Abstract
Impaired vascularity and nerve degeneration are the most important pathophysiological abnormalities of diabetic polyneuropathy (DPN). Therefore, regeneration of both the vascular and nervous systems is required for the treatment of DPN. The neural crest (NC) is a transient embryonic structure in vertebrates that differentiates into a vast range of cells, including peripheral neurons, Schwann cells, and vascular smooth muscle cells. In this study, we investigated the ability of transplantation of NC-like (NCL) cells derived from aged mouse induced pluripotent stem (iPS) cells in the treatment of DPN. iPS cells were induced to differentiate into neural cells by stromal cell-derived inducing activity (SDIA) and subsequently supplemented with bone morphogenetic protein 4 to promote differentiation of NC lineage. After the induction, p75 neurotrophin receptor-positive NCL cells were purified using magnetic-activated cell sorting. Sorted NCL cells differentiated to peripheral neurons, glial cells, and smooth muscle cells by additional SDIA. NCL cells were transplanted into hind limb skeletal muscles of 16-week streptozotocin-diabetic mice. Nerve conduction velocity, current perception threshold, intraepidermal nerve fiber density, sensitivity to thermal stimuli, sciatic nerve blood flow, plantar skin blood flow, and capillary number-to-muscle fiber ratio were evaluated. Four weeks after transplantation, the engrafted cells produced growth factors: nerve growth factor, neurotrophin 3, vascular endothelial growth factor, and basic fibroblast growth factor. It was also confirmed that some engrafted cells differentiated into vascular smooth muscle cells or Schwann cell-like cells at each intrinsic site. The transplantation improved the impaired nerve and vascular functions. These results suggest that transplantation of NCL cells derived from iPS cells could have therapeutic effects on DPN through paracrine actions of growth factors and differentiation into Schwann cell-like cells and vascular smooth muscle cells.
Keywords
Introduction
Diabetic polyneuropathy (DPN) is the most common and intractable complication of diabetes (5, 7, 52). Loss of sensation in the lower limbs at the advanced stage of DPN is a high risk factor for limb amputation, which occurs in 1–2% of diabetic patients (44, 60). Furthermore, various agents developed for DPN do not have therapeutic potential for advanced DPN that is accompanied by severe morphological changes (41, 44, 56, 64). On the other hand, effective therapeutic strategies are reported (8, 10, 28, 51). The major pathophysiological characteristics of DPN are degeneration of nerve fibers and reduced nerve blood flow. Previous studies (15, 24, 26, 45) have reported beneficial effects of vasodilatory agents on DPN. Our previous studies with diabetic rats or mice have demonstrated that local administration of basic fibroblast growth factor (bFGF) and intramuscular injection of cord blood-derived endothelial progenitor cells (EPCs) or bone marrow-derived mesenchymal stem cells (MSCs) ameliorated hypoalgesia, impaired nerve conduction velocity (NCV) and nerve blood flow (NBF) (38, 39, 46). EPCs and MSCs express vascular endothelial growth factor (VEGF) and bFGF (17, 22, 23). Gene transfer of VEGF (45) significantly increased the NCV and NBF as well as the vascular densities in muscles and peripheral nerves, suggesting that the induction of local angiogenesis by VEGF ameliorates experimental DPN. Furthermore, there are many reports about direct neurotrophic effects of bFGF and VEGF (34, 43, 63). Therefore, cells that can express both angiogenic and neurotrophic factors would be suitable for cell transplantation therapy in DPN, and it would be more appropriate if the transplanted cells could differentiate into vascular cells or neural cells.
Induced pluripotent stem (iPS) cells are capable of differentiating into various kinds of cells as needed, and it has been proven in various tissues and cell types that the courses of embryonic stem (ES)/iPS cell differentiation in vitro replicate those of development in utero (2, 19, 40, 49, 50, 65). A procedure of stromal cell-derived inducing activity (SDIA) using PA6 cells followed by exposure to bone morphogenetic protein 4 (BMP4) has been shown to be effective in induction to neural crest (NC) derivatives (33). The NC is a group of cells located in the neural folds at the boundary between the neural and epidermal ectoderm. NC cells differentiate into a vast range of cells, including peripheral neurons, Schwann cells, vascular smooth muscle cells, large vessels, bone and cartilage cells of the maxillofacial region, and odontoblasts (6). Moreover, some NC cells remain at adult stage as stem cells: dorsal root ganglia stem cells, dermal neural crest stem cells, and MSCs (37, 47, 54). Although transplantation of several specific neuron types and glial cell types derived from ES or iPS cells has been experimentally reported to be a promising strategy for the treatment of central nervous impairments (25, 58, 62), there is no report about the transplantation therapy of NC-like (NCL) precursors or NC derivatives from ES or iPS cells in peripheral nervous dysfunction. It has also been shown that transplanted mouse embryonic NC could differentiate to sensory neurons and glial cells at dorsal root ganglion cavity (1). We hypothesized that NCL cells derived from iPS cells would be suitable for cell transplantation therapy from the view point of angiogenesis and neuroregeneration in DPN. This is the first report demonstrating the therapeutic effects of NCL cell transplantation on DPN.
Materials and Methods
Induction of NCL Cells From iPS Cells
We used iPS cells established from bone marrow myeloid cells growing in medium supplemented with granulocyte-macrophage colony-stimulating factor (G-CSF) taken from 21-month-old enhanced green fluorescent protein (EGFP)-C57BL/6 mice (C14-Y01-FM131Osb) carrying pCAG-EGFP (CAG promoter-EGFP) as we have described previously (9). iPS cells were maintained in Dulbecco's modified Eagle's medium (DMEM) (Invitrogen, Carlsbad, CA, USA) containing 10% knockout serum replacement (KSR) (Invitrogen), 1% fetal bovine serum (FBS), nonessential amino acids (Invitrogen), 5.5 mM 2-mercaptoethanol (Invitrogen), 50 U/ml penicillin, and 50 mg/ ml streptomycin (Invitrogen) on mytomycin-C-treated SNL76/7 feeder cells (DS Pharma, Osaka, Japan), which were clonally derived from the spontaneously transformed Sandoz inbred mouse thioguanine- and ouabain-resistant (STO) cell line transfected with a G418 cassette and leukemia inhibitory factor expression construct (32).
