Abstract
Islet cell transplantation as a therapy for type 1 diabetes has been limited by progressive graft loss. Significant immunosuppression including T-cell ablation has been used in an attempt to limit islet rejection. Here, we show that CD3+ lymphocytes depleted of alloreactive T cells selected from a mixed lymphocyte reaction (MLR), where responder BALB/c splenocytes stained with carboxyfluorescein succinimidyl ester (CFSE) were stimulated with irradiated C57BL/6 splenocytes for 5 days, infused into diabetic immunodeficient mice are capable of restoring a broad T-cell repertoire and specifically do not reject islet transplants from the strain (C57BL/6) used in the original depletion. These mice demonstrate reconstitution with CD4+ and CD8+ T cells, the capacity to reject third-party grafts (CBA), and restoration of interferon-γ (IFN-γ) responses to third-party alloantigens. Over time, both forkhead box P3-positive (Foxp3+) T regulatory cells (Tregs) and γδ T cells expand, suggesting a role for peripheral tolerance, in addition to the initial depletion of alloreactive T cells, in long-term islet graft survival. Our results suggest that immune restoration with CD3+ lymphocytes where alloreactive T cells are removed can restore cognate immunity without islet allograft loss and recurrence of diabetes.
Keywords
Introduction
Islet cell transplantation is the only curative therapy for type 1 diabetes (8, 10, 27). Under current immunosuppressive regimens, up to 80% of recipients achieve insulin independence at 1 year. However, the success of this therapy has been limited by ongoing graft loss, with the success rate after 5 years falling to 10% (31). This loss appears in part to be immune mediated due to cellular rejection despite ongoing immunosuppression (28). Further, immunosuppressive treatments have enhanced risks of malignancy and infection (2, 3, 30). Clinically, T-cell-depleting strategies are being used for islet cell transplantation. Anti-cluster of differentiation 54 (CD54) antibody has been used in solid organ transplantation and is part of many islet transplant protocols (4). However, the return of memory T cells requiring ongoing immunosuppression and relative immunodeficiency following immune ablation increases infectious and cancer risks in these patients (25). Thus, the ability to reconstitute the peripheral immune repertoire with memory T cells lacking alloreactivity may allow reduction of immune suppression, limiting infection and cancer risk.
Alloreactive T cells make up around 1–10% of the T-cell pool (33). High-affinity alloreactive T cells proliferate rapidly in response to donor stimulation (23). To achieve tolerance, alloreactive T cells need to be deleted, inactivated, or regulated while an environment that allows for the expansion of nonalloreactive T cells is promoted (16). In both bone marrow transplantation (BMT) and solid organ settings, strategies have been used in vivo and in vitro to specifically deplete allospecific T cells based on surface markers (14, 32). Significant limitations involved in targeting activation markers CD25 and CD69 include nonspecificity, in which other activated cells, including T regulatory cells (Tregs), may be depleted. The escape of activated cells that have downregulated their activation markers may also occur. Rather than target alloreactive T cells through surface markers, it is possible to functionally target alloreactive T cells using cell division (33). We have developed a novel strategy to remove alloreactive T cells in an ex vivo mixed lymphocyte reaction (MLR) system using carboxyfluorescein succinimidyl ester (CFSE) to identify proliferating cells (9, 39). Removal of allostimulated CD4+ dividing cells prolongs donor allograft survival in major histocompatibility complex (MHC) mismatched skin [median survival time (MST) = 69] and heart transplant models in mice (MST > 100), although chronic fibrosis is found in long-term surviving cardiac allografts and skin grafts (9, 39). This was associated with the skin grafts with persistent alloreactive T cells through the indirect pathway but not the direct pathway.
Immune restoration with lymphocyte infusions has been used predominantly in leukemia where donor lymphocytes are given to deliver an antitumor effect while limiting graft-versus-host disease, and lymphocyte infusions have been used to restore immunity in a number of conditions (29). We evaluate full T-cell immune reconstitution with CD3+ T cells where the alloreactive cells have been removed (“pruning”) as a therapeutic strategy to prevent pancreatic islet allograft rejection while maintaining the ability to reject third-party grafts across a major MHC mismatch in diabetic mice.
Materials and Methods
Animals
BALB/c (H-2d), C57BL/6 (H-2b), CBA (H-2k), and severe combined immunodeficient (SCID) (H-2d) mice were obtained from the Animal Resource Centre (Perth, Western Australia, Australia). Female mice aged 8-12 weeks were used in all experiments. All mice were housed in filter-top cages in specific pathogen-free conditions, and animal ethics for this study was approved by the Sydney West Area Health Service Animal Ethics Committee.
