Abstract
Acellular nerve grafting is often inferior as well as an inadequate alternative to autografting for the repair of long gaps in peripheral nerves. Moreover, the injection method is not perfect. During the injection of cells, the syringe can destroy the acellular nerve structure and the limited accumulation of seed cells. To resolve this problem, we constructed a nerve graft by acellular nerve grafting. Bone marrow-mesenchymal stromal cells (BM-MSCs) were affixed with fibrin glue and injected inside or around the graft, which was then used to repair a 15-mm nerve defect in rats. The acellular nerve graft maintained its structure and composition, and its tensile strength was decreased, as determined by two-photon microscopy and a tensile testing device. In vitro, MSCs embedded in fibrin glue survived and secreted growth factors such as nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF). We repaired 15-mm Sprague–Dawley rat sciatic nerve defects using this nerve graft construction, and MSCs injected around the graft helped improve nerve regeneration and functional recovery of peripheral nerve lesions as determined by functional analysis and histology. Therefore, we conclude that supplying MSCs in fibrin glue around acellular nerves is successful in maintaining the nerve structure and can support nerve regeneration similar to the direct injection of MSCs into the acellular nerve for long nerve defects but may avoid destroying the nerve graft. The technique is simple and is another option for stem cell transplantation.
Introduction
The incidence of nerve injuries within 90 days of upper- or lower-limb trauma is 1.64% for patients undergoing surgical intervention (31). A range of surgical approaches have been used to repair different nerve injuries; nerve autografts by far remains the gold standard for peripheral nerve repair (14). However, the drawbacks of using autografts, especially those suitable for treatment of long nerve gaps, include donor site loss of function, formation of potential painful neuromas, structural differences between donor and recipient grafts, and the lack of autograft material. Therefore, finding alternative nerve guide conduits has increased (9,21).
Materials used in the construction of nerve conduits can be categorized as being derived from synthetic polymers or natural materials. Compared with synthetic polymers, natural materials such as extracellular matrix (ECM) molecules purified from tissue possess cell-adhesive or signaling domains, which may present a more biologically relevant microenvironment (13). Biologically derived nerve conduits from veins or muscle tissue have been used with varying degrees of success (7,25). Several absorbable nerve conduits such as collagen and synthetic polyester-based conduits are approved by the US Food and Drug Administration and Conformit Europe for clinical repair of peripheral and cranial nerves (21). Allografts present a viable solution to this challenge; however, the immune response of the host is a major consideration (26), necessitating the decellularization of the grafts prior to use (12). A number of groups have prepared cell-free (acellular) nerve segments with preserved basal lamina tubes using detergent extraction (11,28). Acellular nerve grafts (ANGs) retain the ECM and structural organization of the auto graft, thus presenting a more biologically relevant micro environment suitable for regenerating axons. Acellular nerve grafting is an alternative to autografting for repair of short gaps in peripheral nerves (28,39). However, the outcomes associated with the use of acellular nerve grafting are often inferior to that of autografts, particularly for long lesion gaps (29,34).
To enhance nerve regeneration and functional recovery in the peripheral nervous system, more efforts have been directed toward enhancing acellular nerve grafting with growth-stimulating neurotrophic factors or transplanted cells (1,16,18,27,29,34,38). Our previous research enhanced acellular nerve grafting with hepatocyte growth factor (HGF) and nerve growth factor (NGF) (18,38). Current efforts to use cytokines in clinical settings are limited by methods of administration. Neither bolus injection nor polymeric encapsulation is fully satisfactory. A high-dose bolus injection can lead to systemic or local toxicity. In addition, bolus injection requires multiple administrations due to diminishing bioavailability and therefore maybe costly. However, cell transplantation and incorporation into nerve conduit designs are alternative strategies to neurotrophic factor delivery. Transplanted cells may be tailored to secrete a cocktail of neurotrophic factors to further enhance regeneration (32). However, the injection method is not perfect. During the injection of cells, the syringe can destroy the acellular nerve structure and the limited accumulation of seed cells. Moreover, injecting cells is difficult with acellular grafting of small nerves.
Mesenchymal stromal cells (MSCs) derived from various sources have been found to elute NGF, brain-derived neurotrophic factor (BDNF), glial-derived neurotrophic factor (GDNF), and matrix metalloprotease 2 (MMP2), which degrades chondroitin sulfate proteoglycan (a molecule inhibitory to axonal growth) (6,33). Thus, MSCs may be exploited as a sort of miniature drug factory, releasing factors that result in the desired healing response in injured nervous tissue. Administration of stem cells to the sites of central nervous system injury may be a means of replacing damaged or diseased tissue. This strategy has been used in numerous sources of central nervous system injury (10). In addition, MSCs might exert their efficacy through indirect modulation of the cellular behavior of Schwann cells (SCs) (36).
In the present study, we attempt to fabricate a nerve graft containing regeneration characteristics, using MSCs fixed within fibrin glue and injected around an acellular nerve graft. First, we analyzed the structure and composition, as well as the tensile strength, of acellular nerve grafts. In vitro, we tested the compatibility of encapsulation in fibrin glue with MSCs and then neurotrophic factor release and bioactivity assay. Finally, we compared functional recovery and nerve regeneration of fibrin glue-containing MSCs injected around acellular nerve grafts and those injected into the acellular nerve to bridge a 15-mm sciatic nerve defect in Sprague–Dawley (SD) rats. These scaffolded cells have therapeutic potential in treating nervous system injuries in future studies.
