Abstract
Recently, adipose tissue-derived stem cells (ASCs) were emerged as an alternative, abundant, and easily accessible source of stem cell therapy. Previous studies revealed losartan (an angiotensin II type I receptor blocker) treatment promoted the healing of skeletal muscle by attenuation of the TGF-β signaling pathway, which inhibits muscle differentiation. Therefore, we hypothesized that a combined therapy using ASCs and losartan might dramatically improve the muscle remodeling after muscle injury. To determine the combined effect of losartan with ASC transplantation, we created a muscle laceration mouse model. EGFP-labeled ASCs were locally transplanted to the injured gastrocnemius muscle after muscle laceration. The dramatic muscle regeneration and the remarkably inhibited muscular fibrosis were observed by combined treatment. Transplanted ASCs fused with the injured or differentiating myofibers. Myotube formation was also enhanced by ASC+ satellite coculture and losartan treatment. Thus, the present study indicated that ASC transplantation effect for skeletal muscle injury can be dramatically improved by losartan treatment inducing better niche.
Keywords
Introduction
Muscle injuries and muscle regeneration are challenging problems in aging, traumatology, rare muscle diseases, and sports medicine (1). To date, many investigators have focused on antifibrotic therapy after muscle injury to induce muscle regeneration, which was not the most useful therapy for muscle injury. Recently, stem cells have emerged as an attractive source for regenerative medicine in aging and physical accidents. Although many researchers have realized the enormous possibility of stem cells in the study of organ recovery, the therapeutic efficacy of stem cells still remains a large problem to be solved. Therefore, the development of a new therapy is needed to enhance the efficacy of stem cell therapy for muscle regeneration.
Lately, adipose tissue-derived stem cells (ASCs) have been noted as an alternative source of representative adult stem cells due to their easy isolation and large availability with minimum discomfort (15,29,30,38,40). Moreover, previous studies reported that ASCs also differentiate into several mesenchymal tissues including adipocytes, endothelial cells, myocytes, chondrocytes, and osteocytes (9,16,24,31,38). Therefore, we focused on ASCs as a promising candidate for skeletal muscle regeneration. According to previous studies, chronic injury inducing fibrosis is mainly mediated by transforming growth factor (TGF)-β/Mothers against decapentaplegic homolog (Smad) signaling (10,28). Fibrosis is a common response caused by chronic organ injury, which shows the excessive deposition of extracellular matrix components mainly composed of collagen fibers and various other fibrous proteins (37). Recently, a study reported that a dense collagen network significantly reduces the penetration and efficacy of nanotherapeutics (8), which suggested that muscular fibrosis might disturb the proper migration of transplanted ASCs and deteriorate niche conditions for ASC differentiation. Inspired by that, we have concentrated on a more advanced stem cell therapy accompanying the modulation of niche conditions for maximum stem cell differentiation. Currently, the angiotensin II receptor blocker has been revealed to decrease fibrosis and improve the function of injured organs including the liver, muscles, lungs, and kidneys (7,22,25,35). Therefore, losartan, a representative angiotensin II type 1 receptor blocker, has emerged as a promising antifibrotic agent.
Based on these results, we hypothesized that losartan might dramatically improve the efficacy of stem cell therapy using ASCs for skeletal muscle injury. In order to investigate the effect of a combined therapy using ASCs and losartan and related mechanisms, the present study was performed utilizing a mouse skeletal muscle laceration model.
Materials and Methods
Isolation and Characterization of EGFP-Positive Adipose-Derived Stem Cells for Transplantation
Enhanced green fluorescent protein (EGFP) transgenic C57BL/6-Tg (CAG-EGFP) mice were anesthetized and the inguinal fat pad were harvested and washed with Dulbecco's phosphate-buffered saline (PBS). The fat tissues were then minced finely and digested with 0.075% collagenase type I in a 37°C shaking incubator for 30 min. After digestion, an equal volume of low-glucose Dulbecco's modified Eagle's medium (lgDMEM; Gibco, USA) with 10% fetal bovine serum was added to the digested tissue. After centrifugation at 3,000 rpm for 5 min, the cell pellet was collected carefully and washed with PBS. The cells were plated in culture plates and maintained in the lgDMEM containing 10% fetal bovine serum (FBS). ASCs from passage 3 to passage 5 were harvested and transplanted to the injured muscle area. To demonstrate the phenotype of ASCs, a fluorescence-activated cell sorting (FACS) analysis was performed. Passage 3 ASCs were harvested from the culture dish, and the 5 × 105 cells per 200 μl PBS were suspended. The cells were incubated with fluorescein isothiocyanate (FITC)-conjugated monoclonal antibodies: CD31-FITC, CD34-FITC, CD44-FITC, CD45-FITC, and CD90-FITC (all BD Pharmigen, CA, USA). Isotype-matched normal rat IgG was used as the control.
Animals
Eight-week-old male C57BL/6 mice (n = 40) were used in this study. All mice were obtained from Japan SLC, Inc. The mice were maintained in a room at 22 ± 3°C with a relative humidity of 50 ± 10% and a 12-h light–dark cycle and were given food and water ad libitum. All animal procedures were performed in accordance with the U.S. National Institutes of Health (NIH) guidelines and approved by the Kyungpook National University Institutional Animal Care and Use Committee (IACUC) for the care and use of laboratory animals.