MLR and Cell Sorting
BALB/c splenocytes stained with CFSE (BioScientific Pty. Ltd., Sydney, NSW, Australia) were stimulated with irradiated C57BL/6 splenocytes in an MLR for 5 days as described previously (9). At day 5, cells from the MLR were stained with anti-mouse CD3-phycoerythrin (PE) antibody (BD Pharmingen, North Ryde, NSW, Australia). The cells were sorted into CFSElowCD3+ T cells (dividing CD3+ T cells) and CFSEhighCD3+ T cells (nondividing allodepleted CD3+ T cells) on a FACSDiva (BD Biosciences, North Ryde, NSW, Australia). In the control group, unstimulated naive BALB/c splenocytes were stained with anti-mouse CD3-PE antibody, and naive CD3+ T cells were selected.
Reconstitution and Flow Cytometric Analysis
SCID mice were injected intravenously with 5×105 sorted dividing CD3+ (DCD3), nondividing allodepleted CD3+ (NDACD3), or naive CD3+ T cells. Sorted and harvested cells from the MLR were analyzed on a FACScan (BD Biosciences) for purity and immune profile. Reconstitution of the peripheral blood (PB) and spleens of SCID was confirmed by flow cytometry as described previously (9). Antibodies used included anti-mouse CD3-PE, CD3-peridinin chlorophyll protein (PerCP), CD4-PerCP, CD4-fluorescein isothiocyanate (FITC), CD8-PerCP, CD25-PE, CD69-PE, CD44-PE, CD45-Percp, H2Kb-PE, H2Kd-PE, T-cell receptor γδ (TCRγδ)-PE (BD Pharmingen), and forkhead box P3 (Foxp3)-PE (eBioscience, San Diego, CA, USA).
Islet Isolation and Transplantation
Mouse pancreatic islet cells were isolated by Histopaque (Sigma-Aldrich Pty. Ltd., Castle Hill, NSW, Australia) density gradient after collagenase digestion (Roche, Dee Why, NSW, Australia) as described previously (35). SCID mice were made diabetic with a single injection of streptozotocin (190 mg/kg IP) (Sigma-Aldrich Pty. Ltd., Castle Hill, NSW, Australia) 8 days before transplantation. Blood glucose level (BGL) was measured with glycemic strips and Accucheck III blood glucose monitor system (Roche). Only mice with BGL ≥16 mmol/L were transplanted. Diabetic SCID mice were transplanted with C57BL/6 (donor) or CBA (third-party) islets under the left renal capsule with approximately 800 mouse islets per transplant. Successful islet function was defined by a BGL <10 mmol/L, and allograft rejection by a BGL >16 mmol/L (35).
The Pancreatic Islet Transplant Model
MLR was performed prior to transplantation. On day 5 of the MLR, cells were sorted into DCD3 and NDACD3 T cells, and then 5 × 105 cells were adoptively transferred into SCID mice. SCID mice were injected with streptozotocin 8 days before transplant. The diabetic SCID mice were transplanted with donor C57BL/6 islet allografts. A separate group of recipients that were infused with NDACD3 T cells were transplanted with third-party CBA islet allografts to ascertain the specificity of the response. Nephrectomy was performed >100 days posttransplantation to confirm islet allograft function.
Histological Examination of Islet Allografts
Kidneys containing the islet grafts were removed at selected time points after transplantation. Allografts were frozen in OCT compound (Tissue TCK, Miles Scientific, Elkhart, IN, USA) and stored at −80°C. Cryostat sections (5-7 mm) were used for a routine hematoxylin and eosin (H&E) and immunohistochemical staining of allografts for the detection of insulin secretion and for infiltration of CD3, CD4, and CD8 T cells. For insulin staining, the guinea pig anti-insulin (Zymed, San Francisco, CA, USA) and rabbit anti-guinea pig immunoglobulin (Dako Corp., Noble Park, VIC, Australia) secondary antibodies (35) with the R.T.U. Vectastain universal ABC kit (Vector Laboratories, Inc, Burlingame, CA, USA) were used according to the manufacturer's instructions. For allograft infiltration of T cells, primary anti-mouse CD3 (17A2) (eBioscience), anti-mouse CD4 (L3T4) (BioLegend, San Diego, CA, USA), and anti-mouse CD8 (Ly-2) (BD Pharmingen) were used as described previously (9, 38).
Interferon-g (IFN-γ) Enzyme-Linked Immunospot (ELISPOT) Assay
ELISpotPLUS for mouse interferon-γ (IFN-γ) (MABTECH AB, Nacka Strand, Sweden) was used for detecting IFN-γ production and performed according to the manufacturer's instructions as described previously (9). Briefly, 3×105 splenocytes isolated from previously reconstituted recipient SCID mice of third-party CBA or donor C57BL/6 islet allografts were stimulated with 3×105 C57BL/6 (donor), CBA (third-party), or BALB/c (self) splenocytes in 96-well plates pretreated with IFN-γ antibody. After incubation for 24 h at 37°C, the cells were removed and the plates were washed. A second biotinylated anti-mouse IFN-γ was added for 2 h at room temperature. Plates were washed, streptavidin-ALP was added, and plates were incubated at room temperature for 1 h. Plates were developed with BCIP/ NBT-plus solution. Plates with spots were counted using the computer program AID Elispot6.0iSpot ELISPOT reader (AID GmbH, Strassberg, Germany).