Materials and Methods
Acellular Nerve Graft Preparation
Thirty adult male Wistar rats (Vitalriver, Beijing, China) weighing 250–300 g were killed by intraperitoneal injection with sodium pentobarbital (0.5 ml, 60 mg/ml). The sciatic nerves were excised, removed of peripheral fat and connective tissue, and treated with chemical detergents to become acellular allografts. Acellularization involved use of a previously developed protocol (28). Isolated sciatic nerves were submerged in room temperature distilled water that was replaced several times over a 2-h period. The nerves were then immersed in distilled water containing 3% Triton X-100 for 12 h and then immersed for 24 h in distilled water containing 4% sodium deoxycholate. The extraction procedure was agitated at room temperature and repeated. Finally, tissue segments were washed three times for 15 min each in a solution containing 10 mM phosphate and 50 mM sodium, sterilized for 12 h by use of 60Co (Academy of Military Medical Sciences, Beijing, China), and stored in the same solution at 4°C. All chemicals were purchased from Sigma (St. Louis, MO, USA), unless otherwise noted. All solutions were autoclaved or filter-sterilized before use.
Structure and Compositions of Acellular Nerve Graft
Prepared acellular nerve samples were analyzed by two-photon microscopy (Olympus FV1000MPE multi photon microscope, Olympus, Beijing, China) to detect tissue autofluorescence and second harmonic generation (SHG). Briefly, the acellular nerve was placed on an imaging dish and immersed in phosphate-buffered saline (PBS). Multiphoton excitation was from a titanium/sapphire laser (Maitai, SpectraPhysics, Santa Clara, CA, USA) centered at 780 nm and routed into the scanhead and through the sample objective. Fluorescence emission collected from the sample was detected in a nondescanned configuration with a cooled photomultiplier tube (PMT). Fluorescence emission in the spectral region of 500–650 nm was collected for detection of broadband autofluorescence from the acellular nerve. Data for SHG were collected with 860-nm excitation and a 400-nm bandpass filter in the nondescanned detector path. At each site, the z-stack was obtained with a z-interval of 1- and 200-μm total depths at 40×.
Tensile Testing of Acellular Nerve Graft
Acellular nerve grafts were loaded onto an ElectroForce 3100 Test Instruments (Bose Corporation, Framingham, MA, USA). To avoid slippage during testing, specimens were attached to the tensile tester by stainless steel hooks (Taigang Stainless Steel Co., Shanxi, China) with 4-0 silk sutures (Wuxi Medical Instruments, Wuxi City, China) and ethyl cyanoacrylate adhesive (Gorilla Super Glue, Cincinnati, OH, USA). The strain rate was set to 1%/s. Each specimen was stretched at a constant rate of 0.05 mm/s. The mean specimen length was 5.12 ± 0.44 mm. Each specimen was stretched to complete tensile failure. The data were included when the nerves burst at a location away from the points of connection. During testing, samples were kept moist by applying PBS to the nerve constructs. Normal sciatic nerves were used as controls.
MSC Preparation and Conditioned Medium
Primary cultures of MSCs were obtained from 10 male 4-week-old SD rats (Vitalriver). Briefly, rats were given a lethal dose of phenobarbital, and the tibias and femurs were removed. A 22-gauge needle filled with Dulbecco's modified Eagle's medium (DMEM) was used to flush out whole bone marrow. The recovered cells were then plated in DMEM supplemented with 10% fetal bovine serum (FBS) and penicillin–streptomycin in 25-cm2 tissue culture flasks (Costar, Corning, Corning, NY, USA). After 48 h, the nonadherent cells were removed, and the culture medium was completely replaced. MSCs were used at passage 2. An amount of 10 × 106 of MSCs was diluted with 500 μl fibrinogen plus potassium dihydrogen phosphate. One syringe was filled with this solution, and another was filled with 500 μl thrombin plus calcium chloride. The two syringes were connected to a Y-piece for injection.
To obtain conditioned medium, 5 × 105 of MSCs embedded in fibrin glue and MSCs alone were cultured in six-well plates for 1, 3, 7, and 14 days, when medium was exchanged for DMEM for 24 h. Conditioned medium was collected, filtered through a 20-μm syringe filter apparatus (Millipore, Billerica, MA, USA), and frozen immediately at −80°C. Nonseeded medium was the control medium.
MSC Viability in Fibrin Glue
MSCs embedded in fibrin glue (n = 4 per condition) were placed in the LIVE/DEAD double staining solution (Fluka, Buchs, Switzerland) according to the manufacturer's instructions for 1, 7, 14, and 21 days. After staining, live cells were labeled with fluorescence green by Calcein-AM, and dead cells were labeled red by propidium iodide, which bound to exposed DNA and cannot pass through a viable cell membrane. We obtained the percentage of green cell by counting under a fluorescence microscope (BX-51, Olympus).
Assessment of Neurotrophin Production and Bioactivity
At 1, 3, 7, and 14 days after incubation, medium samples were collected for quantification of neurotrophin release by ELISA (Boster, Wuhan, China, n = 4 per condition). Untreated medium and supernatant from nonencapsulated cells were used as controls. ELISA kits for NGF and BDNF were used according to the manufacturer's protocol (Boster). Average values below the lowest dilution above zero of the standard curve for each kit were considered nondetectable.
