Abstract
Treatment of myocardial infarction (MI) with bone marrow-derived mesenchymal stem cells and recently also adipose-derived stem cells has shown promising results. In contrast to clinical trials and their use of autologous bone marrow-derived cells from the ischemic patient, the animal MI models are often using young donors and young, often immune-compromised, recipient animals. Our objective was to compare bone marrow-derived mesenchymal stem cells with adipose-derived stem cells from an elderly ischemic patient in the treatment of MI using a fully grown non-immune-compromised rat model. Mesenchymal stem cells were isolated from adipose tissue and bone marrow and compared with respect to surface markers and proliferative capability. To compare the regenerative potential of the two stem cell populations, male Sprague–Dawley rats were randomized to receive intramyocardial injections of adipose-derived stem cells, bone marrow-derived mesenchymal stem cells, or phosphate-buffered saline 1 week following induction of MI. After 4 weeks, left ventricular ejection fraction (LVEF) was improved in the adipose-derived stem cell group, and scar wall thickness was greater compared with the saline group. Adipose-derived as well as bone marrow-derived mesenchymal stem cells prevented left ventricular end diastolic dilation. Neither of the cell groups displayed increased angiogenesis in the myocardium compared with the saline group. Adipose-derived stem cells from a human ischemic patient preserved cardiac function following MI, whereas this could not be demonstrated for bone marrow-derived mesenchymal stem cells, with only adipose-derived stem cells leading to an improvement in LVEF. Neither of the stem cell types induced myocardial angiogenesis, raising the question whether donor age and health have an effect on the efficacy of stem cells used in the treatment of MI.
Introduction
Transplantation of mesenchymal stem cells (MSCs) in the treatment of myocardial infarction (MI) has been shown to improve left ventricular function in animal models (21,23,24,30,33). Paracrine effects of the transplanted cells plays a major role in preserving cardiac function (11,28,44), and induction of angiogenesis as well as a strengthened scar area are often reported (5,37). Preconditioning MSCs in a hypoxic environment prior to their use in the treatment of MI improves the effect of stem cell transplantation (20,40), possibly because of increased production of proangiogenic and antiapoptotic cytokines and improved stem cell engraftment (16,17,26,27,40).
MSCs from both bone marrow and adipose tissue have been used in animal infarction models (5,24). Although promising effects have been reported using adipose- derived stem cells (ASCs) compared to bone marrow mononuclear cells from mice (22), the potential of human ASCs needs yet to be determined in the treatment of MI. The MSCs used in many animal trials have been from young animals or of human origin derived from young healthy donors (22,32,36). Given the fact that the conducted human clinical trials have been using autologous transplantation of bone marrow-derived cells (10,19,29,43), the effect of using cells from elderly patients with cardiac ischemia needs to be elucidated. Age and health status of the donor are particularly important, as previous studies have demonstrated a reduced effect on left ventricular ejection fraction (LVEF) and angiogenesis when bone marrow-derived cells were derived from old donors and donors with cardio-myopathy (7,13). In view of the very recent findings that mesenchymal stem cells from adipose tissue have a significantly later onset of senescence than those from bone marrow (41), a head-to-head comparison between human ASCs and bone marrow-derived mesenchymal stem cells (BMSCs) from the same patient needs to be conducted. Furthermore, when xenotransplantations are used in these transplantation models, young immunosuppressed or athymic animals are most often used (5,15,18,21). The effect of immunosuppression on MSCs in the ischemic heart is unknown. However, an often used immunosuppressant, cyclosporine A, is known to have beneficial effects on the myocardial infarct (MI) size in an ischemia– reperfusion model due to its blocking effect on the mitochondrial transition pore (2,12). The use of immunosuppression might not be necessary as MSCs themselves have been shown to have immunosuppressive effects upon transplantation (3,6,38), and human MSCs have previously been used in an immune-competent xenotransplantation model (14,31,39). Hypoxic preconditioning of MSCs before their use in transplantation has previously been shown to enhance MSC survival and efficacy (16,35). In this study, we directly compared hypoxically preconditioned human ASCs and BMSCs from an elderly patient suffering from coronary atherosclerosis, in the treatment of MI, using a rat model. The cell therapy was given to an MI model in immune-competent, fully grown animals, without administration of immunosuppressant drugs.