Animal Model and Experiment Design
All mice were divided into four groups [control group (n = 10), losartan group (n = 10), ASC group (n = 10), and losartan + ASC group (n = 10)]. Local muscle lacerations were performed in the gastrocnemius muscles of all mice. After administering anesthesia, a skin incision was performed in the left leg and the gastrocnemius muscle was exposed. Then the muscle was lacerated at the central part of the muscle using a biopsy punch, which had a diameter of 4 mm. The shape of muscle defect was round, and the size was approximately 4 mm in diameter and 3 mm in thickness. After muscle laceration, the isolated EGFP-positive ASCs (5 × 105 cells/10 μl PBS per animal) were transplanted into the defect of the muscle and then the defect was closed with the fascia and skin. The same volume of PBS without cells was treated to the injured muscle area of the control group and the losartan group. The control group and the ASC group were fed tap water, while the losartan group and the losartan + ASC group were fed losartan (MSD Korea, South Korea) dissolved in tap water at a concentration of 0.6 g/L ad libitum for 2 weeks (7). After 2 weeks, all mice were sacrificed, and the injured gastrocnemius muscles were isolated.
Gross Assessment of Lacerated Muscle
After 2 weeks, the lacerated areas of all mice were evaluated by gross examination. The healing area including the lacerated area was captured using a digital camera. The injured muscles were isolated, and the weight of each muscle sample was measured immediately.
Serum Biochemical Analysis
After 2 weeks, all mice were sacrificed, and serum samples were collected and stored at −70°C for analysis. The serum creatine kinase level was measured by standard enzymatic procedures (Konelab20, Thermo Clinical Labsystems, Vantaa, Finland).
Histopathological Assessment of Lacerated Muscle for Morphological Evaluation of Fibrosis and Muscle Regeneration
Gastrocnemius muscle tissues were rapidly isolated and fixed in 10% neutral buffered formalin. Then the muscle tissues were processed routinely and embedded in paraffin wax. The tissues were cut into 4-μm thickness. The sections were deparaffinized in toluene and rehydrated in a graded alcohol series. The muscle sections were stained with hematoxylin and eosin (H&E) for light microscopic examination. To observe muscle fibrosis, the tissue sections were stained with Masson's trichrome stain. The fibrotic area was quantified using Leica Application Suite software (LAS version 2.8.1, Leica Microsystem) in the 40x fields of each muscle tissue. The number of regenerative (centronucleated) myofibers around the lacerated area was counted in the 200x fields of each muscle tissue.
Immunofluorescence Observation
The isolated muscle tissues were embedded in freezing medium and serially sectioned at 8-μm thickness. The sectioned tissue slides were fixed in 4% paraformaldehyde at 4°C for 20 min and immunostained with the following primary antibodies: myogenin (Santa Cruz Bio technology, CA, USA), myogenic differentiation (MyoD) (Santa Cruz Biotechnology), paired box protein (Pax) 7 (Developmental Studies Hybridoma Bank, Tokyo, Japan), and troponin I (Santa Cruz Biotechnology). Tetramethyl rhodamine isothiocyanate (TRITC)-conjugated anti-mouse IgG for myogenin, MyoD, Pax7, and anti-rabbit IgG antibodies for troponin I were used as secondary antibodies (Invitrogen, CA, USA). 4′,6-Diamidino-2-phenylindole (DAPI, dihydrochloride) (Molecular Probes, Leiden, the Netherlands) staining was performed for nuclear count staining.
Immunoblotting Analysis
The freshly isolated muscle tissues were rapidly frozen and homogenized in RIPA buffer containing 0.1 mmol/L Na3VO4 and protease inhibitor cocktail tablets (Roche, Mannheim, Germany). For the extraction of the soluble proteins, electrophoresis, and immunoblotting, the muscle tissue samples were routinely processed, as described previously (27). After being blocked with a 3% bovine serum albumin in Tris-buffered saline (TBS), the membranes were incubated with the following primary antibodies: anti-myogenin, anti-MyoD, anti-Wnt3a, anti-β-catenin (all from Santa Cruz Biotechnology), and anti-p-Smad3 (Cell Signaling Technology, MA, USA). Equal quantity of loaded protein was confirmed by detecting β-actin expression using a polyclonal mouse anti-β-actin (Santa Cruz Biotechnology). The specific bindings were visualized by using the Super Signal West Dura Extended Duration Substrate (Pierce, IL, USA) and exposing the blots to medical X-ray film (Kodak, Tokyo, Japan).
Reverse Transcription-Polymerase Chain Reaction (RT-PCR) Analysis
Total RNA was isolated from the frozen muscle tissue using Trizol reagent (Invitrogen). Briefly, frozen muscle tissues were homogenized in 1 ml of Trizol reagent. Total RNA was separated from proteins and DNA by adding chloroform and precipitated by isopropanol. The concentration of the total RNA was measured using a Quanti-iT RNA assay kit (Invitrogen) and a Qubit fluorometer (Invitrogen). For cDNA synthesis, the extracted RNA was used as a template for reverse transcription using a commercial RT&GO mastermix (MP Biomedicals, OH, USA) according to the manufacturer's instructions. The synthesized cDNA was amplified by using primers listed in Table 1. PCR products were detected by electrophoresis in 1.5% agarose gel.
Nucleotide Sequence of Primers Used for RT-PCR

See text for gene definitions.
