Abstract
Islet transplantation may be the most efficient therapeutic technique for patients with type 1 diabetes mellitus (T1DM). However, the clinical application of this method is faced with numerous limitations, including isolated islet apoptosis, recipient rejection, and graft vascular reconstruction. Mesenchymal stem cells (MSCs) possess anti-apoptotic, immunomodulatory, and angiogenic properties. Here, we review recent studies on co-culture and co-transplantation of islets with MSCs. We have summarized the methods of preparation of co-transplantation, especially the merits of co-culture, and the effects of co-transplantation. Accumulating experimental evidence shows that co-culture of islets with MSCs promotes islet survival, enhances islet secretory function, and prevascularizes islets through various pretransplant preparations. This review is expected to provide a reference for exploring the use of MSCs for clinical islet co-transplantation.
Introduction
Type 1 diabetes mellitus (T1DM) is an autoimmune disease that leads insulin-producing β cells to death, caused by self-reactive T cells, and represents one of the most frequent chronic diseases in children and adolescents; its incidence has increased in recent years. Cell-based therapies can provide alternatives to lifelong insulin administration in patients with T1DM. In the early stages of the disease, adoptive transfer or in vivo expansion of islet antigen-specific regulatory cell, autologous nonmyeloablative hematopoietic stem cell therapy, and administration of tolerogenic dendritic cells are promising and can be effective in maintaining β-cell function. However, in the late stages of the disease, physiologic glycemic control can only be achieved by replacing degenerated cells 1 . Pancreatic islet transplantation can effectively treat T1DM, which provides a minute-to-minute regulation of glucose levels 2 . However, its clinical application is limited by cell loss that occurs before and after islet transplantation and involves various factors such as oxidative stress, instant blood-mediated inflammatory response (IBMIR), immune responses, and vascular reconstruction difficulties 3 .
Clinically Feasible Mesenchymal Stem Cells
Mesenchymal stem cells (MSCs) are the most commonly used cells in cell therapy. The International Society for Cellular Therapy has defined the criteria for MSCs suitable for cell therapy 4 . They are mainly found in the bone marrow and account for 0.001%–0.01% of its cell population. MSCs can also be isolated and extracted from other tissues (e.g., adipose tissue and umbilical cord blood), and their ability to self-renew is not affected by the extraction process 5 . Clinical experiments have proved that systemic injection of allogeneic umbilical cord MSCs6,7 and bone marrow mesenchymal stem cells (BMSCs) 8 can protect islet β cells and delay the complications of T1DM. Moreover, a recent meta-analysis including bone marrow, umbilical cord, and adipose-derived mesenchymal stem cells (ADMSCs) for the treatment of patients with T1DM confirmed the achievement of good clinical efficacy without serious or chronic adverse effects 9 .
In general, MSCs can be used as a promising therapeutic component. Therefore, MSCs not only are easy to access but also have angiogenic potential and can differentiate into multicellular lineages, such as adipocytes, neurons, and pancreatic β cells 10 , all without teratoma formation 11 . Immunosuppressive and anti-apoptotic activities of MSCs increase β-cell viability and function, promote graft neovascularization, and alleviate autoimmune reactions related to graft and graft rejection 12 . Therefore, MSCs may be considered essential helper cells in islet transplantation.
Current Status of Co-transplantation
Currently, the portal vein is the preferred transplantation site because the procedure is minimally invasive, easy to performed, and has low rates of bleeding and thrombosis 13 . Individuals with diabetes increasingly benefit from islet transplantation therapy; so far, more than 1,500 patients have received intraportal islet transplantation in 40 international islet transplant centers 14 . Although the immunosuppressive regimen reported from Edmonton, Canada, has achieved unprecedented success in achieving insulin independence in islet transplantation 15 , islet transplantation through the portal vein is associated with the risks of portal vein thrombosis, elevated portal vein pressure, and bleeding 16 ; moreover, early inflammatory responses significantly influence islet engraftment and survival after intrahepatic transplantation through many modes. This inflammatory response is now commonly referred to as IBMIR 17 . The characteristics of IBMIR include rapid activation of complement, activation of coagulation system, and infiltration of inflammatory cells, all of which can lead to graft death18,19. Therefore, elevated inflammatory cytokines like interleukin (IL)-1β, IL-γ, tumor necrosis factor alpha (TNF-α), and interferon gamma (IFN-γ) were observed during islet transplantation, and macrophages are also involved in regulating the cell injury of transplanted pancreatic islets 20 .
The main clinical strategies to alleviate the islet damage caused by IBMIR include the following: heparin and etanercept and an inhibitor of TNF-α in clinical practice; an inhibitor of CXC chemokine receptor 1/221,22, low-molecular-weight dextran sulfate 23 , and α1-antitrypsin 24 in clinical studies; and islet encapsulation techniques 25 , an inhibitor of nuclear factor-κB 26 , and graft-site alterations27,28 in preclinical studies. It was observed that concomitant transplantation of islets with MSCs in diabetic rats can prolong the survival of islet grafts 6 . According to reports, currently the biggest problem in islet transplantation is that more than half of transplanted pancreatic islets within hours to 3 days will be significant cell loss very soon after administration, leading to poor engraftment and cell function, which seriously affects long-term efficacy 17 . The following mainly discusses the methods of preparation of co-transplantation, especially the merits of co-culture, and the effects of co-transplantation of MSCs and pancreatic islets.
Merits of Co-culture and Co-transplantation of MSCs and Islets
Before being transplanted, islets are isolated, purified, and cultured; however, this can result in islet damage or death. A major challenge related to islet isolation is hypoxic apoptosis: Islet mitochondria are particularly susceptible to hypoxic stress, and impaired mitochondrial mass and function lead to defects in insulin secretion and a decrease in β-cell survival, eventually reducing islet cell survival and function considerably.