Materials and Methods
The present study conformed to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health. Animal care was provided in accordance with the University of Aarhus guidelines and policies for the use of laboratory animals.
Mesenchymal Stem Cells
This study conforms to the Declaration of Helsinki. All protocols were approved by the local ethics committee in Copenhagen (H-D-2007-0057). Human MSCs were obtained from an 84-year-old male patient with verified coronary artery disease undergoing coronary artery bypass surgery at the Heart Center at Copenhagen University Hospital (Rigshospitalet, Denmark). The patient had a preserved LVEF, normal lung function, hyperlipidemia, and benign prostatic hyperplasia. Medication included a loop diuretic, potassium supplement, acetylic salicylic acid, a statin, a calcium antagonist (dihydropyridine), an angiotensin II receptor blocker in combination with hydro-chlorothiazide, a long-acting nitrate, and finasteride.
Aspiration of 200 ml subcutaneous adipose tissue from the upper abdomen and 40 ml bone marrow from the sternum was performed during bypass surgery. MSCs were isolated from adipose tissue and bone marrow as previously described (8,9,25). Briefly, adipose tissue was washed in phosphate-buffered saline (PBS, Life Technologies, Frederick, MD, USA) and incubated with col-lagenase buffer [0.6 PZ U/ml Collagenase NB6 (Serva Electrophoresis, Heidelberg, Germany) dissolved in Hanks' balanced salt solution (HBSS, Life Technologies)] at 37°C for 60 min. The digested tissue was filtered through a 60-μm Steriflip filter (Millipore, Copenhagen, Denmark) and centrifuged at 400 × g for 10 min, and the pelleted cells were resuspended in Ortho-Mune lysing solution (Hospital Pharmacy, Copenhagen, Denmark) in order to lyse erythrocytes. The cells were seeded, and after 24 h the medium was changed, disposing of all nonadherent cells. The bone marrow aspirate was anticoagulated with heparin (Hospital Pharmacy, Copenhagen, Denmark), and the mononuclear cells were separated with Lymphoprep (Medinor, Copenhagen, Denmark) using density gradient centrifugation. Contaminating erythrocytes were lysed in Ortho-Mune lysing solution. The cells were seeded, and after 24 h the medium was changed, disposing of all non-adherent cells. Growth media contained α-modified Eagle's medium (α-MEM), 1% penicillin, 1% streptomycin, 0.5% gentamicin (all from Invitrogen, Taastrup, Denmark), and 10% fetal calf serum (FCS; Europa Bioproducts Ltd., Cambridge, UK).
To evaluate whether an endothelial phenotype had been achieved, the ability to take up acetylated low-density lipoprotein (LDL) cholesterol labeled with DiI (1,1′-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine perchlorate) (DiI-Ac-LDL) (Biomedical Technologies, Stoughton, MA, USA) was examined. The cells were plated in 48-well plates (Costar, Acton, MA, USA), and after 24 h the medium was changed from standard growth medium to Endothelial Cell Growth Medium 2 (ECGM2) (PromoCell, Heidelberg, Germany). After 10 days of growth, the cells were incubated with DiI-Ac-LDL diluted 1:20 in the ECGM2 for 4 h, washed in media and PBS before fixation with 4% formalin (Bie & Berntsen, Herlev, Denmark), and then observed in a fluorescence microscope (Zeiss Axio Observer Z1; Zeiss, Göttingen, Germany).
Fluorescence-Activated Cell Sorting
Cells were harvested using 5–10-min incubation with acutase (PAA, Laboratories, Pasching, Austria) and incubated with antibodies at 4°C for 30 min. Antibodies were conjugated with allophycocyanin–Alexa 750 (APC-Alexa 750), fluorescein isothiocyanate (FITC), phycoerythrin (PE), or phycoerythrin–Texas Red (ECD). The MSCs were characterized based on a panel of antibodies including cluster of differentiation 13 (CD13)–ECD, CD31–FITC, CD34–APC, CD45–APC–Alexa 750, CD146–PE, CD90– FITC, CD73–PE, and CD166–PE (all from BD Bioscience, Albertslund, Denmark) and CD105–PE (R&D Systems Europe, Abingdon, UK). Analyses were done using 10,000 cells per sample on a Navios flow cytometer (Beckman Coulter, Copenhagen, Denmark), and data analysis was performed using FlowJo 7.2 software (TreeStar, Ashland, OR, USA).
