Abstract
The administration of bone marrow-derived stem cells may provide a new treatment option for patients with heart failure. Transcatheter cell injection may require multi-imaging modalities to optimize delivery. This study sought to evaluate whether endomyocardial injection of mesenchymal precursor cells (MPCs) could be guided by real-time 3D echocardiography (RT3DE) in treating chronic, postinfarction (MI) left ventricular (LV) dysfunction in sheep. Four weeks after induction of an anterior wall myocardial infarction in 39 sheep, allogeneic MPCs in doses of either 25 × 106 (n = 10), 75 × 106 (n = 9), or 225 × 106 (n = 10) cells or nonconditioned control media (n = 10) were administered intramyocardially into infarct and border zone areas using a catheter designed for combined fluoroscopic and RT3DE-guided injections. LV function was assessed before and after injection. Infarct dimension and vascular density were evaluated histologically. RT3DE-guided injection procedures were safe. Compared to controls, the highest dose MPC treatment led to increments in ejection fraction (3±3% in 225M MPCs vs. −5±4% in the control group, p < 0.01) and wall thickening in both infarct (4±4% in 225M MPCs vs. −3±6% in the control group, p = 0.02) and border zones (4±6% in 225M MPCs vs. −8±9% in the control group, p = 0.01). Histology analysis demonstrated significantly higher arteriole density in the infarct and border zones in the highest dose MPC-treated animals compared to the lower dose or control groups. Endomyocardial implantation of MPCs under RT3DE guidance was safe and without observed logistical obstacles. Significant increases in LV performance (ejection fraction and wall thickening) and neovascularization resulted from this technique, and so this technique has important implications for treating patients with postischemic LV dysfunction.
Introduction
Cardiovascular disease continues to be the most prevalent cause of morbidity and mortality worldwide. Acute myocardial infarction (AMI) afflicts 1 million new patients per year, leading to permanent cardiomyocyte loss and, in nearly one third of patients, heart failure on the basis of systolic left ventricular (LV) dysfunction. To avert this problem, the implantation of adult stem cells into postinfarction myocardium has emerged as an attractive treatment strategy. Studies in animal models and clinical trials with autologous or allogeneic bone marrow-derived mesenchymal stem cells (MSCs) indicate that the potential to improve LV function after ischemic injury may be considerable (19,21,22). However, there are methodological obstacles that impede successful delivery of stem cells, therein limiting their survival, biological effects, and potential engraftment (23).
Perivascular stromal precursor antigen 3 (STRO-3)-positive mesenchymal precursor cells (MPCs) are a small fraction of MSCs that demonstrate the capacity for multipotent differentiation and extensive ex vivo proliferation.
STRO-3-positive MPCs have been shown to foster vasculogenesis and improve heart function after myocardial infarction in animal studies (5,12). However, those previous studies utilized an invasive surgical approach. In this study, we aimed to increase the efficacy of catheter-based endomyocardial delivery of allogeneic ovine MPCs in a sheep model of chronic postinfarction LV dysfunction by mapping the target injection site through real-time three-dimensional echocardiographic (RT3DE) delineation of the myocardial scar border and guidance of a novel injection catheter to each specific site. In addition, we sought to evaluate the biological effect of MPC delivery by evaluating the treated histological sections and several echocardiographic parameters 8 weeks following cell delivery.
Materials and Methods
Study Design
The study was in accordance with the NIH guidelines for the care and use of laboratory animals and was approved by the Institutional Animal Care and Use Committee (IACUC) of the Jack H. Skirball Center for Cardiovascular Research of the Cardiovascular Research Foundation. Acute myocardial infarction (MI) was induced in Dorsett hybrid female sheep (35±5 kg, Animal Biotech Industries, Danboro, PA, USA) by 90-min balloon occlusion of the midsegment of the left anterior descending artery (LAD). Four weeks following MI, animals were randomly assigned to either allogeneic MPCs at three different doses (25 × 106 cells, n = 10; 75 × 106 cells, n = 9; or 225 × 106 cells, n = 10) or assigned to be a control (nonconditioned media, n = 10), and then the cells or media were administered transendocardially into the infarct and periinfarct regions using RT3DE guidance. LV function was measured by two-dimensional (2D) echocardiography prior to (baseline) and at 8 weeks (chronic) after study intervention. Blood samples were collected from the external jugular vein before (baseline), immediately after cell injection, and at 8 weeks follow-up. All collected whole blood and serum samples were sent to Antech Diagnostics (New Hyde Park, NY, USA) for hematology and serum chemistry analysis, including creatine kinase (CK). All animals were euthanized, and myocardial tissue was harvested for histology at the study's conclusion.