Isolation of Satellite Cells
The gastrocnemius muscles were isolated from the injured or normal hindlimbs of 12-week-old C57BL/6J mice. The isolated muscle tissue was dissociated with 0.1% collagenase for 1 h, 0.2% dispase for 1 h, and 0.1% trypsin for 30 min. The preplate technique was used to isolate the slowly adhering satellite cells from the rapidly adhering cells as described previously (11). Slowly adhering satellite cells in the last two steps of preplating process, PP5 and PP6, requiring 6–8 days for the generation, were obtained and suspended in 35-mm tissue culture dishes coated with collagen.
Coculture with Adipose-Derived Stem Cells and Satellite Cells
For direct coculture, the myogenic satellite cells were plated onto six-well plates with a density of 2 × 104 cells/ cm2 in growth medium (lgDMEM, 10% FBS, 1% antibiotics). After 1 day, EGFP-positive ASCs were seeded on the same area of the six-well plates with a density of 1 × 104 cells/cm2. After one additional day, the medium was changed to differentiation medium (high-glucose (hg) DMEM, 5% horse serum, 1% antibiotics). From day 1 to day 7, the cocultured cells were treated daily with losartan (10 μM), angiotensin II (1 μM; Sigma-Aldrich, USA), and transforming growth factor (TGF)-β (4 ng/μl; R&D Systems, USA) for 7 days. The cells were also observed microscopically, fixed for immunofluorescence, or lysed for RT-PCR analysis.
Effect of Losartan, TGF-β1, and ALK5 Inhibitor on Myogenesis In Vitro
Nonadherent satellite cells (preplate passages 5–6) were seeded on 0.1% gelatin-coated six-well plate (1 × 106 cells/well) supplemented with hgDMEM with 10% FBS and 5% horse serum made with sterile, deionized water (growth medium), and grown for 2 days in the presence or absence of losartan (10 μM) with or without TGF-β1 (0.025 ng/ml) or activin receptor-like kinase 5 (ALK5; TGF-β1 receptor) inhibitor (LY364947, 2 μM; Sigma-Aldrich).
Statistical Analysis
All results taken from each group were shown as mean ± SD. The statistical significance between two experimental groups was determined with a Student's t test. The statistical significance among three or more experimental groups was determined by one-way analysis of variance (ANOVA) and Bonferroni post hoc test. All analyses were performed using GraphPad InStat (version 3.05, GraphPad Software, Inc.) The statistical significance value was set at p < 0.05 or p < 0.01.
Results
Characterization of ASCs
For characterization of the ASCs using a FACS analysis, ASCs showed positive reactions for the stromal cell-related makers CD44 and CD90, whereas the cells were negative for CD31 (endothelial cell-related marker) as well as CD34 and CD45 (hematopoietic cell-related marker) (Fig. 1A). In the RT-PCR analysis, the freshly isolated ASCs showed no expression of MyoD, myogenin, and myogenic factor 5 (Myf5), which demonstrated that ASCs do not express muscle differentiation-related genes (Fig. 1B).

Characterization of adipose tissue-derived stem cells (ASCs) and representative gross findings of the lacerated muscle at week 2 after muscle laceration. Characterization of isolated and cultured passage 3 ASCs was performed by fluorescence-activated cell sorting (FACS) analysis and RT-PCR analysis. (A) The FACS analysis was performed using cluster of differentiation (CD) markers including CD31-FITC, CD34-FITC, CD44-FITC, CD45-FITC, and CD90-FITC antibodies. The cells showed a positive reaction for stromal cell-related makers, which are CD44 and CD90, whereas the cells were negative for CD31 (endothelial cell-related marker) and CD34 and CD45 (hematopoietic cell-related marker). (B) RT-PCR analysis of the cultured passage 3 ASCs. There was no expression of myogenic markers in cultured ASCs. (C) Losartan + ASC group shows dramatically improved muscle regeneration. (D) The moist muscle weight value also indicates significantly increased values compared with control lacerated gastrocnemius muscle tissues. (E) Serum creatine kinase levels showed the most significantly decreased levels in the losartan + ASC group. Data are shown as mean ± SD (∗p < 0.05, ∗∗p < 0.01).
Morphological and Serum Biochemical Regeneration of the Lacerated Skeletal Muscle
At 2 weeks after laceration, the lacerated regions were remarkably recessed in the losartan-treated group and the ASC-treated group. Surprisingly, the gastrocnemius muscles of the losartan + ASC group had recovered enough to appear to be almost the same as the intact gastrocnemius muscle of right hind limb. In the measurement of the moist weight of each isolated muscle, there was a significant increase in the losartan + ASC group compared to the control group. Surprisingly, the weight value of the losartan + ASC group was almost the same with the intact right gastrocnemius muscle (Fig. 1C, D). To confirm the morphological muscle regeneration, a serum biochemical analysis was performed for the measurement of the serum creatine kinase level. As expected, the losartan + ASC group showed the largest decreased in serum creatine kinase levels (Fig. 1E), confirming that the combined treatment using losartan and ASCs dramatically promoted muscle regeneration.
Histopathological Evaluation of Muscle Fibrosis and Regeneration
In Masson's trichrome staining for the assessment of fibrosis, the greatest amount of ameliorated muscle fibrosis was observed in the losartan + ASC group, which indicates that the combined treatment with losartan and ASCs can induce a synergistic antifibrotic effect in muscle injury (Fig. 2A, B). The result of H&E staining revealed that the losartan + ASC group had the highest number of regenerative (centronucleated) myofibers compared with the other groups (Fig. 2C, D), which indicated that significantly elevated muscle regeneration was induced by the losartan and ASC combined treatment. Moreover, stromal cells differentiating into regenerative muscle fibers were observed in the losartan + ASC group.

