Abstract
Currently, islet cells are transplanted into the liver via portal vein infusion. One disadvantage of this approach is that it is not possible to adequately biopsy the islets in the liver to assess for rejection. Islet transplantation (Tx) into the gastric submucosal space (GSMS) can be performed endoscopically and has the potential advantage of histological evaluation by endoscopic biopsy. The aim of this study was to determine whether a representative allograft sample could be obtained endoscopically. We performed islet Tx into the GSMS in nonimmunosup-pressed pigs using simple endoscopic submucosal injection. Islets were transplanted at four sites. Endoscopic ultrasonography and biopsy of the transplanted islets at two sites by modified endoscopic submucosal dissection were carried out successfully in all pigs 5 days after islet Tx. Tissue obtained at both biopsy and necropsy (including full-thickness sections of the gastric wall around the sites of the remaining islets and biopsies) were examined by histology and immunohistochemistry to confirm the presence of the islet grafts and any features of rejection. Representative allograft sampling was successfully obtained from all biopsy sites. All biopsies included islets with insulin-positive staining. There was significant CD3+ and CD68+ cell infiltration in the islet masses obtained at biopsy and from sections taken at necropsy, with similar histopathological features. Endoscopic biopsy of islet allografts in the GSMS is feasible, provides accurate histopathological data, and would provide a significant advance if translated into clinical practice.
Introduction
Worldwide, there are millions of diabetic patients who might benefit from islet allotransplantation (alloTx). In the US alone, there are at least an estimated 2 million people with type 1 diabetes and a further approximately 20 million with type 2 diabetes. The number of people developling type 1 diabetes in the US each year is currently estimated to be more than a quarter of a million (22,41,44).
The liver is the most commonly used site for islet Tx. The Tx of deceased human donor islets into the portal vein (PV) in patients with type 1 diabetes has been followed by encouraging results (9,32,33,38). However, islet cell Tx into the PV is suboptimal for several reasons. (i) There is a significant incidence of morbidity from hemorrhage and/or thrombosis (28,42). (ii) There is an immediate loss of a large mass of islets (estimated at 60–80%) through an inflammatory response known as the instant blood-mediated inflammatory reaction (IBMIR) (3,6,21,37). (iii) In the majority of patients, there is a steady loss of normoglycemia over the succeeding 5 years, necessitating a return to insulin therapy; this loss could possibly be prevented if IBMIR could be avoided (as only a borderline number of islets survive the IBMIR) (30). (iv) Biopsy of the islets in the liver is not possible, and therefore, the cause of loss of islet function (e.g., acute rejection) cannot be fully assessed.
There are several theoretical reasons why the gastric submucosal space (GSMS) may be advantageous as a site for islet Tx (7). (i) It has a similar embryonic origin as the pancreas (10,24). (ii) It has a rich vascular supply of oxygen and nutrients. (iii) Like the pancreas, its venous drainage is into the portal blood stream (38). In addition, islet Tx into the GSMS would avoid (iv) IBMIR and (v) the complications of the percutaneous transhepatic catheter procedure necessary when islets are transplanted into the PV (39). Finally, (vi) the GSMS is easily accessible for endoscopic islet Tx (7), and it may be possible to biopsy allografts in the GSMS, which would be an immense advantage over islet Tx into the PV. A pilot report in a large animal model demonstrated that allotransplanted islets can engraft in the GSMS and that endoscopic islet alloTx can be carried out safely (7).
With regard to the biopsy of islets transplanted into the GSMS, endoscopic ultrasonography (EUS) is able to accurately visualize the submucosa (11,17,20,25). EUS can indicate the difference between a tumor and normal healthy tissue. The tumor can be biopsied by endoscopic submucosal dissection (ESD) and/or endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA). These methods could be used to identify and biopsy the islet mass after islet Tx into the GSMS.
The aims of the present study were to determine whether (i) the transplant sites could be identified by EUS and (ii) whether ESD allows submucosal sampling of the allograft for histological assessment.