To detect bioactivity of conditioned medium from MSC-embedded fibrin glue, rat pheochromocytoma (PC12) cells (Shanghai Institute for Cell Research, Chinese Academy of Sciences, Shanghai, China) were cultured in Roswell Park Memorial Institute (RPMI)-1640 medium with 10% horse serum and 5% FBS. Cells were plated at low density into each well of four 4-well chamber slides (NUNC, Life Technologies, Paisley, UK) coated with poly-d-lysine and allowed to attach. Then slides were switched to one of the experimental conditions: 7- and 14-day conditioned medium, DMEM with 1% FBS (50 ng/ml NGF), and medium (with 1% FBS added). Every 2 days, fresh medium was changed for 10 days, and slides were imaged live under phase contrast microscopy. Ten high-powered fields per well were quantified by an observer blinded to the experimental condition, and the total number of neurite-bearing cells/field was quantified (neurites were projections greater than 1× the cell body diameter).
Animals
We used adult male SD and Wistar rats (Vitalriver), weighing 250 and 300 g, respectively. The experimental protocol was approved by the institutional care and use committee of our institution. The animals were placed in a temperature- and humidity-controlled room with a 12-h light/12-h dark cycle and allowed free access to standard rat chow and water.
Experimental Groups and Surgical Methods In Vivo
In total, 52 SD rats were divided into four groups (n = 13 each) for autografting, acellular allografting with fibrin glue-containing MSCs injected inside the graft, acellular allografting with fibrin glue-containing MSCs injected around the graft, or acellular allografting with fibrin glue injected alone. Rats were anesthetized by 0.6-ml injection of sodium pentobarbital (60 mg/ml). The sciatic nerve on the rat's left leg was exposed, and a 12-mm segment was removed from the midthigh level and replaced with a 15-mm-long extracted nerve segment. The grafts were sutured to the epineurium of the proximal and distal nerve stumps with two sutures (12–0 Ethilon, Ethicon Inc., Sommerville, NJ, USA) at each junction. For autografting (autograft), a 15-mm-long sciatic nerve was excised and then grafted in the reverse direction between the two nerve stumps. To avoid retraction of nerve ends, the nerve ends of the prepared 15-mm segment were affixed with 12-0 nylon sutures before being excised. For acellular allografting with MSCs injected inside the proximal and distal part of the graft (MSCs-iANG), the prepared acellular nerve graft was implanted, and 0.01 ml fibrin glue containing 5 × 105 MSCs was injected into the graft. For acellular allografting with MSCs injected around the graft (MSCs-aANG), the prepared acellular nerve graft was implanted, and 0.1 ml fibrin glue containing 5 × 105 MSCs was injected around the graft. For acellular allografting with fibrin glue alone (ANG), the acellular nerve graft was implanted with 0.1 ml fibrin glue injected around the graft. The wound was then closed in layers. The animal experiment was divided into two periods: five animals in each group were killed 2 weeks after surgery, and the rest of the animals were killed 12 weeks after surgery. There were eight animals in each group 12 weeks after surgery, which were used for sensory and motor functional analysis and histology.
Evaluation of Growth of Axons
In vivo, we traced grafted MSCs labeled with the fluorescent dye PKH26 according to the manufacturer's protocol. Briefly, cultured cells were detached with use of trypsin, washed in serum-free medium, and resuspended in 1 ml of the dilution buffer from the manufacturer's labeling kit. The cell suspension was mixed with an equal volume of the labeling solution containing 4 × 10−6M PKH26 in the dilution buffer and incubated for 4 min at room temperature. The reaction was terminated by adding 2 ml of FBS, and cells were washed three times. MSCs labeled with PKH26 were used for analyzing the distribution of seed cells in acellular nerve grafts. After 2 weeks, samples of the graft and attached nerve stumps were obtained for immunofluorescence assay to assess early regeneration in the growth distance of axons.
Longitudinal distal sections 10 μm thick were cut and directly observed by fluorescence microscopy. After nonspecific immunoreaction was blocked, the samples were incubated in solution of antineurofilament (NF) antibody (1:200) (Sigma) overnight at 4°C, then washed three times with PBS, and incubated in biotinylated anti-mouse rabbit IgG solution (1:200; Zhongshan Corp., Beijing, China) for 1 h at room temperature, and then washed three times with PBS, and the distance of axon growth in each group was observed on fluorescence microscopy.
Functional Evaluation of Nerve Regeneration In Vivo
Sensory and motor functional analyses were performed 12 weeks after surgery. Von Frey hair sensitivity testing was adapted from Chaplan et al. (4) to evaluate sensory function recovery. During sensitivity testing, the rats were placed in plastic cages and allowed to acclimatize for 10 min. Cages were equipped with a wire mesh bottom, which allowed full access to the midplantar hind paw testing area. Before and after surgery, testing was performed by touching the paw area with a series of six von Frey hairs with logarithmically incremental stiffness (4, 6, 8, 10, 15, and 26 g) (Stoelting, Wood Dale, IL, USA). Each hair was applied to the surface of the paw until buckling was noted and then held for 6–8 s. Testing started with the 4-g hair and continued with ascending thickness of hairs. A positive response was noted if the paw was sharply withdrawn. A negative response was recorded with no paw withdrawal. Following a positive response, the previous weaker stimulus was tested. A total of six responses were obtained to determine the 50% withdrawal response.
