Abstract
Both enzymatic dissociation of cells prior to needle-based injections and poor vascularization of myocardial infarct areas are two important contributors to cell death and impede the efficacy of cardiac cell therapy. Because these limitations could be overcome by scaffolds ensuring cell cohesiveness and codelivery of angiogenic cells, we used a chronic rat model of myocardial infarction to assess the long-term (6 months) effects of the epicardial delivery of a composite collagen-based patch harboring both cardiomyogenesis-targeted human embryonic SSEA-1+ (stem cell-derived stage-specific embryonic antigen-1 positive) cardiovascular progenitors and autologous (rat) adipose tissue-derived angiogenesis-targeted stromal cells (n = 27). Cell-free patches served as controls (n = 28). Serial follow-up echocardiographic measurements of left ventricular ejection fraction (LVEF) showed that the composite patch group yielded a significantly better preservation of left ventricular function that was sustained over time as compared with controls, and this pattern persisted when the assessment was restricted to the subgroup of rats with initial LVEFs below 50%. The composite patch group was also associated with significantly less fibrosis and more vessels in the infarct area. However, although human progenitors expressing cardiac markers were present in the patches before implantation, none of them could be subsequently identified in the grafted tissue. These data confirm the efficacy of epicardial scaffolds as cell carriers for ensuring long-term functional benefits and suggest that these effects are likely related to paracrine effects and call for optimizing cross-talks between codelivered cell populations to achieve the ultimate goal of myocardial regeneration.
Keywords
Introduction
One of the main lessons drawn from the accumulated experience with cell therapy is that, regardless of the cell type, the efficacy of the procedure is severely hampered by the low rate of engraftment due to cell losses during transfer and subsequent death of the initially retained cells (30,32). The conventional injection-based mode of cell delivery likely plays an important role in these events for at least two reasons: First, multiple intramyocardial injections cause leakage of cells through puncture holes and washout through the lymphatic and venous circulations; second, injection first requires a proteolytic dissociation of cells to suspend them in the vector solution, which results in loss of their anchoring to an extracellular matrix (26) and sets the stage for an apoptotic mode of cell death (anoikis) (33). Because it provides a direct control over the heart, cardiac surgery offers the unique opportunity to overcome these two limitations by shifting from cell injection to cell seeding onto epicardially delivered scaffolds, which have the dual advantage of enhancing minimally invasive cell transfer (18) and providing a template for cells to form a cohesive sheet. In addition to injection, ischemia is another major contributor to cell death and can be addressed by cotransplantation of cells featuring trophic properties (15,31) targeted at improving graft survival (4). Although several studies have now documented the benefits of these composite epicardial cell-loaded biomaterials, their clinical relevance is challenged by (i) the use of scaffold-free cell constructs (18,28,29), which, because of their intrinsic frailty, may be hazardous to manipulate outside of a laboratory environment; (ii) the use of noninfarcted animal models (29); and (iii) the relatively short-term follow-up periods (9,27). In an attempt to address these issues, the present study leveraged our previous findings of improved outcomes after patch delivery (10,11) and stromal cell cotransplantation (1,24) to assess the long-term (6 months) results obtained, in a rat model of chronic myocardial infarction, with the epicardial delivery of mechanically resistant collagen films coseeded with human embryonic stem cell (ESC)-derived cardiovascular progenitors and autologous (rat) adipose-derived stromal cells (ADSCs).
Materials and Methods
Animals
Fifty-five female Wistar rats (Janvier Labs, Genest Saint Isle, France) and nine Rowett nude (rnu/rnu) female rats (Harlan, Gannat, France) weighing an average of 200–250 g were used in this study. All procedures were approved by our institutional ethics committee and complied with the European legislation (European Commission Directive 86/609/EEC) on animal care.