Materials and Methods
Animals
Four Large White, Landrace, wild-type, female, non-diabetic, nonimmunosuppressed pigs (weighing 11.4±1.0 kg, aged 8 weeks) were used as recipients (Wally Whippo, Enon Valley, PA, USA), and two female pigs (approximately 250 kg, aged 3.5 years) were used as the source of islets. In all recipients, intravascular catheters (Saint-Gobain Performance Plastics, Valley Forge, PA, USA) were inserted into both right jugular veins for blood withdrawal and drug infusion. All received cefazolin (250 mg IV, 2× daily; APP Pharmaceuticals, LLC, Schaumburg, IL, USA) and famotidine (2.5 mg IV, 2× daily; APP Pharmaceuticals, LLC) throughout the course of the experiment.
All animal care procedures were in accordance with the US National Institutes of Health guidelines for the use and care of laboratory animals. All protocols were approved by the University of Pittsburgh Institutional Animal Care and Use Committee.
Donor Pancreatectomy and Islet Isolation
Under full inhalational anesthesia, the abdomen was opened in the midline. After perfusion of the pancreas in situ with Hank's balanced salt solution (Mediatech Inc., Manassas, VA, USA), pancreatectomy was performed without warm ischemia. Islet isolation and overnight culture were carried out as described previously (2). Approximately 350,000 IEQ were obtained from each donor pancreas.
Islet Viability
Islet viability was assessed by the double fluorescent calcein-AM and propidium iodide stain (Molecular Probes, Eugene, OR, USA) (1,2).
Endoscopic Islet Transplantation
Under full inhalational anesthesia, using an endoscope (GIF-2T160, Olympus Medical Systems Corp., Tokyo, Japan), endoscopic submucosal injection was performed using a disposable injection needle (needle diameter 23G, MN-200U-0423, Olympus). Specifically, the islets (15,000 IEQ/kg) were injected into the GSMS at four different sites in the posterior and anterior gastric antrum (Fig. 1). A three-way connector (MX4341L Medex, Dublin, OH, USA) was attached to the needle to facilitate delivery of the islets. Immediately before infusion, the islets were resuspended in 0.5 ml Connaught Medical Research Laboratories medium (CMRL)-1066 (Mediatech, Manassas, VA, USA) supplemented with 1% heat-inactivated donor pig serum. The injection of islets was preceded (to prime the catheter) with the same preparation but supplemented with 5% heat-inactivated donor pig serum. The islet injection was followed by the injection of 1 ml CMRL-1066 supplemented with 1% heat-inactivated donor serum to flush the needle and ensure complete delivery. Two islet masses were injected into the anterior wall and two into the posterior wall of the antrum. To provide some indication of the approximate sites of the islet transplants, two marking clips (HX-201LR-135, Olympus) were applied to the gastric mucosa approximately 20 mm on either side of each islet mass, one clip toward the minor curvature of the stomach and the other toward the major curvature.
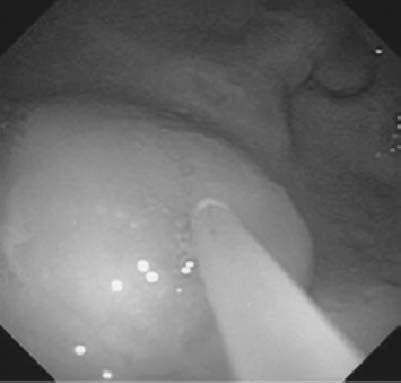
Endoscopic view of the submucosal bleb created as donor islets are injected into the gastric submucosal space (GSMS) of a recipient pig.
Follow-Up
Although the recipient pigs had not been rendered diabetic, monitoring of blood glucose was carried out twice daily with a True Track system glucometer (Home Diagnostics, Fort Lauderdale, FL, USA) to measure a fasting overnight level and a semifasting evening level.
