Abstract
Mesenchymal stromal cell (MSC) immunosuppressive properties have been applied to treat graft-versus-host disease (GVHD) in allogeneic hematopoietic stem cell transplants (HSCTs). We have previously demonstrated that MSC infusions early after haplo-HSCT prevent GVHD in a haploidentical-HSCT mouse model. Now, we investigated the impact that MSCs' immunosuppressive properties have on the graft-versus-leukemia (GVL) effect. First, to mimic a chronic myeloid leukemia (CML) relapse after a haploidentical HSCT, lethally irradiated mice were coinfused with haploidentical donor bone marrow cells plus syngenic hematopoietic progenitors transduced with a retroviral vector encoding both the
Keywords
Introduction
Graft-versus-host disease (GVHD) is a frequent and potentially severe complication of allogeneic hematopoietic stem cell transplants (HSCT) not always controlled with the conventional immunosuppressive treatments (26). One of the recently developed alternatives to treat severe GVHD is the mesenchymal stromal cell (MSC) infusion (12,22,23). These multipotential nonhematopoietic progenitor cells have important immunosuppressive properties over most of the immune system cells, including T lymphocytes and dendritic cells, two of the main cell populations involved in GVHD (14,25,31,32,37). The infusion of MSCs in corticoid-resistant GVHD patients results in a modulation of the severity of this complication in an important proportion of the treated patients (2,12,22,23,36).
We have previously shown that the phenotype and immunoregulatory properties of adipose tissue-derived MSCs (Ad-MSCs) are similar to bone marrow-derived MSCs (41). Additionally, MSCs are able to prevent the development of GVHD in a mouse model of haploidentical bone marrow transplant. In these experiments, a significant increase in the survival of mice receiving the infusion of Ad-MSCs on days 0, 7, and 14 after transplant was observed compared to mice that were not infused with MSCs or that received MSCs later after transplant or that were treated with a single MSC infusion on day 0 (41).
It is well known that there is a strong correlation between GVHD and graft-versus-leukemia effect (GVL) in which the donor's T lymphocytes recognize tumoral antigens as nonself and destroy them (17–19). In this way, the GVL effect contributes to the eradication of residual malignant disease and reduces relapses after allogeneic HSCT. Chronic myeloid leukemia (CML) responds better to delayed lymphocyte infusions, a GVL-based therapy used for the treatment of CML relapses after allo-HSCT. In most studies, conducted before Imatinib became the first choice of treatment, between 60% and 80% of the treated patients obtained responses, although frequently associated with the development of severe GVHD (17,19).
Although MSCs are currently applied to patients with corticoid-resistant GVHD and no evident adverse consequences have been associated with their use, the MSC's impact upon the GVL effect remains to be elucidated. In this respect, previous studies showed that the subcutaneous injection of melanoma cells in allogeneic immunocompetent mice led to tumor growth only if coinjected with MSCs (8,9). Another experimental work demonstrated increased metastatic potency of a human breast carcinoma cell line in xenografts (15). Additionally, one small pilot study showed a higher relapse rate in oncohematologic patients cotransplanted with HSCs and MSCs to prevent GVHD, both obtained from human leukocyte antigen (HLA)-identical sibling donors (28). This repercussion, however, was not detected in other studies that coinfused MSCs to prevent GVHD and HSCs in oncohematologic patients, either in nonmyeloablative HSCT with HLA-mismatched donors (3) or with HLA-identical sibling donors after myeloablative therapy (20). Additionally, no evident increase in the recurrence rates of malignancies was observed after MSCs were infused to treat severe GVHD in the very high number of patients included in several clinical trials (16,22,27,35,40).