Myocardial Infarction Protocol
MI was induced by percutaneous balloon occlusion of the coronary artery and was followed by reperfusion. In brief, under general anesthesia [1–3% isoflurane (Butler Schein Animal Health, Dublin, OH, USA) via facemask], a balloon catheter (3.0 × 8 mm, Voyager, Abbott Vascular, Santa Clara, CA, USA) was advanced into a position distal to the first diagonal branch of the LAD. The balloon was inflated, and total occlusion of LAD was confirmed by angiography during 90 min of inflation. Intravenous metoprolol (1–2 mg bolus; Hospira, Lake Forest, IL, USA) and lidocaine (4 mg/kg bolus; Cardinal Health, McGaw Park, IL, USA) were administered during periods of balloon occlusion and reperfusion. Animals were allowed to recover from exposure to the anesthetic 2 h after coronary artery reperfusion.
Cell Preparation
STRO-3-positive MPCs were derived from bone marrow aspirates of male crossbred sheep using previously described techniques (5,12). Cells were cryopre-served in 42% Profreeze (Lonza, Mapleton, IL, USA), 50% alpha minimum essential medium (Sigma-Aldrich Co., St. Louis, MO, USA), 7.5% dimethyl sulfoxide (Sigma-Aldrich) mixture in 25, 75, or 225 × 106 cells/4 ml ampoules. Recovery of the frozen preparations was achieved by rapid thawing, and cell suspensions were kept on ice until administered. A 100-μl aliquot was removed and diluted at 1:2,000 ratio in 0.4% trypan blue/phosphate-buffered saline (Sigma-Aldrich) to assess cell viability and cell number using a hemocytometer (Bright-Line, Hausser Scientific, Horsham, PA, USA). Control injections consisted of identical media composition as above but did not contain MPCs and were nonconditioned.
Endomyocardial Cell Delivery Procedure
Endomyocardial injections were administered at 4 weeks after MI creation. Animals were intubated and anesthetized as previously described above. Intravenous heparin (Hospira) was given to maintain an activated clotting time of 200–300 s. Baseline echocardiograms were obtained in the right lateral decubitus position to identify and characterize the infarct and periinfarct target areas. Using the short-axis view, four cross sections were analyzed from the papillary muscle level down to the apex (Fig. 1, left). LV wall thickness was measured at the border of the scar and at the middle portion of the infarct area in all views. From this, 16 injection sites were planned. Segments that were dyskinetic or with a wall thickness of ≤6 mm (especially the LV apex) were excluded as injection sites.
A pigtail catheter was then placed in the left ventricle for biplane ventriculography and additional visualization of hypokinetic segments. The injection catheter (Abbott Vascular) is an 8 Fr, multicomponent device containing elements for single-plane, 180° tip deflection and for biological injection. The latter component is composed of a unibody 27-ga nitinol needle with a 45° bevel at the distal aspect and can be advanced beyond the catheter tip to predetermined lengths (3–6 mm). The proximal aspect of the device is composed of a handle on which manual control adjustment of catheter curvature and needle extension are mounted, as well as separate portals for catheter irrigation and transneedle injection. Unique to the device are echogenic marker elements at the distal segment of the catheter tip. The catheter was introduced through a long sheath into the left ventricle and then guided to each individual target injection site under fluoroscopy. Location of the tip of the injection catheter was then confirmed using an iE33 X3-1 3D echocardiography transducer (Philips Medical Systems, Bothell, WA, USA). Images were acquired, rotated, and cropped to confirm the location of the catheter tip in relation to the target infarct region (Fig. 1, right) for each injection site. Needle extension length was adjusted to 50±20% of myocardial wall thickness. Upon confirmation of its location, the needle was deployed into the infarct area, and 200 μl of MPCs or control media was delivered over a 30-s period and repeated to distribute a total of 16 injections over the infarct area and border zone (3.2 ml total volume) in each animal. At the completion of the procedure, 2D echocardiography was performed to assess periprocedural complications.