Endoscopic Detection and Biopsy of Transplanted Islets
EUS and biopsy of the transplanted islets were carried out in all pigs 5 days after islet Tx (since recipient pigs were not administered any immunosuppressive therapy, and therefore, acute rejection was likely to develop rapidly). Under full inhalational anesthesia, a therapeutic endoscope (GIF-2T160, Olympus) was used to examine the sites of islet Tx, indicated by elevated lesions similar to submucosal tumors (Fig. 2A). EUS was then used to confirm the location of the four islet masses. After filling the lumen of the stomach with water, scanning with an ultrasonic miniature probe at 20 MHz (UM-3R, Olympus) showed solid masses of mixed hyperechoic/hypoechoic echogenicity in the submucosal layer (Fig. 2B). After the identification of the site of the islet mass to be biopsied, saline (2–5 ml) was injected around the islets to create a submucosal bleb, and the biopsy was carried out using a modified technique of endoscopic submucosal dissection (ESD). The raised mucosal bleb was incised with a Flexknife (KD-630L, Olympus) to allow access to the submucosal space. An IT Knife2 (KD-611L, Olympus) was then used to cut around the site of the graft using a PulseCut slow, 40 W (ESG-100, Olympus) (Fig. 3A). A biopsy of the graft was taken by snaring the dissected tissue (by electric cutting using a PulseCut slow, 40 W), including the submucosal tissue (Fig. 3B). The biopsied tissue was then removed with Grasping Forceps (FG-42L-1, Olympus) (Fig. 3C and D). Two of the four islet masses in each pig were biopsied using the modified ESD technique, and the histopathology of the graft was examined.
Euthanasia and Necropsy
The recipient pig was immediately euthanized by pentobarbital (200 mg/kg IV; Schering-Plough Animal Health Corp., Union, NJ, USA), and the abdomen was opened. The wall of the stomach was carefully examined to determine whether perforation or other complications had occurred. Gastric wall tissue around the two sites of biopsy was taken for histological examination to determine the exact depth of the biopsies. Tissue (full-thickness gastric wall) was also taken (that included the two islet masses that had not been biopsied) for the histopathological examination to compare with the information obtained from the histopathological examination of the biopsied islets.
Histopathologic Examination
Tissues obtained at both biopsy and necropsy were fixed in 10% formalin (Fisher Scientific, Fair Lawn, NJ, USA), and paraffin sections were stained with hematoxylin and eosin (H&E; Anatech Ltd., Battle Ceek, MI, USA), monoclonal mouse anti-human insulin antibody (clone HB125, Biogenex, San Ramon, CA, USA), polyclonal rabbit anti-swine glucagon antibody (Biogenex), polyclonal rabbit antihuman cluster of differentiation 3 (CD3) antibody (Dako, Glostrup, Denmark), monoclonal mouse anti-human CD20 antibody (clone L26, Dako), and monoclonal mouse anti-pig CD68 antibody (clone BA4D5, AbD Serotec, Oxford, UK). Unfortunately, we were unable to identify antibodies that stained for swine CD4+ and CD8+ cells. This was followed by blocking, as previously described (36).
Results
Endoscopic Islet Tx
Islets were successfully transplanted in four sites in the GSMS of the antrum in each of the recipient pigs. All pigs remained normoglycemic throughout the 5 days of follow-up.
Endoscopic Detection and Biopsy of Transplanted Islets
The four islet masses in each pig were identified clearly by direct endoscopic vision and confirmed by EUS (Fig. 2A and B). Each islet mass was approximately 10±3 mm in diameter (Fig. 2B). Biopsies of two of the four masses in each pig were obtained without complication. The tissue obtained from each site (including the islet mass) was approximately 20 mm in diameter (Fig. 3D).

Endoscopic profile of transplanted islet mass. (A) Endoscopic appearance of the gastric submucosa at site of islet mass (black arrow) 5 days after islet transplantation (Tx). (B) Endoscopic ultrasound identifying the islet mass (white arrows) in the GSMS 5 days after islet Tx.

Endoscopic view as a biopsy of an islet mass is taken 5 days after islet Tx. (A) After identification of the site of the islet mass to be biopsied, saline (2–5 ml) was injected to create a submucosal bleb. The raised mucosal bleb has been incised with a Flex knife to allow access to the submucosal space. An IT Knife2 was then used to cut around the site of the graft. (B) A biopsy of the graft is being taken by snaring the dissected tissue, including the submucosal tissue. (C) The biopsied tissue is being removed with forceps. (D) The removed specimen was 23 × 12 mm.
Euthanasia and Necropsy
At necropsy, there was no evidence of perforation or bleeding at the biopsy sites. Examination of the remaining stomach wall at necropsy clearly indicated the sites of endoscopic biopsy by the presence of small ulcers. The remaining islet masses (not biopsied) could also be identified visually.