To evaluate motor function recovery, the tension of the triceps surae muscle was detected at 12 weeks after surgery. Eight animals in each group were anesthetized by peritoneal injection of sodium pentobarbital (0.6 ml, 60 mg/ml). The triceps surae muscles from both operated and nonoperated sides of rats were dissected and detached from the bone at their terminal point. The terminal point was connected to a muscle tension transducer (BL-420F biological function experimental system, Chengdu Taimeng, Sichuan, China) with the use of a 3-0 suture thread (Wuxi Medical Instruments). Stimulating electrodes were placed at the proximal end of the sciatic nerve, and the sciatic nerve was stimulated supramaximally. The greatest muscular contraction tension was recorded. Muscle tension on the operated side was expressed as a percentage of that on the nonoperated side.
Histomorphometric Assessment of Nerves In Vivo
At the end of functional evaluation, the lesions of sciatic nerves and contralateral nerves were harvested. The midportion of the nerve graft was sliced transversely to determine the morphology of the regenerated nerve fibers. After nonspecific immunoreaction was blocked, 10-μm-thick samples were incubated in solution of anti-NF antibody (1:200; Sigma) and anti-p75 protein antibody (low-affinity nerve growth factor receptor; 1:100; Sigma) overnight at room temperature, then washed three times with PBS, and incubated in biotinylated anti-mouse rabbit IgG solution (1:200) for 1 h. They were washed three times with PBS and then labeled with secondary antibody.
A 2-mm segment of the midportion of the nerve graft and 5 mm distal to the lesion site was dissected. The segments were fixed in 2% glutaraldehyde for 2 days, dehydrated, and plastic embedded. Transverse sections of semithin (0.5 μm) and ultrathin (50 nm) sections were obtained by use of an ultramicrotome (Leica EMUC-6, Wetzlar, Germany). Semithin sections were stained with 1% toluidine blue for 1 h and examined by light microscopy. Photomicrographs were obtained for measuring the cross-sectional area of the whole nerve and morphological analysis. Morphological evaluation involved use of ImagePro Plus (Media Cybernetics, Bethesda, MD, USA), and the extrapolated total number of nerve fibers (TNFe) and fiber diameter distribution were obtained. The ultrathin sections were stained with 1% uranyl acetate and examined by use of a Philips CM120 transmission electron microscope (Amsterdam, Netherlands) equipped with an image acquisition system.
Weighing Triceps Surae Muscles and Masson Trichrome Staining
The triceps surae muscles were harvested from the experimental and control sides and weighed. A conservation ratio of muscle to mass was recorded for each animal by dividing the experimental side muscle mass by the control side muscle mass. Then specimens were fixed in 4% paraformaldehyde. The midportions of the triceps surae muscles were transversally cut into 5-μm-thick paraffin sections and stained with Masson trichrome to measure the mean cross-sectional areas of the fibers. The data for the cross-sectional area of the gastrocnemius muscle were measured by use of ImagePro Plus.
Statistical Analysis
Results are expressed as mean ± standard deviation (SD), including the nonparametric von Frey hair sensitivity data for clarity. The statistical significance of the observed differences was analyzed by t tests or one-way analysis of variance (ANOVA) followed by the Student–Newman–Keuls test. The results of the von Frey hair sensitivity testing was analyzed by Kruskal–Wallis and Nemenyi tests. A value of p < 0.05 was considered to indicate significant differences.
Results
Structure and Composition of Acellular Nerve Grafts
To determine the influence of decellularization on acellular nerves, two-photon microscopy was used to produce a 3D reconstruction of the acellular nerve viewed from a 0 angle to a depth of 200 μm. Autofluorescence was combined with SHG to show the relative makeup of the acellular nerve. SHG microscopy was used to observe fibrillar collagen (Fig. 1A) in this 3D reconstructed z-projection (collapsed z-stack) and other ECM (Fig. 1B). The 3D structure of acellular nerve was not severely destroyed during decellularization process.

Two-photon images of rat acellular nerve viewed to a depth of 200 μm. Field of view is 320 μm. Second harmonic generation microscopy was used to observe fibrillar collagen (A) in this 3D reconstruction, autofluorescence of elastin and other extracellular matrix (B), merged (C).
Tensile Testing of Acellular Nerve Graft
Average lengths and widths of the specimens as tested did not differ among groups. The acellular nerve specimens withstood an average ultimate stress of 1,622 ± 63 kPa, and normal nerve specimens withstood an average ultimate stress of 2,834 ± 297 kPa (p < 0.01). The average ultimate strains for the experimental and control groups were 0.54 ± 0.13 and 0.76 ± 0.11, respectively (p < 0.05). The Young's modulus did not significantly differ between experimental and control groups (5,654 ± 1,710 and 4,964 ± 1,014 kPa, respectively). Mechanical work to failure was 2.61 ± 1.68 × 10−3 J and 4.30 ± 0.67 × 10−3 J for the experimental and control groups, respectively (p < 0.05) (Table 1).
Tensile Testing of Acellular Nerve and Normal Sciatic Nerves

p < 0.01,
p < 0.05 compared with the control group. N, newtons; kPa, kilopascals; J, joules.