For differentiation, iPS cells were transferred into dishes confluent with PA6 cells (RIKEN Cell Bank, Kobe, Japan) and maintained in differentiation medium: Glasgow minimum essential medium (Invitrogen) containing 0.1 mM nonessential amino acids, 1 mM pyruvate (Sigma-Aldrich, St. Louis, MO, USA), 0.1 mM 2-mercaptoethanol, and 10% KSR. The day on which iPS cells were cocultured with PA6 cells was defined as day 0. Medium change was performed everyday. During days 5–9, 0.5 nM BMP4 (Sigma-Aldrich) was added to coculturing dishes (33). For immunostaining, differentiated cells were stained with rabbit anti-p75 low-affinity neurotrophin receptor (p75NTR) antibody (Advanced-Targeting-System, San Diego, CA, USA) and then with Alexa Fluor 594-coupled goat anti-rabbit IgG antibody (1:200; Invitrogen) at day 12. For fluorescence-activated cell sorting (FACS) analysis, differentiated cells were stained with rabbit anti-p75NTR antibody followed by allophycocyanin (APC)-coupled goat anti-rabbit IgG antibody (Beckman Coulter, Fullerton, CA, USA). The immunostained cells were analyzed by FACSCanto (Becton Dickinson, Franklin Lakes, NJ, USA).
Cell Separation and Evaluation of Differential Ability of NCL Cells
Differentiated cells were detached enzymatically (trypsin-EDTA; Invitrogen) and reacted with rabbit p75NTR antibody. These cells were separated with a magnetic-activated cell sorting (MACS) (Miltenyi Biotec, BergischGladbach, Germany) system according to the manufacturer's instructions. The sorted cells were transferred again into dishes confluent with PA6 cells. They were maintained with the differentiation medium. Fourteen days after coculturing with PA6 cells, they were stained with the following antibodies: mouse anti-α-smooth muscle actin (α-SMA) antibody (1:200; Sigma-Aldrich), mouse anti-peripherin antibody (1:200; Millipore, Billerica, MA, USA), mouse anti-β-III-tubulin antibody (1:200; Sigma-Aldrich), and rabbit anti-S100-β antibody (1:10; AbD Serotec, Oxford, UK). After rinsing with phosphate-buffered saline (PBS; Nissui Pharma, Tokyo, Japan), the cells were incubated for 60 min at room temperature with Alexa Fluor 594-coupled goat anti-rabbit or anti-mouse IgG antibody (1:200; Invitrogen).
Real-Time Reverse Transcription-PCR (RT-PCR) Analysis
Total RNA was isolated from cultured iPS cells, PA6 cells, sorted NCL cells, and immortalized Schwann cells (IMS32) using Isogen (Nippon Gene, Toyama, Japan). IMS32, established by long-term culture of adult mouse dorsal root ganglia (DRG) and peripheral nerves (61), was a kind gift from Dr. Kazuhiro Watabe (Jikei University School of Medicine, Tokyo, Japan). RNA was reverse-transcribed into cDNA by ReverTraAceqPCR RT kit (Toyobo, Osaka, Japan) according to the manufacturer's instructions. Primers were designed by Primer3 software (http://frodo.wi.mit.edu/) and tested for specificity with NCBI-BLAST (http://www.ncbi.nlm.nih.gov/tools/primer-blast/). Primer sequences are shown in Table 1. Real-time quantitative PCR was performed and monitored using the Mx3000P QPCR System (Stratagene Agilent Technologies, Santa Clara, CA, USA) with SYBR Green I as a double-stranded DNA-specific dye according to the manufacturer's instructions (Applied Biosystems, Foster City, CA, USA). The PCR products were analyzed by aga-rose gel (Wako, Osaka, Japan)/ethidium bromide (Sigma-Aldrich) to confirm these predicted lengths. Relative quantity was calculated by the ΔΔCt method.
Primers Used in Real-Time RT-PCR Analysis

AP-2, transcription factor activating enhancer binding protein 2; dHand, deciduum, heart, autonomic nervous system and neural crest derivatives-expressed protein 2; PAX3, paired box 3; SOX10, sex determining region Y box; p75, low-affinity p75 neurotrophic receptor; NGF, nerve growth factor; NT-3, neurotrophin 3; VEGF, vascular endothelial growth factor; bFGF, basic fibroblast growth factor.
Cell Culture and Preparation of Conditioned Media
IMS32 cells were cultured in DMEM with 5% FBS as described previously (13). When the cells reached ~70% confluency, they were maintained in DMEM with 2% FBS containing 5.5 mmol/L d-glucose (NG) or 30 mmol/L d-glucose (HG). After a 3-day culture, the cells were maintained in serum-free DMEM containing NG or HG. After 24 h, culture media were collected, concentrated 10 times using 10-kDa centrifugal filters (Amicom Ultra-15, Nihon Millipore, Tokyo, Japan), and frozen at −80°C until use. We defined these media as NG-IMS media or HG-IMS media.
PA6 cells were cultured in α-minimum essential medium (Invitrogen) supplemented with 10% FBS, penicillin, and streptomycin. At confluence, the media were replaced with serum free media and collected 24 h later. The collected conditioned media were concentrated 10 times using 10-kDa centrifugal filters and frozen at −80°C until use. Sorted NCL cells were also cultured in the serum-free differentiation media for 24 h, and the media were collected and concentrated as described above. SNL76/7 cells were cultured in DMEM supplemented with 10% FBS, penicillin, and streptomycin.
Primary Culture of Dorsal Root Ganglion (DRG) Neurons and Evaluation of Neurite Outgrowth
DRG neuron cultures were prepared from 5-week-old male C57BL/6 mice (Chubu Kagaku Shizai, Nagoya, Japan) as previously described (13). In brief, DRGs were collected, dissociated by collagenase (Wako Pure Chemical, Osaka, Japan), and diluted in medium consisting of F-12 media, 10 mM glucose, and 30 nM selenium. Isolated DRG neurons were seeded on glass coverslips (Matsunami, Osaka, Japan) coated with poly-l-lysine (Sigma-Aldrich). DRG neurons were cultured for 24 h in 1,000 μl of F-12 media with PA6 cell-conditioned media (PA6-CM) or NCL cell-conditioned media (NCL-CM). To evaluate the effects of NCL-CM on impaired neurite outgrowth under the diabetic condition, DRG neurons were cultured in HG-IMS media and NG-IMS media that were diluted one-tenth with F-12 media. DRG neurons were immunostained with rabbit polyclonal anti-neurofilament heavy-chain antibody (1:5,000; Nihon Millipore). After rinsing with PBS, the cells were incubated for 60 min at room temperature with Alexa Fluor 488-coupled goat anti-rabbit IgG antibody (1:200; Invitrogen). Coverslips were counterstained with 4′,6-diamidino-2-phenylindole (DAPI) (Merck, Tokyo, Japan). Images were captured by a CCD camera (DP70, Olympus Optical, Tokyo, Japan) using a fluorescence microscope (BX51, Olympus Optical). Neurite outgrowth was observed in 10–20 neurons per coverslip and evaluated by a computed image analysis system as previously described (Angiogenesis Image Analyzer Ver. 2, KURABO Industries, Osaka, Japan) (57).
