-like cells were consistently observed in each of the four independent experiments.
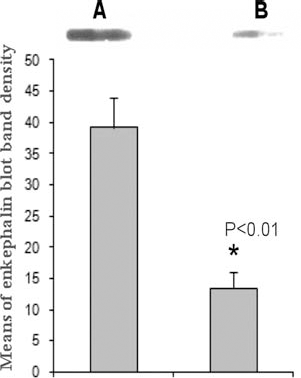
The secretion of Met-enkephalin opioid peptides of chromaffin-like cells (1 × 105 cells) in serum-free culture was examined by an immunoblot at 1 week (passage 2) after cell reprogramming. Naive hMSCs (1 × 105 cells) with the same number of passages served as controls. Twenty-four hours after culturing, the medium sample was collected and purified for immunoblot assay. The quantification of immunoblot band density was assessed by densitometric analysis using the NIH image program (ImageJ). Data were expressed as the mean±SEM of four independent experiments. Statistical analysis showed that the Met-enkephalin secretion of chromaffin-like cells (A) is significantly augmented compared to that of naive hMSCs (B) for the same number of cells under the same culture condition (*p < 0.01, Student's t test).
Immunocytochemical Examination of Chromaffin-Like Cells
Following cell reprogramming, morphological changes in hMSCs were observed within the first few days, that is, reprogrammed hMSCs became smaller and rounder. Five days later, the cells reverted to fibroblast-like shapes. These cells expanded in culture at a speed slightly slower (doubling time: about 84 h) in the first week and recovered to a normal dividing rate (doubling time: about 72 h) similar to that of naive hMSCs. Two weeks after cell reprogramming, immunocytochemical examination showed that most of the resultant chromaffin-like cells (≥90%) expressed a strong immunoreactivity for Met-enkephalin and TH (Fig. 3), specific cytoplasmic markers for adrenal chromaffin cells. Interestingly, BrdU-positive staining was detected in a subpopulation of chromaffin-like cells (data not shown), suggesting that some chromaffin-like cells may have retained a similar proliferative capability as that of hMSCs and could be expandable in cell cultures. These chromaffin-like cells remained stable phenotypes and expanded in cultures with an average cell doubling time of about 72 h up to 1 month, undergoing about eight passages. We did not maintain the culture of the cells past the 1-month time point after cell reprogramming.

Fluorescence immunocytochemical staining was performed on cultured chromaffin-like cells 2 weeks post-cell reprogramming (passage 4). Naive hMSCs with the same number of passages served as controls. Micrographs showed that chromaffin-like cells expressed strong immunoreactivity for tyrosine hydroxylase (a) and Met-enkephalin (c), while naive hMSCs expressed weak immunoreactivity for tyrosine hydroxylase (b) and Met-enkephalin (d). DAPI stained the nucleus of all cells. Consistent results were received in three independent experiments. Scale bar: 20 μm.
Analgesic Effects of Chromaffin-Like Cells
The analgesic effects of the chromaffin-like cells to a noxious thermal stimulus were investigated with the Ad and C fiber-mediated foot withdrawal latency tests in rats. Transplantation of chromaffin-like cells into the subarachnoid space of rats produced remarkable analgesic effects on both Ad and C fiber-mediated responses, which were evoked by a high and a low heating rate, respectively, and increased foot withdrawal latencies for 3 weeks in the absence of immunosuppression, with significant increases compared to the baselines of foot withdrawal latencies for 2 weeks (Fig. 4) (p < 0.01). The analgesic effects of chromaffin-like cells were greater for C fiber- than for Ad fibermediated responses. The maximum analgesic effects were observed in the first week following cell transplantation, and analgesic efficacy declined gradually with time. Control rats with naive hMSC transplantation showed similar response latencies to those measured previously in rats without cell transplantation (data not shown). No adverse effect was observed in the rats either with chromaffin-like cell or with naive hMSC transplantation.