Representative histological observation of lacerated gastrocnemius muscle. The losartan + ASC group shows the most decreased fibrosis and the highest number of regenerative myofibers among all groups. (A) Losartan + ASC combined treatments revealed the most ameliorated muscular fibrosis induced by muscle laceration. The muscle tissues were stained by H&E and Masson's trichrome stain. Scale bars: 100 μm. (B) Histological area of muscular fibrosis. The fibrotic area was measured by computer analysis using the Leica Application Suite (Version 2.8.1) software in all mice. (C) The losartan + ASC group showed the most enhanced muscle regeneration showing the highest number of centronucleated regenerative myofibers. Scale bars: 50 μm. In higher magnification, the spindle-shaped stromal cells differentiating into eosinophilic myoblast-like cells were observed in the losartan + ASC group (black arrows). H&E stain. Scale bar: 20 μm. (D) The number of regenerative myofibers was dramatically increased in the losartan + ASC group compared to the other groups. Regenerative myofibers were counted around lacerated area in 200x fields of all muscle tissue. (E) Confirmation of enhanced green fluorescent protein (EGFP)-labeled ASC transplantation to the lacerated area. (A) Transplantation of EGFP-labeled ASCs was observed in lacerated areas of gastrocnemius muscles. Scale bar: 50 μm. (B) The EGFP-labeled ASCs were fusing to injured muscle fibers and regenerative muscle fibers. EGFP-positive muscle fibers were also detected, and all these EGFP-positive cells (white arrows) were observed at 480-nm wavelength of a confocal microscope. Scale bar: 20 μm. (C) The EGFP-positive cells (black arrows) fusing to regenerative muscle fibers were also observed in histopathological observation of other serial sections. H&E stain. Scale bar: 40 μm. Data are shown as mean ± SD (∗p < 0.05, ∗∗p < 0.01).
Confirmation of Transplanted EGFP-Labeled ASCs in Lacerated Gastrocnemius Muscle
To confirm if the injected EGFP-labeled ASCs were transplanted to the injured area of the muscle, the skeletal muscle tissues were analyzed using a confocal microscope. Interestingly, the injected ASCs were observed at the wave length of 488 nm, which showed that the EGFP-positive cells were incorporated into the regenerative skeletal muscle fibers. EGFP-positive muscle fibers were also present in the injured muscle tissue. In the histopathological observation via H&E staining, the EGFP-positive cells fusing to the injured muscle fibers were identified as mesenchymal-like or fibroblast-like cells, which were larger than normal satellite cells (Fig. 2E).
Combined Treatment Using ASCs and Losartan Dramatically Increase Myogenin and MyoD Expression in Muscle Laceration
In the immunofluorescent assessment, the losartan + ASC group showed a greater increase in myogenin expression levels in the injured or regenerative skeletal muscle fibers compared with those of the losartan group and the ASC group (Fig. 3A). In the RT-PCR analysis, the losartan + ASC group also exhibited the highest level of myogenin mRNA (Fig. 3B), which revealed a similar pattern with the results of immunofluorescence and immunoblot analysis (Fig. 3C). In the protein expression level of MyoD (Fig. 3D), there was no significant increase in the losartan-treated group, while the losartan-treated group as well as the ASC-treated group revealed significantly elevated MyoD mRNA levels (Fig. 3E). The ASC group and the losartan + ASC group showed significantly increased protein expression levels when compared with the control group (Fig. 3F). Surprisingly, numerous EGFP-positive muscle fibers were observed in the losartan + ASC group, whereas only a few EGFP-positive cells infiltrating the injured skeletal muscle fibers were observed in the ASC group (Fig. 3), which indicates that losartan might play an important role in niche formation for muscle differentiation.

Dramatically increased myogenin and myogenic differentiation (MyoD) expression by losartan + ASC combination treatment. (A) Immunofluorescent observation of myogenin expression in lacerated muscle tissue. The losartan + ASC group revealed the strongest expression level of myogenin. Scale bars: 50 μm. (B) RT-PCR analysis for myogenin mRNA expression in the lacerated muscle tissues. Graph represents myogenin mRNA expression levels relative to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) in all groups. (C) Immunoblot analysis for myogenin expression level in lacerated muscle tissues. Graph shows the relative band density to β-actin. (D) Immunofluorescent finding of MyoD expression in lacerated muscle tissue. Scale bars: 50 μm. (E) RT-PCR analysis for MyoD mRNA expression level in lacerated muscle tissues. Graph represents MyoD mRNA expression levels relative to that of GAPDH. (F) Immunoblot analysis for MyoD expression levels in lacerated muscle tissues. Graph shows the relative band density to β-actin. Data are shown as mean ± SD (∗p < 0.05, ∗∗p < 0.01).
Pax7 Expression Was Significantly Upregulated by Losartan and ASC Combined Treatment
To evaluate the effect of the combined treatment on the self-renewal of skeletal muscle progenitor cells, Pax7 expression level was measured by immunofluorescent staining. Dramatically elevated Pax7 expression was observed in the losartan + ASC group (Fig. 4A), while by RT-PCR, only the losartan + ASC group indicated significantly upregulated Pax7 mRNA levels whereas there was no significant increase of Pax7 mRNA levels in the losartan group and the ASC group compared to the control-L group (Fig. 4B).