Proliferation Assay
Proliferation capability was measured essentially as previously described (45). Briefly, ASCs and BMSCs were seeded at a density of 300/cm2 in 48-well plates and cultured at both 1% and 20% oxygen. On days 0, 2, 4, 6, 8, and 10, the cells were lysed in 200 μl 0.02% sodium dodecyl sulfate (SDS; Sigma-Aldrich, Brøndby, Denmark) in DNase-free water (Life Technologies), after which the cell number in the samples was determined using the PicoGreen dsDNA Quantitation Kit (Invitrogen). Aliquots of 100 μl cell lysate were mixed with 100 μl PicoGreen diluted 1:200 in 1× TE buffer, placed in a black microtiter plate (Thermo Fisher Scientific, Rockford, IL, USA), and incubated on a rocking shaker for 10 min. Fluorescence was measured using a Wallac 1420 Victor Multilabel Counter (PerkinElmer, Hvidovre, Denmark) with excitation at 485 nm and emission at 535 nm. A standard curve based on known Lambda DNA (CyQuant; Molecular Probes, OR, USA) concentrations was used to calculate sample DNA concentrations. The number of cells was calculated using a theoretical calculated value of 6.6 pg DNA per cell. Based on the exponential phase of the growth curve, the cell doubling time was calculated. The experiment was performed twice as two independent experiments, each in triplicate.
Apoptosis Assay
To determine if hypoxic culture affected the cells with respect to apoptosis, ASCs and BMSCs were seeded at a density of 104 cells/cm2 in 24-well culture plates (Corning, Amsterdam, The Netherlands) and cultured at 20% oxygen for 24 h, after which half the plates were transferred to 1% oxygen. After 24 h of additional culture, the medium was removed, and cells were washed twice with PBS. Then the cells were incubated with 2.5 mm/ml Hoechst 33342 (Invitrogen) to visualize nuclei and 1 ng/ml YO-PRO-1 (Invitrogen) to identify apoptotic cells. The stains were dissolved in PBS, and cells were incubated with the dyes at 37°C for 1 h. At the end of the incubation, the cells were washed twice with PBS, fixed in 4% formalin, and analyzed with fluorescence microscopy (Zeiss Axio Observer Z1). The level of apoptosis in each well was determined by evaluating four image fields at 20× magnification. The analysis was carried out through two independent experiments, each performed in triplicate.
Immunoblotting
ASCs were lysed in 50 mM Tris, 150 mM NaCl, 2 mM EDTA, 0.5% NP40, 0.5 mM phenylmethylsulfonyl flu oride, 50 mM NaF, 0.04% β-mercaptoethanol, 1 mM Na3VO4, and 1 Complete Mini Protease Inhibitor Cocktail (Roche Diagnostics, Hvidovre, Denmark). Protein concentrations in lysates were measured using the BCA Protein Assay Kit (Thermo Fisher Scientific). Samples of 40–60 μg total protein were denatured by heat, separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE; Life Technologies), and transferred to nitrocellulose membranes (Invitrogen, Taastrup, Denmark) using the iBlot transfer equipment (Invitrogen) according to standard protocols. Membranes were incubated overnight at 4°C with primary antibodies rabbit polyclonal to hypoxia-inducible factor 1 (HIF-1α; Abcam, Cambridge, UK) 1:500 and mouse monoclonal to β-actin (Sigma-Aldrich) 1:10,000. Membranes were then incubated 1 h at room temperature with horseradish peroxidase (HRP)-conjugated secondary antibodies rabbit polyclonal anti-mouse (Dako, Glostrup, Denmark) and goat polyclonal anti-rabbit (Santa Cruz Biotechnology, Heidelberg, Germany). PBS containing 5% skimmed milk (Sigma-Aldrich) and 1% Tween 20 (Sigma-Aldrich) was used for all dilutions. Finally, target proteins were visualized using enhanced chemiluminescence (Amersham ECL Plus; GE Healthcare Europe, Brøndby, Denmark), and signal acquisition was accomplished using a Kodak Image Station 4000 mm Pro (Carestream Health Denmark, Skovlunde, Denmark).