Animals and Induction of Diabetes
Five-week-old male C57BL6/J mice (Chubu Kagakushizai, Nagoya, Japan) with an initial body weight of 24–26 g were allowed to adapt to the experimental animal facility for 7 days. Diabetes was induced by intraperitoneal injection of streptozotocin (STZ) (150 mg/kg; Sigma-Aldrich). Control mice received an equal volume of citric acid buffer. Mice with plasma glucose concentrations of >16 mM were selected as the STZ-induced diabetic group. The Nagoya University Institutional Animal Care and Use Committee approved all the protocols of this experiment.
Transplantation of NCL Cells
Normal and diabetic mice were classified into four groups: normal mice treated with saline (N-S), normal mice treated with NCL cells (N-NCL), diabetic mice treated with saline (DM-S), and diabetic mice treated with NCL cells (DM-NCL) (n = 10 in each group). Sixteen weeks after the induction of diabetes, 1 × 105 cells/limb of NCL cells in 0.2 ml of saline (Otsuka Pharma, Tokyo, Japan) were injected at 10 separate sites into the right thigh and soleus muscles of N-NCL and DM-NCL. Left hind limb muscles were treated with saline alone. Four weeks later, the following parameters were bilaterally measured.
Measurement of Current Perception Threshold (CPT)
To evaluate the nociceptive threshold, CPT was measured in 16- and 20-week STZ-induced diabetic and age-matched normal mice using a CPT/LAB neurometer (Neurotron, Denver, CO, USA) according to the method by Himeno et al. (13) with minor modifications. The electrodes (SRE-0405-8; Neurotron) for stimulation were attached to plantar surfaces. Each mouse was kept in a Ballman cage (Natsume Seisakusho, Tokyo, Japan) suitable for light restraint to keep awake. Three transcutaneous sine wave stimuli with different frequencies (2,000, 250, and 5 Hz) were applied to the plantar surfaces. The intensity of each stimulation was gradually increased automatically (increments of 0.01 mA for 5 and 250 Hz, increments of 0.02 mA for 2,000 Hz). The minimum intensity at which a mouse withdrew its paw was defined as the CPT. Six consecutive measurements were conducted at each frequency.
Nerve Conduction Velocity (NCV)
Mice anesthetized with pentobarbital (Kyoritsu Pharma, Tokyo, Japan) were placed on a heated pad in a room maintained at 25°C to ensure a constant rectal temperature of 37°C. Motor nerve conduction velocity (MNCV) was determined between the sciatic notch and ankle with a Neuropak NEM-3102 instrument (Nihon-Koden, Osaka, Japan) as previously described (38, 39, 46). The sensory nerve conduction velocity (SNCV) was measured between the knee and ankle with retrograde stimulation.
Tissue Collection
The mice were anesthetized with sodium pentobarbital (5 mg/100 g) and perfused with 50 ml of 4% paraformaldehyde fixative or Zamboni's fixative (Both Wako). After perfusion, soleus muscles and sole skin were excised and fixed in 4% paraformaldehyde or Zamboni's fixative at 4°C overnight. Specimens were immersed in PBS containing 20% sucrose (Wako) embedded in O.C.T. compound (Sakura Finetechnical, Tokyo, Japan) and cut into 5-μm sections with a sliding cryostat (CM1800, Leica Microsystems AG, Wetzler, Germany).
Measurement of Intraepidermal Nerve Fiber Densities (IENFDs)
After 3×5-min microwave irradiation with citrate buffer (pH 6.0; Sigma-Aldrich) for antigen retrieval, cryostat sections of sole skin were blocked with 5% skim milk (Meiji Milk, Tokyo, Japan), and the rabbit polyclonal anti-protein-gene-product 9.5 (PGP 9.5) antibody (1:500; Millipore) was applied to the sections at 4°C overnight. After washing, Alexa Fluor 594-coupled goat anti-rabbit IgG antibody (1:200; Invitrogen) was loaded for 1 h at room temperature. Sections were counterstained with DAPI (Merck). Images were captured by a CCD camera (DP70, Olympus Optical) using a fluorescence microscope (BX51, Olympus Optical). Nerve fibers stained with anti-PGP 9.5 antibody were counted as previously reported (13). In brief, each individual nerve fiber with branching inside the epidermis was counted as one, and a nerve fiber with branching in the dermis was counted separately. Six fields from each section were randomly selected for the IENFDs. IENFDs were derived and expressed as epidermal nerve fiber numbers per length of the epidermal basement membrane (fibers/mm).
Thermal Plantar Test
Paw withdrawal response to thermal stimuli of radiant heat was measured using a testing apparatus (Plantar test, 7370; Ugo Basile, Comerio, Italy). Radiant heat was beamed onto the plantar surface of the hind paw. The paw withdrawal latencies were measured five times per session, separated by a minimum interval of 10 min. Paw withdrawals due to locomotion or weight shifting were not counted. Data are expressed as paw withdrawal latency in seconds.
Sciatic Nerve Blood Flow (SNBF) and Plantar Skin Blood Flow (PSBF)
SNBF and PSBF were measured by laser Doppler flow-metry (FLO-N1; Omega wave Inc., Tokyo, Japan). To measure SNBF, the thigh skin of an anesthetized mouse was cut along the femur, and then incision through the fascia was carefully performed to expose the sciatic nerve. Five minutes after this procedure, the blood flow was measured by a laser Doppler probe, placed 1 mm above the sciatic nerve. To determine PSBF, three different spots of plantar skin were measured by the probe, placed 1 mm above the skin. During this measurement, the mouse was placed on a heated pad in a room maintained at 25°C to ensure a constant rectal temperature of 37°C.