Anti-apoptotic
The first challenge to extracted isolated islets is hypoxia loss. Islet death and apoptosis are mainly due to damage during the isolation and purification, prolonged separation time, and lack of microvascular regeneration in the early stage of transplantation (Fig. 1A). MSCs can secrete various bioactive substances that reduce the adverse effects of injury factors on other cells and maintain cell growth and vitality29,30. For example, MSCs improve survival and function of islets by cell-to-cell contact mediated by the adhesion molecule N-cadherin 31 . Many studies have used it as seed cells to repair transplanted pancreatic injury. However, the specific mechanism by which MSCs play an important role in enhancing the anti-hypoxic damage ability of islets is currently unclear. Rackham et al., using an in vitro co-culture system, observed that MSCs transferred mitochondria to islet β-cells, demonstrating that this was associated with an increase in the β-cell’s mitochondrial oxygen consumption and enhanced glucose-induced insulin secretion. Moreover, they also found that mitochondrial transfer from human MSCs to human islets was more extensive than that from rodent MSCs to rodent pancreatic islets 32 . It is most likely because isolated human islets were exposed to more extreme cellular stressors, which initiated mitochondrial transfer to human islets. Chen et al. found that MSCs also attenuated hypoxia-mediated endoplasmic reticulum stress via miR21 and inhibited p38 mitogen-activated protein kinases (MAPK) phosphorylation to protect β-cells from apoptosis 33 . In addition, MSCs also can promote pancreatic hypoxia tolerance via upregulation of hypoxia-inducible factor α 34 . This suggests that the main way to interfere with islet mitochondrial metabolism is to resist apoptosis.
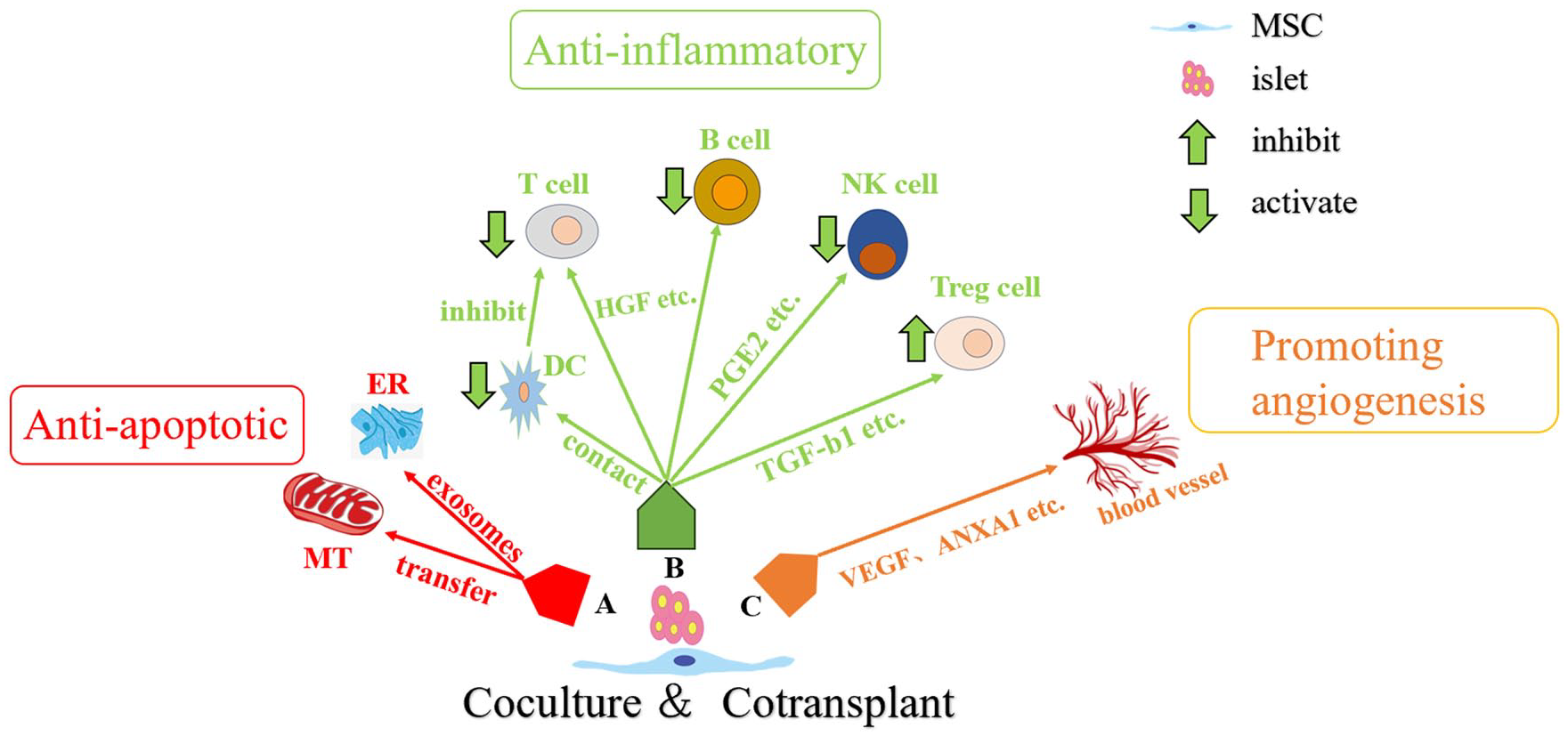
Schematic representation of plausible mechanisms by which MSCs are co-cultured and co-transplanted with islets. (A) MSCs exert anti-apoptotic effects by transferring mitochondria and secreting exosomes into pancreatic islet cells. (B) MSCs exert anti-inflammatory effects by directly contacting immune cells or regulating immune cells through cytokines. (C) MSCs promote angiogenesis through cytokines. MT, mitochondria; ER, endoplasmic reticulum; DC, dendritic cell; NK cell, natural killer cell; Treg cell, regulatory T cell; HGF, hepatic growth factor; PEG, prostaglandin; TGF, transforming growth factor; VEGF, vascular endothelial growth factor; ANXA1, annexin A1.