MSC Viability in Fibrin Glue
MSCs embedded in fibrin glue were observed at 0 day (Fig. 2A) and 3 days (Fig. 2B) in culture. We then investigated the viability of encapsulated MSCs and MSCs after different culture times (up to 21 days) using LIVE/DEAD staining. The viable cells were reported in the graph as percentage of viable cells with respect to day 0 (set to 100%). We observed a steady but significant (p < 0.05) decrease in viability during the 21 days. The viability of FG + MSCs and MSCs also significantly differed at 21 days (p < 0.01) (Fig. 2C).
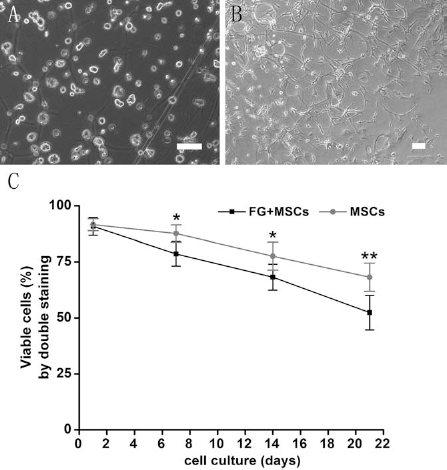
MSCs embedded in fibrin glue culture. Mesenchymal stem cells (MSCs) embedded in fibrin glue culture at 0 day (A) and 3 days (B). Scale bars: 100 μm. Viability of cultured MSCs adherent (gray lines) and encapsulated (black lines) in fibrin glue (FG) as determined by LIVE/DEAD staining (C). *p < 0.05, **p < 0.01 versus day 1.
MSCs Embedded in Fibrin Glue Secrete Bioactive Neurotrophins
ELISA analysis of conditioned medium demonstrated detectable levels of neurotrophin secretion by MSCs embedded in fibrin glue and MSCs alone. The amount of NGF secreted from unencapsulated and fibrin-embedded cells was between 20 and 70 pg/million cells/day depending on the time point. NGF levels for unencapsulated and fibrin-embedded MSCs were higher at day 7 than at initial and distal times; no other significant differences were found except at day 1 (Fig. 3A). At 1 and 3 days, BDNF secretion from unencapsulated and fibrin-embedded MSCs was higher than at other times (between 20 and 130 pg/million cells/day). With increasing time, BDNF secretion quickly decreased, with no significant differences between cell types (Fig. 3B).

Quantification and effect of neurotrophic factors. Quantification of protein content of nerve growth factor (NGF) (A) and brain-derived neurotrophic factor (BDNF) (B) in a medium of MSCs embedded in FG. MSCs embedded in FG secrete bioactive neurotrophins into culture medium. Pheochromocytoma (PC12) cells cultured in 7-day (C) or 14-day (D) conditioned medium, medium containing NGF (E), or unconditioned medium (F) for 10 days. Quantification of neurite outgrowth from PC12 cells grown in conditioned medium (G). Data are mean ± SD. **p < 0.01, compared with unconditioned medium. Scale bars: 50 μm.
Bioactivity of Conditioned Medium
As a functional bioassay of secreted neurotrophins, PC12 cells were cultured in 7-day (Fig. 3C) and 14-day (Fig. 3D) conditioned medium, DMEM with 1% FBS (50 ng/ml NGF) (Fig. 3E), and unconditioned medium (Fig. 3F) (with 1% FBS added). Results of a bioactivity assay revealed that MSCs embedded in fibrin glue with 7- and 14-day conditioned media elicited robust neurite outgrowth to the level matching that of the NGF-positive control, with significantly fewer neurite-bearing cells in unconditioned medium (p < 0.01) (Fig. 3G).
Growth Distance of Axons
Animals (n = 5 in each group) were sacrificed to measure the length of axonal regeneration 2 weeks after grafting surgery. MSCs labeled with PKH26 were used for analyzing the distribution of seed cells in acellular nerve grafts. In acellular allografting containing MSCs inside the graft, seed cell distributed accumulated in the longitudinal section of the nerve graft (Fig. 4A). However, in acellular allografting containing MSCs around the graft, MSCs were arranged along the longitudinal acellular nerve, which resembled bands of Büngner (Fig. 4B). The MSCs inside or around the acellular nerve were shown to be stable and with viable morphology. The mean axonal regeneration was significantly greater in the nerve repair with autografting and with MSCs inside or around the acellular grafting than with acellular grafting alone (p < 0.01), with no significant differences between grafts with MSCs injected inside or around the acellular grafting (Fig. 4C).

Distribution of PKH26-labeled MSCs in acellular allografting. Distribution of PKH26-labeled MSCs in acellular allografting with MSCs injected inside (A) and around (B) the graft 14 days after transplant. Scale bars: 100 μm. Quantification of growth of axons in different groups 14 days after grafting (C). MSCs-iANG, MSCs injected inside the graft; MSCs-aANG, MSCs injected around the graft; ANG, acellular grafting alone. **p < 0.01 compared with acellular nerve grafting alone.
Functional Evaluation
In evaluating sensory function recovery, the Von Frey response was significantly lower with autografting than acellular nerve grafting after week 12 (p < 0.05), with no significant difference observed for the other groups (Fig. 5A). To evaluate motor function recovery, the tension of triceps surae muscles was significantly lower with acellular nerve grafting than with the other three treatments (p < 0.05), with no significant differences between grafts with MSCs injected inside or around acellular grafts (Fig. 5B).