Significantly upregulated paired box protein 7 (Pax7) expression level by combined treatment and significantly increased Wnt3a and transforming growth factor (TGF)-β signaling in ASC-transplanted muscle tissues (A) Immunofluorescent staining of Pax7 expression in the lacerated muscle tissues. Scale bars: 50 μm. (B) RT-PCR analysis for Pax7 mRNA expression levels in lacerated muscle tissues. Graph indicates Pax7 mRNA expression level relative to that of GAPDH. (C) Immunoblotting analysis of the Wnt3a, β-catenin, and phosphorylated Mothers against decapentaplegic homolog 3 (p-Smad3). Losartan treatment downregulated the p-Smad3 expression of ASC-transplanted muscle tissue. Graph indicates relative expression levels of Wnt3a (D), β-catenin (E), and p-Smad3 (F) to β-actin expression. Data are shown as mean ± SD (∗p < 0.05, ∗∗p < 0.01).
Combination of Losartan and ASCs Promotes Muscle Differentiation and Downregulation of TGF-β Expression
ASC injections induced the elevation of Wnt3a, β-catenin, and p-Smad3 protein expression, which suggests that ASC transplantation might stimulate the Wnt/β-catenin pathway and TGF-β/Smad signaling (Fig. 4C–F). The losartan + ASC group revealed significantly inhibited p-Smad3 expression compared with the ASC group, which indicated that losartan might play a pivotal role in niche formation by suppressing upregulated TGF-β/ Smad signaling (Fig. 4F).
Losartan Dramatically Upregulated MyoD and Myogenin Expression Inducing Myotube Formation in Coculture of Satellite Cells and ASCs
Isolated satellite cells were observed to form myotubes with myogenic differentiation (Fig. 5A), confirming that the isolated cells were myogenic cells. We also demonstrated that the mRNA expressions of Pax3, Pax7, Delta-like 1 (Dlk1), and Notch1 were decreased, whereas those of Wnt3a, dishevelled (Dvl), lymphoid enhancer-binding factor 1 (LEF1), T-cell-specific transcription factor (TCF), and myogenin increased throughout the myogenic differentiation of the isolated satellite cells, which suggests that Wnt signaling increases whereas Notch is downregulated with myogenic differentiation (Fig. 5B, C). To confirm the results from the in vivo experiments and to evaluate the effect of losartan on the differentiation of satellite cells and ASCs, isolated satellite cells and passage 3 ASCs were cocultured for 1 day in growth medium and for 7 days in differentiation medium. On day 3, we started to observe the formation of contracting myotubes. The coculture of satellite cells and ASCs showed a more increased number of myotubes and more activated satellite cell morphology compared with pure culture of satellite cells (data not shown). On day 7, we could detect remarkably increased myotube formation in the losartan-treated wells compared with non-losartan-treated wells (Fig. 6A). The cells were harvested, and RT-PCR analysis was performed to observe the MyoD and myogenin gene expression level. As expected, in the losartan-treated cells, strongly elevated MyoD and myogenin mRNA levels were observed (Fig. 6B). To confirm whether the EGFP-labeled ASCs could differentiate into muscle fibers, the cells cocultured for 7 days were fixed, and immunofluorescent staining was performed using an antibody for troponin I, a marker of myogenic differentiation, and visualized using a confocal microscope. Interestingly, we detected that EGFP-positive ASCs and myotubes were double-positive for both EGFP and troponin I. Moreover, to our surprise, we also observed the fusion of EGFP-positive ASCs to the myotubes expressing troponin I (Fig. 6C). These results suggest the possibility that ASCs can differentiate into the myotubes and fuse to the myotubes. Additionally, we observed that TGF-β inhibited muscle differentiation and self-renewal capacity by downregulating stemness factors [Krüppel-like factor 4 (Klf4), myelocytomatosis viral oncogene homolog (cMyc), Nanog], self-renewal factors [Pax7, Dlk1, Notch1, glycogen synthase kinase 3β (Gsk3b)], and myogenic differentiation factors (MyoD, myogenin, β-catenin) in isolated satellite cells (Fig. 7A, B).

Myogenic differentiation of isolated satellite cells. (A) Isolated satellite cells show numerous myotube formation. Scale bars: 100 μm. (B, C) The isolated satellite cells revealed high levels of self-renewal related mRNA expression including Pax3, Pax7, Delta-like 1, and Notch1 at the early stage, whereas myogenic differentiation mRNA expression, such as desmin, myogenin, and Wnt signaling, was remarkably increased at late stage. Data are shown as mean ± SD (∗p < 0.05, ∗∗p < 0.01).