Anti-inflammatory
The first challenge to islets transplanted into recipients is the attack by the host immune system. The lack of donor antigens, low levels of human leukocyte antigen (HLA) class I, and absence of HLA class II make MSCs a suitable source for transplantation35,36. Very early on, Duprez et al. 37 suggested that the adhesion capacity of MSCs could be utilized to create composite MSC-islets, which would allow for MSC-islet transplantation. This may have beneficial immunosuppressive effects for optimizing islet transplantation. It was reported that MSCs can produce immune-privileged sites when co-transplanted with pancreatic islets without any systemic effects on the recipient’s immune system 38 . A study suggested that MSCs were able to enhance insulin secretion from islets in vitro and maintain heme oxygenase-1 and ferritin H protein levels in a context of cytokine exposure 39 . In addition, MSCs can exert immunomodulatory effects by altering the phenotype of different immune cells, including dendritic cells, T cells, B cells, natural killer cells, and regulatory cells40,41. MSCs act on T cells by physically hindering the contact of antigen-presenting cells or through intercellular contacts hindering dendritic cells’ maturation indirectly inhibiting T-cell activation. Moreover, MSCs are able to inhibit T-cell reactivity and evade cytotoxic T-cell-mediated apoptosis via soluble factors such as transforming growth factor b1 and hepatic growth factor or nitric oxide and downregulation of pro-inflammatory cytokines 42 . Among these, transforming growth factor b1 plays a full role in immunomodulation of MSCs, including a role in regulatory cell induction and/or expansion 43 . In addition, Ishida et al. 44 preactivated MSCs with cytokines (IFN-γ, TNF-α, and IL-1β) and then co-transplanted them with pancreatic islets and found that preactivated MSCs inhibited post-transplanted natural killer cells from pancreatic islets in the portal vein through secretion of prostaglandin 2, thereby improving islet transplantation survival (Fig. 1B). It can be seen that MSCs can to some extent be resistant to inflammatory responses, including IBMIR, but the exact mechanisms involved are complex and unknown and need to be further explored.
Promoting Angiogenesis
The greatest challenge to the long-term survival of islets graft is angiogenesis. Because the isolated islet cells cut off oxygenation of the microvasculature, the transplant is temporarily dependent on the diffusion of nutrients and oxygen to ensure survival. Neovascularization is completed after about 2 weeks, and further remodeling takes up to 3 months. A growing number of experiments have shown that MSCs had the potential to promote angiogenesis (Fig. 1C). These are through the expression and release of different pro-angiogenic cytokines, including vascular endothelial growth factor (VEGF), fibroblast growth factor, transforming growth factor β, annexin A1, matrix metalloproteinases, and angiopoietin-131,45–47. Wang et al. 48 demonstrated that MSCs promoted the expression of islet insulin gene enhancer binding protein 1 (ISL1) and led to an increase in VEGF in islet grafts. Subsequently, MSCs transfected with high ISL1 expression have been utilized to further demonstrate that ISL1 promotes the release of exosomes of aniline from BMSCs, secretion of Inhibin beta A chain and caffeine, and reduction of streptozotocin (STZ)-induced islet apoptosis 49 . In addition, it has also been shown in many studies that islet and MSC co-transplantation shortens the post-transplantation ischemic period by increasing neovascularization50,51.
Overall, MSCs co-transplanted with islets are clinically promising therapeutic approaches because of their potential to be anti-apoptotic, anti-inflammatory, and pro-angiogenic through the secretion of trophic factors. However, it remains to be further verified whether the same effects are observed in clinical trials in particular.
Preparation of Co-transplantation
In recent years, MSCs have been co-cultured with islets by using various approaches. Although they can improve secretory function and survival rate, they all have their own advantages and disadvantages. Here, we discuss co-culture systems that have been studied.