Myocardial Infarction Model, Transplantation, and Experimental Groups
Rats were anaesthetized with isoflurane (Baxter, Maurepas, France), 3% at induction and 2% for maintenance, and tracheally ventilated at a rate of 70/min and with a 0.2-ml average insufflate volume (Alphalab, Minerve, France). Analgesia was performed for 2 days after surgery with a 10 mg/kg subcutaneous injection of ketoprofen (Merial, Lyon, France). The heart was exposed through a left thoracotomy and the left coronary artery was permanently ligated between the pulmonary artery trunk and the left atrial appendage. One month after myocardial infarction, rats underwent a baseline echocardiographic assessment of left ventricular (LV) function. Following a median sternotomy, animals were then randomly allocated to receive an epicardial collagen patch (Gelfilm®, Pfizer, Groton, CT, USA) made of autologous ADSCs and human ESC-derived stage-specific embryonic antigen-1 positive (SSEA-1+) cardiovascular progenitors (treated, n = 27) or a cell-free patch (controls, n = 28). Echocardiographic measurements were then serially repeated every month until 6 months after transplantation. After 2 months, 10 animals (six controls and four treated) were euthanized with an overdose of isoflurane for histologic studies. Immunosuppression was achieved by continuous delivery of cyclosporine (5 mg/kg per day) by subcutaneously implanted Alzet® micro-osmotic pumps (Model 2ML4, Durect Corporation, Cupertino, CA, USA) and started the day of transplantation. In light of the 2-month histological results showing the absence of the transplanted human progenitor cells (see the following), it was interrupted from this time point onward. This negative finding also prompted the implementation of an additional series of nude rats (n = 9), which underwent myocardial infarction according to the same protocol and were then transplanted with a collagen patch coseeded with human ESC-derived SSEA-1+ cardiovascular progenitors and human ADSCs.
Preparation of ESC-Derived Cardiovascular Progenitors
Human ESCs from the I6 cell line (generously provided by J. Itskovitz-Eldor, Technion Institute, Haïfa, Israel) were cultured in a defined medium (NutriStem®, Biological Industries, Kibbutz Beit Haemek, Israel) on human foreskin fibroblasts (generously provided by Dr. O. Damour, Hospital E. Herriot, Lyon, France). Cell colonies were dissociated into small clumps every 5 days using collagenase type II (Invitrogen, Carlsbad, CA, USA). The cells were treated for 4 days with 10 ng/ml of bone morphogenetic protein 2 (BMP-2; Life Technologies, Lyon, France) in the presence of 1 μM SU5402 (Millipore, Molsheim, France), a fibroblast growth factor (FGF) receptor inhibitor, in Roswell Park Memorial Institute (RPMI)/B27 medium (Life Technologies) to induce cardiac specification. For sorting, trypsinized cells (Life Technologies) were incubated with gentle occasional agitation at room temperature for 30 min in the presence of anti-SSEA-1 antibodycoated microbeads (40 μl/107 cells, Miltenyi Biotec, Paris, France) in Dulbecco's phosphate-buffered saline (D-PBS; Life Technologies) supplemented with 0.5% (w/v) bovine serum albumin (BSA; Life Technologies) and 2 mM ethylenediaminetetraacetic acid (EDTA; Sigma-Aldrich, Lyon, France). Cells were transferred to an L50 Miltenyi cartridge (Miltenyi Biotec) set on the magnet. Cells were washed three times with 3 ml of D-PBS/BSA/EDTA mixture and eluted from the column after removing the magnet using 3 ml of D-PBS/BSA/EDTA mixture. We have previously shown by flow cytometry performed after 48 h that this procedure achieved a purity rate of 95% of SSEA-1+ cells (2).
Isolation and Preparation of ADSCs
Autologous cells from the stromal vascular fraction were isolated from inguinal subcutaneous adipose tissue at the time of myocardial infarction in each rat. After enzymatic digestion in Dulbecco's modified Eagle's medium with F12 supplement (DMEM-F12; Invitrogen) supplemented with 2 mg/ml collagenase (Serva, Reno, NV, USA) and BSA (2%) for 30 min at 37°C and filtration, mature adipocytes were separated from the stromal fraction by centrifugation at 600 × g for 10 min. The pellet was incubated in erythrocyte lysis buffer (StemCell Technologies, Vancouver, BC, Canada) and then washed in PBS and finally resuspended in DMEM-F12 supplemented with 10% fetal bovine serum (FBS; Life Technologies) for cell seeding and culture in 5% CO2 until subconfluence. Subconfluent ADSCs were obtained after 6 days (22). In the additional series of nude rats, human ADSCs were obtained from patients undergoing elective abdominal dermolipectomy after informed patient consent according to the bioethics law No. 2004-800 of August 6, 2004, and processed as described above. Human ADSCs were cultured in minimum essential medium with α modification (α-MEM; Life Technologies) supplemented with 2% human plasma enriched with human platelet growth factors (EFS-PM, Toulouse, France).