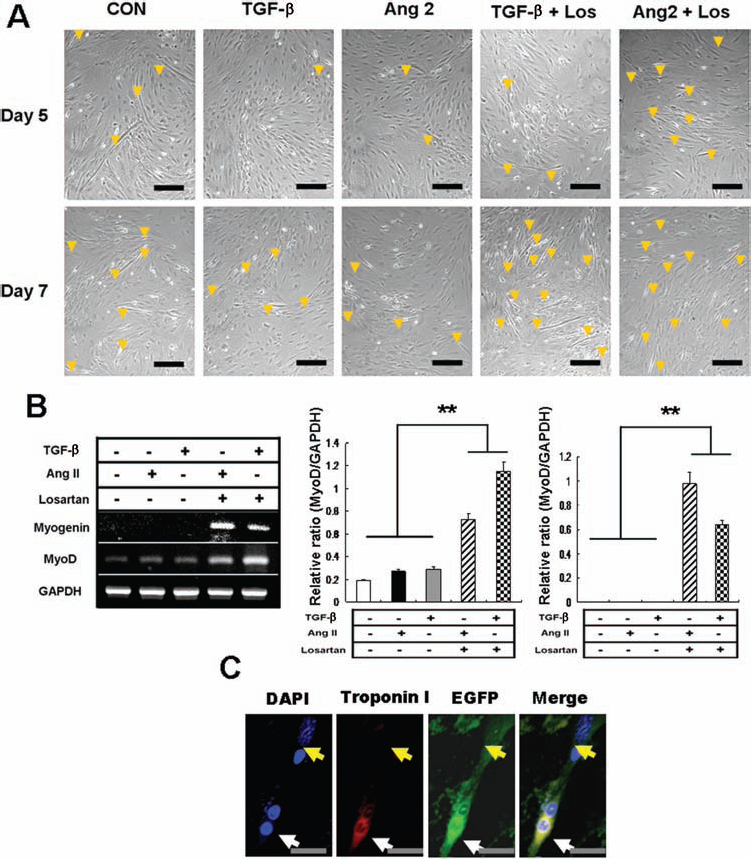
Promoted formation of myotubes by losartan treatment in cocultures. (A) Isolated satellite cells from hind limb of wild-type C57BL/6 mice and passage 3 ASCs isolated from inguinal adipose tissue of EGFP transgenic mice were cocultured for 7 days. Remarkably increased formation of myotubes was observed in losartan-treated well at day 7. Scale bars: 50 μm. (B) Total RNA was extracted from cocultured cells for RT-PCR analysis. The losartan-treated cells show dramatically increased mRNA levels of myogenin and MyoD. (C) Incorporation of ASCs into differentiating myotubes. The myogenic differentiation showing myotube formations was confirmed by immunofluorescent observation using troponin I antibody (red). The EGFP-labeled (green) ASCs (yellow arrow) are being incorporated into an EGFP-positive (green) myotube (white arrow). The EGFP-positive myotube also indicates positive reaction for troponin I antibody showing double EGFP/troponin I positivity. Scale bars: 10 μm. Data are shown as mean ± SD (∗p < 0.05, ∗∗p < 0.01).

(A) Suppression of stemness factors [Krüppel-like factor 4 (Klf4), myelocytomatosis viral oncogene homolog (cMyc), Nanog], self-renewal factors [Pax7, Dlk1, Notch1, glycogen synthase kinase 3b (Gsk3b)], and (B) myogenic differentiation factors (MyoD, myogenin, β-catenin) by TGF-β treatment in RT-PCR analysis using isolated satellite cells. Data are shown as mean ± SD (∗p < 0.05, ∗∗p < 0.01).
Losartan Stimulates Self-Renewal and Myogenic Differentiation of Satellite Cells
Next, we investigated the effects of losartan in isolated satellite cells. Losartan upregulated the mRNA expressions of promyogenic factors such as Pax7, Notch1, Wnt3A, MyoD, desmin, and myogenin as compared with those of the control (Fig. 8A). Moreover, losartan overcame the antimyogenic effects of TGF-β1 through restoring the mRNA expressions of Notch1, Pax7, desmin, and MyoD. Concomitantly, losartan neutralized the effects of TGF-β1 on the mRNA expressions of Klf4 and Nanog. The inhibitor (LY364947) of ALK5 directly blocks TGF-β signaling. Therefore, we investigated the effects of the ALK5 inhibitor on mRNA expressions related to myogenesis (Fig. 8B). The combination of ALK5 inhibitor with losartan activated Wnt signaling, which is suppressed by the former, leading to increased myogenic differentiation. In the view of the mRNA expression levels, losartan functioned as an antagonist of TGF-β1 by upregulating those suppressed genes by TGF-β1 and thereby resulting in an increase in both self-renewal and myogenic differentiation of satellite cells.

Enhanced self-renewal and myogenic differentiation of satellite cells by losartan treatment. (A) Nonadherent satellite cells were seeded on 0.1% gelatin-coated six-well plates (1 × 106 cells/well) and grown for 2 days in the presence or absence of losartan (10 μM) with or without TGF-β1 (0.025 ng/ml) or activin receptor-like kinase 5 (ALK5) inhibitor (2 μM). The effect of TGF-β1 and losartan on mRNA expression related with self-renewal and myogenic differentiation of isolated satellite cells. (B) The effect of ALK5 inhibitor and losartan on myogenic differentiation of isolated satellite cells. (C) Promoted myogenic differentiation of myoblasts without losing its stemness. Myoblasts were seeded on 0.1% gelatin-coated six-well plates (1 × 106 cells/well) and grown for 7 days in the presence of or absence of losartan (10 μM). Total RNA was isolated from cells on each indicated day of incubation, and the differences among mRNA expressions on the basis of incubation time were determined by the RT-PCR analysis. The expression of GAPDH indicates equal loading of cDNA. The RT-PCR analysis shows that losartan increases myogenic differentiation (Pax7, MyoD, myogenin, Delta, Notch1, Wnt3a, β-catenin) and stemness (Klf4, Nanog, Rex1) of satellite cells. Data are shown as mean ± SD (∗p < 0.05, ∗∗p < 0.01).