Direct Co-culture System
In the direct co-culture system, direct contact co-culture systems are available in plain petri dishes and temperature-responsive petri dishes (Fig. 2A, B)
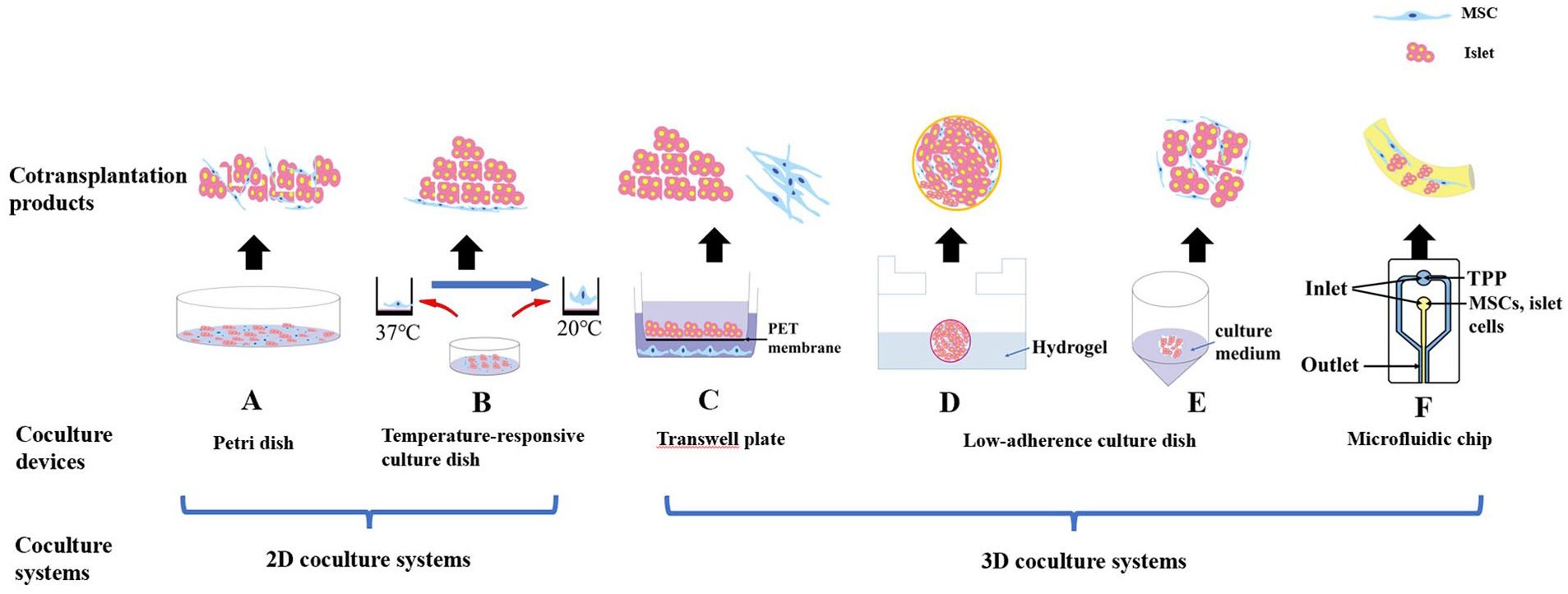
Pancreatic islets and MSCs co-culture systems. (A) Islets and MSCs were simply co-cultured to form a graft mixture by petri dishes. (B) Islets were co-cultured with MSCs to form a graft mixture by temperature-appropriate petri dishes. (C) Islets were co-cultured with MSCs to form a graft mixture by transwell dishes. (D) Islets were co-cultured with MSCs to form 3D stem cell spheroids by scaffold-based co-culture system. (E) Islets were co-cultured with MSCs to form neo-islets by a co-culture system without scaffolds. (F) Islets were co-cultured with MSCs to form alginate micro-encapsulated islets by a microfluidic chip.
Culturing by temperature-responsive culture dishes or plain petri dishes is considered as two-dimensional (2D) culture method. In the 2D co-culture method, islet cells and MSCs are added to a culture medium and grown along a 2D plane, extending from the surface of a glass or plastic petri dish 52 . In addition, the temperature-responsive culture dish has a coating of temperature-responsive polymers on the surface. At temperatures >32°C, the surface is hydrophobic and highly suitable for cell adhesion and growth. In contrast, at temperatures ≤20°C, the surface becomes hydrophilic and swollen, naturally releasing adhering cells, without the need for enzymatic digestion or treatment 53 .
There is also a more efficient direct co-cultivation method. Myrick et al. 54 have designed a micropyramid-patterned and oxygen-permeable-bottomed dish for high-density culture of pancreatic islets. This platform supported the high oxygen demand of islets at a seeding density 2- to 3-fold higher than the conventional islet culture, demonstrating improved islet morphology, metabolism, and function.
The 2D co-culture method is easy to operate, and the culture equipment is relatively affordable. However, this co-culture method cannot maintain the original characteristics of the cells for a long time and cannot accurately describe and simulate the rich environment and complex processes in vivo, which may mislead the effect of transplantation in vivo.
Three-Dimensional Co-culture System
In the three-dimensional (3D) co-culture methods, pancreatic islet cells and MSCs are placed into a 3D scaffold or low-adherence culture dish such that they grow and interact as 3D islet–MSC complexes in a 3D stereospecific space 52 .
Scaffold-based co-culture system
The 3D scaffold of the co-culture system is usually made of biocompatible materials, especially hydrogels, which typically support cell growth, simulate extracellular matrix, and promote tissue formation for cell attachment (Fig. 2D).
Duprez et al. 37 discovered that, in the process of suspension co-culture, MSCs rapidly adhered to the islets and spread out to uniformly cover the islet surface, forming a composite structure of MSC-islets. Bal et al. 55 prepared microspheres containing islet cells and MSCs using the hanging-drop method in the following steps: (1) transferred islet cells and MSC suspensions with different proportions into the suspension plate to form a suspension; (2) cells in suspension formed microspheres under the action of gravity and surface tension; and (3) transferred the microspheres containing cells from the suspension plate to a low adsorption culture dish for cultivation. The islet cells were noted to continue to survive and secrete insulin when the heterospheroids were exposed to proinflammatory factors. Moreover, they confirmed that transforming growth factor β1 and glucagon-like peptide 1 secreted by MSCs enhance the islets’ secretory function and immunoprotective effects. In addition, Yu et al. 56 prepared 3D stem cell spheroids by adding MSCs with human umbilical vein endothelial cell (HUVEC) suspension in hydrogel. The 3D stem cell spheroids were noted to secrete VEGF, insulin-like growth factor 1, and hepatocyte growth factor.