ELISA Analysis of Conditioned Media
ASCs were seeded in 12-well plates (Costar, Acton, MA, USA) with 10,000 cells/cm2. After 3 days, half of the cells were exposed to 1% oxygen for 24 h, and the other half of the cells were left at 20% oxygen. Before the last 24 h of culture, the medium was changed to α-MEM with 0.2% FCS. After 24 h of culture in serum-reduced medium, the medium was harvested and the cells were lysed in 0.02% sodium dodecyl sulfate in DNase-free water, after which the cell number in the samples was determined using the PicoGreen dsDNA Quantitation Kit. Fluorescence was measured with excitation at 485 nm and emission at 535 nm. The collected medium was analyzed using a commercially available ELISA vascular endothe-lial growth factor (VEGF) kit (R&D Systems Europe). The experiments were conducted twice as two independent experiments, each in quadruplicate.
Culture of Rat Aortic Rings
Rat aortic rings were derived from thoracic aortas asep-tically removed from four 8-month-old male Sprague– Dawley rats (Taconic Europe, Ry, Denmark) after eutha-nization using CO2. The aortas were trimmed, removing fibroadipose tissue. The vessels were cross-sectioned into rings of 2 mm in length, and the rings were washed in five consecutive baths of serum-free α-MEM to remove blood clots. The rings were placed in a 50-μl drop of growth factor-reduced Matrigel (BD Biosciences, Albertslund, Denmark) at the bottom of a 48-well plate (Costar) with the luminal axis parallel to the bottom of the culture well. The culture plate was incubated at 37°C for 30 min to allow cross-linking of the Matrigel, then 300 μl conditioned media from MSCs cultured at different oxygen concentrations were added to each well. After 4 days, the number of endothelial outgrowths from the aorta rings was counted using a phase-contrast microscope (Zeiss Axio Observer Z1). The experiments were conducted twice as two independent experiments.
Animals and Cell Transplantation
All protocols were performed in accordance with and approved by the Danish Animal Research Committee (2007/561-1337). Animals were anesthetized with a mixture of ketamine (50 mg/kg) (Intervet, Ballerup, Denmark), xylazine (5 mg/kg) (Bayer HealthCare, Animal Health, Copenhagen, Denmark), and acepromazine (0.75 mg/kg) (Pharmaxim, Helsingborg, Sweden), intubated and ventilated with a mixture of room air, oxygen, and isoflurane 0.5% (Baxter, Allerød, Denmark) at 70 breaths per minute (depth of anesthesia was tested applying a painful stimulus). The heart was exposed through a left-side thoracotomy, and MI was induced in 45 male Sprague–Dawley rats (5 months old, weight 450–550 g; Taconic Europe) by ligation of the left anterior descending artery. Throughout surgery, the animals were kept on a small animal temperature-controlled (rectal probe) heating plate (Föhr Medical Instruments GMBH, Seeheim, Germany). Mortality was 40% with all deaths occurring within the first 24 h after induction of the MI. After 1 week and prior to the first echocardiography, the 27 surviving rats were randomized to receive ASCs, BMSCs, or PBS. After echocardiography and before receiving cells, one animal in the ASC group and two animals in the PBS group were excluded due to an ejection fraction above 75%. Transplantation was done by intramyocardial injection using a 30-gauge needle (Hamilton, Reno, N V, USA). The cells were suspended in PBS and injected at three places in the peri-infarct zone with a total transplantation of 1 × 106 cells. All cells were cultured for 24 h in 1% oxygen prior to transplantation. After surgery, the animals received buprenorphine (0.1 mg/kg SC) (Schering-Plough, Ballerup, Denmark) twice daily for 3 days.
Echocardiography
Rats were anesthetized with a mixture of ketamine, xylazine, and acepromazine. Echocardiography was performed 7 and 35 days after MI using Vivid i (GE Healthcare) equipment fitted with a 10-MHz probe. Parasternal short-and long-axis pictures were obtained; from these pictures, the ejection fraction was calculated using the Teichholz formula. Rats have an echocardiographic measured ejection fraction of approximately 85% (4,42); therefore, based on echocardiography performed before treatment, only animals with an ejection fraction below 75% were included in the analysis, leaving the following groups: ASCs (n = 8), BMSCs (n = 9), and PBS (n = 7). All echocardio graphic analyses were performed by an experienced operator blinded to the treatment protocol using the EchoPAC software (GE Healthcare).