Preparation and Characterization of Composite Patches
The collagen films were cut into 1-cm2 squares and first seeded with 300 × 103 ADSCs in DMEM-F12 supplemented with 10% FBS. Three days later, an equal number of ESC-derived SSEA-1+ cardiovascular progenitors were pipetted onto the patch and the coculture was continued for 7 days in NutriStem®. After 3 (ADSCs-seeded patches) and 10 days (composite ADSCs-SSEA-1+ patches) of culture, the number of viable cells was quantified on a Malassez cell (Sigma-Aldrich) by the trypan blue (Life Technologies) exclusion method. SSEA-1+ human cells were quantified using the Quantifiler™ Human DNA quantification kit (Applied Biosystems, Paris, France). Additional patches were processed for immunocytochemical labeling using antibodies against markers for a specific nuclear human protein (lamin A/C, Novocastra, Leica Biosystems, Nanterre, France), ESC-derived progenitor cells (SSEA-1, Cell Signaling Technology, Danvers, MA, USA), and cardiac-committed cells [NK2 homeobox 5 (Nkx 2.5; R&D Systems, Abingdon, Oxfordshire, UK) and T-Box transcription factor 5 (Tbx5; Abcam, Paris, France)]. For in vivo implantations, patches were laid over the macroscopically identified infarct area and secured to its epicardial edges by two to three polypropylene 7-0 sutures.
Assessment of LV Function
Pre- and posttransplantation cardiac function was evaluated by transthoracic echocardiography (Sequoia 516, equipped with a 13-MHz phased array linear 15L8 probe; Siemens, Saint-Denis, France) in animals sedated with 2% isoflurane (Sigma-Aldrich). Parasternal two-dimensional long-axis views allowed measurement of LV end-diastolic surface (LVEDS), LV end-systolic surface (LVESS), LV end-diastolic length (LVEDL), and LV end-systolic length (LVESL). LV end-diastolic volume (LVEDV) and LV end-systolic volume (LVESV) were calculated as (8/37π) × (area2/length). LV ejection fraction (LVEF, %) was calculated as [(LVEDV – LVESV)/(100 × LVEDV)]. All measurements were made on digital loops in triplicate and averaged by an investigator blinded to the treatment group.
Histological and Immunohistochemical Assessment
After the 2-month echocardiographic assessment, randomly selected hearts in each group were separated into two halves by a short-axis section through the mid-portion of the infarcted area. The blocks were immediately fixed in Tissue-TeK Optimal Temperature Cutting medium (Sakura Finetek USA Inc., Torrance, CA, USA) and frozen in liquid nitrogen until they were sliced into 8-μm-thick cryosections using a cryostat (CM 1850, Leica, Wetzlar, Germany). A total of 108 and 72 sections per heart were generated in the control and composite patch groups, respectively, and at least six fields per section randomly spanning the entire infarct area were then analyzed with a microscope (Leica DM 2000) equipped with a digital camera (Qicam, Qimaging, Surrey, BC, Canada). Hematoxylin and eosin (Dako, Les Ulis, France) staining was used to delineate the area of infarction. The presence of human cells was assessed by immunofluorescence using a human-specific antibody directed against lamin A/C. The specific binding of primary antibodies was revealed using fluorescein isothiocyanate (FITC)- or Texas red-conjugated secondary antibodies (Life Technologies). Endothelial cells were immunostained with a rat-specific antibody [rat endothelial cell antigen (RECA), clone HIS52, Serotec, Colmar, France] conjugated with a biotinylated anti-mouse IgG secondary antibody (Vector Labs, Burlingame, CA, USA). Angiogenesis was then computed by manually counting RECA-positive cells. The extent of fibrosis was assessed by Sirius red (RAL Diagnostics, Bordeaux, France) staining at 5× magnification. Digital images were then processed with Metamorph software (Universal Imaging Corporation, Downingtown, PA, USA), and fibrosis was expressed as the ratio between the area of scar tissue and the LV area. Nuclei were counterstained with hematoxylin or 4′,6-diamidino-2-phenylindole (DAPI; Life Technologies). In the additional series of infarcted nude rats transplanted with the composite collagen patches, hearts were serially explanted at 2 days, 8 days, 15 days, 1 month, and 2 months after the procedure and immunohistochemically stained for lamin A/C. Moreover, lamin A/C-positive cells were costained with antibodies directed against α-actinin (Sigma-Aldrich), Nkx 2.5, caveolin-1 (Santa Cruz, Dallas, TX, USA), and vimentin (Progen Biotechnik, Heidelberg, Germany).