Polylactic acid-co-glycolic acid (PLGA) is a copolymer with excellent biodegradability, biocompatibility, and biosafety 57 . Kim et al. 58 prepared PLGA–MSC complexes by encapsulating hypoxia-pretreated MSCs with PLGA scaffolds; then, they implanted the complexes into the subcutaneous space of mice for prevascularization. Finally, the islets were transplanted into the scaffold holes. At 100 days after islet transplantation, the PLGA bed was covered by tight connective tissue and abundant blood vessels. Moreover, micropatterned membranes with MSCs attached in a subcutaneous rat model can improve implant prevascularization 59 .
CellSaic is a 3D-structure consisting of cells combined with a new bioabsorbable material and a recombinant protein for medical use. Kogawa et al. 60 prepared MSC CellSaic platforms and combined them with microcapsule–mesh bag pockets. The authors noted that the inflammatory response around the capsule was low in the early stages of transplantation—until up to 14 days after transplantation.
Co-culture system without scaffolds
This co-culture method forms a 3D mechanism through the extracellular matrix secreted by the cells themselves, instead of relying on external support materials, which more closely resembles the structure of tissues in their natural state. The devices commonly used are the ultra-low adsorption surface and transwell plates.
A low-adherence culture dish contains a hydrogel covalently bonded to the surface. This hydrogel minimizes cell attachment, cell activation, protein uptake, and enzyme activation without having a toxic effect on the cultured cells 52 . For example, MSCs and islet cells were co-cultured at a 1:1 ratio in ultra-low-adhesion surface culture dishes and neo-islets (NIs) formed overnight (Fig. 2E)61,62.
A transwell plate consists of a transwell chamber (ie, upper chamber) and a culture plate (ie, lower chamber); the upper and lower chambers are separated by a polycarbonate membrane. Because the polycarbonate membrane is permeable, the components in the lower chamber can affect the cells in the upper chamber 63 . Jung et al. 64 made islets co-cultured with MSCs in the Transwell system, the levels of monocyte chemoattractant protein-1 and TNF-α were decreased, and VEGF levels were increased at 4 weeks. Wei et al. 34 added MSCs and pancreatic islets to the lower and upper chamber, respectively, and co-cultured them with hypoxia-inducible factor 1α activator or inhibitor under hypoxia or normoxia. Their results demonstrated both methods enhance islet- secretion function and survival. however, direct contact co-culture demonstrated stronger effects. This co-culture method essentially uses various growth factors from MSCs to culture islets (Fig. 2C). This method can separate the islets from the MSCs, which can protect the function and activity of the isolated islets to a certain extent. This may be a way to protect the islets before our future islet transplantation.
Microfluidic chip co-culture system
Microfluidic chip co-culture system is a special 3D co-culture approach. A microfluidic chip, also called a lab-on-a-chip, is a device that is mainly used for manipulation of fluids in a micrometer-scale space (Fig. 2F). This chip has been used for establishing islet cell–MSC co-culture models because it enables precise control and testing of the whole co-culture system 65 . Moreover, MSCs have the ability to migrate to pancreatic islets and provide an apparent overall preservation for islet function via the microfluidic device 66 . Laporte et al. 67 developed a microfluidic card and then embedded MSCs with Arg-Gly-Asp (RGD)-rich alginate and pancreatic islets to form a microcapsule composite product. Their results demonstrated that the favorable in vitro composite environment within the RGD-rich alginate capsules enriched with MSCs improved pancreatic islet survival and function better than conventional alginate capsules alone. This effect may be related to the specific cytoprotective function MSCs elicit through paracrine effectors such as growth factors and cytokines 68 . Alternatively, the composite capsule (i.e., the mixture of MSCs and RGD-rich alginate) may impart to the micro-environment mechanical properties that are beneficial to the islet cells 69 .
The 3D co-culture method can better simulate the micro-environment of the cells in vivo, which can make up for the shortcomings of 2D co-culture. These co-culture devices provide a 3D environment similar to in vivo, allowing pancreatic islets and MSCs to grow and interact more closely to natural conditions. In other words, the use of a 3D co-culture system can enable MSCs to protect isolated pancreatic islets while also preparing for transplantation, thereby maximizing graft survival. These strategies might overcome the limitation of liver as a transplant site by making other organs available as a possible transplant site.
Sites for Islets/MSCs Co-transplantation
Liver
The liver and portal vein have been identified as preferred sites for islet transplantation because of their easy accessibility and associated low morbidity. However, further research is needed on the efficacy and safety of co-transplantation of pancreatic islets and MSCs 70 . More and more animal experiments have shown that co-transplantation of pancreatic islets with MSCs through the portal vein is an effective and safe method.
Gou et al. 71 co-infused human pancreatic islets with human ADMSCs into the liver of nonobese diabetic/severe combined immunodeficient (NOD/SCID) mice induced STZ. They noted that >90% of the mice receiving co-infusion of islets and ADMSCs achieved and maintained euglycemia. Moreover, inflammation-associated chemokines were significantly reduced in mouse serum. Notably, ADMSCs and islet-secreting cells with ADMSC characteristics were also detected in the mice 60 days after transplantation. MSCs are formed to encapsulate the pancreatic islets on the basis of co-cultivation, forming heterogeneous spheres. They can co-localize with the pancreatic islets in the liver, which can prevent the initiation of IBMIR after transplantation 51 . Ishida et al. 44 precultured BMSCs with inflammatory cytokines (IL-γ, TNF-α, and IL-1β), mixed BMSCs with pancreatic islets using a syringe, and slowly injected them into the portal vein via adipose tissue in rats.