Histology
After the animals were sacrificed, the hearts were excised, fixed in formalin (10%) and after dehydration, embedded in paraffin. Infarction size was determined ste-reologically on five Masson trichrome (Bie & Berntsen)-stained sections from each animal. The diameter of the thinnest part of the left ventricular wall was measured in four sections and included in the analysis of scar wall thickness. Vascularization was quantified in the peri-infarct area using sections stained with α-smooth muscle actin antibody (Dako), stereologically determining the area covered by vessels in five 1 × 0.5-mm areas and one control area in the noninfarct area. In order to detect remaining human cells, the sections were incubated with human-specific anti-mitochondria antibody (Millipore, Copenhagen, Denmark).
BNP Analysis
Blood was drawn when the animals were sacrificed and, after separating serum, analysis for rat B-type (brain) natriuretic peptide-45 (BNP-45) was performed using a commercially available ELISA kit [AssayMax, Rat BNP-45 (rBNP-45) ELISA kit; Assaypro, St. Charles, MO, USA].
Statistics
Data were analyzed using SigmaPlot 11.0 software (Systat Software, Erkrath, Germany). When comparing more than two groups, a one-way analysis of variance (ANOVA) test combined with a Bonferroni post hoc test was used, and when comparing two samples, a Student's t test was used. As BNP and infarct wall thickness data were not normally distributed, a Kruskall–Wallis oneway analysis of variance on ranks test combined with a Dunn post hoc test was used. Normal distributed data are presented as means ± standard error of the mean (SEM); nonnormal distributed data are presented using box-plots denoting median and lower and upper quartiles. A two-sided value of p < 0.05 was considered statistically significant.
Results
Characterization of MSCs
Both ASCs and BMSCs were positive for the surface markers CD13, CD73, CD90, CD105, and CD166 and were negative for the surface markers CD31 and CD45. A small subpopulation of both ASCs and BMSCs was positive for CD34 (16%). The pericyte-related surface marker CD146 was expressed only in a subpopulation of ASCs (27%), whereas 87% of the BMSCs were positive for CD146 (Fig. 1A). MSCs stimulated with ECGM2 for 10 days gained the ability to take up DiI-Ac-LDL; hypoxic exposure did not affect this ability (Fig. 1B).
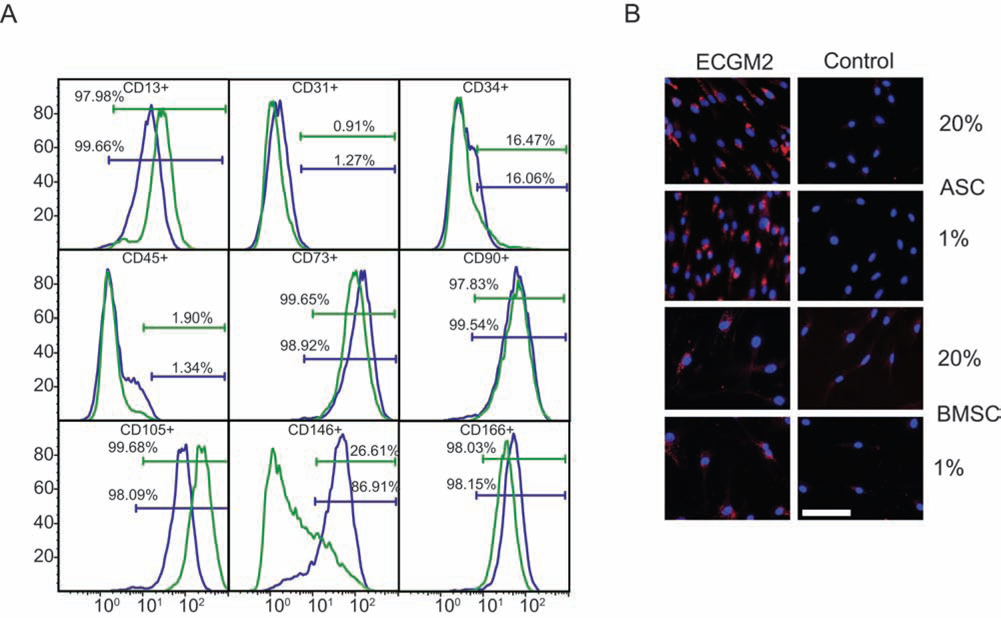
Flow cytometric characterization of cultured human ASCs and BMSCs at passage 2 cultured at 20% oxygen. (A) Representative histograms of positive surface markers. ASCs in green and BMSCs in blue. (B) Representative images of MSCs cultured 10 days in ECGM2 and incubated with DiI-Ac-LDL. Scale bar: 100 μm. ASCs, adipose-derived stem cells; BMSCs, bone marrow-derived mes-enchymal stem cells; ECGM2, endothelial cell growth medium 2; DiI-Ac-LDL, 1,1′-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine perchlorate-labeled acetylated low-density lipoprotein cholesterol; CD13, cluster of differentiation 13.