Capillary Number-to-Muscle Fiber Ratio (CMR)
CMR were calculated as previously reported (39, 46) with minor modification. In brief, the sections of soleus muscles fixed with paraformaldehyde were used for immunostaining. The vascular capillaries were stained by Alexa Fluor 594-conjugated isolectin Griffonia simplicifolia-IB4 (GS-IB4; Invitrogen) and were counted under a fluorescence microscope (BX51, Olympus Optical), and images were obtained by a CCD camera (DP70, Olympus Optical). The muscle fibers were concomitantly counted to determine the CMR. Five fields from each section were randomly selected for capillary counts.
Tracing of NCL Cells In Vivo
Transplanted cells were evaluated by immunohistochemical analysis 2 and 4 weeks after transplantation. For the detailed characterization, transplanted cells settled in the soleus muscles were stained with the following antibodies at 4°C overnight: mouse anti-α-SMA antibody (1:200; Sigma-Aldrich), rabbit anti-S100-β antibody (1:10; AbD Serotec), mouse anti-VEGF antibody (1:50; Santa-Cruz Biotechnology, Santa-Cruz, CA, USA), rabbit anti-bFGF antibody (1:50; Santa-Cruz Biotechnology), mouse antinerve growth factor (NGF) antibody (1:100; Millipore), and rabbit anti-neurotrophin-3 (NT-3) antibody (1:50; Santa-Cruz Biotechnology). After washing, the following secondary antibodies were loaded for 1 h at room temperature in a dark box: Alexa Fluor 594-coupled goat anti-rabbit or anti-mouse IgG antibody (1:200; Invitrogen).
Measurements of NGF and VEGF Contents in Sciatic Nerves and DRGs
The NGF and VEGF levels of sciatic nerves and DRGs were measured in duplicate for each sample using quantitative sandwich enzyme immunoassay NGF ELISA kits (Abnova, Taipei, Taiwan) and VEGF ELISA kits (R&D Systems, Minneapolis, MN, USA) according to the manufacturer's instructions. Results were expressed as picograms per milligram of protein in sciatic nerves and DRGs. Absorbance from colorimetric reactions with horseradish peroxidase and 3,3′,5,5′-tetramethyl benzidine was determined by a Powerscan HT microplate reader (Dainippon Pharmaceutical, Osaka, Japan) and converted to a NGF or VEGF protein level by normalizing to a standard curve.
Statistical Analysis
All the group values were expressed as means ± SD. Statistical analyses were made by one-way ANOVA, with the Bonferroni correction for multiple comparisons. All analyses were performed by personnel unaware of the animal identities.
Results
Differentiation of iPS Cells to NCL Cells
Undifferentiated GFP-positive iPS cells (Fig. 1A) were cultured on dishes with PA6 cells as described previously (20). Under these conditions, more than half of colonies extended neurite-like processes and were immunopositive for p75NTR, suggesting a neural crest cell lineage (Fig. 1B, C). We assessed the appearance of p75NTR-positive (p75+) cells by fluorescence-activated cell sorting (FACS). p75+ cells appeared after 4 days of culture and peaked on day 12. About 50–80% of differentiated cells expressed p75NTR from low to high levels at day 12 (Fig. 1D). Based on these findings, we used MACS to isolate p75+ cells from the bulk cultures at day 12. To analyze their differentiation ability in vitro, p75+ cells were transferred to the dishes on which PA6 cells were cultured. After 14 days of coculturing, we observed three major differentiated cell types: β-III-tubulin- or peripherin-positive neurons (10.0 ± 2.4% of GFP-positive cells), S100-β-positive glial cells (7.7 ± 1.6%), and α-SMA-positive smooth muscle cells (11.4 ± 1.3%) (Fig. 1E–I). In addition, we confirmed the expression of NC markers—sex determining region Y box 10 (SOX10), paired box 3 (PAX3), Snail, transcription factor activating enhancer binding protein 2 (AP-2), and deciduum, heart, autonomic nervous system and neural crest derivative-expressed protein 2 (dHand)—in p75+ cells by RT-PCR (Fig. 2A). Furthermore, it was revealed that the p75+ NCL cells expressed growth factors: NGF, NT-3, VEGF, and bFGF (Fig. 2B). Each growth factor mRNA expression in NCL cells was significantly increased compared with that in undifferentiated iPS cells. The NGF mRNA expression level of NCL cells was comparable with that of IMS32 Schwann cells, which have been proven to be rich in several neurotrophic factors (16, 46), and the NT-3 mRNA expression level of NCL cells was higher than that of the IMS32 cells. The expression levels of bFGF and VEGF-A of NCL cells were comparable to those of PA6 cells, a mouse MSC line (Fig. 2B).

Induction of NCL cells from aged mouse iPS cells. (A) Green fluorescent protein (GFP)-positive induced pluripotent stem (iPS) cell. (B) Phase-contrast photographs of mouse iPS cell colonies cocultured with PA6 cells. Neurite-like processes were extended from the colonies over time. (C) Expression of low-affinity neurotrophin receptor (p75NTR) (red), a cell surface marker of neural crest cells, at day 12. (D) Fluorescence-activated cell sorting (FACS) analysis of differentiated cells was conducted after 4, 8, and 12 days of coculture. Cells were labeled with allophycocyanin (APC)-conjugated antibodies for p75NTR. Scale bars: 100 μm. (E–H) Ability of the p75NTR-positive cells to differentiate into neural crest derivatives in vitro. After the further induction of purified p75NTR-positive cells on PA6 cells, a number of differentiated cells expressed β-III-tubulin and peripherin (red) and extended neurite-like processes (E, F). Many of the induced cells were immunostained with anti-S100-β antibody (red) (G) and anti-α-smooth muscle actin (α-SMA) antibody (red) (H). Nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI; blue). Scale bars: 100 μm. (I) Quantification of neuronal, glial, and smooth muscle cell differentiation in vitro expansion. NCL, neural crest-like.