Lee et al. 72 placed ADMSCs on temperature-responsive culture dishes to prepare ADMSC slices, which were then seeded with pancreatic islets, co-cultured to form islet/ADMSC sheets, and finally grafted onto the surface of the murine liver. Blood glucose levels immediately returned to normal in the liver surface. Related studies indicate that a relatively small mass of islet cell sheets was enough to achieve normoglycemia in diabetic mice when the liver surface was selected as the transplantation site 73 .
Recently, Kenyon et al. used crab-eating monkeys with T1DM as the recipients and donors of the BMSCs while islets were from third-part monkeys. The authors performed intrahepatic infusion of grafts composed of BMSCs co-cultured with islets and infusions of BMSCs on postoperative day 5, 11, 18, and 28. The post-transplantation results indicated that infusion of recipient-derived BMSCs and subsequent intravenous (IV) infusions over the first posttransplant month enhanced metabolic outcomes and better rejection-free and overall transplant outcomes 74 . This study was the first to report outcomes potentially the closest to those of human allogeneic islet transplantation. In another study, after introducing neonatal porcine islets into monkeys with BMSCs from monkeys via the mesocolic vein and then injecting intravenously BMSCs after co-transplantation at weeks 1, 3, 5, and 7, it was found that BMSCs can promote the development and maturation of neonatal porcine islets 75 . Therefore, injecting a certain amount of BMSCs can improve the transplantation effect after co-transplantation via the portal vein.
In 2018, Wang et al. 76 conducted the first clinical trial in which autologous pancreatic islets were transplanted with autologous BMSCs into the portal vein of patients who underwent pancreatectomy via the mesocolic vein; BMSC patients showed significantly higher physical quality of life (QOL) with better pain relief at 6 months than controls. Although they are chronic pancreatitis patients rather than patients with T1DM, this also indicates that co-transplantation of MSCs and islets may be a safe and feasible strategy to improve the efficacy of islet transplantation. Moreover, this program can also be used to solve the more complicated problem of islet allograft transplantation and treat patients with T1DM.
Greater Omentum
Although comparatively an invasive procedure, omentum has emerged as an attractive alternative site because it is highly vascularized with portal venous drainage, allowing insulin production by transplanted cells to mimic endogenous islet physiology, which is critical for the post-islet engraftment. The greater omentum has been demonstrated to be a clinically effective, feasible islet transplantation site 77 . Navaei-Nigjeh et al. 78 used Wistar rats for allogeneic transplantation. First, they co-cultured ADMSCs directly or indirectly with rat islet cells for 28 days and then co-transplanted islets with or without ADMSCs into the greater omentum. The authors noted earlier appearance of islet-like clusters, more endothelial cells, and higher vascular density in direct co-culture than in indirect co-culture, possibly because islets must be in direct contact with MSCs to secrete insulin. Furthermore, they determined that the islet mass necessary to reverse diabetes had been significantly reduced. It may also be that ADMSCs provide a micro-environment conducive to islet cell repair and growth. In the study by Navaei-Nigjeh et al., 79 for the first time, ADMSCs and islets were co-encapsulated in chitosan microfibers generated through microfluidics and then transplanted into murine mesentery, which demonstrated that compared with unencapsulated ADMSCs, co-encapsulated ADMSCs and islets in chitosan microfibers demonstrated higher viability, insulin secretion, and islet-specific gene expression.
Naqvi et al. 80 co-cultured BALB/c mouse BMSCs with endothelial progenitor cells and mouse pancreatic islets at different ratios to form 3D spheroids and then co-transplanted them into the large omental pouch of mice with diabetes. The results indicated that the mice returned to normoglycemia after 5 days of transplantation, and the grafts secreted mouse c-peptide and angiogenic factors.
Peritoneum
Kokorev et al. 81 used Titanium Nickel (TiNi) scaffold containing rat BMSCs to transplant islets into the abdominal cavity of diabetes rats. The results showed that in the TiNi scaffold, combined with cell transplantation group, the normalization of biochemical parameters such as blood glucose and glycated hemoglobin occurred earlier than it did with allogeneic islet cell transplantation alone and was associated with an increase in animal lifespan. Transplantation of allogeneic BMSCs on cellular scaffolds eliminates immune system dysfunction and restores disturbed homeostasis in the body, leading to regeneration of pancreatic islets. In addition, MSCs along with allograft islets were embedded in a hydrogel composite and transplanted intraperitoneally, and MSCs co-transplanted with allograft decreased pro-inflammatory cytokines 82 . Razavi et al. 83 first placed bioscaffold pieces in murine internal epididymal fat pads and then co-transplanted murine islet cells co-cultured for 24 h with murine ADMSCs or murine pancreatic islet cells into the abdominal bioscaffolds. The mice with the seeded bioscaffolds achieved euglycemia by day 2 and demonstrated a significant increase in survival rate and insulin index compared with the control mice.
Montanari et al. 84 co-cultured porcine islet cells with juvenile porcine pancreatic MSCs for 3 days to form islet cell clusters (ICCs) and then co-transplanted that into the peritoneum of mice; this led to a significant increase in insulin secretion compared with the mice that received ICCs cultured alone. Moreover, pancreatic MSCs derived from juvenile porcine exocrine pancreas improved ICC differentiation potential and insulin secretion in vivo and in vitro through direct cell–cell contact.