Sensory and motor functional evaluation 12 weeks after grafting. (A) Vo n Frey testing. (B) Tension of the triceps surae muscle. *p < 0.05, **p < 0.01 compared with acellular grafting (ANG) alone.
Histology Analysis of Nerves
Light microscopy of the regenerated tissues 12 weeks after implantation revealed that nerves regenerated across the defect in all implanted groups (n = 8 in each group). The midportion of the regenerated segment revealed axons and Schwann cells confirmed by double immunofluorescence (Fig. 6). The immunopositivity of P75 and NF-200 proteins was monitored, respectively, as markers of Schwann cells and axons in the bridged and regenerating nerves.

Immunofluorescence double staining of the midportion of the regenerated segment revealing axons and Schwann cells. Immunopositivity of neurofilament (NF)-200 and low-affinity nerve growth factor receptor (P75) proteins were monitored as markers of axons and Schwann cells, respectively, in the bridged and regenerating nerves. MSCs-iANG, MSCs injected inside the graft; MSCs-aANG, MSCs injected around the graft; ANG, acellular grafting alone. Scale bars: 50 μm.
Figure 7 shows representative images of midportion and distal sections used for analysis. Autograft-repaired nerves had the characteristic appearance of regenerated nerves, with slightly larger, more thickly myelinated axons than the other three groups. Interestingly, MSCs injected inside and around the acellular nerve graft allowed a similar degree of regeneration. Transmission electron micrographs of the sciatic nerve trunk 5 mm distal to the suture site of each repair group showed regenerated myelinated fibers dispersed in clusters and numerous unmyelinated fibers.

Midportion of the nerve graft and 5 mm distal to the lesion site stained with toluidine blue. Toluidine blue staining of the midportion of the nerve graft (central; A, D, G, J) and 5 mm distal to the lesion site (distal; B, E, H, K) for the previously described groups. Scale bars: 50 μm. Ultrathin sections distal to the lesion site examined by Philips CM120 transmission electron microscope (TEM; C, F, I, L). Scale bars: 2 μm.
This disparity was most significantly reflected in measures of cross-sectional area and total fiber count of nerve (Fig. 8). The cross-sectional area for autografting and MSCs injected inside or around the acellular nerve graft was nearly double that of the acellular nerve grafting alone in the central section, but the cross-sectional area with autografting was higher in the distal section than that for the three other treatments (p < 0.01) (Fig. 8A). The number of myelinated nerve fibers that regenerated through the cross-suture repair site into the distal nerve was highest in the autograft group (4,225 ± 276, p < 0.05), with no significant difference between grafting with MSCs injected inside or around the acellular nerve graft (both 3,216 ± 493 and 2,869 ± 436) (Fig. 8B). Acellular nerve grafting alone (1,787 ± 416 fibers) differed significantly from the other three treatments (p < 0.05). Furthermore, a plot of frequency of fiber diameters displayed a narrow peak of smaller diameter fibers in the proximal stump compared to the distal stump, with the autograft group showing a more broad distribution of larger diameter fibers (Fig. 8C), which is indicative of greater maturity.
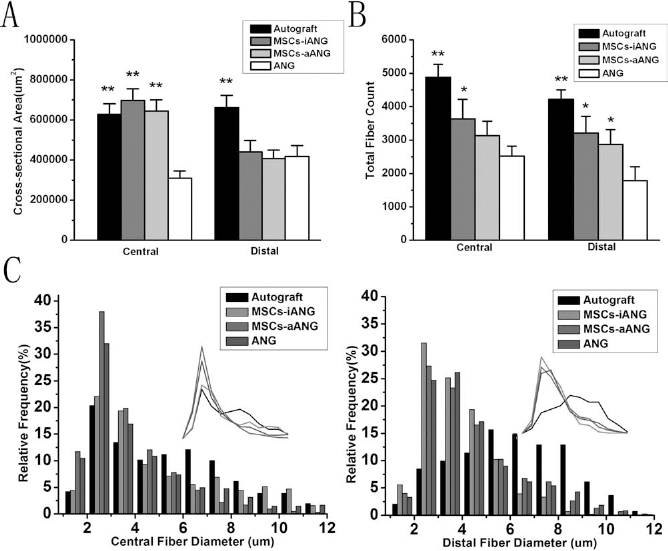
Sciatic nerve morphology 12 weeks postgrafting. Cross-sectional area (A) and total fiber count (B) of sciatic nerves 12 weeks postgrafting. Plots of frequency of fiber diameters showing a narrow peak of smaller diameter fibers in the central stump (C) compared with the distal stump (D). *p < 0.05, **p < 0.01 compared with acellular grafting alone.
Weighing Triceps Surae Muscles and Masson Trichrome Staining
At 12 weeks postsurgery, the relative weight of the triceps surae muscles markedly decreased to varying degrees in all four groups and was especially small with acellular nerve grafting alone. The relative wet weight ratio showed significant differences among the four groups (p < 0.05), with no significant differences between grafts with MSCs injected inside or around the acellular graft (Fig. 9A). In the autograft group, muscle fibers on the experimental side were polygonal and uniform in size with a mean cross-sectional area of 963 ± 141 μm2 and were significantly larger than those with acellular nerve grafting alone (p < 0.01), with no significant difference among muscle fibers between grafts with MSCs injected inside or around acellular grafts, which was significantly different from those of acellular nerve grafting alone (p < 0.01) (Fig. 9B).