Real-Time RT-PCR of TCR Vβ Repertoire and CDR3 Spectratyping of TCR Vβ Families
Real-time reverse transcription-polymerase chain reaction (RT-PCR) of TCR Vβ (TRBV) repertoire (22 families) by TaqMan Gene Expression Assay (Applied Biosystems, Carlsbad, CA, USA) and complementary determining region 3 (CDR3) spectratyping of these TRBV families on the ABI Prism 310 Genetic Analyzer (Applied Biosystems) were used to analyze the TRBV repertoire diversity in T-cell subsets and to screen clonal expansion of T cells as described previously (9, 39). TRBV repertoires and CDR3 spectratyping were assessed in splenocytes of reconstituted DCD3 and NDACD3 T-cell recipient SCID mice transplanted with donor C57BL/6 islet allografts.
Statistical Analysis
PRISM 4.0 software (GraphPad, San Diego, CA, USA) was used for statistical analysis for comparisons of matched groups to detect significance. The log-rank test was employed for comparison of survival data between groups. Survival is shown by MST. ANOVA comparison of means was used to compare the percentage of CD3+ T cells and proportion of different types of T cells in vivo; p < 0.05 was considered significant. ELISPOT data were compared by two-tailed Student's t test with a Bonferroni correction for multiple analyses; p < 0.05 was considered significant for single comparisons, p < 0.025 for two comparisons, etc.
Results
MLR Cell Phenotype
On day 5 of the MLR, we analyzed the immune phenotype of cultured cells prior to sorting. The responding BALB/c CD3+ T cells were segregated into two populations, DCD3 and NDACD3 T cells (Fig. 1A), on day 5 of MLR. The percentages of NDACD3, nondividing allodepleted CD4+ (NDACD4), and nondividing allode-pleted CD8+ T cells were 43.6+4.1%, 45.3±1.0%, and 38.3±5.4% (Fig. 1A-C), respectively.
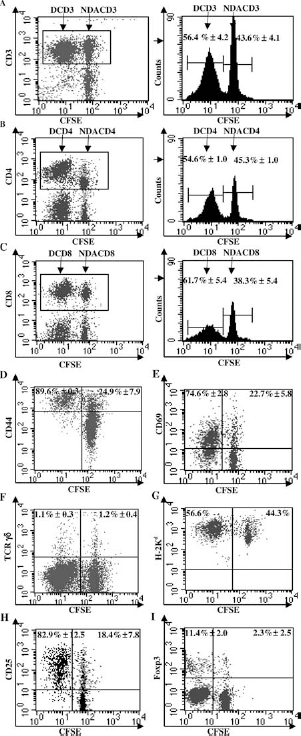
Flow cytometric analysis of MLR cell phenotype. Flow cytometric analysis showing the percentages of (A) low carboxyfluorescein succinimidyl ester cluster of differentiation 3 positive [CFSElowCD3+; dividing CD3 (DCD3)] (56.4+4.2%) and CFSEhighCD3+ (nondividing allodepleted CD3; NDACD3) (43.6±4.1%) T cells, (B) DCD4 (54.6±1.0%) and NDACD4 (45.3±1.0%) T cells, and (C) DCD8 (61.7±5.4%) and NDACD8 T cells (38.3±5.4%) relative to total CD3+, CD4+, or CD8+ T cells. After gating on CD3+ T cells, (D) the percentages of CD44+ were 89.6±0.3% of DCD3 and 24.9±7.9% of NDACD3 T cells, and (E) the percentages of CD69+ were 74.6±2.8% of DCD3 and 22.7±5.8% of NDACD3 T cells. (F) The percentages of T-cell receptor γδ+ were 1.1±0.3% in DCD3 and 1.2±0.4% in NDACD3 T cells. (G) No stimulatory H-2Kd- cells were found in both DCD3 and NDACD3 T cells. After gating on CD4+ T cells, (H) the percentages of CD25+ were 82.9±12.5% of DCD4 and 18.4±7.8% of NDACD4 T cells, and (I) the percentages of forkhead box P3 positive (Foxp3+) were 11.4±2.0% of DCD4 and 2.3±2.5% of NDACD4 T cells [data are representative of three separate mixed lymphocyte reaction (MLR) experiments].
DCD3 T cells expressed higher levels of CD44 (89.6±0.3%) (Fig. 1D) and CD69 (74.6±2.8%) (Fig. 1E) than NDACD3 T cells. The nondividing CD3+CD44+ and CD3+CD69+ proportions were 24.9±7.9% (Fig. 1D) and 22.7±5.8% (Fig. 1E) of total NDACD3 T cells. There were similar percentages of γδ+ T cells in both DCD3 (1.1±0.3%) and NDACD3 (1.2±0.4%) T-cell populations (Fig. 1F). H-2Kd cells (Fig. 1G), but not stimulatory H-2Kb cells (data not shown), were found in either the DCD3 or NDACD3 T-cell populations after MLR. Dividing CD4+ (DCD4) T cells had higher levels of the activation marker CD25 (82.9±12.5%), compared to NDACD4 T cells (18.4±7.8%) (Fig. 1H). Interestingly, there was a relative decrease in CD4+Foxp3+ Tregs (2.3±2.5%) in the NDACD4 T cells after the MLR (Fig. 1I) compared to naive BALB/c (15.3±3.1%) (data not shown).