Representative images for echocardiographic-guided endomyocardial cell delivery. Allogeneic mesenchymal progenitor cells (MPCs) or control media injections were administered to infarct or border regions using 2D and 3D echocardiography. Left: 2D images of injection mapping technique. Upper frame is a long-axis image from which four separate short-axis planes were chosen to identify the target region. The lower frame is a short-axis image at the level of plane #2. (A to D) Injection sites from septal to anterolateral walls, respectively. Right: single frame images from 3D acquisitions of catheter positions corresponding to injection sites A to D. White arrows identify the tip of the injection catheter.
Quantitative Echocardiography Protocol
Two-dimensional echocardiography images for quantitative assessments were acquired in the right lateralc decubitus position using a 5-MHz probe (iE33, Philips Medical Systems) from standard parasternal long- and short-axis planes. LV end-diastolic diameter (EDD) and end-systolic diameter (ESD) were determined from short-axis planes; end-diastolic volume (EDV) and end-systolic volume (ESV) were calculated using the Simpson's method from long-axis planes. LV ejection fraction (EF) was calculated using a standard formula: EF = [(EDV – ESV)/EDV]×100. Wall motion score indices (WMSIs) were analyzed using a 16-segment model (20). Each segment is assigned a score based on its contractility: normal = 1, hypokinesis = 2, akinesis = 3, and dyskinesis = 4.
Regional LV function was evaluated by speckle tracking echocardiography (STE). Two-dimensional strain images were acquired at three different short-axis levels of the LV (base, mid, and apex) at a high frame rate (80–100 frames/s), and at least three consecutive cardiac cycles were recorded for offline analysis (QLab 6.0 software, Philips). When a cardiac cycle with good quality image was selected, a region of interest for speckle tracking was defined first at end diastole using a semiautomated border detection method. The locations of the tracking points extended from endocardial to epicardial borders were adjusted, and then, the segmental myocardial strain curves were automatically generated by the system. For each animal, peak systolic circumferential strain, radial strain, and wall-thickening parameters were automatically generated and exported by the system.
Histology Protocol
At termination, the heart was removed and pressure perfused with saline followed by 10% buffered formalin (Cardinal Health). Hearts were sliced at 1-cm intervals from the apex to the base to assess infarct area and LV wall thickness in both normal and infarct regions. Digital images of gross myocardial slices with a linear scale were used for morphometric analysis using calibrated IPLab software (Scanalytics, Rockville, MD, USA). LV area measurements were performed on each of the serially sliced LV segments and summed to give the total LV area. Areas of infarct were also measured on gross slices to generate a total percentage of LV with infarct. A total of 17 myocardial sections were taken at three different levels (apex, mid, and base) (1) and analyzed with hematoxylin and eosin, Masson's trichrome, and Movat pentachrome (all from Sigma-Aldrich).
Immunohistochemical identification of endothelial cells lining vessels on myocardial sections was performed using biotinylated lectin (Dolichosbiflorus, DBA, Sigma-Aldrich; dilution 1:20). A monoclonal antibody directed against smooth muscle actin (SMA) clone 1A4 (Sigma Aldrich, dilution 1:4,000) was used for the identification of smooth muscle cells for arterioles. Staining procedures were performed using a DakoAutostainer Plus system (Carpinteria, CA, USA). Quantitative analysis of vessel densities were performed on each animal in three regions: the myocardial infarction, a border region, and a region remote from infarct tissue. Total vessel density measurements (on lectin-stained slides) were performed in two fields at 200× magnification; arterioles (diameter < 100 μm) were counted (on SMA-stained slides with adjacent Movat sections) in two fields at 100× magnification in each of the three regions of interest (14). All muscular vessel counts were averaged for each animal and expressed as the number of vessels/mm2.
Statistical Analysis
All statistical analyses were performed using MATLAB (R2008a, The Mathworks, Natick, MA, USA). All measurements were tabulated as mean±SD. Quantitative differences between treatment groups were evaluated by ANOVA and, if a difference was found, were compared using Dunnett's test, holding media injection as the control. Post hoc pairwise comparisons between treatment groups were performed using the Tukey–Kramer method. A value of p < 0.05 was considered statistically significant.