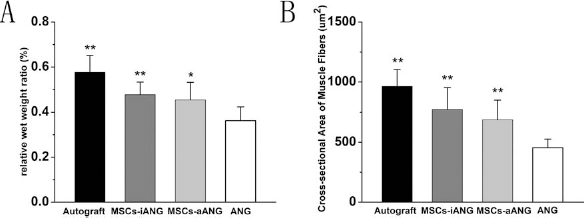
Muscle fiber morphology 12 weeks postgrafting. Comparison of relative wet weight ratio (the experimental side to control side muscle mass) (A) and cross-sectional areas of muscle fibers (B). *p < 0.05, **p < 0.01 compared with acellular grafting alone.
Discussion
In this study, we constructed a novel nerve graft by acellular nerve grafting with MSCs affixed with fibrin glue injected around the nerve grafting. The procedure maintained the structure and composition of the nerve graft. MSCs embedded in fibrin glue could survive and secrete growth factors. Therefore, injecting MSCs around the graft may help improve nerve regeneration and functional recovery of peripheral nerve lesions. Compared to MSCs directly injected into the acellular nerve, injecting MSCs around acellular nerves is easy, is comparable, maintains nerve structure, and is another option for stem cell transplantation to repair long nerve defects.
The strongest connective tissue layers in peripheral nerves are the perineurium and, to a lesser extent, the epineurium. Alterations in the epineurium and perineurium ECM composition are likely to have significant effects on the tensile strength of acellular nerve constructs (3,19). After multiple decellularization attempts, we found that the tensile strength of the peripheral nerve was overall decreased by the acellularization process compared with normal nerve. The ultimate strain was decreased relative to fresh nerve. The grafting groups did not differ in Young's modulus, indicating that the stiffness of the materials was equivalent. The acellularization process may have partially removed one or more components of the ECM to reduce the amount of energy able to be absorbed before tensile failure. Nerve grafts are tension free by design (20). Therefore, whether such a biomaterial has substantial tensile strength must be demonstrated before its clinical use. Using two-photon microscopy, we confirmed that the acellular rat nerve maintained the major components and the structure of natural nerve. The acellular nerve ECM retains enough of the natural nerve collagen and laminin network. As expected, the decellularization process allowed for retention of the basic nerve structure and compositions.
Acellular nerve grafting is an alternative to autografting for repair of short gaps in peripheral nerves (28,39). However, outcomes associated with the use of acellular nerve grafting are often inferior to that of autografts, particularly over long lesion gaps. Compared with autografts, acellular nerves lack MSCs, which are a rich source of neurotrophins and cytokines that support axonal regeneration and recruit other cells into the injury site. To enhance nerve regeneration and functional recovery in the peripheral nervous system, our previous research showed enhanced acellular nerve grafting with neurotrophins (18,38). Li et al. (18) found that adenoviral transfection with hepatocyte growth factor has potent angiogenic and neuroregenerative effects in allogenic grafting of peripheral nerves. Yu et al. (38) found that microspheres of NGF that are fixed around an acellular nerve graft can sustain the release of bioactive NGF for nerve regeneration in rats. However, several neurotrophic factors are required for the regulation of neuronal differentiation, survival, and nerve regeneration after nerve injury (15). MSCs elute neurotrophic factors and indirectly modulate the cellular behavior (36). Yang et al. (37) showed that MSC-conditioned medium enhanced axon growth and neurogenesis in cultured dorsal root ganglia explants, augmented cell survival of and expression of nerve factor and growth-associated protein 43 (GAP-43) by cultured dorsal root ganglion (DRG) neurons, promoted cell survival and proliferation of cultured MSCs, and increased the expression of NGF, BDNF, and basic fibroblast growth factor (bFGF) in cultured SCs.
A crucial issue is the effect of the encapsulation procedure on the growth and activity of cells (8,30,35). Therefore, we determined the viability of MSCs encapsulated in fibrin glue. While the apparent viability appeared to be reduced compared to unencapsulated MSCs, it is worth noting that, since the encapsulated cells did not migrate very far, there is a limited 3D environment for growth and limited nutrition after 21 days, which may contribute to the reduced viability. It is worth noting that more than 50% of the MSCs encapsulated in fibrin glue remained viable throughout 3 weeks of in vitro culture. Moreover, our data reveal that MSCs, even when embedded in fibrin glue, secrete NGF and BDNF into the surrounding medium, and a study published since the acceptance of this article agrees with the paracrine action of fibrin glue-embedded MSCs (17). Neurotrophic factors are released rapidly from fibrin glue (2). Quantitative analysis of constitutive neurotrophic factor release from MSCs is rarely reported in the literature, despite the popular theory that release of these proteins and their effects on compromised host tissue is a potential mechanism of the cells' restorative capacity. Release of the therapeutically active molecule is needed at the level of allowing exchange of nutrients and waste and release of the therapeutic agents. Therefore, that MSCs embedded in fibrin glue can survive and secrete growth factors is of interest.