Long-Term Islet Allograft Survival After Reconstitution with NDACD3 T Cells
To test whether NDACD3 T cells could allow islet allograft survival, SCID mice reconstituted with either DCD3 or NDACD3 T cells were transplanted with donor C57BL/6 islet allografts. SCID mouse recipients (n = 7) of C57BL/6 islet allografts reconstituted with NDACD3 T cells demonstrated prolonged islet allograft survival (MST > 100 days) compared with SCID mouse recipients (n = 5) reconstituted with DCD3 T cells that rejected C57BL/6 islet allografts (MST = 6 days, p < 0.05) (Fig. 2). Control SCID mice (n = 3) reconstituted with naive CD3+ T cells rejected their C57BL/6 islet allografts (MST = 32 days p < 0.05) (Fig. 2). Further, SCID mice (n = 2) reconstituted with NDACD3 T cells rejected third-party CBA islet allografts (MST = 10.5 days, p < 0.05) (Fig. 2). This demonstrates that NDACD3 T cells do not reject MHC mismatched islet allografts while retaining the ability to reject third-party allografts.

Reconstitution with NDACD3 T cells led to long-term specific islet allograft survival. Severe combined immunodeficient (SCID) mice reconstituted with NDACD3 show long-term specific C57BL/6 islet allograft survival (white circle) [median survival time (MST) >100 days, n = 7] compared to DCD3-SCID mice that rejected C57BL/6 islet allografts (black triangle) (MST = 6 days, n = 5) (p < 0.05), naive CD3-SCID mice that rejected C57BL/6 islet allografts (white square) (MST = 32 days, n = 3) (p < 0.05), and NDACD3-SCID mice rejected third-party CBA islet allografts (black diamond) (MST = 10.5 days, n = 2) (p < 0.05) (data are from three separate experiments).
Histology Demonstrates Intact Islet Grafts with Limited Infiltrate in the NDACD3 Group
Long-term islet allografts were evaluated for the presence of infiltrating CD3+, CD4+, and CD8+ T cells and insulin by immunohistochemistry (Fig. 3). Histology of C57BL/6 islet allografts from SCID mice reconstituted with DCD3 T cells demonstrated massive cellular infiltration (Fig. 3A, C, E, G, I) and loss of insulin staining (Fig. 3C). In contrast, long-term islet allografts had no T-cell infiltrates, and a large number of intact islets were found in long-term allografts, with an occasional T cells at the periphery of the islet allografts (Fig. 3B, D, F, H, J), showing strong staining for insulin (Fig. 3D).

Histological assessment of islet allografts by H&E staining and immunohistochemical staining for islet insulin content, infiltrating CD3+ T cells, CD4+ T cells, and CD8+ T cells. (A) H&E, (C) insulin, (E) CD3+, (G) CD4+, and (I) CD8+ are islet allografts from SCID recipient reconstituted with DCD3 and loss of graft function was observed 22 days posttransplantation. (B) H&E, (D) insulin, (F) CD3+, (H) CD4+, and (J) CD8+ are long-term islet allografts from SCID reconstituted with NDACD3 on day 84 posttransplantation. (A, C, E, G, I) There was greater cellular infiltration and no insulin staining in rejecting C57BL/6 allografts. (B, D, F, H, J) No cellular infiltration, minimal T cells surrounding the edge of allografts, and a large number of intact islets present with intense insulin staining in long-term allografts.
Successful T-Cell Reconstitution in All Groups
To determine whether T-cell reconstitution was successful, we examined SCID mouse peripheral blood (PB) and spleen by flow cytometry for the percentage of CD3+ T cells in the CD45+ subset. A control dot blot from the PB of SCID mice before reconstitution is shown (Fig. 4A). Representative dot blots show the CD3+CD45+ T cells in the PB of SCID mice reconstituted with naive CD3+ (6.9%), DCD3 (5.2%), and NDACD3 (7.4%) T cells 16 days after reconstitution (Fig. 4B–D). Although there was a difference initially (16 days after reconstitution), they were similar 24 days after reconstitution in all groups (Fig. 4E). Further, after transplantation, there was also no significant difference in reconstitution between groups (Fig. 4F). The percentage of CD3+CD45+ increased over time (Fig. 4E, F). The equivalent level of reconstitution between groups suggests that differences in survival are not due to differences in reconstitution.