MSC Response to Hypoxia
The 24-h hypoxic exposure did not affect the frequency of apoptotic cells, which in normoxia and hypoxia was 5.4% and 3.9% for ASCs, respectively, and 3.7% and 2.5% for BMSCs, respectively. Statistical analysis did not reveal any significant difference between the oxygen conditions. After 24 h of hypoxic culture, ASCs extensively stabilized HIF-1α compared to ASCs in normoxic culture, while this was not evident for BMSCs (Fig. 2A). ASCs demonstrated a significantly shorter doubling time than BMSCs when cultured at 1% oxygen (Fig. 2B). Culturing the cells for 24 h at 1% oxygen led to a statistically significant increased VEGF secretion from both ASCs and BMSCs compared with cells cultured at 20% oxygen. BMSCs secreted statistically significant more VEGF per cell than ASCs both under 20% and 1% oxygen (Fig. 2C). Conditioned media resulted in a statistically significant increased angiogenic response in a rat aortic ring model when rings were incubated with conditioned media from ASCs cultured at 1% oxygen compared with both conditioned media from ASCs and BMSCs cultured at 20% oxygen, whereas there was no difference in angiogenic response when conditioned media from ASCs and BMSCs cultured at 1% oxygen was compared (Fig. 2D and E).

Response of human ASCs and BMSCs to hypoxic culture. (A) Detection of hypoxia-inducible factor (HIF)-1α stabilization by immunoblotting. (B) Proliferation capacity expressed as cell doubling times. (C) Vascular endothelial growth factor (VEGF) secretion in response to 24-h culture at 20% and 1% oxygen, respectively. (D) Angiogenic response from a rat aortic ring model incubated with conditioned media from human ASCs and BMSCs cultured at 20% and 1% oxygen. (E) Representative phase-contrast image of the angiogenic response from a rat aortic ring model. Scale bar: 200 μm. Values are represented as the mean and SEM. *p < 0.05.
Effect of MSCs on Myocardial Infarction
Echocardiographic measurements of LVEF were conducted prior to cell treatment (ASC: 54.7 ± 12.3%; BMSC: 61.1 ± 9.0%; PBS: 60.4 ± 8.8%, p = 0.40) and 4 weeks after cell treatment (ASC: 65.6 ± 15.2%; BMSC: 56.6 ± 21.1%; PBS: 58.1 ± 18.5%, p = 0.59) (Fig. 3A). The differences in LVEF before and after treatment were compared between the treatment groups, and no significant difference was found (p = 0.12) (Fig. 3B). Within the ASC group, there was a significant improvement in ejection fraction, whereas this was not evident for the BMSC and PBS groups (Fig. 3B). The left ventricular end diastolic diameter (LVEDD) was significantly increased in the PBS group (Fig. 3C), whereas the two cell-treated groups showed no significant LVEDD dilatation. None of the treatment groups demonstrated a significant increase in left ventricular end systolic diameter (LVESD) (Fig. 3D). BNP in serum 4 weeks after cell or PBS treatment did not differ between groups (Fig. 3E). Histologically, there was no significant difference in infarct size between the three treatment groups (Fig. 4A). The thickness of the scar at its thinnest part in four sections was measured, and wall diameter in the ASC group was greater compared to the PBS group (Fig. 4B). The degree of vascularization in the peri-infarct areas as well as the noninfarcted areas did not differ between groups (Fig. 4C). Using an antibody directed against human mitochondria, it was possible to detect remaining human ASCs or BMSCs in the hearts transplanted with MSCs (Fig. 4D), the detected cells were placed in groups of rounded cells with a high nucleus to cytoplasm ratio, and there were no morphological signs of differentiation.