Effects of intrathecal transplantation of a single dose (1 × 105) of chromaffin-like cells on foot withdrawal latencies evoked by low (C nociceptor: 0.9°C/s, squares) and high (Ad nociceptor: 6.5°C/s, diamonds) heating rates on the dorsal surface of the feet in rats (n = 6). Foot withdrawal latencies were measured before cell transplantation (baseline) and remeasured 1 week and then weekly at 1-week intervals following cell transplantation. Data from baseline and response latencies at different time points after cell transplantation were expressed as the mean±SEM and compared using t tests. Follow-up analysis was performed by Bonferroni post hoc tests. Results showed that transplantation of chromaffin-like cells increased foot withdrawal latencies evoked by both high and low heating rates for at least 3 weeks, with significant increases compared to baseline for 2 weeks (*p < 0.01, t tests and Bonferroni correction).
Discussion
Cellular extract-based cell reprogramming technology makes it possible to develop new types of cells by reprogramming one type of cells with extracts derived from a chosen targeted cell type to furnish reprogrammed cells with the phenotypic characteristics of the target cells (3,5,8). For example, incubation of a cell line, 293T fibroblasts, in the nuclear and cytoplasmic extracts of human T-cells resulted in the reprogrammed fibroblast cells taking on T-cell properties, expressing T-cell-specific surface molecules, and assembling the interleukin-2 receptor in response to T-cell receptor CD3 stimulation—a complex regulatory function (8). By using a similar approach, a related study demonstrated the induction of the pancreas-specific genes pancreatic and duodenal homeobox 1 (Pdx1) and insulin in rat primary fibroblasts treated with an extract of rat insulinoma cells (6). In the current studies, we reprogrammed hMSCs with nuclear and cytoplasmic extracts from porcine chromaffin cells, and the reprogrammed hMSCs demonstrated phenotypic and functional characteristics similar to those of true chromaffin cells.
As previous studies have shown, hMSCs are a subset of self-renewing multipotent stem cells and are capable of differentiating into various mesenchymal cell lineages, including bone, cartilage, fat, tendon, and other connective tissues (18,26,28). Several studies have reported that hMSCs can also transdifferentiate into a diverse family of cell types unrelated to their phenotypic embryonic origin, including muscle and hepatocytes (14,19,27), as well as neural cells (2,30,35). Recent studies, including ours, have revealed that hMSCs display an inherent gene expression of hPPE and spontaneously release a low level of Met-enkephalin in culture (29,41). These cells are preferred candidates for our targeted cell reprogramming not only because of their latent capability to produce analgesic substances and plasticity for multitransdifferentiation but also because they are relatively easy to isolate from an individual's own tissues and are able to be expanded in culture with a regular doubling time as well as low levels of senescence during repeated passages. We choose porcine adrenal chromaffin cells as reprogramming materials because these cells share characteristics with human adrenal chromaffin cells in many respects, such as synthesizing and releasing opioid peptides and other pain-inhibitory compounds, including enkephalins and catecholamines (16,40). Also, compared with bovine chromaffin cells, porcine chromaffin cells are more potent in producing analgesia as transplants (16). In addition, porcine chromaffin cells are considered a safe source of cells that are readily available in large quantities—in fact, pigs have been cloned and are now being bred for wholeorgan transplantation in humans (21,22).