Our present study aimed to shed some light on whether MSCs used to prevent GVHD have an impact on the GVL effect and, consequently, whether infusion of these cells increased leukemic relapses after HSCT. Since GVL effect has its highest efficacy when applied to treat CML, in this work we developed a CML relapse-like mouse model by means of the cotransplantation of syngenic HSCs that were previously transduced with retroviral vector carrying the human oncogene breakpoint cluster region/V-abl Abelson murine leukemia viral oncogene homolog 1 (
Materials and Methods
Retroviral Production
The retroviral vector murine stem cell virus-internal ribosome entry
Hematopoietic Progenitor Infections
Bone marrow (BM) cells were harvested from femurs and tibias of female B6D2F1 mice and subjected to a negative hematopoietic lineage selection with the lineage cell depletion kit (Miltenyi Biotec, Auburn, CA, USA) according to the manufacturer's instructions. Lineage negative (Lin-) cells, enriched in hematopoietic progenitors, were cultured for 48 h in 20% FCS IMDM, 100 ng/ml murine stem cell factor (SCF; Preprotech, Rocky Hill, NJ, USA), and 100 ng/ml human interleukin-11 (IL-11; R&D Systems, Minneapolis, MN, USA). Nontreated culture plates were coated with 20 mg/ml retronectin (Takara Bio, Inc., Shiga, Japan) for 2 h at room temperature (RT) and washed before use. Supernatants were preloaded for 4 h, at 37°C, and washed before the infection. In the first infection cycle, 5×105 Lin- cells/ml and supernatants were added at a V/V proportion. After 24 h, a second infection cycle was performed. After 24 h, the expression of NGFR was analyzed by flow cytometry (see
Mice
B6D2F1 [H2b/d, cluster of differentiation 45.2 (CD45.2)] and C57Bl/6 (H2b/b, CD45.1) mice, aging 10-12 weeks, were housed and bred at the CIEMAT Laboratory Animals Facility (Registration No. 28079-21 A) from breeding pairs originally obtained from Jackson Lab (Bar Harbor, ME, USA). Mice were routinely screened for pathogens, in accordance with FELASA (Federation of European Laboratory Animal Science Associations) procedures, and received water (50 mm filtered and UV irradiated) and food (SAFE R04 25 KGy gamma-irradiated) ad libitum.
All experimental procedures were carried according to European convention ETS123 and Spanish L32/2007 and RD1201/2005 laws and regulations. Procedures were approved by our Animal Experimentation Ethical Committee according to all external and internal biosafety and bioethics guidelines.
Generation and Characterization of Mouse Ad-MSCs
To generate mouse Ad-MSCs, adipose tissue from B6D2F1 mice epiploon was cut into pieces, digested for 2 h with DMEM plus 1 mg/ml of collagenase A (Roche Diagnostics GmbH, Germany), centrifuged, 40 μm-filtrated, and cultured in flasks (Nalge Nunc International, Penfield, NY, USA) at 1.6×105 cells/cm2 in MesenCult medium and supplements for mouse cells (Stem Cell Technologies, Vancouver, BC, Canada) at 37°C, 5% CO2. Nonadherent cells were discarded after 24 h. Fresh medium was added and replaced twice a week. At 80% confluence, adherent cells were trypsinized, washed, and seeded at 4×103 cells/cm2. In all the experiments, MSCs were used at six to eight passages.
By immunophenotype analysis (see
Flow Cytometry Analysis
To analyze the phenotype by flow cytometry, Ad-MSCs were harvested by the addition of 0.05% trypsin-EDTA, washed, and resuspended in 1% bovine serum albumin (BSA) supplemented PBS (all Sigma-Aldrich, St. Louis, MO, USA), incubated in the dark at 4°C, 30 m with monoclonal antibodies, and washed. The analysis included CD34, CD45.1, CD45.2, CD90, CD117, CD105, CD73, CD166, CD29, and CD31 expression (BD Biosciences, San Jose, CA, USA).
Antibodies used to analyze
Chronic Myeloid Leukemia Relapse Mouse Model
On day 0, 107 BM cells from C57Bl/6 haploidentical donor mice were intravenously infused into B6D2F1 recipients, lethally irradiated with 2×6 Gy doses spaced 24 h apart. To mimic a posttransplant leukemic relapse arising from residual leukemic disease mice were additionally infused with a fixed dose of 105 Lin-
Recipient Mice and Cell Populations Infused in Each Study and Control Groups

The irradiated B6D2F1 recipients of the BM transplant control group (Table 1; HSCT,
Graft-Versus-Leukemia Effect Mouse Model
Mice BM did not contain enough T lymphocytes to produce GVHD after haploidentical HSCT, and GVHD only consistently developed after the infusion of 2×107 splenocytes. In this work, to generate a GVL mouse model (Table 1; GVL,
For GVHD group development, 107 haploidentical BM cells and 2.5×107 haploidentical splenocytes from C57Bl/6 mice were transplanted into lethally irradiated B6D2F1 recipients (Table 1; GVHD,
Ad-MSCs Impact on GVL Effect Mouse Model
To evaluate the impact that Ad-MSCs used to prevent GVHD have on the GVL effect, in the GVL mouse model (105 Lin- syngenic
In one experiment, five mice of the GVL group mice received the three Ad-MSCs doses plus four additional 2-2.5 × 105 Ad-MSCs infusions on days 19, 22, 24, and 25 in order to enhance the MSC's immunosuppressive effects.