Statistical Analysis
Data are summarized using mean (95% CI) or median (quartiles) values as appropriate. Changes in LV function variables were compared between treatment groups using a mixed model repeated-measures analysis of variancecovariance with time, treatment group, and their interaction. The mean percentage of fibrosis and the mean number of RECA-positive cells per square millimeter were compared among groups using a mixed model analysis of variance–covariance, taking into account the intraheart correlation. For all analyses, a two-tailed p value of < 0.05 was considered statistically significant. Analyses were conducted using SAS 9.2 (Statistical Analysis System, Cary, NC, USA).
Results
Characterization of Composite Cell Patches Before Transplantation
In order to prepare composite cell patches, we first cultured 300 × 103 ADSCs on collagen films for 3 days, after which an equal number of ESC-derived SSEA-1+cardiovascular progenitors were seeded on them and continued to be cultured for 7 days. Three days after ADSC seeding, scanning electron microscopy imaging demonstrated that the cells were attached on the non-porous collagen surface and formed a carpet of cells well connected with each other (Fig. 1). At 3 days, the number of viable ADSCs on collagen films was 93 ± 7 × 103(n = 3); after ESC-derived SSEA-1+ seeding and an additional 7-day culture period, their number increased to 290 ± 50 × 103 (n = 3). At the end of the 10-day culture, based on DNA quantification, the number of human cells was estimated at 150 ± 40 × 103 (n = 3); these cells were visualized using a human-specific lamin A/C antibody (Fig. 2A and B) and found to be cardiac committed, as demonstrated by positive immunostaining for Tbx5 and Nkx 2.5 (Fig. 2C–F).

Scanning electron microscopy images of ADSCs seeded on collagen film. Three days after seeding of 300 × 103 ADSCs and before the subsequent deposit of ESC-derived SSEA-1+ cells, the collagen patch (1 cm2) represents a carpet of cells well interconnected with each other. Scale bar: 40 μm (A), 10 μm (B).

In vitro characterization of the composite cell patch. Human ESC-derived SSEA-1+ cardiovascular progenitors were visualized using a human specific lamin A/C antibody (A and B, green fluorescence); their cardiac commitment is demonstrated by a positive staining for Tbx5 (C and D, green fluorescence) and Nkx 2.5 (E and F, green fluorescence). Combined green fluorescence and DAPI-stained nuclei (blue) are shown in (B), (D), and (F). Scale bar: 50 μm (A–D); 25 μm (E–F).
LV Function Assessment
LVEF did not significantly differ at baseline between the two groups. However, after transplantation, changes were markedly different with a sustained increase over time in the cell-seeded patch group, whereas after an initial drop, values tended to remain stable in the control group (p = 0.0008) (Fig. 3A). The numbers of animals available for these functional measurements were 28, 27, 20, 9, 9, 8, and 7 at baseline, 1, 2, 3, 4, 5, and 6 months, respectively, in the control group; corresponding numbers in the treated group were 27, 22, 15, 7, 6, 3 and 3. Fractional shortening changes displayed similar patterns with a significant difference (p = 0.0039) between the two groups over the 6-month follow-up period. Of note, the better preservation of LV function in the treated group was still manifest when the assessment was restricted to the subgroup of animals (n = 20 in each arm) whose baseline LVEF was below 50% (p = 0.0005) (Fig. 3B). LV volumes did not significantly differ between the two groups (data not shown). Likewise, there was no difference in survival curves (data not shown).
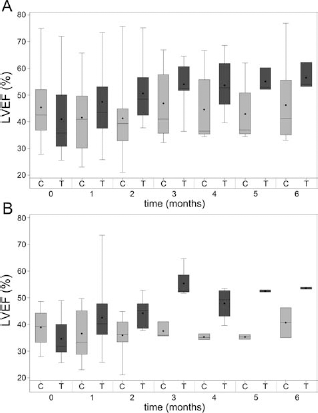
Box plot representation of LVEF. (A) Box plot representation of LVEF during the 6-month treatment period. (B) The same data set in the subgroup of animals whose baseline LVEF was below 50%. Values are presented as percentages. The boxes represent the lower to upper quartile range, the error bars represent the entire range, the horizontal bar represents the median value, and dot points represent mean values. C, control; T, treated groups; p = 0.0008 (A), p = 0.0005 (B).