Westenfelder et al. 62 injected canine NIs comprising equal amounts of dog MSCs and dog islet cells into the abdomen of NOD/SCID mice with diabetes. The results for the first time demonstrated that administration of a single dose of canine neo-islets (cNIs) improved glycemic control, and a second dose of cNIs restored normal blood glucose levels in these mice. Later, Gooch et al. 85 injected cNIs into the pet dog abdomen with ultrasound guidance. These results indicated that administration of a single cNI dose in pet dogs improved glycemic control, and the second dose of hyperglycemic insulin normalized blood glucose levels. In other words, cNIs may establish euglycemia and insulin independence in canines with diabetes. Experiments have shown that human NIs also have the same transplantation effects, which is definitively significant as this will further strengthen the justification for the currently planned conduct of a scientifically based clinical trial in human study subjects with T1DM.
Subcutaneous
Subcutaneous transplantation of islets is simple and easy to monitor and poses low complication risk 86 . However, the lack of vascularization in the subcutaneous space hinders the supply of nutrients and oxygen, influencing the effect of grafts. To address this limitation, strategies to induce angiogenesis are essential to support graft islet implantation and achieve normoglycemia.
Wang et al. 87 combined SD rat ADMSCs and HUVECs to construct 3D islet microtissues. In the islets, they improved secretion function, promoted extracellular matrix proteins, and established a vascular network. The pancreatic islet microtissues were biocompatible, and the islets in the 3D microtissues had normal secretory function and achieved euglycemia by day 3 after implantation and maintain stable blood glucose in mice for a long period. Kim et al. 88 also proposed a simple but effective subcutaneous colocalization modality: cell–islet colocalizing composite. By optimizing the culture conditions, endothelial-like MSCs (EMSCs) were differentiated from mouse BMSCs; then, islets were coated with EMSCs or BMSCs. In particular, the MSCs were evenly coated on the surface of porcine islets (>85%). The results indicated that both the BMSC–islet and EMSC–islet composites significantly reduced fragmentation and increased islet mass in porcine pancreatic islets alone. Finally, in a prevascularized deviceless (DL) subcutaneous transplantation method, ADMSCs and islet cells were colocalized in the subcutaneous spaces 86 . The DL subcutaneous space was prepared by implanting a nylon catheter under the skin of mice and then transplanting islets and ADMSCs that had been co-cultured for 24 h into the DL subcutaneous space or renal capsule, which demonstrated that DL subcutaneous space in combination with ADMSCs achieved euglycemia 11 weeks after transplantation but prevented deleterious graft fibrosis overgrowth 89 .
Yamashita et al. 90 co-cultured porcine islet cells with pig ADMSCs to prepare islet cell–ADMSC sheets and then co-transplanted them subcutaneously into the waist and buttocks of pigs with T1DM. The results showed that compared with islets alone, the cell sheets led to higher islet viability, significantly increased secretion of the vascularizing cytokine IL8, and effective restoration of blood glucose levels within 2 weeks in the pigs. This was the first study to describe blood glucose normalization after subcutaneous islet transplantation in larger animals.
Renal Capsule
Although renal peritoneal transplantation site is less difficult to perform the procedure on and less likely to result in rejection, the smaller capacity makes it difficult to meet the needs of larger patients, and it is more susceptible to islet damage and necrosis. Wang et al. 49 transfected BMSCs with ISL1 adenovirus to obtain BMSC-ISL1; then, the authors injected these cells with islet cells in the renal capsule of rats with diabetes. Co-transplantation of the islet cells with BMSCs-ISL1 increased graft survival and prevented graft apoptosis and thus achieved euglycemia by day 5. They also noted the BMSCs-ISL1-conditioned medium, which was by BMSCs overexpressing ISL1, exerts a protective effect on pancreatic islet cells via various nutritional factors, preventing the safety issues typically caused by stem cell transplantation, and may therefore be applied in clinical practice.
When BMSCs were used to co-culture with islets and then transplanted on the renal peritoneum, the BMSCs were noted to significantly improve the function of transplanted islets, protect islets from apoptosis, and promote graft blood flow reconstruction after the two co-cultured and then co-transplanted45,48. Laporte et al. 67 reported that transplantation of islets without prior co-cultivation with ADMSCs was not conducive to transplantation. A study 91 found that SCID mouse islet cells, HUVECs, and human ADMSCs suspended in a culture medium could autonomously form vascularized islets. The results demonstrated that vascularized islet transplantation induces early reperfusion and promotes transplantation. Razavi et al. 92 co-cultured ADMSCs and islet cells both from C57BL/6 mice and then coated both using media such as sodium alginate, then co-transplanted them under the renal peritoneum of mice with diabetes, and finally, treated them with pulsed focused ultrasound in vivo. The results demonstrated that this technique improved the overall survival and function of the transplanted islets, with a corresponding increase in angiogenesis and decrease in inflammation.
Many studies have explored the effects of both co-culture and co-transplantation of MSCs and islets in various animal models of TIDM. The above is a summary of the main results of co-transplantation of islets and MSCs into rodents, large animals, and humans. Specific transplantation information, including recipient species, number of sources of islets and MSCs, and major transplantation results can be found in Table 1.
Summary of Co-transplantation Sites.