Successful T-cell reconstitution in SCID mice in all groups. Dot plot showing (A) the absence of CD3+ T cells in control unreconstituted SCID mice (0.5 ± 0.1%, n = 3), and the proportion of CD3+CD45+ T cells in peripheral blood (PB) of SCID reconstituted with (B) naive CD3 (CD3) (6.9%), (C) DCD3 (5.2%), and (D) NDACD3T cells (7.4%) day 16. (E) Graph showing the percentage of CD3+CD45+ T cells in PB of SCID reconstituted with naive CD3+ (5.3 ± 1.9%) (p < 0.05) (n = 3), DCD3 (4.2 ± 2.6%) (p < 0.05) (n = 9), and NDACD3 (8.3 ± 2.1%) (n = 11) on day 16 of reconstitution, and naive CD3+ (5.3 ± 1.3%) (n = 3), DCD3 (3.5 ± 0.5%) (n = 6), and NDACD3 (5.8 ± 1.4%) (n = 3) on day 24 of reconstitution. (F) Percentage of CD3+CD45+ T cells in PB and spleens of receipt SCID mice reconstituted with naive CD3+ T cells on days 33–38 posttransplantation (13.9 ± 6.4% in PB, 26.5 ± 13.3% in spleens) (n = 3), DCD3 (8.4 ± 4.6% in PB, 12.4 ± 6.7% in spleens) (n = 5) and NDACD3 (10.8 ± 7.2% in PB, 22.2 ± 16.3% in spleens) (n = 4) on days 7–26 posttransplantation, and NDACD3 on day ≥84 posttransplantation (18.0 ± 13.4% in PB, 15.5 ± 6.2% in spleens) (n = 4 in PB, n = 3 in spleen) (data are from three separate experiments).
CD4+ and CD8+ T-Cell Subsets in All Groups of SCID Mouse Recipients
To investigate whether there were differences among T-cell subsets of reconstitution, CD4+ and CD8+ T cells were assessed in the PB and spleen of SCID mouse recipients. Both CD4+ and CD8+ T-cell subsets were reconstituted in SCID mice in all groups (Table 1). There was a similar proportion of the CD8+ T-cell subset in the DCD3 (4.7 ± 4.5% in PB and 4.9 ± 4.4% in spleen) and NDACD3 T cells (4.6 ± 1.6% in PB and 3.9 ± 2.0% in spleen) of SCID mouse recipients (Table 1). Although there was an initial decrease in the CD8+ T-cell subset in the DCD3 and NDACD3 groups as compared with naive CD3+ T-cell reconstituted SCID mouse recipients (17.0 ± 4.7% in PB and 19.2 ± 7.1% in spleen) and naive BALB/c mice (21.7 ± 6.0% in PB and 24.2 ± 8.0% in spleen), over time the proportion of CD8+ T cells in CD3+ T-cell reconstituted SCID mouse recipients surviving long-term was restored (12.6 ± 7.0% in PB and 16.8 ± 8.3% in spleen) (Table 1). The equivalent level of reconstitution between groups suggests that differences in survival are not due to differences in reconstitution of CD4+ and CD8+ T-cell populations.
Proportion of CD4+ and CD8+ T Cells in Recipient SCID Mice After Transplantation
Days posttransplantation (all data from three separate experiments). CD4, cluster of differentiation 4; SCID, severe combined immunodeficient; PB, peripheral blood; DCD3, dividing CD3+ cells; NDACD3, nondividing allodepleted CD3+ cells.
Diversity of the T-Cell Repertoires After Reconstitution in Both the DCD3 and NDACD3 Groups
In order to investigate the diversity of the T-cell repertoire after reconstitution, TCR Vβ (TRBV) repertoires and CDR3 spectratyping of TRBV families were assessed in both spleens of SCID mouse recipients of C57BL/6 allografts reconstituted with DCD3 T cells (DCD3-C57BL/6) and NDACD3 T cells (NDACD3-C57BL/6) (Fig. 5). Both groups were reconstituted with almost a full repertoire of TRBV families (except TRBV23) (Fig. 5A). However, CDR3 spectratyping for CDR3 length of the five TRBV families that had greater than 5% (Fig. 5B) and the other 16 TRBV families (less than 5%) (data not shown) of the repertoire showed a broad Gaussian distribution in all mice reconstituted with NDACD3 T cells. This suggests a very diverse TCR repertoire as compared to the mice reconstituted with DCD3 T cells that showed skewed CDR3 distributions and fewer peaks suggestive of a degree of TCR repertoire restriction and oligoclonality.

Almost complete TRBV restoration after reconstitution in both the DCD3 and NDACD3 groups of SCID mice. (A) Real-time RT-PCR showed T-cell receptor Vβ (TRBV) repertoires in spleens of receipt SCID mice of C57BL/6 allografts reconstituted with DCD3 T cells at the time of rejection and NDACD3 T cells on day ≥84 posttransplantation (a pool of three mice for each group). Twenty-one of 22 TRBV families (except TRBV23) were detected in both groups. (B) Complementary determining region 3 (CDR3) spectratyping of TRBV families, from highly represented families (>5%) in both spleens of DCD3-SCID mice at the time of rejection and NDACD3-SCID mice on day ≥84 posttransplantation (3 mice each group), shows diversity in the NDACD3 group versus oligoclonality and skewing in the DCD3 group.