Histological and Immunohistochemical Assessment
At the 2-month time point, we failed to identify persisting human cells in the grafted area as all examined sections (18 sections per heart) were negative for the human-specific marker lamin A/C. For this reason, it was deemed futile to continue immunosuppression, which was then stopped. Fibrosis was significantly reduced in the composite patch group compared with controls: 20.6% (18.1%; 23.1%) versus 28.6% (25.2%; 32.1%) (p < 0.0001) (Fig. 4A). The number of RECA-positive cells per square millimeter did not differ between groups in the normal myocardium [control: 1751 (1658; 1845); treated: 1661 (1536; 1785)] or the border zone [control: 549 (511; 587); treated: 637 (576; 697)]; conversely, angiogenesis was significantly increased in the infarct zone covered by the composite cell patch [417 (377; 456) RECA-positive cells/mm2] compared with the cell-free patch control [248 (222; 273), p = 0.0024] (Fig. 4B).

Box plot representation of fibrosis (A) and number of vessels (B) of control and treated hearts. Two months after treatment, 18 sections per heart were stained by Sirius red (fibrosis) or immunostained with a rat-specific endothelial cell antibody (vessels). The area of fibrosis and number of vessels were quantified using Metamorph software. Fibrosis was expressed as the ratio between the area of scar tissue and the LV area. The number of vessels was expressed as vessels per square millimeter of the infarct zone. C, control; T, treated groups. Nb, number.
To check whether the absence of human cardiac progenitors was not due to an immune response outweighing our cyclosporine delivery regimen, a separate set of experiments was performed in infarcted nude rats receiving patches coseeded with human SSEA-1+ progenitors and human ADSCs. Fairly extensive epicardial clusters of lamin-positive cells could then be detected until 2 months after the transplantation (102 positive sections out of 144 that were examined), at which time the animals were sacrificed (Fig. 5), but, again, none of these human cells coexpressed cardiomyocyte-specific markers (Tbx5 and Nkx 2.5) (data not shown). Indeed, these engrafted cells expressed markers of the mesenchymal and endothelial lineages, as demonstrated by their positive staining for vimentin and caveolin-1, respectively (Fig. 6).
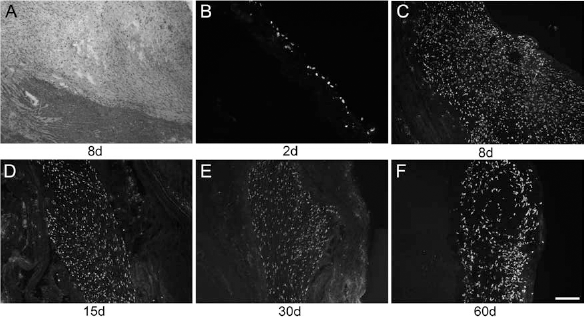
Detection of grafted cells. Human SSEA-1+ cardiovascular progenitor and human ADSC-seeded collagen patch visualized by hematoxylin and eosin staining (A) 8 days after delivery in a Rowett nude (rnu/rnu) rat. Grafted human cells were then serially identified using human-specific lamin A/C immunostaining at 2 (B), 8 (C), 15 (D), 30 (E), and 60 days (F) after implantation. Human cells were consistently identified by the formation of fairly extensive epicardial clusters. d, days. Scale bar: 50 μm (A–E); 25 μm (F).

Characterization of engrafted cells. Sixty days after epicardial delivery of human SSEA-1+ cardiovascular progenitor and human ADSCs seeded onto a collagen patch in a rnu/rnu rat, the heart sections were costained with anti-lamin A/C (A, red fluorescence) and anti-caveolin-1 (B, green fluorescence) or with anti-lamin A/C (D, red fluorescence) and vimentin (E, green fluorescence). Combined red and green fluorescence and DAPI-stained nuclei (blue) are shown in (C) and (F). Arrowheads indicate costained cells, whereas arrows indicate costained cells presented in the inset. Insets show higher magnification (4×) of a costained cell. Scale bar: 50 μm.