Expression of neural crest cell marker, neurotrophic factor, and angiogenic factor in p75NTR-positive cells. (A) RT-PCR analysis of p75NTR-positive cells. (B) Neurotrophic factor and angiogenic factor expressions in the undifferentiated iPS cells (iPS), PA6 cells (PA6), p75NTR-positive cells (p75+), and immortalized mouse Schwann cells (IMS32). The mRNA levels of these factors were normalized to that of β-actin, and the mRNA levels in these cells were expressed relative to those in undifferentiated mouse iPS cells. Results are means ± SD. #p < 0.05 versus undifferentiated mouse iPS cells, *p < 0.05 versus IMS32 cells, †p < 0.05 versus PA6 cells. PAX3, paired box 3; Sox10, sex determining region Y box; AP-2, transcription factor activating enhancer binding protein 2; dHand, deciduum, heart, autonomic nervous system and neural crest derivatives-expressed protein 2; Ngf, nerve growth factor; Nt3, neurotrophin 3; Vegfa, vascular endothelial growth factor a; Fgf2, basic fibroblast growth factor 2.
NCL Cell-Conditioned Media Promoted Neurite Outgrowth of DRG Neurons
DRG neurons (stained with neurofilament heavy chain immunofluorescence) extended neuritis, and the extensions were promoted in PA6 mesenchymal stem cell-conditioned media (Fig. 3A). Furthermore, neurite outgrowths were remarkably promoted in the presence of NCL-CM compared with those in the presence of PA6-CM (Fig. 3A). Total length (TL) and joint number (JN) of neurites were significantly increased by NCL-CM (control: JN, 39.8 ± 15.2/ cell; TL, 778 ± 332 μm/cell; PA6-CM cells: JN, 60.4 ± 24.5; TL, 914 ± 464; NCL-CM: JN, 119.9 ± 51.5; TL, 2306 ± 856; JN and TL: control vs. NCL-CM, p < 0.0001; PA6-CM vs. NCL-CM, p < 0.0001) (Fig. 3B). DRG neurons cultured with HG-IMS media, which mimicked the diabetic state, had shorter neurites and smaller joint numbers compared with those cultured with NG-IMS media, which mimicked the nondiabetic normal state. The impaired neurite outgrowth of DRG neurons cultured with HG-IMS media was improved in the presence of NCL-CM (NG-IMS: JN, 124.7 ± 27.4/cell; TL, 2295.3 ± 383.6 μm/cell; HG-IMS: JN, 90.0 ± 8.13; TL, 1560.4 ± 241.5; NG-IMS + NCL-CM: JN, 147.0 ± 17.27; TL, 2643.0 ± 187.71; HG-IMS + NCL-CM: JN, 114.9 ± 9.63; TL, 2070.0 ± 271.7; JN: NG-IMS vs. HG-IMS, p < 0.0001; HG-IMS vs. HG-IMS + NCL-CM, p = 0.0008; TL: NG-IMS vs. HG-IMS, p = 0.0004; HG-IMS vs. HG-IMS + NCL-CM, p = 0.011) (Fig. 3B).

Neurite outgrowth of DRG neurons. (A) Representative fluorescence micrographs of dorsal root ganglion (DRG) neurons stained with neurofilament heavy chain and cultured in the absence or presence of PA6 cell- or NCL cell-conditioned culture media. (B) NCL cell-conditioned media significantly promoted total neurite length and increased joint number of neurite. Results are means ± SD. #p < 0.05 versus control group, *p < 0.05 versus PA6 cell-conditioned media, **p < 0.05 versus NG-IMS, †p < 0.05 versus HG-IMS. Scale bars: 50 μm. CNT, F-12 control media; PA6, PA6-conditioned media; NC, neural crest-like cell-conditioned media; HG-IMS, IMS media obtained from IMS32 cultured with 30 mM d-glucose; NG-IMS, IMS media obtained from IMS cultured with 5.5 mM d-glucose.
Body Weights and Blood Glucose Levels
At 16 weeks, diabetic mice showed severe hyperglycemia and significantly reduced body weight gain. Random blood glucose levels measured during the experimental period were not significantly changed in any group. NCL cell transplantation did not alter the body weights or blood glucose levels in the normal or diabetic groups (Table 2).
Body Weight and Blood Glucose in Normal and Diabetic Mice
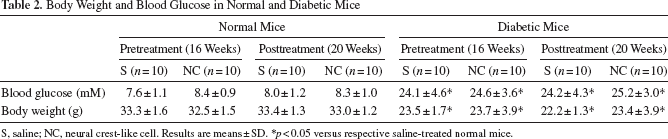
S, saline; NC, neural crest-like cell. Results are means ± SD.
p < 0.05 versus respective saline-treated normal mice.
Impaired Nerve Functions in Diabetic Mice Were Significantly Ameliorated by the Transplantation of NCL Cells
We evaluated sensory functions by CPT. In 16-week diabetic mice, CPTs at 5, 250, and 2,000 Hz were significantly increased compared with those in normal mice [5 Hz: normal mice (N), 47.0 ± 8.2 μA; diabetic mice (DM), 89.5 ± 12.5, p < 0.0001; 250 Hz: N, 47.0 ± 8.8; DM, 101.0 ± 13.9, p < 0.0001; 2,000 Hz: N, 107.0 ± 8.5; DM, 167.2 ± 52.0, p < 0.0001], representing hypoalgesia in diabetic mice. Four weeks after transplantation of NCL cells, these deficits in sensation were significantly improved in diabetic mice (DM-NCL) compared to those in diabetic mice treated with saline (DM-S) (5 Hz: DM-S, 86.0 ± 26.0; DM-NCL, 59.3 ± 11.8, p < 0.0001; 250 Hz: DM-S, 95.0 ± 19.0; DM-NCL, 59.0 ± 12.6, p < 0.0001; 2,000 Hz: DM-S, 189.7 ± 31.2; DM-NCL, 130.8 ± 30.8, p < 0.0001) (Fig. 4A–C).

Sensory and motor nerve functions. Measurements of current perception thresholds (CPTs) at 5 (A), 250 (B), and 2,000 Hz (C) by a neurometer were performed before and 4 weeks after transplantation. CPTs for all pulses were significantly increased in the diabetic group, and these deficits were significantly improved by NCL cells. Motor nerve conduction velocity (MNCV; D) and sensory nerve conduction velocity (SNCV; E) were measured before and after transplantation of NCL cells. The MNCV and SNCV of 16-week diabetic mice were significantly delayed compared with those of normal mice. The delay in MNCV and SNCV was significantly restored by NCL cell transplantation. (F, G) Intraepidermal nerve fiber densities (IENFDs). IENFDs were decreased in diabetic mice, and this decrement was ameliorated by NCL cell transplantation. Arrows indicate intraepidermal nerve fibers by immunohistological staining for protein gene product 9.5 (PGP 9.5). N, normal mice; DM, diabetic mice; S, saline; NC, NCL cells; contra, saline-injected limb of NCL cell-transplanted mice; Pre, pretransplantation; Post, posttransplantation. Results are means ± SD. #p < 0.05 versus N, *p < 0.05 versus N-S, †p < 0.05 versus DM-S. Scale bars: 50 μm.