Increased Expression of Foxp3 and the Proportion of γδ T Cells in Mice with Long-Term Islet Transplants
We noted two potential mechanisms for regulation in addition to the removal of alloreactive T cells. First, an expansion of Tregs over time in the long-term islet allograft recipients; second, an expansion of CD3+γδ+ T cells. In order to determine whether Tregs had a role in prolonging allograft survival in SCID mice reconstituted with NDACD3 T cells, the populations of CD4+CD25+ and CD4+Foxp3+ were assessed in all groups. After MLR, there was a relative loss of CD4+Foxp3+ Tregs in both DCD4 and NDACD4 T cells (Fig. 1I). High levels of CD25 in the DCD4 T cells (82.9 ± 12.5%) and also significant expression of CD25 in the NDACD4 T cells (18.4 ± 7.8%) after MLR were observed, reflecting its role as an activation marker (Fig. 1H). The proportions of CD4+CD25+ T cells in PB of SCID mouse recipients of the DCD3-C57BL/6 group at the time of rejection (5.3 ± 1.1%) and the NDACD3 group at matching time points (4.3 ± 1.4%) (Fig. 6) were similar and significantly less than that seen in the MLR. However, in SCID mouse recipients of NDACD3-C57BL/6, the proportion of CD25+ T cells increased and graft survival increased over time (11.0 ± 3.1%) (Fig. 6A).
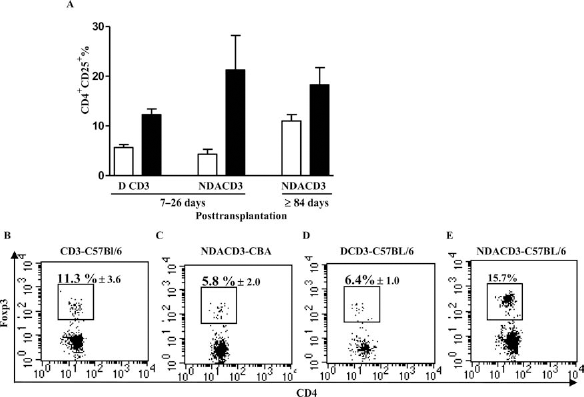
Islet allograft survival is associated with increased expression of Foxp3. (A) The proportion of CD4+CD25+ T cells in PB and spleens of recipient SCID mice of C57BL/6 allografts reconstituted with DCD3 (5.3 ± 1.1% in PB, 12.2 ± 1.6% in spleen) (n = 4) (at the time of rejection) and NDACD3 (4.3 ± 1.4% in PB, 21.3 ± 9.8% in spleen) (n = 2) (the match time point) on days 7–26 posttransplantation, and NDACD3T cells on day ≥84 posttransplantation (11.0 ± 3.1% in PB, 18.2 ± 8.4% in spleen) (n = 3). The proportion of CD4+CD25+ T cells was increased in the PB of the NDACD3-C57BL/6 group (≥84 days) compared to the DCD3-C57BL/6 and NDACD3-C57BL/6 groups (7–26 days). (B) The proportion of CD4+Foxp3+ Tregs was highest in the spleen of the (E) NDACD3-C57BL/6 group on day 26 posttransplantation (15.7%) compared to the (B) CD3-C57BL/6 (11.3 ± 3.6%) (n = 3), (D) DCD3-C57BL/6 (6.4 ± 1.0%) (n = 3) (at matched time points), and (C) NDACD3-CBA groups (5.8 ± 2.0%) (n = 2) at the time of rejection.
We further found that the expression of Foxp3+ in total CD4+ T cells was the highest in SCID mouse recipients of C57BL/6 allografts reconstituted with NDACD3 T cells (15.7%) (Fig. 6E) compared to those reconstituted with DCD3 (6.4 ± 1.0%) (Fig. 6D) or naive CD3+ T cells (11.3 ± 3.6%) (Fig. 6B), or in SCID mouse recipients of third-party CBA allografts reconstituted with NDACD3 (NDACD3-CBA) (5.8+2.0%) (Fig. 6C) at the time of rejection or matching survival time. Therefore, SCID mouse recipients reconstituted with NDACD3 had a higher proportion of Tregs, suggesting that selective expansion of Tregs had a role in the long-term acceptance of islet allografts.