Histopathology of the Biopsied Islets and the Remaining Islet Masses
Microscopic examination of all biopsies demonstrated that the tissue obtained included the gastric submucosa (not shown). H&E staining showed significant polymorphonuclear and mononuclear cell infiltration and fibrosis in the islet masses. In addition, early neovasculization of islets was confirmed (Figs. 4 and 5). Insulin-positive cells were few in areas of severe cell infiltration of biopsy or of necropsy samples (Figs. 4C and 5C). Infiltrates were largely CD3+ T-cells (Figs. 4D and 5D) and CD68+ macrophages (Figs. 4E and 5E), with a smaller number of polymorphonuclear neutrophils. However, in areas with mild cell infiltration, biopsied islets and the remaining islet masses (not biopsied) showed multiple insulin-positive (Fig. 6A and C) and glucagon-positive (Fig. 6B and D) islet cells. Although CD20+ cells could be observed in the mucosal layer (not shown), they were not detected within the islet masses. Appearances in all respects were very similar between biopsied islet masses (Figs. 4 and 6A, B) and islet masses examined after necropsy (Figs. 5 and 6C, D).

Histopathological appearance of an area of severe cellular infiltration of biopsies taken 5 days after islet Tx into the GSMS. (A) Islet mass in the GSMS [hematoxylin and eosin (H&E), 40×; scale bar: 200 μm]. (B) Polymorphonuclear and mononuclear cell infiltrate in islet mass (H&E, 100×; scale bar: 100 μm). (C) Few insulin-positive β-cells were identified in an area of severe cellular infiltration of the islet mass. Islets were infiltrated particularly by (D) cluster of differentiation 3-positive (CD3+) and (E) CD68+ cells (200×; scale bar: 50 μm).

Histopathological appearance of an area of severe cellular infiltration of the remaining nonbiopsied islets (at necropsy, in the same recipient pig as in Fig. 4) 5 days after islet Tx into the GSMS. (A) Islet mass in the GSMS (H&E, 40×; scale bar: 200 μm). (B) Polymorphonuclear and mononuclear cell infiltrate in islet mass (H&E, 100×; scale bar: 100 μm). (C) Few insulin-positive β-cells were identified whenever severe cell infiltration of the islet mass was present. Islets were infiltrated particularly by (D) CD3+ and (E) CD68+ cells (200×; scale bar: 50 μm). Similar histopathological findings were detected between biopsied islets (Fig. 4) and the remaining nonbiopsied islets.
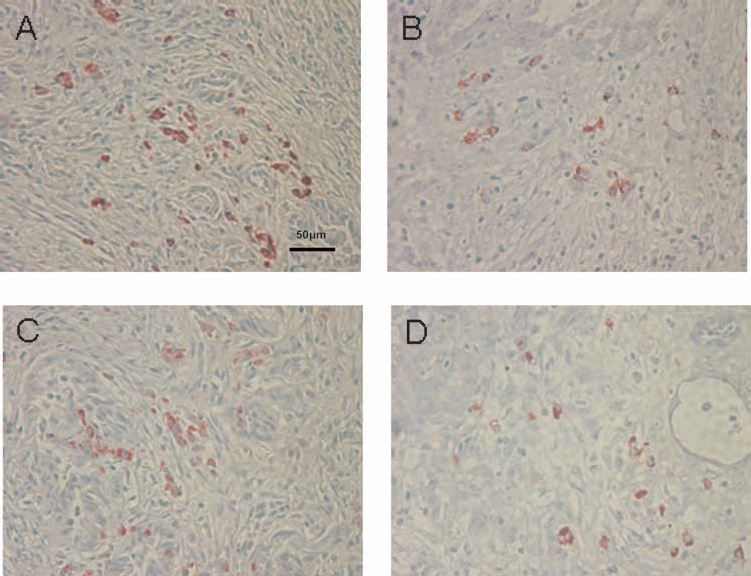
Histopathological appearance of an area of mild cellular infiltration of islet biopsies and nonbiopsied islets 5 days after islet Tx into the GSMS. (A) Insulin-positive β-cells and (B) glucagon-positive β-cells in an area of mild cellular infiltration in a biopsy of an islet mass in the GSMS. (C) Insulin-positive β-cells and (D) glucagon-positive cells in an area of mild cellular infiltration of an islet mass in the GSMS that had not been biopsied. Multiple insulin- and glucagon-positive cells were present in areas of mild cell infiltration compared to areas of severe cell infiltration (200×; scale bar: 50 μm).