Discussion
The three main findings of this study are summarized as follows: (i) The epicardial delivery of a composite patch harboring both ESC-derived cardiovascular progenitors and autologous ADSCs provides a functional benefit that is sustained over time; (ii) this benefit seems to involve paracrine effects leading to reduction in fibrosis and increased angiogenesis because there was no evidence for a donor cell-derived cardiomyogenesis; (iii) the failure of the ESC-derived cardiovascular progenitors to survive challenges the putative tolerogenic effect of the codelivered ADSCs.
Few data are available on the long-term effects of cardiac cell therapy in rodent models, particularly when biomaterials are combined with cells, and a recent comprehensive review of the relevant literature indicates, in this setting, the longest follow-up period of only 13 weeks (21). The present study tried to address this issue by implementing a serial follow-up of the animals until 6 months after transplantation. The results show that rats receiving the composite patches demonstrated a better functional recovery than the controls. This improvement was consistent over the entire study period even though the reduced number of surviving animals at the latest time points precludes meaningful statistical analyses. In a separate set of experiments in which both cardiac progenitor and adipose tissue-derived human cells were similarly patch delivered in nude rats to avoid the confounding effect of an immune response, a noticeable number of transplanted laminpositive human cells could still be identified after 2 months, but the negativity of concurrent staining for cardiac markers indicates that these cells were indeed the ADSCs that expressed mesenchymal and endothelial lineage markers. Together, these data support the previous findings of our laboratory (10,11) and others (6,14,20) that epicardial patches are efficient cell delivery vehicles compared with conventional needle-based injections. There is compelling evidence that these benefits primarily stem from maintenance of cell-to-cell and cell-to-matrix connections that are conversely disrupted when cells are proteolytically dissociated for injections (11,28).
In the present study, the improved functional recovery following patch implantation was paralleled by a reduction of fibrosis and an increase in angiogenesis. The discrepancy between these findings and the lack of donor-derived new cardiomyocytes (see the following) strongly supports a predominant involvement of paracrine mechanisms likely mediated by ADSCs (25), although the failure to identify cardiac progenitors even early after transplantation in the nude rat experiments does not exclude that they can also exert paracrine effects (7). One could argue that if cells act primarily as short-term sources of biomolecules, their long-term persistence in the transplanted tissue may not be so critical. This reasoning, however, does not apply when the transplantation entails the use of cells not only behaving as biofactories but also capable of differentiation into force-generating cardiomyocytes intended to boost the contractile machinery of the infarct-related zones.
This objective is rationalized by the limited efficacy of adult cells in cardiac cell therapy clinical trials, which suggests that paracrine mechanisms may not be powerful enough to effect a true regeneration of extensively scarred myocardium. Among cells endowed with a cardiomyogenic differentiation potential, endogenous cardiac stem/progenitor cells generate a great deal of interest. However, despite the promising results reported by the interim analysis assessing the effects of c-kit+ cardiac stem cells in patients (3), serious doubts persist over the real presence of cardiac progenitor cells in the human diseased heart beyond the first months of life (23), and it is indeed surprising that the V-Kit Hardy-Zuckerman 4 feline sarcoma viral oncogene homolog positive (c-kit+) cardiac stem cells used in the above-mentioned trial are reported to express not more than 1.1% of the myocyte, endothelial, and smooth muscle cell lineage markers. For this reason, we have rather focused on human ESCs whose pluripotent state allows for a directed and controlled differentiation toward the desired lineage. In this study, we used SSEA-1+ cardiovascular progenitors that have been previously shown to be able to differentiate into cardiomyocytes and to repopulate substantial areas of scarred myocardium in a nonhuman primate infarction model (2). However, although human donor cells were present in cultured patches prior to transplantation, we failed to find them after the procedure.