γδ T cells have been shown in a variety of models to be capable of regulatory functions. This subset of predominantly tissue-based T cells can have both regulatory and inflammatory properties (11, 12, 40). We found a high level of γδ+ T cells in the PB from SCID mice with long-term islet allografts (Fig. 7). On day 15 posttransplantation, SCID mouse recipients of C57BL/6 allografts reconstituted with NDACD3 T cells showed a normal proportion of γδ+ T cells in PB (1.0%) (Fig. 7E), which was similar to that found in naive BALB/c mice (1.2%) (Fig. 7A). Similar proportions of γδ+ T cells were found in the DCD3 (1.1+0.3%) and NDACD3 T cells (1.2+0.4%) after MLR (Fig. 1F). In long-term survivors posttransplant, γδ+ T cells as a proportion of CD3+ T cells were increased significantly in SCID mouse recipients of C57BL/6 allografts reconstituted with NDACD3 T cells (10.4+3.6%) (Fig. 7F). This increase was significantly greater than that seen in all other groups at the time of rejection, including SCID mouse recipients of C57BL/6 allografts reconstituted with naive CD3+ T cells (2.6±1.2%) (p < 0.05) (Fig. 7B) or dividing CD3+ T cells (2.1±0.8%) (p < 0.05) (Fig. 7D) and SCID mouse recipients of third-party CBA allografts reconstituted with nondividing CD3+ T cells (1.9+0.3%) (p < 0.05) (Fig. 7C). This suggests that the γδ T cells may have a role in prolonging allograft survival in this model.

Islet allograft survival is associated with a high proportion of γδ T cells. Flow cytometric analysis of γδ+ T cells showed (A) the proportion of γδ+CD3+ T cells was 1.4% in PB in naive BALB/c mice. γδ T cells were significantly increased in PB of (F) NDACD3-C57BL/6 recipient SCID mice on day >84 posttransplantation (10.4+0.6%) (n = 3) compared to the (B) CD3-C57BL/6 group (2.6±1.2%) (n = 3) (p < 0.05), (C) NDACD3-CBA third-party group (1.9±0.3%) (n = 2) (p < 0.05), and (D) DCD3-C57BL/6 group (2.1±0.8%) (n = 4) (p < 0.05) at the time of rejection (days 7-22 posttransplantation) and to the (E) NDACD3-C57BL/6 group on day 15 posttransplantation (1.0%).
Immune Response to Donors and Third Party
IFN-γ ELISPOT assay was used to determine the number of IFN-γ T cells induced by direct alloactivation (Fig. 8). In SCID mouse recipients of C57BL/6 allografts reconstituted with NDACD3 T cells, the number of IFN-γ spots was lower after donor stimulation compared to third party (Fig. 8D). In contrast, the IFN-γ response in recipients of C57BL/6 allografts reconstituted with naive (Fig. 8A) or DCD3 (Fig. 8C) T cells was stronger to donor stimulation than third party. Interestingly, mice reconstituted with NDCD3 T cells that received third-party CBA allografts mounted strong responses against both CBA and the stimulator strain C57BL/6 (Fig. 8B).

Reduced IFN-γ production in the NDACD3 group. ELISPOT assay for interferon (IFN)-γ production by splenocytes from CD3-C57BL/6, DCD3-C57BL/6, and NDACD3-CB A recipient mice at the time of allograft rejection and recipient mice with functional C57BL/6 allografts reconstituted with NDACD3 T cells on day >100. In all four groups, there were significant differences in IFN-γ production between cells stimulated by allogeneic C57BL/6 and syngeneic BALB/c (*p < 0.05, **p < 0.001) and between allogeneic third-party CBA (3rd) and BALB/c (*p < 0.05). (A) Increasing IFN-γ production was found in response to the stimulator C57BL/6 strain compared with the third-party CBA strain in responder cells from both recipient SCID mice of C57BL/6 allografts reconstituted with naive CD3+ (195 ± 30.5 vs. 131 ± 22.6 spots per 3×105 cells) and (C) DCD3 T cells (138 ± 11.5 vs. 99 ± 21.9 spots per 3×105 cells). (B) There was a similar amount of IFN-γ production when stimulated by either allogeneic stimulator C57BL/6 (285.0 ± 43.1 spots per 3×105 cells) or CBA (303 ± 4.9 spots per 3×105 cells) strain in the third-party NDACD3-CBA group. (D) There was reduced IFN-γ production in response to the stimulator C57BL/6 strain (48 ± 5.6 spots per 3×105 cells) compared with the third-party CBA strain (69 ± 12.0 spots per 3×105 cells) in the NDACD3-C57BL/6 group.
Discussion
We have shown that immune reconstitution with CD4+ T cells depleted of alloreactive CD4 T cells delayed skin graft rejection and allowed long-term cardiac graft survival, but with the development of chronic rejection showing significant fibrosis (9, 39). Here, long-term islet allograft survival and treatment of diabetes were achieved across a major MHC mismatch in mice reconstituted with allodepleted CD3+ T cells in vitro with restoration of diverse CD4+ and CD8+ T-cell repertoires that retain the ability to reject third-party allografts without the need for additional immunosuppression. The lack of fibrosis in islets as compared to that found in skin and heart models suggests that there may be additional pathways of immune regulation in this model potentially related to the broader set of T cells used for reconstitution.