Discussion
Tx into the liver through the PV is the current technique for islet alloTx (8,29,38). However, following islet Tx into the liver, it is not possible to histologically assess for rejection (38). Previous studies have established the potential value of the GSMS as a site for islet Tx (4,7,43), but no study has yet demonstrated the ability to successfully biopsy islets transplanted into this site. In a previous attempt, endoscopic biopsy using forceps was not successful in identifying any islets (43). The present report, therefore, is the first to describe a successful technique for endoscopic biopsy of islets in the GSMS.
EUS is the procedure of choice for evaluating sub-epithelial lesions of the gastrointestinal tract (11,17,20, 25). Therefore, after initial identification by direct vision through an endoscope, EUS was used to confirm the location of the islet masses in the GSMS. In preliminary experiments, we performed EUS after injection of saline into the GSMS to investigate whether a small mass (0.5 ml, 5 mm in diameter), which is similar in size to an islet mass, could be detected. EUS clearly detected 0.5 ml of saline injected into the GSMS (not shown). In the present study, scanning with a 20-MHz miniature probe confirmed the presence of the islet masses (10 mm diameter) in the GSMS. EUS showed mixed hyperechoic/hypoechoic masses in the submucosal layer at the site of islet Tx, which probably correlated with the presence of cell infiltration and fibrotic changes. The specific existence of islets could not be confirmed by EUS.
The injection of India ink has been used as a permanent marker of a specific site in the gastrointestinal tract that requires follow-up examination. After injection (1– 2 ml) into the submucosa, the ink has been reported to remain for up to 15 months in pigs (34) and 24 months or longer in humans (35). However, in our previous experiments, we demonstrated that India ink injected close to islets appeared to be detrimental with regard to islet viability and function both in vitro and in vivo. In in vitro experiments, we noticed that islets exposed to Indian Ink (short exposure, less than 15 min) prior to dynamic perfusion released lower amounts of insulin under basal as well as stimulated conditions, compared to untreated islets from the same donor (data not shown). In a mouse model of islet transplantations, diabetic immunodeficient recipients of islets exposed to Indian ink did not achieve or achieved delayed islet function following kidney capsule islet transplantation, in comparison to animals that received the same number of islets not exposed to Indian ink as control (data not shown). Furthermore, with time, the ink dissipated throughout the submucosa, obscuring identification of the original site of injection.
Following intraportal islet Tx, a histological assessment of the allograft cannot be reliably obtained. In one study, it was only possible to identify insulin-positive cells within the liver parenchyma in one of six insulin-independent patients (17%) using ultrasound-directed 18-gauge needle liver biopsies (36). EUS-FNA is a technique where intra- and extramural lesions can be sampled under direct endoscopic ultrasound guidance. However, EUS-FNA is generally performed using a smaller gauge needle (19–25 gauge) compared with that used for liver biopsy (15,40). Therefore, it may be difficult to obtain an adequate cytologic yield to assess for rejection.
Endoscopic mucosal resection (EMR) is currently an accepted method for removal of superficial low-risk gastrointestinal malignancies. A limitation of EMR is the piecemeal resection technique required in the case of large lesions, which leads to difficulty in making an accurate histopathological assessment (19,23). In a preliminary study by us, EMR provided only mucosa, rather than representative submucosa (data not shown).
Because of these limitations with cytological and/or histological sampling, we used a modified ESD technique to biopsy the islet grafts. ESD is frequently performed in Japan for the diagnosis and treatment of early phase gastrointestinal malignancies. This technique has been developed to enable en bloc endoscopic resection of large tumors (12,26,27,31). A previous report documented the feasibility of ESD in obtaining diagnostic and therapeutic yields of gastric submucosal tumors (14).