Several hypotheses can be put forward to account for this lack of engraftment. First, the cross-talk between the two cell populations may have impaired the progenitor viability. When cultured onto irradiated mouse embryonic fibroblasts, SSEA-1+ cardiovascular progenitors proliferate and cluster, a prerequisite for their differentiation into cardiomyocytes. However, in this study, they were cocultured with mitotically active ADSCs, as demonstrated by the rapid coverage of the patch by these cells after only a few days. This might have deprived the progenitors of some of the growth factors present in the medium during the preimplantation phase of patch culture (because of a competitive uptake by the ADSCs), thereby mitigating their ability to subsequently grow and survive. Thus, although the concept of coculturing two synergistically acting cell populations looks sound, the expected benefit from these interactions likely requires an appropriate match between the candidate cells. A second likely reason for the lack of identifiable human cells could be rejection of the xenograft in this rat model. Because of the planned long duration of the follow-up (6 months), concerns over daily injections of cyclosporine led us to use subcutaneously implanted micro-osmotic pumps preset to release the drug in a controlled fashion. However, random checks of serum drug levels unexpectedly revealed extremely variable values, thereby raising the possibility of an inappropriate handling of the expected immune response to the human progenitor cells. Although this response could have been addressed by the tolerogenic effect of ADSCs, which we have demonstrated in vitro under culture conditions similar to those used in the present in vivo experiments (5), this happened not to be the case as cotransplantation of ADSCs failed to protect the SSEA-1+ cardiovascular progenitors from a likely rejection. This observation is indeed consistent with previous findings from our laboratory showing that mesenchymal stem cells codelivered with ESC cardiac-committed derivatives improve graft viability through trophic rather than immunoprotective effects (24). Whether the stromal cells had an adverse effect on the immunosuppressive efficacy of cyclosporine, similar to that reported for tacrolimus and rapamycin (12), is another unsettled issue. Nevertheless, whereas it still looks sound to complement the transplantation of cardiomyogenesis-targeted cells by stromal cells for enhancing graft viability, it seems hazardous to exclusively rely on these support stromal cells to control rejection, particularly in view of a potential milieu-induced phenotypical switch of the engrafted mesenchymal cells from an immunoprotective to an immune-enhancing pattern (16).
It is fair, however, to acknowledge that this rejection hypothesis could only partially explain the lack of cardiac progenitor cell engraftment because the nude rat experiments showed that even in this setting, these cells could not yet be identified in contrast to ADSCs. Although one might conclude that the disappearance of the cardiac progenitors makes them useless and that patches made of ADSCs exclusively should be effective enough to paracrinally improve postinfarct LV function (13), this assumption must be tempered by other findings from the same group (17) showing that the efficacy of composite sheets made of ESC-derived cardiomyocytes, endothelial cells, and mural cells are indeed largely contributed by the presence of the cardiomyocytes even though the engraftment of the latter cells starts to decrease early after transplantation. Clearly, the mechanism of action and fate of the two populations (ADSCs and cardiac progenitors) used in the present study would have been more accurately dissected by studying patches made of each of these populations alone. However, this was unfortunately not possible because the early developmental stage of the SSEA-1+ cardiac progenitors, and the attendant immaturity of their adhesion molecule expression, prevented their direct attachment to the collagen film and required them to be seeded onto the collagen-bound layer of ADSCs. Similar observations have been made by Matsuura et al. (19), who failed to generate sheets of mouse ESC-derived cardiomyocytes alone and were forced to coculture them with fibroblasts to form the constructs.
In summary, these data support the effectiveness of epicardial patches for ensuring a sustained improvement of postinfarction myocardial function. Although our data do not allow us to determine the respective contribution of the two patch-seeded cell populations to this positive outcome, the persistence of ADSCs after 2 months in the nude rat experiments contrasting with the lack of ESC-derived cardiac progenitors suggests that, at least, the former likely played a major paracrine role. Because generation of new cardiomyocytes remains a logical goal for further improving postinfarction function, these data justify the ongoing search for optimizing the persistent engraftment of ESC-derived cardiac progenitors through preimplantation prosurvival strategies, optimal match with cotransplanted trophic cells, and more efficient control of immune alloresponses (8).
Footnotes
Acknowledgments
This work was supported by INSERM, the LeDucq Foundation (SHAPEHEART network II CVD 02), the LabEx_REVIVE (ANR-10-LABX-73), the Agence de la Biomedecine, the Fondation Desmarets, and the Fondation de l'Avenir (grant numbers ET9-547 and ET2-658). H.H. was supported by fellowships from the Agence Universitaire de la Francophonie, the Fondation LeDucq, and the Foundation BNP Paribas. The authors declare no conflicts of interest.