Echocardiographic and BNP measurements of the effects of MSC transplantation in a rat model of myocardial infarction. (A) Time line of animal experiments (not drawn to scale) and representative echocardiographic pictures of the left ventricle in parasternal short-axis view and M-mode. (B) LVEF% before and after cell treatment. (C) LVEDD difference after–before. (D) LVESD difference after–before. (E) Comparative analyses of rat BNP using ELISA. ASCs (n = 8), BMSCs (n = 9), and PBS (n = 7). Values are represented as the mean and SEM (B, C, D) and as boxplots (E) showing lower quartile, median, and upper quartile. PBS, phosphate-buffered saline; ASCs, adipose-derived stem cells; BMSCs, bone marrow-derived mesenchymal stem cells; MSCs, mesenchymal stem cells; LVEF%, left ventricular ejection fraction %; LVEDD, left ventricular end diastolic diameter; LVESD, left ventricular end systolic diameter; BNP, brain natriuretic peptide; ELISA, enzyme-linked immunosorbent assay. *p < 0.05.

Histological examinations of rat hearts 4 weeks after treatment of myocardial infarction. (A) Left: Stereological quantification of infarct size using consecutive sections stained with Masson trichrome stain. Right: Masson trichrome-stained section displaying myocardial infarction (blue area). (B) Scar thickness measured in consecutive sections. (C) Left: Vascularization in infarction and non-infarction areas. Right: Myocardium with a-smooth muscle actin-stained vessels. Scale bar: 200 μm. (D) Myocardium with remaining donor cells indicated by arrows. Scale bar: 40 μm. ASCs (n = 8), BMSCs (n = 9), and PBS (n = 7). Values are represented as the mean and SEM (A, C) and as boxplots (B) showing lower quartile, median, and upper quartile. Whiskers denote 9th and 91st percentile, and outliers are shown with dots (*p < 0.05).
Discussion
In the present study, we demonstrate that ASCs from a more than 80-year-old patient with ischemic coronary disease ameliorated heart failure following MI in a rat model. The LVEF was improved during a 4-week follow-up period, and scar wall thickness was greater compared with a saline control group. In contrast, BMSCs from the same patient did not result in any significant improvement in these parameters. Our findings are in line with Mazo et al., who, by transplanting murine cells to an immuno-suppressed rat MI model, demonstrated that ASCs were superior to bone marrow mononuclear cells in the treatment of heart failure following MI (22).
We could not find an increased angiogenic response in vivo after cell treatment compared with PBS treatment. This is in contrast to the findings by Cai et al., who used human ASCs from a healthy 50-year-old donor in an athymic rat model of MI, injecting the cells 1 h after creation of the infarction (5). This group demonstrated an increased density of α-smooth muscle actin-positive vessels in the peri-infarct area 4 weeks after treatment. The present study used human ASCs and BMSCs from an 84-year-old patient suffering from coronary atherosclerosis in a nonimmunocompromised rat model with injection of cells 1 week after creation of the infarction and a follow-up period of 4 weeks. It is uncertain whether these differences in experimental setup and donor characteristics may explain the lack of angiogenic response after MSCs transplantation, but age and disease of the donor may influence the efficacy of MSCs. Thus, Fan et al. found that transplanted BMSCs from young donors generated a significantly better angiogenic response in recipient myocardium after MI compared with both BMSCs from elderly donors and control medium (7). This is in accordance with our findings showing lack of change in vascular density in the myocardium after transplantation of ASCs and BMSCs from an elderly patient suffering from coronary atherosclerosis. Furthermore, Fan et al. demonstrated that BMSCs from elderly donors were not able to improve LVEF compared with control medium (7). This supports our findings demonstrating no effect on LVEF after transplantation of BMSCs from the elderly patient suffering from coronary atherosclerosis. Heeschen et al. compared the angiogenic potential of the bone marrow mononuclear fraction from elderly patients suffering from ischemic cardiomyopathy with that of young healthy donors in an ischemic hindlimb model and found that cells from young healthy donors performed significantly better (13). This corroborates our findings demonstrating that neither ASCs nor BMSCs from an elderly patient suffering from coronary atherosclerosis generated a significant angiogenic response in the infarcted myocardium compared with control PBS.