The MNCV and SNCV of 16-week diabetic mice were significantly delayed compared with those of normal mice (MNCV: N, 46.6 ± 2.3 m/s; DM, 33.7 ± 2.2, p < 0.0001; SNCV: N, 45.0 ± 2.5; DM, 32.5 ± 3.0, p < 0.0001). The delay in MNCV and SNCV was significantly restored by NCL cell transplantation (MNCV: DM-S, 35.3 ± 3.7; DM-NCL, 39.4 ± 3.4, p = 0.0066; SNCV: DM-S, 31.7 ± 3.3; DM-NCL, 37.6 ± 1.5, p < 0.0001) (Fig. 4D, E).
Nerve Fibers in Epidermis Were Regenerated by NCL Cell Transplantation
Intraepidermal nerve fibers were evident in both epidermis and dermis of foot skin by fluorescence imaging of PGP 9.5. Although IENFDs were decreased in diabetic mice (N: 55.0 ± 7.7 fibers/mm, DM: 25.4 ± 5.7, p < 0.0001), this decrement was partially but significantly ameliorated by NCL cell transplantation (DM-NCL: 40.4 ± 4.5, DM-S: 26.4 ± 4.9, p = 0.0008) (Fig. 4F, G).
NCL Cell Transplantation Improved the Sensitivity to the Thermal Stimuli
Response to thermal stimulation was measured by exposing the plantar region of paw to the radiant heat. The delay of withdrawal response time was observed in 16-week diabetic mice compared with those of normal mice (N: 9.2 ± 0.7 s, DM: 18.3 ± 0.9, p < 0.0001). The delay was partially restored by NCL cell transplantation (DM-S: 18.3 ± 0.9, DM-NCL: 13.1 ± 1.4, p < 0.0001) (Fig. 5).

Thermal plantar test. The delay of withdrawal response time was observed in 16-week diabetic mice compared with that in normal mice, and this delay was ameliorated by NCL cell transplantation. N, normal mice; DM, diabetic mice; S, saline; NC, NCL cells; contra, saline-injected limb of NCL cell-transplanted mice. Results are means ± SD. #p < 0.05 versus N, *p < 0.05 versus N-S, †p < 0.05 versus DM-S.
NCL Cell Transplantation Improved Decreased SNBF and PSBF
The SNBFs in 16-week diabetic mice were significantly decreased compared to those in normal mice (N: 23.7 ± 1.3 ml/min/100 g, DM: 16.1 ± 1.9, p < 0.0001), and the decrease was ameliorated by NCL cell transplantation (DM-NCL: 21.3 ± 1.8, DM-S: 16.1 ± 1.1, p < 0.0001) (Fig. 6A). The PSBFs in 16-week diabetic mice were significantly decreased compared to those in normal mice (N: 24.4 ± 1.7 ml/min/100 g, DM: 17.2 ± 1.4, p < 0.0001), and the decrease was ameliorated by NCL cell transplantation (DM-NCL: 20.1 ± 2.6, DM-S: 17.3 ± 1.7, p = 0.0027) (Fig. 6B).
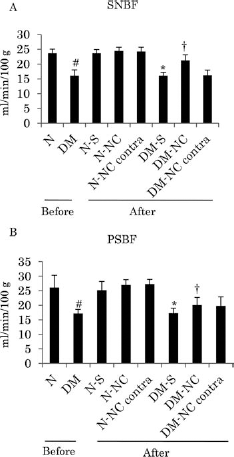
Sciatic nerve blood flow and plantar skin blood flow. (A) The Sciatic nerve blood flows (SNBFs) in 16-week diabetic mice were significantly decreased compared with those in normal mice, and this decrease was ameliorated by NCL cell transplantation. (B) The plantar skin blood flows (PSBFs) in 16-week diabetic mice were significantly decreased compared with those in normal mice, and this decrease was ameliorated by NCL cell transplantation. N, normal mice; DM, diabetic mice; S, saline; NC, NCL cells; contra, saline-injected limb of NCL cell-transplanted mice. Results are means ± SD. #p < 0.05 versus N, *p < 0.05 versus N-S, †p < 0.05 versus DM-S.
CMR Was Recovered by NCL Cells
Quantitative analyses revealed that the CMRs (shown by isolectin GS-IB4 staining) in the hind limb skeletal muscles of diabetic mice were significantly reduced compared to those in normal mice (DM: 0.48 ± 0.11, N: 1.04 ± 0.16, p < 0.0001). Transplantation of NCL cells significantly augmented the ratio compared to saline injection (DM-NCL: 0.84 ± 0.16, DM-S: 0.48 ± 0.11, p = 0.0004) (Fig. 7A, B).

Capillary number-to-muscle fiber ratio. The capillary number-to-muscle fiber ratio (CMR) in diabetic mice was significantly reduced compared with that of normal mice. Transplantation of NCL cells significantly augmented the ratio compared with the saline-injected diabetic mice. Arrowheads indicate vascular cells detected by immunohistological staining for isolectin GS IB-4 (A, B). N, normal mice; DM, diabetic mice; S, saline; NC, NCL cells; contra, saline-injected limb of NCL cell-transplanted mice; Pre, pretransplantation; Post, posttransplantation. Results are means ± SD. #p < 0.05 versus N, *p < 0.05 versus N-S, †p < 0.05 versus DM-S. Scale bars: 50 μm.
Differentiation Into Schwann Cell-Like Cells and Vascular Smooth Muscle Cells and Paracrine Actions of Transplanted Cells
Two weeks after transplantation, most of the surviving transplanted cells expressing green fluorescent protein (GFP) were found between muscle fibers around primary injected sites without forming any specific tissue structure. These engrafted cells could be immunohistochemically detected by antibodies of growth factors: NGF, NT-3, VEGF, and bFGF (Fig. 8A–D). Although it was difficult to trace these cells 4 weeks after transplantation, there were still some cells coexpressing GFP and these growth factors (Fig. 8E–H).