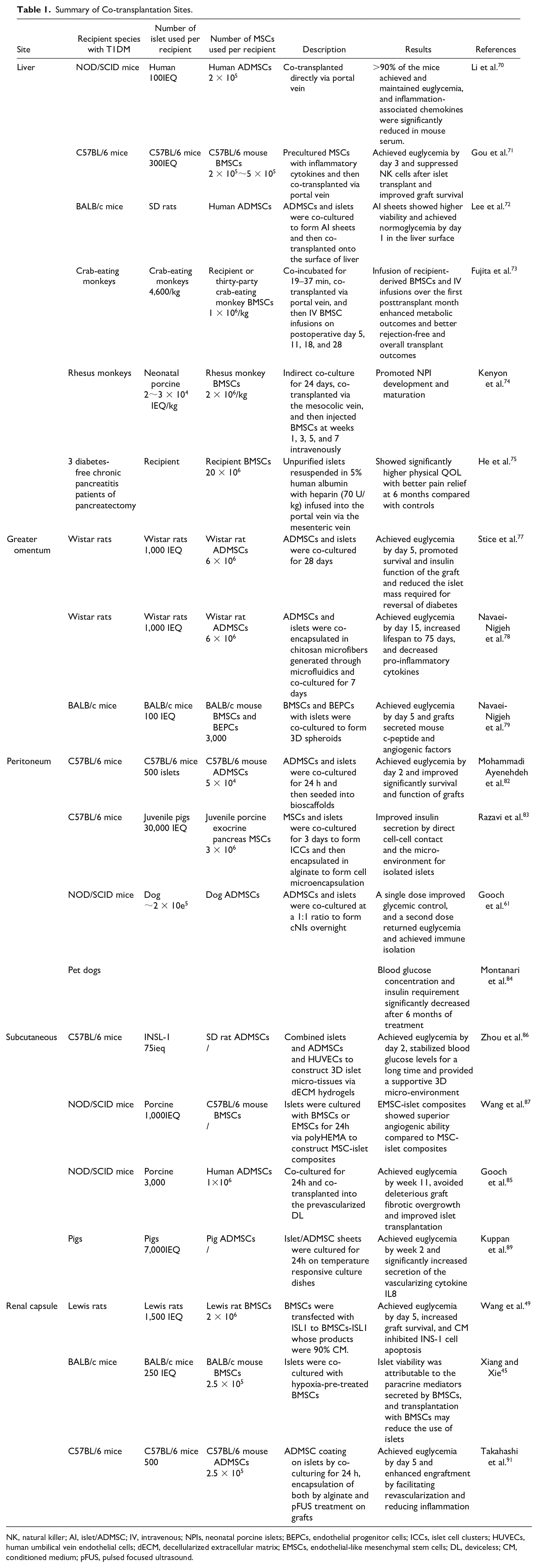
NK, natural killer; AI, islet/ADMSC; IV, intravenous; NPIs, neonatal porcine islets; BEPCs, endothelial progenitor cells; ICCs, islet cell clusters; HUVECs, human umbilical vein endothelial cells; dECM, decellularized extracellular matrix; EMSCs, endothelial-like mesenchymal stem cells; DL, deviceless; CM, conditioned medium; pFUS, pulsed focused ultrasound.
Discussion and Outlook
Islet transplantation is an emerging organ transplantation technique that solves the problem of insufficient insulin secretion and significantly improves the QOL of diabetic patients. MSC transplantation is a positive development in clinical treatment. Based on the characteristics of the MSCs, more and more experiences indicated that co-culture of pancreatic islets and MSCs had significantly higher survival and increased insulin levels, insulin secretion index, IL-6 levels, and VEGF secretion, than islets cultured alone; moreover, direct contact co-culture tends to demonstrate larger islet cell–protective effects than indirect contact co-culture 93 . A study suggested that co-culturing of islets with MSCs prior to transplantation improves graft function, and that these effects require 48–72 h of co-culture time 94 . However, the optimal co-culture time may be different depending on the method of co-culture. Currently available transplantation sites, besides the portal vein, are the greater omentum, peritoneum, subcutaneous, and renal capsule, which are also promising sites with clinical co-transplantation of pancreatic islets and MSCs. Therefore, we can also develop different co-transplantation methods to improve more avenues for clinical treatment of diabetic patients depending on the transplantation site. Also, re-transplantation of MSCs after co-transplantation has been shown to improve graft outcome.
Regardless of the type of method used, co-culture of islets with MSCs enhances the anti-apoptotic and secretory abilities and increases the prevascularization capability of the resulting graft. Moreover, co-transplantation of islets with MSCs increases graft survival, viability, and angiogenic capacity regardless of the co-transplantation method or site used. So far, islet co-culture with MSCs followed by co-transplantation is a clear plan for islet transplantation.
It is important to note that co-transplantation of pancreatic islets and MSCs still faces some challenges and limitations. For example, the source, preparation, and quality control of both need to be further studied and standardized. Embryonic stem cells or induced pluripotent stem cells, as an unlimited source for in vitro generation of β cells, may obviate the shortage of pancreas/islet donors, but further improvements need to be made to increase the safety and efficacy of them for use in a clinical setting 1 . In addition, the use of this strategy for the treatment of T1DM patients is still in the research stage, and the clinical application needs further validation and improvement.
With the ongoing advancements in islet and MSC co-transplantation technology, their joint transplantation is now nearer to clinical application. In the future, through the continuous exploration of pretransplantation preparations and the optimization of the corresponding co-culture techniques, possibly coupled with regular postoperative injections of MSCs, islet morphology and function can be better maintained, and graft survival can be promoted, thus gradually improving co-transplantation effects.
Footnotes
Author Contributions
L.M., L.WP., and Y.YW. performed the literature review and wrote the manuscript with the consultation of C.JB., F.BZ, and X.ZR. G.HJ. provided writing ideas and funding support. All authors have read and agreed to the published version of the manuscript.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Guangxi Science and Technology Base and Talent Project (grant number AD22035122), the Funding for Innovative Team for Cultivating High-level Talents in Clinical Research of Cell and Tissue Engineering (grant number 04B23008B8), and the Funding for Chinese University Industry University Research Innovation Fund—Huatong Guokang Medical Research Special Project (grant number 2023HT024).