Losartan Promotes Differentiation of Satellite Cells Without Losing Their Stem Cell Properties
The mRNA expressions of Klf4, Nanog, and reduced expression 1 (Rex1), which are stemness factors, along with Pax7 and Notch1, which are related to the self-renewal of satellite cells, were maintained longer in cultures of satellite cells in the presence of losartan than in the absence of it (Fig. 8C). Furthermore, mRNA expressions of Wnt3a and β-catenin, which are related to the differentiation of satellite cells into myocytes, were more increased in cultures with losartan than without it. At 7 days, the expressions of stemness factors were still shown while differentiation factors were not, indicating that the self-renewal of satellite cells for regeneration after the death of differentiated cells due to aging was more activated in cultures with losartan.
Discussion
In the present study, we demonstrated for the first time the synergistic effect of therapy combining the use of losartan and ASCs. Consistently with previous studies (14,24,32), isolated ASCs were positive for stromal cell markers and negative for endothelial cell markers and hematopoietic cell markers, showing that the isolated ASCs originated from mesenchyme (Fig. 1A). It has been shown that a few mesenchymal spindle cells differentiate to myoblast-like cells characterized by a central nucleus and prominent eosinophilic cytoplasm (Fig. 2C, arrow), which made us speculate that ASCs might have the potential for muscular differentiation. EGFP-positive cells were observed around the injured muscle fibers, and a few muscle fibers were EGFP positive. We also observed that the EGFP-positive ASCs fuse to the injured muscle fibers (Fig. 2E). Based on these results, we confirmed that the transplanted ASCs were incorporated into the healing area.
Skeletal muscle differentiation of isolated satellite cell was remarkably promoted by ASC coculture (data not shown), which suggested that ASCs can incorporate into skeletal myotubes. We also checked the effect of losartan on myotube formation in coculture experiments. While the skeletal myotube formation was inhibited by TGF-β or angiotensin II treatments, losartan treatment not only promoted skeletal myotube formation but also significantly increased the expression level of skeletal muscle differentiation markers in cocultured cells (Fig. 6A, B). These results indicate that the incorporation of ASCs into skeletal myotubes can be promoted by an improved stem cell niche via losartan treatment. Then we pose the question: is it possible for ASCs to differentiate into skeletal myotubes? In the present study, we observed ASC-like spindle cells differentiating into myoblasts (Fig. 2C, arrow) as well as ASC-like mesenchymal cells fusing to injured or regenerative skeletal muscle fibers (Fig. 2E) in microscopic findings of ASC-transplanted muscle tissue via H&E stain. Interestingly, in the cocultures of EGFP-labeled ASCs with isolated satellite cells, we have observed an almost similar phenomenon as that of the in vivo result showing EGFP-positive ASCs fusing to EGFP-positive differentiating skeletal myotubes (Fig. 6C). Since we used only EGFP-negative satellite cells, we concluded that the EGFP-positive myotubes originated from EGFP-positive ASCs. Therefore, these results suggest that ASCs can convert to myogenic phenotype-forming skeletal myotubes and ASCs also can incorporate into skeletal myotube formation by fusing to differentiating myotubes. A recent previous study revealed that adipose tissue-derived mesenchymal stem cells have skeletal myogenic potential by being directly cocultured with myoblasts (9). Consistent with the previous study, our results also indicated the myogenic differentiation of ASCs into skeletal myotubes and the fusion of ASCs to differentiating skeletal muscle fiber. Therefore, it is thought that transplanted ASCs might incorporate into skeletal muscle differentiation by direct contact with myogenic satellite cells.
Wnt signaling has been known to enhance myogenic proliferation and differentiation in muscle development (3). In the present study, we found that ASC transplantation induced an upregulation of Wnt/β-catenin signaling (Fig. 4C). According to other studies, however, Wnt signaling is also increased in fibrosis status, suggesting that Wnt signaling is involved with fibrogenesis (6,12,18,22,23). Additionally, it has recently been demonstrated that Wnt/β-catenin signaling influences TGF-β signaling by way of modulating Smad3 protein stability (12). Since TGF-β has been known to be the most important factor in fibrogenesis (28), we speculated that the transplanted ASCs may have the potential to differentiate into fibroblasts or myofibroblasts to induce muscle fibrosis. Thus, we have checked p-Smad3 expression levels in lacerated muscle tissues by immunoblot analysis. Interestingly, the ASC group showed the most increased p-Smad3 expression level, whereas the losartan + ASC group exhibited significantly lower p-Smad3 expression level compared with that of the ASC group (Fig. 4C). Since we induced surgical muscle injury via muscle laceration inducing muscular fibrosis, p-Smad3 expression has been dramatically increased, suggesting upregulation of TGF-β signaling pathway. This abnormally increased TGF-β signaling induces poor and bad microenvironment for skeletal muscle differentiation. Therefore, it seems that the upregulated TGF-β signaling leads transplanted ASCs to differentiation into myofibroblasts or blood vessels, which inhibits ASC differentiation into skeletal muscle fibers. However, promoted angiogenesis by ASC transplantation may reinforce skeletal muscle regeneration indirectly (13,21,34), although a few direct fusions of ASCs into injured skeletal muscle fibers were also observed. Moreover, several previous studies demonstrated that TGF-β signaling plays a critical role in the differentiation of stromal mesenchymal stem cells into various mesenchymal tissues including bone, cartilage, vessels, and fat tissues (14,30). Especially, Salazar et al. demonstrated that mesenchymal stem cells contain similar TGF-β level to the level produced by lung fibroblasts playing an important role in pulmonary fibrosis and activation of TGF-β in MSCs upregulated collagen gene expression (30). Additionally, they also reported that high level of TGF-β produced by MSCs and fibroblasts can induce matrix production (30). Thus, upregulation of p-Smad3 by ASC transplantation seems to be associated with promoted various mesenchymal differentiation including fibroblasts, myofibroblasts, and vessel differentiation in the present study.