Differentiation of transplanted NCL cells into Schwann cell-like cells or vascular smooth muscle cells and paracrine actions of NCL cells. Two weeks after transplantation, transplanted cells expressed NGF (A), NT-3 (B), VEGF (C), and bFGF (D). Four weeks after transplantation, transplanted NCL cells expressed NGF (E), NT-3 (F), VEGF (G), and bFGF (H) or differentiated into S100-β-positive Schwann cell-like cells (I) on muscle fibers and α-smooth muscle actin (α-SMA)-positive vascular smooth muscle cells in vessel walls (J). The α-SMA-positive grafted cells also produced VEGF (G). Nuclei were stained with DAPI (blue). Scale bars: 50 μm.
Under careful observation, several GFP-positive cells could be detected at points distant from transplanted sites and formed an axon-like shape on muscle fibers, and these cells expressed S100-β, a marker of astrocytes and Schwann cells (Fig. 8I). Furthermore, some of the transplanted NCL cells were also incorporated into blood vessels among muscle fibers and expressed α-SMA, a marker of vascular smooth muscle cells (Fig. 8J). It was also confirmed that these grafted cells forming vessels produced VEGF (Fig. 8G). We could not find a teratoma or other mass of transplanted cells throughout our observations.
NGF and VEGF Contents in Sciatic Nerves and DRGs
NGF and VEGF contents in the sciatic nerve of the saline-injected side of diabetic mice were significantly reduced compared with those of normal mice (2 weeks after the transplantation: NGF, N-S: 874.1 ± 39.5 pg/mg protein, DM-S: 401.2 ± 16.6, p < 0.0001; VEGF, N-S: 87.0 ± 4.6 pg/ mg protein, DM-S: 48.6 ± 6.0, p < 0.0001; 4 weeks after the transplantation: NGF, N-S: 879.6 ± 38.6, DM-S: 382.6 ± 17.0, p < 0.0001; VEGF, N-S: 90.6 ± 2.9, DM-S: 47.6 ± 6.2, p < 0.0001). The transplantation of NCL cells improved the reduction of NGF and VEGF contents in diabetic mice (2 weeks: NGF, DM-NCL: 875.8 ± 41.6, p < 0.0001 vs. DM-S; VEGF, DM-NCL: 91.4 ± 2.6, p < 0.0001 vs. DM-S; 4 weeks; NGF, DM-NCL: 619.2 ± 25.6, p < 0.0001 vs. DM-S; VEGF, DM-NCL: 71.8 ± 4.4, p < 0.0001 vs. DM-S) (Fig. 9A, B).

Evaluations of NGF and VEGF protein contents in sciatic nerve and DRG by enzyme-linked immunosorbent assay. (A, B) NGF and VEGF contents in sciatic nerve were significantly decreased in the saline-injected side of diabetic mice compared with those of normal mice, and this decrease was ameliorated by NCL cell transplantation. (C, D) NGF and VEGF contents in DRG were significantly decreased in the saline-injected side of diabetic mice compared with those of normal mice, and these decreases were not ameliorated by NCL cell transplantation. Protein contents were expressed as picograms per milligram protein. N, normal mice; DM, diabetic mice; S, saline; NC, NCL cells. 2 weeks, 2 weeks after transplantation; 4 weeks, 4 weeks after transplantation. Results are means ± SD. #p < 0.05 versus N-S, †p < 0.05 versus DM-S, n = 5 in each group.
NGF and VEGF contents in DRGs were significantly decreased in the saline-injected side of diabetic mice compared with those of normal mice (2 weeks: NGF, N-S: 909.6 ± 25.2, DM-S: 411.0 ± 28.4, p < 0.0001; VEGF, N-S: 86.4 ± 4.3, DM-S: 48.6 ± 5.1, p < 0.0001; 4weeks: NGF, N-S: 907.0 ± 26.8, DM-S: 404.2 ± 13.1, p < 0.0001; VEGF, N-S: 88.6 ± 5.6, DM-S: 49.0 ± 4.3, p < 0.0001). The transplantation of NCL cells did not show any effects on the NGF and VEGF contents in DRG (Fig. 9C, D).
Discussion
In this study, we investigated whether NCL cells derived from aged mouse iPS cells have therapeutic effects on DPN. Firstly, we succeeded in inducing NCL cells from iPS cells. These cells expressed NC cell markers (p75NTR, SOX10, PAX3, Snail, AP-2, and dHand) and growth factors (NGF, NT-3, bFGF, and VEGF). The mRNA expressions of these growth factors in NCL cells were significantly increased compared to those in undifferentiated iPS cells, and NCL cell-conditioned media enhanced the neurite outgrowth of DRG neurons. Purified NCL cells had abilities to differentiate into NC derivatives: peripheral neurons, glial cells, and vascular smooth muscle cells. Secondly, we demonstrated that transplantation of NCL cells improved several impairments of peripheral neural functions and vascular abnormalities of diabetic mice: the increased CPTs in the plantar pedis, delayed NCVs in hind limbs, the delay of response to thermal stimuli, decreased nerve fiber densities of epidermis in the plantar skin, decreased sciatic nerve blood flow and plantar skin blood flow, and decreased capillary numbers in soleus muscle. Thirdly, transplanted NCL cells survived supplying growth factors and differentiated into Schwann cell-like cells and vascular smooth muscle cells at each intrinsic placement.
Because diabetic patients are relatively old, the use of iPS cells for personalized clinical cell therapy will require the establishment of iPS cells from autologous tissue of elderly patients. Therefore, in our present experiments, we used iPS cells reprogrammed from aged mouse. Although chromosomal aberrations have been observed in iPS derived from aged human (4, 31), the iPS cells we used have been previously confirmed the integrity of chromosome and the pluripotent differentiation potential (9). The inducing activity present on the surface of stromal cells has been named stromal cell-derived inducing activity (SDIA) (20). It has been described that SDIA using PA6 stromal cells is appropriate for neuronal induction (20). Furthermore, late exposure to BMP4 in the course of mouse ES cell differentiation combined with SDIA of PA6 has been reported to be efficient to induce NC (33), and this efficiency has been also proven on human ES cells by Jiang et al. (18). Jiang and his coworkers showed that the efficiency of NC differentiation from human ES cells can be dramatically improved by combining SDIA coculture of PA6 with FACS-based isolation of the p75+ cell population. In our study, we could efficiently induce and isolate p75+ cells by using the SDIA protocol with late BMP4 exposure and MACS. The induced p75+ cells here expressed several NC markers and differentiated into peripheral neurons, glial cells, and smooth muscle cells, so the cells could be comparable to embryonic NC cells.
