Abstract
The liver is currently the site of choice for clinical islet transplantation, even though many alternative implantation sites have lately been proposed as more ideal for graft survival. The suggested sites, for example intramuscular space, omentum, bone marrow, and spleen, are sometimes difficult to compare due to differences in animal model, islet isolation procedure, and islet quality. In addition, the variation in transplanted islet mass is vast. The aim of this commentary is to review alternative implantation sites tested experimentally as well as in clinical islet transplantation. Although many sites have been investigated, none have convincingly proved better suited for clinical islet transplantation than intraportal injection to the liver, regardless of whether it is autologous or allogeneic transplantation. However, in order to fully evaluate upcoming bioengineering techniques, such as scaffolds containing insulin-producing cells derived from stem cells, the need of an alternative site has arisen to enable cellular monitoring, which currently cannot be achieved within the liver.
Introduction
Islet transplantation (IT) is currently offered to a small subset of type 1 diabetes (T1D) patients to replace destroyed beta cells and thereby avoid glycemic lability. IT is a minimally invasive procedure with increasingly better outcome due to progress in the islet isolation technique and immunosuppressive regime 1 . Allogeneic IT is used for T1D patients, whereas autologous IT is performed in the treatment of chronic pancreatitis or following pancreatectomy 2 . Both types of transplantation are performed by an injection of islets into the portal vein for engraftment into the liver, which has been the site of choice since the beginning of clinical IT 1,3 . The outcome of islet autotransplants demonstrate superior long-lasting graft function even if the transplanted islet mass sometimes is lower than in allogeneic transplantations 4 .
Currently, insulin-independent success rate in allogeneic IT is reported in up to 50% of patients after 5 years 1,5 . Although some patients revert to exogenous insulin therapy, 87.5% achieved freedom of severe hypoglycemic events 1 year after IT and reported improved quality of life 6 .
Clinical IT faces two major challenges. First, it is desirable to decrease the need for immunosuppressive treatment in order to expand the group of patients that could be candidates for IT. Second, a lack of donors will arise if the only source of islets comes from brain-dead organ donors. New strategies for beta cell replacement might solve both problems, using encapsulation of insulin-producing cells and stem cells. Promising stem cell programs are developed, but clinical translation of such advanced therapy medicinal products requires safety studies with graft monitoring and retrieval possibilities. Questions regarding the liver as the future implantation site of choice have therefore been raised on several occasions over the last decades. We have previously demonstrated engraftment difficulties after intraportal transplantation, including reduced vasculature, hypoxia, and amyloid formation, which contribute to islet mass reduction and loss of function 7 . The aim of this commentary is to highlight the conditions of alternative sites for future beta cell replacement.
Advantages of the Intrahepatic Site for Clinical Transplantation
The clinical procedure of percutaneous intraportal infusion of islets into the liver is associated with low morbidity and low risk of adverse events, such as bleeding (7%) and portal thrombosis (3.7%) 8 . In order to further reduce the risk of complications, it is recommended to restrict the total volume of transplanted tissue and to avoid rapid increases in portal pressure 9 . At the same time, islet volume is a known predictor that positively correlates to transplantation outcome 10 . A unique consequence of intraportal injection is the scattering of islets throughout hepatic sinusoids, avoiding clusters, which form in every other transplant site and may initially impair diffusion of oxygen and nutrients. On the other hand, this makes the graft very difficult to monitor by imaging techniques, and means that biopsies are difficult to obtain 11 .
The liver is the main target organ for insulin, and intrahepatic islets can therefore mimic the physiological pancreatic insulin secretion as opposed to systemic insulin release 12 . In experimental studies, the liver also appears to be more efficient than bone marrow 13 and kidney subcapsular site 14 due to immunological factors. Islets grafts within the liver lasted a longer time before rejection compared with islet grafts at the kidney subcapsular site in rats 13 . Likewise, a lower count of recruited lymphocytes has been reported in islet grafts in liver than in islet grafts in bone marrow 14 .
Challenges with the Intrahepatic Site for Clinical Transplantation
After transplantation, beta cell survival can be impaired by acute and/or chronic factors. Immediately when islets are injected, direct contact with the vascular system exposes them to an instant blood-mediated reaction (IBMIR) which contributes to early graft loss 15 –17 . In addition, there is a substantial risk of beta cell death due to acute hypoxia. Even after revascularization, oxygen delivery to intrahepatic islets is inferior compared with native islets in the pancreas, leading to prevailing hypoxia 18 –21 . Better outcomes have been demonstrated with selective transplantation of smaller islets, which exhibit a lower oxygen demand 10 . Amyloid formation, which is associated with type 2 diabetes, has been found in intraportal islet grafts 22 . In our recent study, 27% of islets contained amyloid 1 month after transplantation 7 . Furthermore, gluco-lipotoxicity from surrounding hepatocytes has been shown to damage transplanted islets 23 –25 . Although there are controversial findings, Robertson et al. have demonstrated how glycogen metabolism and glucose flux in the hepatic site inhibit secretion of glucagon by transplanted alpha cells, which leads to hyperinsulinemia and hypoglycemia in both autologous and allogeneic settings 26 –28 . Kupffer cells are specific to the liver site and have also been shown to be detrimental for islet allograft survival at this site 29 . Another concern regarding the hepatic site which has been postulated is that portal blood contains potentially harmful concentrations of immunosuppressive agents 30 . Notably, however, intrahepatic islets only sense to glucose stimulation through the hepatic artery after their revascularization; therefore, the relevance of drug-related effects due to increased concentrations in portal blood can be debated 31 .