Results
Procedure-Related Outcomes
Fifty-six animals underwent AMI induction, 17 (29%) succumbed during the induction procedure, and 39 survived to enrollment at 4 weeks post-MI (baseline). Injected MPCs were highly viable: 97.3±0.9% in the 25 × 106 group, 96.9±0.6% in the 75 × 106 group, and 97.0±0.9% in the 225 × 106 group, resulting in total, viable cells injected with 23.9±1.6, 72.9±4.7, and 217.0±6.5 × 106, respectively.
At the time of study intervention, the infarct, its surrounding border area, and the catheter tip were readily covisualized by RT3DE (Fig. 1). By study design, regional distribution of injections closely followed infarct location: anteroseptum (wall motion score 3) received 41%, anterior (wall motion score 2–3) 32%, and border zones (inferoseptum and anterolateral wall) (motion score 2–3) received 24% of injections. The average procedure time was 40±11 min. Aside from brief periods of ventricular ectopy, there were no catheter-related complications (myocardial perforation, pericardial effusion, valvular or conduction system trauma) detectable by intra- or postprocedure electrocardiography (ECG) and echocardiography. Moreover, while not a prespecified measure of success of a study intervention, we noted distinct changes in wall thickness after intramyocardial injection in several animals, the most striking example of which is displayed in Figure 2. No significant changes in CK levels were noted compared to baseline values (data not shown).

Still frames of transthoracic 2D diastolic images of the left ventricle before and after MPC injection. Top: long-axis images. Bottom: short-axis images. Left: preinjection. Right: postinjection. An increase in wall thickness (arrows) is seen in the regions of injections (anteroseptum).
Myocardial Performance Endpoints
The hemodynamic parameters summarized in Table 1 indicate that mean aortic and, with the exception of the 75 × 106 group, LV systolic pressures did not change significantly between baseline and chronic measurements. The LVEDP decreased in the 225 × 106 group (baseline, 14.1±3.0 mmHg; chronic, 10.9±2.6 mmHg, p < 0.01), without significant changes observed at other doses.
Hemodynamic and Echocardiography Parameters Assessed at Baseline (4 Weeks Post-MI) and 8 Weeks Following Treatment

MI, myocardial infarction; MAP, mean aortic pressure; LVSP, left ventricle systolic pressure; LVEDP, left ventricle end-diastolic pressure; EF, ejection fraction; EDV, end-diastolic volume; ESV, end-systolic volume. Values are means±SD.
p < 0.05 versus 4 weeks post-MI;
p < 0.05 versus control group.
Four weeks after acute injury, LVEF declined and LV dilatation progressively increased in all groups (preinfarct data not shown). Compared to post-MI baseline, EF significantly fell in the controls, while it notably increased in the 225 × 106 group (p = 0.0004 vs. control) (Table 1) (Fig. 3A). Correspondingly, EDV and ESV significantly rose (p < 0.01) in the controls, as did EDV in the 225 × 106 group. WMSI was decreased in the 225M MPC-treated group (1.39±0.14 vs. 1.36±0.15, p = 0.018) at 8 weeks following cell transplantation but showed no difference in the control and low-dose MPC groups (Table 2). At post-MI baseline, all groups demonstrated decreased wall thickening in the infarct (anterior and anteroseptal) and border areas (inferoseptal and anterolateral) compared to remote areas (Table 2). At 8 weeks after MPC administration, wall thickening in the infarct and border zones increased (4±4% and 4±6%, respectively) in the 225M MPC group compared to the control and other MPC groups (Fig. 3B).
Wall Motion Score Index and Wall Thickening (%) Assessed at Baseline (4 Weeks Post-MI) and 8 Weeks Following Treatment
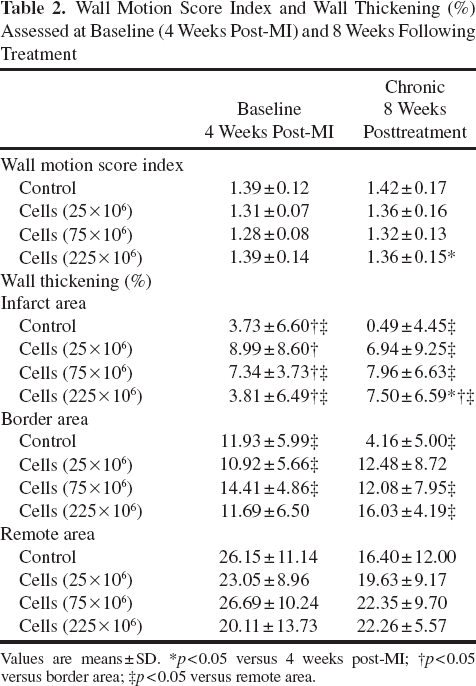
Values are means ± SD.
p < 0.05 versus 4 weeks post-MI
p < 0.05 versus border area;
p< 0.05 versus remote area.