Trophic mechanisms have been reported to be impaired in the development of DPN (42), and several therapies employing growth factors have been proposed. Herpes simplex virus-mediated gene transfer of NGF protected against sensory deficits in diabetic mice (11) and NT-3 has been shown to restore abnormalities in mitochondrial depolarization in diabetic models in parallel with improvements in sensory conduction velocity (14). However, the application of NGF in a clinical trial did not succeed in the amelioration of DPN because of the emergence of pain (3). Efforts have been made to establish an efficient delivery or production system for these trophic factors (12, 21, 36, 55). In this study, we presumed that paracrine actions of NCL cells through supporting trophic factors would induce a beneficial effect on DPN, which was supported by an in vitro study using primary DRG neurons. Neurite outgrowth of DRG neurons was promoted in the presence of NCL cell-conditioned media compared to that in the absence of these media. These results might indicate that NCL cells exerted supportive or regenerative effects on peripheral sensory neurons by releasing trophic factors such as NGF, NT-3, VEGF, and bFGF.
bFGF or VEGF has the independent ability to induce angiogenesis, and the use of bFGF and VEGF in a coordinated manner has been shown to induce functional neovascularization (29). Furthermore, bFGF and VEGF have been reported to have neurosupportive effects (38, 45, 46). Our study revealed that the capillary density in the hind limb skeletal muscles was significantly lower in STZ-induced diabetic mice than in normal mice, and NCL cell transplantation ameliorated the decreased capillary density in diabetic mice. The level of therapeutic vasculogenesis induced by NCL cell transplantation was similar to that by EPC or MSC transplantation, as previously reported (39, 46). Neovascularization induced by the transplantation of NCL cells would be mediated both through the actions of angiogenic factors such as VEGF and bFGF secreted from NCL cells and through differentiation of transplanted NCL cells into vascular smooth muscle cells, which would explain the improvement of blood flows of sciatic nerve and plantar skin.
We evaluated the sensory nerve functions using a CPT/LAB neurometer. The neurometer is now widely and clinically used to evaluate the effects of analgesic drugs and peripheral nerve functions in various painful neuropathies including DPN (30, 59). In this study, after 16 weeks of diabetes, hypoalgesia at 2,000, 250, and 5 Hz was observed in the diabetic mice, and transplantation of NCL cells improved these abnormalities. Depletion of IENFDs, representing the peripheral terminals of nociceptive fibers, appears to be an early index of DPN (48, 53). NCL cell transplantation ameliorated the decreased IENFDs in diabetic mice, which explained the restoration of sensory functions. Additionally, we evaluated MNCV and SNCV that represent relatively large fiber functions. Both the delayed MNCV and SNCV in diabetic mice were improved by NCL cell transplantation, indicating that NCL cells had therapeutic effects on both impaired motor and sensory nerve functions. The therapeutic effects of NCL cell transplantation on these neuropathophysiological changes would be mediated, in part, through the supply of various neurotrophic factors, such as NGF and NT-3. Moreover, it can be speculated that VEGF and bFGF supplied by transplanted NCL cells contributed to the improvement of NCVs through the amelioration of the microcirculation or direct actions on axons and Schwann cells.
In the in vitro study, NCL cells had the capacity to differentiate into peripheral neurons, glial cells, and smooth muscle cells. In the in vivo study, transplanted NCL cells survived, migrated, and differentiated into Schwann celllike cells or vascular smooth muscle cells. Past studies have clarified some molecular signals related to the development and the differentiation of neural crest and its derivatives, for example, wingless-type mouse mammary tumor virus (MMTV) integration site family (Wnt), FGF2, BMPs, Pax3, Forkhead box D3 (Foxd3), NGF, brain-derived neurotrophic factor (BDNF), and NT-3 (27, 35). Furthermore, a reproducibility of the differentiation in in vitro experiments using neural crest stem cells or ES cells has been reported. According to these previous studies, we used BMP4 for induction of neural crest and confirmed expressions of growth factors that were important to maintain themselves. However, there is no report about a differentiation pattern of those induced neural crest (like) cells after transplantation into adult vertebrates. and our study might give a clue to resolve this problem. Therefore, further studies are required to address the in vivo behavior of induced neural crest cells.
NCL cell transplantation has several outstanding problems including potential risks of teratoma formation and unexpected lineage differentiation caused from contaminations of undifferentiated cells or other germ layers. In our study, however, NCL cells did not show such characteristics or tumorigenic proliferation. The reason for the absence of any tumor formation in this study could be the relative small number of transplanted cells or the long-term induction into neuroectoderm by SDIA using PA6. Although we could find a lot of GFP-positive cells in vivo, it was hard to find a GFP-positive Schwann cell-like cells and vascular smooth muscle cells. Intra-arterial injection of NCL cells might achieve a successful differentiation into vascular smooth muscle cells. NCL cells with shorter or longer induction might acquire superior ability to migrate to farther appropriate microenvironments. Further studies are needed to clarify these questions.
In conclusion, we first demonstrated the beneficial effects of transplantation of NCL cells on DPN by using GFP-positive iPS cells. NCL cells may have practical advantages for regenerative medicine, such as their easy accessibility and differentiation potential. The advantage of NCL cells for transplantation therapy actions is that they can exert the dual actions of cell replacement and supplying growth factors, both of which are required in the treatment of DPN. Although further studies designed to reveal additional useful aspects of NCL cell transplantation on DPN might be required, the transplantation of NCL cells appears to be a promising therapeutic strategy for DPN.
Footnotes
Acknowledgments
This research was supported in part by Grant-in-Aid for Scientific Research (21592506) from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) and in part by the “Strategic Research AGU-Platform Formation (2008–2012)” Project for Private Universities: matching fund subsidy from MEXT of Japan. The authors thank Ms. Michiko Yamada and Ms. Mayumi Katagiri (Department of Endocrinology and Diabetes, Nagoya University Graduate School of Medicine) for technical assistance. The authors declare no conflict of interest.