Recently, MSCs were considered an alternative to SCs, and their use has overcome the difficulties encountered with autologous SCs. There have been encouraging results for bone marrow stem cell transplantation in the rat sciatic nerve transection model (5,10). We found axon regeneration faster with MSCs injected inside or around acellular grafting than with acellular grafting alone during the first 14 days, but slower than with autografting. Furthermore, stem cells penetrated into the regenerated nerve at the site of stumps in grafts with MSCs injected around the graft. In addition, we demonstrated the promotion of repair of 15-mm nerve defects by supplementing with MSCs evidenced by [1] improved sensory and motor function as measured by 50% withdrawal response and tension of the triceps surae muscle, [2] increased cross-sectional area of the whole nerve and total fiber count, and [3] reduced triceps surae muscle atrophy as measured by muscle weight and increased cross-sectional areas of muscle fibers. The cross-sectional area for autografting and MSCs injected inside or around the acellular nerve graft was nearly double that with acellular nerve grafting alone in the central section. The supplemental MSCs had no significant effect on the cross-sectional area in the distal section compared with acellular nerve grafting alone. However, supplemental MSCs can increase the total fiber count in the central and the distal section compared with acellular nerve grafting alone. Moreover, we found no difference in angiogenesis and fibrosis when the cells were seeded inside or outside of the nerve graft. Thus, the MSCs maintained the acellular nerve grafting structure and promoted axonal regeneration through the acellular nerve grafting whether transplanted inside or around the acellular nerve graft. Supplemental MSCs might be responsible for their promoting effects on peripheral nerve regeneration. The success might be due to both the expression of trophic factors by the MSCs leading to axonal growth and differentiation of MSCs into Schwann-like cells causing myelin reformation in both groups. Mohammadi et al. (22) reported on a 10-mm sciatic nerve defect bridged with a vein graft filled with bone marrow stromal cells (BMSCs) or omental adipose-derived nucleated cells. These two kinds of stem cells have a similar effect on enhancing sciatic nerve regeneration in terms of functional study, morphometric indices, and immunohistochemistry at 4, 8, and 12 weeks after surgery. Nijhuis et al. (23) showed that BMSC-filled venous grafts could bridge a 20-mm neural gap. Vein grafts with BMSCs had better functional nerve regeneration than grafts with nothing or saline, as measured by somatosensory-evoked potentials, toe spread, pin prick, and gastrocnemius muscle index. Results of this study indicate that supplementing MSCs around acellular nerve grafts has great potential to promote regeneration of peripheral nerve injury and has the same effect as supplementing MSCs inside the grafts.
Rather than atrophy, the contractile strength of the gastrocnemius muscle in response to proximal sciatic stimulation would have been a much better indicator of improved regeneration (4). Sensory and motor functional analyses were performed 12 weeks after the operation. Previously, von Frey hair testing has been used to quantify neuropathic pain in a complete sciatic nerve transection model. We observed increased sensory nerve regeneration leading to decreased threshold value with autografting. A decrease in threshold value indicates decreased resistance to applied force, thus leading to increased sensitivity. This finding was associated with low muscle mass values for each group, apparently due to decreased axonal muscle reinnervation. Both grafting rat groups were able to lift their feet in response to stimulus, thus displaying decreased resistance to applied force and increased sensitivity and thus successful axonal connections leading to an increase in sensory nerve regeneration. This situation in turn leads to an increase in axon muscle reinnervation and a subsequent increase in tension of the triceps surae muscle and overall muscle mass.
In the present study, we directly transplanted MSCs around acellular nerve grafts. The procedure facilitated nerve regeneration and avoided destroying the acellular nerve structure. This method was not limited by the number of seed cells and the diameter of nerves, especially small nerves. Pan et al. (24) also found that rat amniotic fluid MSCs embedded in fibrin glue delivered to sciatic nerves after crush injury may facilitate regeneration by amniotic fluid MSC-secreted neurotrophic factors. Supplementing MSCs around the nerve conduit may be another option for treatment of nerve injury. The success might be due to both expression of trophic factors by the MSCs leading to axonal growth and to the differentiation of MSCs into Schwann-like cells causing myelin reformation. Our data support that fibrin glue-encapsulated MSCs may have an important role in tissue engineering and may be useful for improving a regenerative medicine approach for tissue repair based on MSCs.
Conclusions
In this study, we created a nerve graft model of acellular nerve grafting with bone marrow MSCs affixed with fibrin glue, which maintained the structure and composition of the nerve but decreased tensile strength. MSCs embedded in fibrin glue and injected around the graft can survive and secrete growth factors and may help improve nerve regeneration and functional recovery of peripheral nerve lesions without destroying the acellular nerve graft. Compared to MSCs directly injected into the acellular nerve, injecting MSCs around acellular nerves is easy, maintains the nerve structure, and supports nerve regeneration. The technique is simple and is another option for stem cell transplantation. This nerve graft may be a substitute for autologous nerve grafting. Using these nerve regeneration guides in larger animal models to repair long nerve gaps needs further work.
Footnotes
Acknowledgments
Special thanks to Xu Guo for the histology and animal surgery. The authors gratefully thank Jifeng Chen for assistance in cell encapsulation training. This project was supported by Beijing Metropolis Beijing Nova Program (201115); National Natural Science Foundation of China (31170946, 31100696, 31300805); National High Technology Research and Development Program of China (2012AA020502); People's Liberation Army 12th five-year plan period (BWS11J025); China Postdoctoral Science Foundation (2013M532134), and National Basic Research Program of China (973 Program, 2012CB518106, 2014CB542201). The authors declare no conflict of interest.