Consistent with previous studies (29,41), our results demonstrate that naive hMSCs are able to express a low level of hPPE genes and to release a basal level of Met-enkephalins into a serum-free culture medium. However, chromaffin cell extract-based cell reprogramming significantly increases the expression of hPPE and the production and release of analgesic molecule Met-enkephalin, a neurotransmitter that plays a major role in analgesia by activating peripheral opioid receptors, in reprogrammed hMSCs (p < 0.01) (Figs. 1 and 2). As shown by our immunocytochemical examination, about 90% of the resultant chromaffin-like cells expressed strong Met-enkephalin and TH (Fig. 3), key markers of chromaffin cells. We did not examine the levels of catecholamines released by chromaffin-like cells in this study. However, TH is an enzyme controlling the rate-limiting step of catecholamine biosynthesis and is specifically found in the cytoplasmic matrix of cells containing catecholamines (31,36); positive immunoreactivity for TH may suggest that these chromaffin-like cells have the potential to produce catecholamines. More interestingly, BrdU immunoreactivity was detected in a subpopulation of chromaffin-like cells (30%), suggesting that these cells may have retained the proliferative properties of hMSCs and may make these cells even more valuable because generation of dividing cells that are expandable in culture would provide a sufficient quantity of such cells for targeted use. In practice, the analgesic potential and secretion activity of chromaffin-like cells for enkephalins can be further manipulated in continuous cultures and thus become more powerful by using targeted gene transfection and/or cell fusion techniques, as demonstrated in our previous studies (39,41). As revealed by RT-PCR, immunocytochemistry, and immunoblot analyses in our studies, reprogrammed hMSCs demonstrated similar phenotypic and functional characteristics of chromaffin cells by targeted cell reprogramming. Although the mechanism for cell reprogramming is not fully understood, it is suggested that nuclear and cytoplasmic extracts may contain regulatory components that mediate alterations in the gene expression profile of the target genome and promote the nuclear importation of nuclear regulatory components (7,10). It is likely that chromaffin cell extract-based cell reprogramming switches hMSCs from a mesenchymal program to a chromaffin-like program.
We further investigated the analgesic effects of chromaffin-like cells in vivo by spinal transplantation of the cells into the subarachnoid space of the rats. As shown in Figure 4, transplantation of chromaffin-like cells produced remarkable analgesic effects and significantly increased the foot withdrawal latencies mediated by both high (Ad nociceptor) and low (C nociceptor) heating rates (p < 0.01) (Fig. 4). The analgesic effects of chromaffin-like cells lasted for 3 weeks in rats without immunosuppression. Although we did not make observations on the secretion of chromaffin-like cells past the 1-week time point of cell reprogramming in our in vitro studies, the analgesic effects of chromaffin-like cells demonstrated in living animals suggest that these cells can continuously release analgesic substances in vivo after transplantation. In addition, a more robust analgesic potential of chromaffin-like cell grafts compared to that of the hPPE-transfected NT2 (human neuron-committed teratocarcinoma) cell grafts as reported in our previous studies was found (17), that is, a low number of chromaffin-like cells (1 × 105) produced similar analgesia effects to that of a high number of hPPE-transfected NT2 cells (1 × 107) in the same animal models. The duration of the analgesic effects produced by chromaffin-like cells was similar to that of the porcine chromaffin cells reported previously (16), with a time-dependent gradual decline in analgesic efficacy. Although we did not perform histological examination for the grafted cell fate in this study, these results suggest that host immune responses to the transplanted xenogeneic cells may occur. As demonstrated by our previous study (23), only few xenogeneic cells could be detected in the transplanted animals without immunosuppression, suggesting that administration of immunosuppressants is necessary to maintain long-term survival and long-lasting analgesic effects of xenogeneic cell grafts. Further studies are warranted to determine the survival rate, phenotypic stability, and secretion activity of chromaffin-like cells in vivo by using autologous cell transplantation in related animal pain models.
It is common knowledge that use of immunosuppressants can have severe side effects, including tumor formation, and should be avoided when possible. Transplantation of cells derived from individuals' autologous tissues into the same individuals would be safe and immunocompatible compared to xeno- and allotransplants. In practice, hMSCs can be derived from a patient's own tissues and not only could be prepared to become isogeneic through this cell reprogramming approach but also could avoid immunological rejection through autologous cell transplantation. Thus, autologous chromaffin-like cells could be the most desirable alternative to adrenal chromaffin cells for potential therapeutic purposes. Robust and long-lasting analgesic effects of autologous chromaffin-like cells are expected because these cells would be spared from immune responses, thereby improving the therapeutic efficacy of the transplanted cells.
Footnotes
Acknowledgment
This study was supported by the Charles H. and Bertha L. Boothroyd Foundation Award and DOD Grant PR100499P1 to T. Qu. The authors declare no conflict of interest.