Ejection fraction and wall thickening changes from 4 weeks post-MI to 8 weeks after treatment in four treatment groups. Top: ejection fraction changes; bottom: wall thickening changes in infarct and border zones (mean±SD, *p < 0.05 vs. control group; †p < 0.05 vs. 225M MPC group). MI, myocardial infarction; EF, ejection fraction.
Regional circumferential and radial strain changes from baseline are shown in Table 3. Compared to the control group, the highest dose of MPC treatment (225 × 106 cells) significantly improved strains in the anterolateral wall.
Regional Strain Changes From Baseline (4 Weeks Post-MI) to 8 Weeks After Treatment in Four Treatment Groups
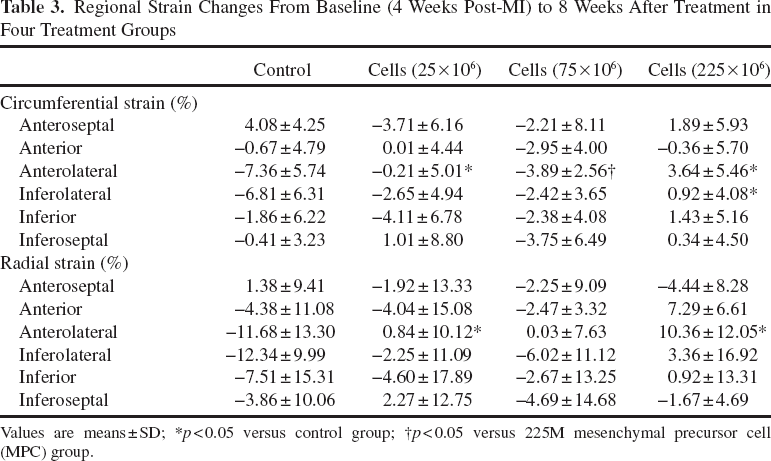
Values are means ± SD;
p < 0.05 versus control group
p < 0.05 versus 225M mesenchymal precursor cell (MPC) group.
Histological Analysis 8 Weeks Following MPC Injection
There were no significant differences between the four treatment groups in infarct size as a percentage of gross measurements (control, 14.4±11%; 25× 106, 12.9±10%; 75 × 106, 11.3±5%; 225 × 106, 12.9±9%; p = NS), nor in wall thickness in either infarct or remote regions. Evidence of MPC survival was absent in all 39 animals. SMA staining revealed a significant difference in arteriole density between the four treatment groups both in infarct and border zones (Table 4) (Fig. 4). The 225 × 106 group showed the highest arteriole density (79.76±21.68 vessels/mm2), significantly higher than that seen in the control group (54.37±19.71 vessels/mm2, p < 0.05). Similar results were observed within the border area when comparing the 225 × 106 and control groups (25.97±5.69 vessels/ mm2 vs. 17.96±6.96 vessels/mm2, respectively, p < 0.05). Several myocardial sections showed evidence of chronic inflammatory foci (giant cells) in both control and MPC-treated animals.
Arteriole Densities Based on SMA-Positive Immunostains Expressed as Vessels/mm2

Values are means ± SD. SMA, smooth muscle actin
p < 0.05 versus control group
p < 0.05 versus 225M MPC group.