Interestingly, we found that BMSCs secreted a significantly larger amount of VEGF into the culture medium than ASCs, but this conditioned medium did not result in a better angiogenic response in an in vitro rat aortic ring model. We even found a trend toward conditioned media from ASCs cultured at 1% oxygen being superior to conditioned media from BMSC cultured at 1% oxygen. This indicates that the VEGF concentration is not the sole regulator of the angiogenic response and that other secreted factors from the MSCs must be important in eliciting a widespread angiogenic response. This is in line with our previous findings, demonstrating that conditioned media from human ASCs in prolonged culture (13 days), compared with short-term culture (1 day) at 1% oxygen, contained significantly more VEGF but did not elicit a more extensive angiogenic response in a rat aortic ring model (26). These findings correspond with the fact that angiogenesis is a highly complex process involving a plethora of growth factors and different cell types, with VEGF being essential for the initiation of angiogenesis but far from being solely responsible for the extent of the angiogenic response (1).
We were not able to demonstrate any difference in stereologically quantified infarct size among the three treatment groups. An ASC treatment effect on infarction size could be hidden from this analysis given the fact that the echocardiographic LVEF measurements showed a trend toward the ASC group having the largest infarctions before cell treatment. Thus, it could be expected that the ASC group had the largest infarctions measured by stereology, but the groups demonstrated similar infarction sizes, and this could cover a de facto effect of ASC treatment reducing the infarction size to the level of that demonstrated in BMSC- and PBS-treated animals.
Our study comprises some limitations. Only one patient was used as donor of the MSCs; this excludes the possible effect of interdonor variability but also limits the applicability of the results to the general ischemic patient. Thus, while this study support the general observation that ASCs are at least as good and, in some cases, more effective than BMSCs in clinical applications (34), a larger clinical trial comparing ASCs and BMSCs is needed to confirm the results in this study. Also, in evaluating the cardiac function, a small animal magnetic resonance imaging scanner would have added strength to the conclusions. In order to avoid any bias from the operator, the analysis of the echo-cardiograms was performed by an experienced operator blinded to the treatment protocol. Finally, we used healthy nonimmunosuppressed animals in this study. It is possible that a host immune response could affect the outcome of stem cell transplantation. With these caveats in mind, there are several clinical implications of this study. First, that the use of hypoxically preconditioned ASCs in treating ische-mic disease seems to offer at least as efficient a therapeutic paradigm as that of BMSCs in terms of positive effects on cardiac function and remodeling. Furthermore, the use of ASCs compared with BMSCs will reduce the need for in vitro proliferation time as ASCs can be harvested in large numbers and with less discomfort for the patient. Also, while these data support a previous finding that ASCs are less prone to diminish in numbers and efficacy with aging compared with BMSCs, the characteristics of MSCs from elderly patients suffering from coronary atherosclerosis need to be determined.
In conclusion, hypoxically preconditioned ASCs from the elderly patient suffering from coronary atherosclerosis improved LVEF following MI, whereas this could not be demonstrated using BMSCs from the same patient. The observed effect was not based on an increased density of α-smooth muscle actin-positive vessels associated with ASC transplantation but could, to some degree, be related to remodeling of the scar tissue preventing left ventricular dilation.
Footnotes
Acknowledgments
This work was supported in part by grants from the danish Heart Foundation (08-4-R64-A2020-B584-22460, 07-4-B584-A1453-22364, and 10-04-R78-A2925-22599), Brødrene Hartmanns Foundation, the A.P. Møller Foundation for the Advancement of Medical Science, Købmand Sven Hansen and Hustru Ina Hansens Foundation, and the Danish Medical Association Research Fund. Jeppe Grøndahl Rasmussen was supported by a grant from the Faculty of Health Science, Aarhus University. The authors thank Bente Wormstrup, Helle Skjødt Møller, Simone Elkjær Riis, Louise Hansen, and Mette Skov Mikkelsen for technical assistance. The authors alone are responsible for the content and writing of the paper. The authors declare no conflict of interest.