In the present study, we performed islet graft biopsy by the ESD technique using an IT Knife2 at the site of the islet mass, which was identified by both direct visual means and by EUS. Using the same knife, we dissected around the islet mass. However, to undermine the islet mass, we used a snaring technique (by electric cutting using a PulseCut slow, 40 W), which allowed the biopsy to be carried out more rapidly than by classical ESD. The dissection time using the ESD technique is longer than using the snaring technique. Cauterization of the tissue was less when using the snaring technique, thus, reducing potential injury to the islets.
Histological examination of the biopsies demonstrated insulin- and glucagon-positive islets infiltrated by CD3+ and CD68+ cells (with peripheral CD20+ cells); the appearances were identical to those in the nonbiopsied islet sites excised at necropsy. Early neovasculization of the islets was also seen. ESD, therefore, appears capable of obtaining representative allograft specimens. The islet grafts in the GSMS were fragmented, with no intact islets and considerable cellular infiltration. The features suggested that rejection of the grafts had developed within 5 days after Tx, correlating with the fact that the pigs had received no immunosuppressive therapy.
Potential complications of ESD include bleeding and perforation (12,26). Significant bleeding can result in termination of the procedure prior to obtaining the appropriate tissue sample. Bleeding might be problematic particularly if a punch biopsy without cauterization had been performed. In the present small study, however, ESD was completed without complication. The absence of perforation and bleeding was confirmed at necropsy immediately following ESD.
There are several reasons why the antrum was chosen as the site for Tx in this study. The stomach has a similar embryonic origin as the pancreas (10,24), and the submucosal space of the antrum is the most common ectopic site for pancreatic tissue (5,18). The upper and middle parts of the stomach may prove to be more difficult locations to perform ESD (16). Furthermore, the antrum wall is thicker than other gastric sites, increasing the safety of ESD.
In future studies, in order to mimic the clinical situation, diabetes will be induced by streptozotocin in the recipient pigs (13), and longer periods of post-Tx observation will be carried out. We are hopeful that endoscopic biopsy will provide evidence of allograft rejection, which correlates with changes in blood glucose levels and insulin requirements after islet alloTx. This could ultimately influence clinical decision making regarding immunosuppressant dosing and care of the post-Tx patient. However, it is yet to be determined whether islet transplant sites in the GSMS can be identified by endoscopy and/or EUS several weeks or months following Tx. In addition, a comparison of the techniques of ESD and EUS-FNA to investigate whether adequate samples of the islets are obtained to evaluate the histolopathological features will be necessary; ESD has not been accepted worldwide, as it is a difficult technique, and specific training is necessary.
In summary, this is the first study to demonstrate successful endoscopic sampling after islet Tx into the GSMS. EUS and ESD proved valuable techniques to allow this aim to be achieved. Further studies are warranted to confirm these preliminary results, specifically with the goal of obtaining biopsies after longer periods of observation post-Tx. We believe these approaches would provide a significant advance if translated into clinical practice.
Footnotes
Acknowledgments
This study was supported by a grant from the Olympus Corporation of the Americas Educational and Research Grant Committee (H.H.) and in part by NIH grant RO3 (AI096296, H.H.). Minoru Fujita, M.D., Ph.D., is the recipient of Kawasaki Sukenobu Memorial Fund of Kawasaki Medical School for Research Study, Japan, and Kawasaki Medical School Alumni Association Fund for Foreign Study, Japan. Eefje M. Dons, M.D., is the recipient of fellowships from the Ter Meulen Fund of the Royal Netherlands Academy of Arts and Sciences and the Stichting Professor Michael van Vloten Fund, The Netherlands. The authors thank Mr. Masahiro Ashizuka and Mr. Shoichi Matsui for technical help in performing EUS and ESD and Drs. Martin N. Wijkstrom, Massimo Trucco, David Ayares, and Fadi G. Lakkis for support and advice. Author's specific contributions: M.F. participated in the performance of the research and in the writing of article. E.M.D., R.B., C.L., G.K., and B.E. participated in the performance of the research and in the review of the article. G.J.E., J.H., and K.H. participated in the research design and in the review of the article. D.K.C.C. participated in the research design and in the writing of article. K.M.M. and H.H. participated in the research design, in the performance of the research, and in the writing of the article. The authors declare no conflict of interest.