Alternative Implantation Sites and Comparative Studies
Comparing engraftment efficacy for different transplant sites is challenging due, for example, to variations in transplanted islet mass, especially when using human islets. Table 1 lists experimental studies comparing alternative sites to the liver in the commonly used C57Bl/6 mouse model.
Experimental Studies Comparing Alternative Islet Transplant Sites to Liver in C57Bl/6 Mice.
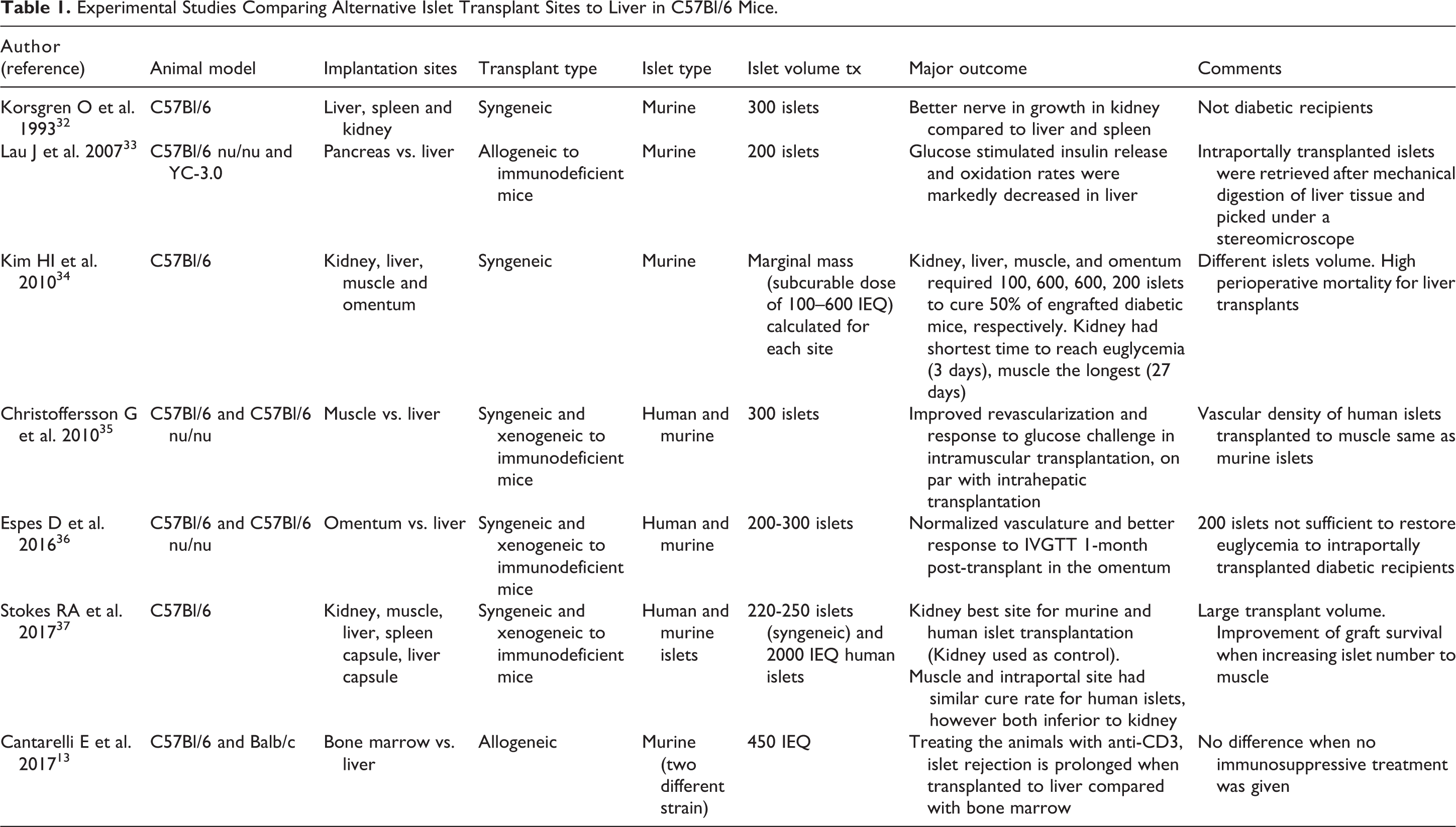
Table 2 gives a summary of alternative sites used in the clinical setting, for both allogeneic and autologous IT.