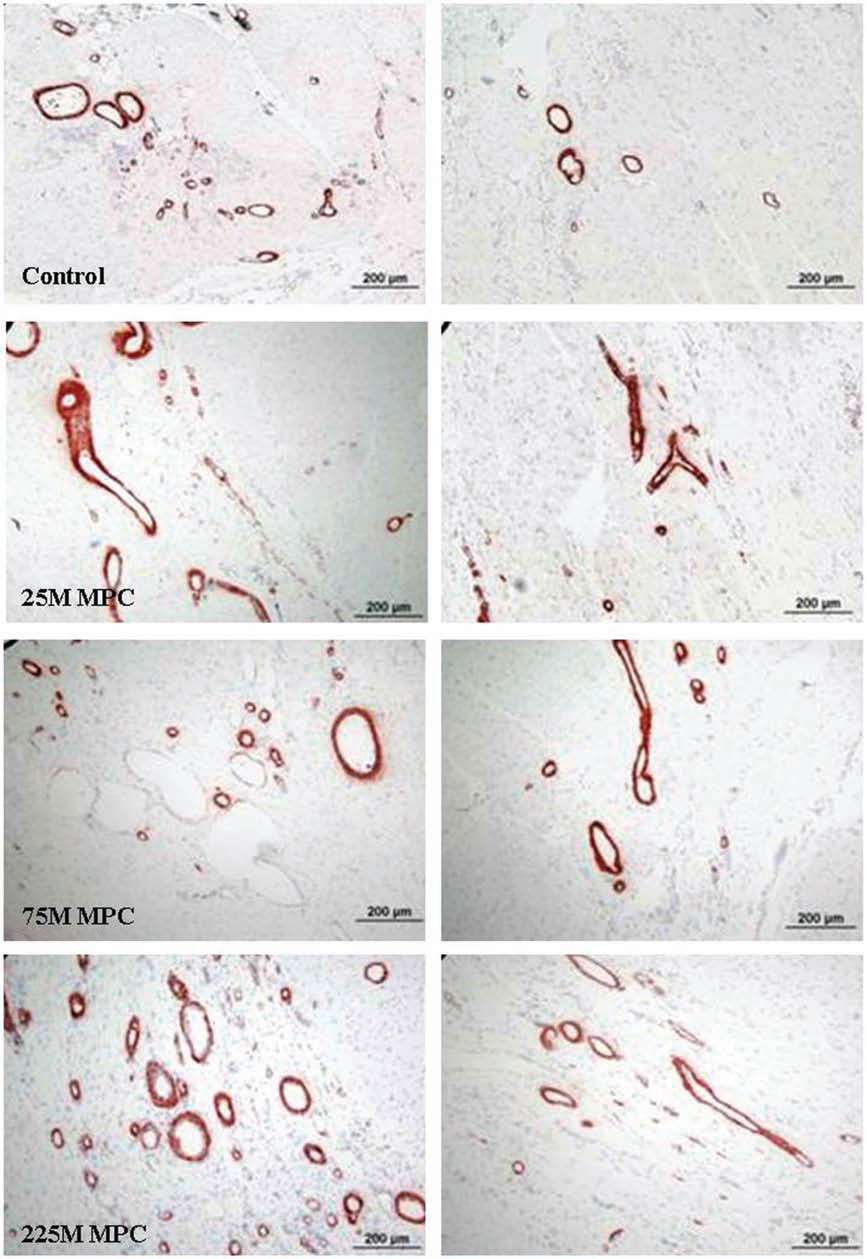
Representative images (10×) of smooth muscle actin-stained sections from the infarct, border zones associated with treatment groups. Left: infarct zone; right: border zone. Compared to the control group, higher arteriole density was observed in both border and infarct regions in the highest dose MPC-treated group (225M MPCs).
Discussion
The prevalence of heart failure continues to increase each year (10), despite significant advances in therapeutics. To address this trend, the potential of stem cell-facilitated repair and regeneration in treating advanced LV dysfunction is being explored. Techniques for applying progenitor cells to the ventricular wall have involved their direct injection under surgical visualization or using catheter-based techniques and most often with biological products in the form of a cell suspension (3,16). Animal and clinical studies have demonstrated the safety and efficacy of these approaches (8,9,11,13,17,25), each having its own attributes and drawbacks (23). The goals of the present study were (1) to determine whether MPCs given by catheter injection would lead to measurable improvements in LV function, as had been demonstrated with the same cells given surgically, and (2) to determine the utility of a novel catheter system to deliver MPCs using a target-enhanced, real-time 3D echocardiography-directed approach.