TGF-β1 has been well known as an antimyogenic factor associated with fibrosis, which reduces skeletal muscle regeneration and leads to skeletal muscle degeneration, especially in skeletal muscular dystrophy (1,4,5). The use of angiotensin II type I receptor blockade, losartan, revealed to reduce TGF-β1 signaling, resulting in promising effects in skeletal muscle regeneration in mouse models of Marfan syndrome and muscular dystrophy (7). Therefore, we speculated that the myogenic potential can be enhanced by losartan treatment, which might induce the synergistic effects of ASC therapy for skeletal muscle injury. To further assess for the effect of losartan on isolated satellite cells, the expression of skeletal myogenic factors was suppressed by TGF-β (Fig. 7) and the losartan treatment restored it by inhibiting TGF-β pathway. Furthermore, losartan also increased the expression of stemness factors related to the self-renewal of stem cells maintaining high expression levels of Wnt3a and β-catenin (Fig. 8A, C). These results indicate that losartan promote the skeletal muscle differentiation of satellite cells without decreasing stemness.
Pax7 was well known to increase after acute muscle injury (20). This increased Pax7 plays a pivotal role in satellite cell self-renewal for skeletal muscle regeneration and development (20,26). In the present study, we showed losartan treatment increased Pax7 mRNA expression level in isolated satellite cells (Fig. 8A). However, this was not seen in vivo data, which indicated no significant difference among control, losartan, and ASC sample groups (Fig. 4A, B). Since Pax7 has been known to express specifically in proliferating satellite cells, these results suggest that single ASC or losartan treatment cannot efficiently improve self-renewal in vivo experiment. However, in combined treatment, it seems that losartan not only induce better niche condition for transplanted ASC to differentiate into proliferating myogenic satellite cells expressing Pax7 but also increase the fusion of transplanted ASC into injured or regenerative skeletal muscle fibers.
Angiotensin II has been known to promote fibrosis by activating TGF-β signaling in various organs including skeletal muscle, cardiac muscle, and the liver (7,22,36). Especially, in skeletal muscle and cardiac muscle, losartan plays an important role in inhibition of fibrosis as well as the promotion of muscle regeneration. However, losartan treatment cannot be the essential therapy for genetic muscular disease such as Becker muscular dystrophy and Duchenne muscular dystrophy. To overcome this problem, many researchers have tried to use stem cell therapy for muscle disease. To date, several types of mesenchymal adult stem cells including peripheral blood-derived cells, endothelial progenitor cells (EPCs), bone marrow-derived cells (BMCs), and ASCs have been tried for stem cell therapy to restore skeletal muscle function (1,12,19,32). Among them, ASCs have been regarded as an alternative source of adult mesenchymal stem cells owing to their abundance and ability to be easily isolated. However, in many previous studies, mesenchymal stem cells including ASCs mainly differentiated into blood vessels or revealed therapeutic effects indirectly by secreting growth factors (17,19,24). A few previous studies also showed that increased angiotensin II and TGF-β induce differentiation of ASCs to smooth muscle-like cells (16,39). Furthermore, Shi et al. revealed that angiotensin II induces vascular endothelial growth factor synthesis in mesenchymal stem cells (33). The studies mentioned above suggest that ASCs easily differentiate into blood vessels or myofibroblastlike cells, especially in chronically injured areas secreting high levels of TGF-β. In our study, we demonstrated that the normalization of increased TGF-β signaling by losartan treatment improves the stem cell niche in injured skeletal muscles and promotes skeletal differentiation of the transplanted ASCs.
To summarize, we concluded that losartan promoted skeletal myogenic differentiation of transplanted ASCs by enhancing the microenvironment condition induced by inhibited TGF-β signaling pathway and increased differentiation capacity of myogenic cells (Fig. 9). Losartan is an FDA-approved drug, which is commercially available, well-tolerated, safe, and commonly employed in clinical practice (2,4). Additionally, ASCs are also known to be easily isolated from patient's fat tissue. Therefore, it is strongly believed that combined therapy using ASCs and losartan can be easily achieved clinically and can be potential antifibrotic therapy for skeletal muscle injury.

Scheme for proposed mechanism of losartan treatment improving stem cell niche in ASC transplantation. The scheme indicates how losartan improves the microenvironment of ASCs in muscle regeneration. TGF-β signaling is activated by ASC transplantation, and losartan improves the niche for muscle differentiation of stem cells including ASCs and satellite cells by downregulating the increased TGF-β signaling.
Footnotes
Acknowledgments
This research was supported by Basic Science Research Program through the National Research Foundation (NRF) of Korea funded by the Ministry of Education, Science, and Technology (2010-0022587). The authors declare no conflicts of interest.