Implantation Sites Used in Clinical Allogeneic and Autologous Islet Transplantation.
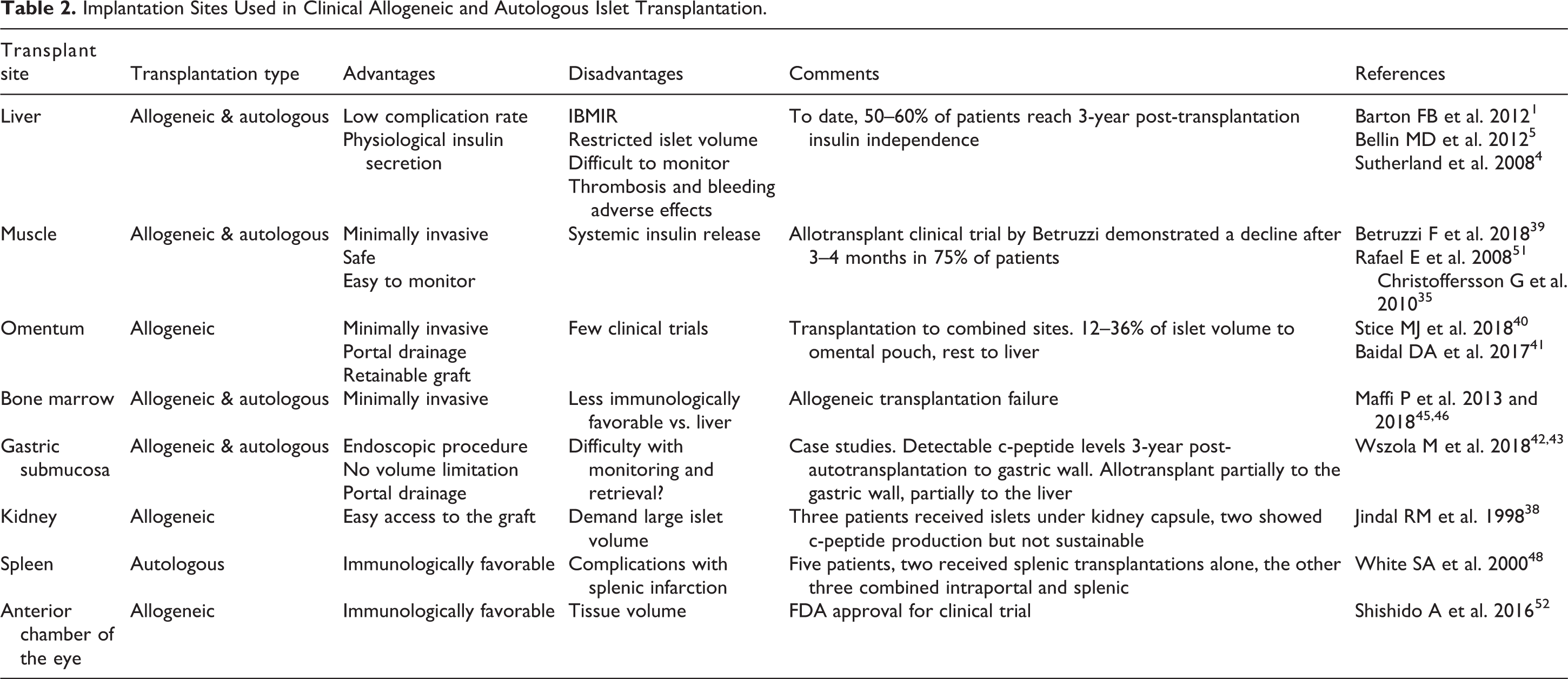
Concluding Remarks
Many different sites have been evaluated for beta cell replacement in the experimental setting and found superior in different aspects when compared with intraportal IT. Also, in the clinical setting a number of sites have been evaluated, but so far none have reported improved outcome when compared with the intrahepatic site. Even though IT is minimally invasive, especially compared with whole pancreas transplantation, the different sites used are associated with specific technical complications and different volume capacities. Upcoming bioengineering techniques of stem cells and scaffolds, including the use of auxiliary stem cells such as mesenchymal stem cells, neural crest stem cells, or endothelial progenitor cells, may be used to advance the field of beta cell replacement 53 –57 . However, monitoring possibilities and safety issues make the intrahepatic site inappropriate for this purpose. Changing site could contribute to better graft survival by avoiding, for example, IBMIR and lipotoxicity. However, solutions to obtain scattering of graft tissue are then needed. We consider scattering of the tissue at implantation to be the most important reason why the liver works so well clinically.
Footnotes
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