As compared to vascularized grafts, islet grafts showed long-term survival without evidence of chronic rejection even under the more stringent barrier of MHC mismatched CD3+ T-cell reconstitution containing both CD4+ and CD8+ subsets. The better outcomes for islets than previously found in heart or skin transplants following “pruning” has a number of possible explanations. The islets themselves express predominantly class I antigens as compared to vascularized or skin grafts where class II antigens are highly expressed on endothelium (1, 26, 36). However, both CD4+ and CD8+ T cells have been shown to be capable of rejecting islet allografts, making this a less likely explanation. It appears that in islet allograft rejection, CD4+ and CD8+ T cells work together, with CD4+ cells providing help for CD8+ cells through either the direct or indirect pathway (21). In NOD mice, CD4+ T cells alone can mediate rejection (21). Histologically, we find both CD4+ and CD8+ T cells in rejecting allografts in mice receiving DCD3 T cells, suggesting a role for both in rejection, whereas in mice that have received NDACD3 T cells, there is no cell infiltrate and a minimal amount of CD3+ T cells around the allografts.
An additional explanation for long-term allograft survival is inadequate reconstitution or function of the adoptively transferred lymphocytes. However, mice reconstituted with either dividing or nondividing subsets demonstrated homeostatic proliferation with CFSE dye dilution over time (data not shown) and reconstitution of both CD4+ and CD8+ subsets. The functionality of these cells was demonstrated by their capacity to reject third-party grafts and also to generate IFN-γ as part of the rejection response as shown by ELISPOT. Therefore, even with the added functionality caused by homeostatic proliferation, nondividing T cells did not reject grafts from the stimulator strain while retaining the capacity to reject third-party grafts (9).
The development of immune regulation, which was not found in the skin and cardiac models, might also explain the long-term tolerance to the islet allografts. We have previously noted differences in tolerance in equivalent MHC mismatches between kidney (tolerant) and heart (rejected) in the C57BL/6 to B10.BR model (37). We noted two potential mechanisms for regulation in addition to the removal of alloreactive T cells. This may be explained by the broader (CD3+) T-cell repertoire in this study as compared to (CD4+) in the skin and heart studies. The first is the expansion of Tregs over time in the long-term islet allograft recipients; this is a mechanism well described for maintenance of tolerance and may explain the lack of chronic rejection (34). Foxp3+ Tregs can transfer tolerance to mouse islet allografts between recipients (6, 7, 24). Elevated Foxp3 has been shown in tolerant liver allografts (19) and cardiac allografts (17) in different animal models. Foxp3 staining showed Foxp3+ cells in the rejecting grafts and occasional cells at the edge of long-term tolerant grafts (data not shown). This is consistent with the data from thymidine kinase depletion in islet cell transplantation where Treg expansion is also found (5). Our finding that there is increased expression of Foxp3+ Tregs in SCID mice reconstituted with NDACD3 T cells with donor allografts compared with other groups in vivo and NDACD3 after MLR suggests that induced Tregs or expanded naturally occurring Tregs may play a role in islet allograft survival in the model.
The second is the expansion of CD3+γδ+ T cells. This subset of predominantly tissue-based T cells can have both regulatory and inflammatory properties (11, 12, 40). Intraepithelial γδ+ T cells were required for tolerance induction using oral insulin to prevent diabetes in the nonobese diabetic (NOD) mouse (20). In human studies of allograft tolerance, increased numbers of γδ+ T cells are found in the peripheral circulation of both adult and pediatric liver allograft recipients (15, 22). We have also identified regulatory subsets of γδ+ T cells in models of nephritis (40). Similarly, in this islet transplant model, these cells may potentially play a role in islet tolerance. Currently, there are a number of immune ablative strategies and cell therapy strategies, including mixed chimerism and facilitator cells, to achieve human tolerance. The removal of alloreactive T cells by pruning may assist these strategies in human cell and organ transplantation (13, 18).
In conclusion, immune reconstitution with CD3+ lymphocytes depleted of alloreactive T cells promoted long-term allograft survival without rejection despite restoration of CD4+ and CD8+ T cells and a broad T-cell repertoire associated with expansion of γδ+ T cells and Foxp3+ Tregs. This may potentially allow profound lymphocyte depletion at the time of islet cell transplantation followed by reconstitution of the T-cell repertoire with T cells that have been “pruned” to allow restoration of normal immune function without the need for strong immunosuppression.
Footnotes
Acknowledgments
We thank the staff of Westmead Hospital Animal House for animal care, and the flow unit of Westmead Millennium Institute for sorting. This study was supported by a program grant from the Juvenile Diabetes Research Foundation (Grant 4-2006-1025) and National Health and Medical Research Council (NHMRC) of Australia (Grant 512246) and (NHMRC) Training Fellowship APP1013185 (to M.H.). The authors declare no conflict of interest.