Previous studies have suggested that MPCs, a subset of bone marrow stromal stem cells, can improve LV function in the setting of acute MI animal models (5,12). In the present study, we confirmed that administering allogeneic MPCs into chronically infarcted myocardium improves both LV global and regional functions. Unlike the findings from another large animal study (5), the present study demonstrated that the high-dose MPC group (225 × 106 cells) showed the highest improvement in LVEDP, EF, WMSI, and wall thickening in both infarct and border areas. These contrasting observations may have arisen from one or more of the methodological differences of two studies (present vs. previous): type of MI model (ischemia/reperfusion vs. permanent ligation), timing of cell delivery (28 days vs. within 1 h), method of delivery (catheter-based endoventricular vs. surgical-based epicardial), and injected regions (combination infract and border zone vs. border zone only). Even though the literature does not clearly favor one method over the other as to the effect on LV function, support for delayed administration of higher cell doses can be found in both a recent meta-analysis of preclinical studies (26) and a small clinical study (27) in keeping with the findings from our study. Additionally, it is our belief that translational studies using percutaneous, ischemia/reperfusion models more closely resemble and apply to clinical conditions.
The cellular and molecular mechanisms by which mesenchymal cells influence LV performance in the post-MI setting are severalfold, with paracrine (4,15), engraftment (19), and cardiostimulatory (6) mechanisms invoked in various acute and chronic models. In our study, evidence of enhanced arteriogenesis occurring in both the infarcted and border zones after cell injection was readily apparent. These findings are similar to those in previous studies in which MPCs were applied surgically within chronic ischemic tissue (5,12) and found to favorably impact myocardial healing and contractility.
The procedural success of catheter-based delivery is presently assessed according to safety parameters, rather than those of efficacy. While both rely on imaging modality to approximate spatial interrelationships between ventricular wall and catheter tip, determining that a given injection achieved its purpose, including appropriate distribution of injections across target areas, has been a challenge. Realtime visualization of the catheter tip and ventricular wall enables the methodical distribution of biological agents over specific targeted segments. From the perspective of clinical application, both radiography and guidance methods that employ adjunctive imaging fall short of these expectations. Real-time assessment of regional wall motion is difficult with fluoroscopy (including biplane imaging), especially over time as extracardiac landmarks shift with patient movement and other variables. Moreover, without marking the biological preparation with radiocontrast, the success of injections is impossible to ascertain. Alternatively, electromechanical mapping technology of endocardial topography is very useful for infarct delineation and regional targeting (2,17,18,24). However, it remains a costly, investigational technique that also has not been shown to identify successful injections in regions of myocardial fibrosis. Echocardiography is an accepted, clinically accessible imaging technique useful in the evaluation of myocardial function after myocardial infarction. High-resolution RT3DE is a relatively new iteration capable of generating detailed images of the LV endocardial surface, as well as MI size and healing (7). The major results of the current study show that delivery of MPCs using RT3DE guidance is a safe and effective innovative method of cell transplantation. In contrast to fluoroscopy alone, RT3DE allows the identification of the infarct and border zone, and it is possible to confidently guide injections into these regions of interest with high accuracy. The length of the needle was calibrated, and the location of the catheter tip was always confirmed before injection, with a mean procedural time that was relatively short (40 min) and improved as the number of cases increased (data not shown). In our study, we did not see any myocardial complications related to the catheter use. Additionally, as demonstrated in one notable example from our series, the determination of a successful injection may be possible with online 2D echocardiography; further studies will be required to confirm the utility of this image modality for such a purpose. Therefore, the combination of 3D real-time echocardiography and catheter-based injection is feasible.
In conclusion, transendocardial targeted injection of MPCs using RT3DE guidance effectively increases neovascularization and improves EF by increasing LV regional wall motions in an ovine model of heart failure. The results from this study suggested that RT3DE-guided catheter-based injections might provide a quick, safe, and effective method for percutaneous delivery of cells.
Footnotes
Acknowledgments
The authors would like to acknowledge Alyssa Flynn and Diane Ordanes for their support with animal care. This study was sponsored, in part, by Abbott Vascular (Santa Clara, CA) and Angioblast Systems, Inc. (New York, NY). Alexander Sheehy and Igor Polyakov are current employees of Abbott Vascular. Michael Schuster and Silviu Itescu are current employees of Angioblast Systems, Inc. At the time of the study, Timothy Martens was an employee of Angioblast Systems, Inc. At the time of the study, Shubhayu Basu was an employee of Abbott Vascular, Inc. At the time of the study, Juan F. Granada and Warren Sherman were consultants of Abbott Vascular, Inc. There are no other disclosures pertinent to this study.