We included a GVHD prevention control group to assure Ad-MSCs ability to prevent GVHD development, in which three standard Ad-MSCs doses were intraperitoneally injected on days 0, 7, and 14 into GVHD group mice (Table 1; GVHD + MSCs;
Follow-Up of the Experimental Groups
Six independent experiments were conducted. Each one of them included all the above described study and control groups. Animals were controlled every 2-3 days for clinical state and survival. Around 3 weeks after transplants, when hematopoietic reconstitution allowed it, mice of all the groups in each experiment were simultaneously weekly bled for blood counts and formulae (Hematological Analyzer Abacus Junior Vet, Diatron Messtechnik Ges.m.B.H., Ameisgasse, Austria) and for flow cytometry analysis of hematopoietic cell lineages and detection of NGFR+ cells. When a bad health state was detected, animals were euthanized by CO2 inhalation. In mice that received Lin-
Histology Analysis and Immunohistochemistry NGFR Detection
Mice were killed by CO2 inhalation; organs (spleen, lung, liver, bone marrow, skin, gut) were removed and fixed in formalin at RT before being embedded in paraffin and sectioned. To determine changes in tissue architecture, sections were stained with H&E.
For immunohistochemistry, sections were deparaffinized, rehydrated, and subjected to heat-induced antigen retrieval. For endogenous peroxidase inhibition, sections were treated with hydrogen peroxide (Sigma-Aldrich) for 1 h at RT and incubated O/N at 4°C with specific primary antibody against NGFR (ab8874, Abcam, Cambridge, UK) (1:500 dilution). After washing, cells were incubated with biotinylated donkey anti-rabbit IgG secondary antibody (1:1,000 dilution; Jackson Immuno-Research Laboratories, West Grove, PA, USA). Immunohistochemical staining was done with ABC Vectastain Elite Kits (Vector Laboratories, Peterborough, UK) according to the manufacturer's instructions. Slides were developed with diaminobenzidine (Dako, Glostrup, Denmark) and counterstained with hematoxylin.
Images were studied with Nikon, Eclipse E400 microscope with Nikon Plan 4×0.10, 10×0.25, 20×0.40, and 40×0.65 objective lenses (Nikon, Kingston Upon Thames Surrey, UK). Histology pictures were obtained with a Leica DFC420 camera with Leica Application Suite acquisition software (Leica, Milton Keynes, Buckinghamshire, UK).
T Lymphocytes In Vitro Response to Bone Marrow BCR/ABL-Transduced Lin– Cells
Ad-MSCs from B6D2F1 mice were seeded in P-96 flat-bottommed plates (105 and 104 per well; Nunc, Wiesbaden, Germany) and left to adhere for 18 h before 40 Gy irradiation to avoid their proliferation while preserving their viability. Spleens obtained from C57Bl/6 mice were disaggregated, treated with red blood cell lysing buffer (Sigma), and splenocytes resuspended at 106/ml concentration in 20% FCS-supplemented DMEM. Bone marrow Lin- nontransduced and
Statistical Analysis
Data statistical analysis was performed by using a one-way analysis of variance on rank with Tukey's post hoc test. Results are expressed as mean ± standard error of the mean and were considered significant if the
Results
BCR/ABL Retrovirally Induced Leukemic Relapse in a Mouse Model of Haploidentical HSC Transplantation
In a first set of experiments, Lin- BM cells from B6D2F1 (H2b/d) mice were transduced with a retroviral vector that carries the p210

Development of a retrovirally induced CML relapse-like mouse model. (A) Experimental design to generate a chronic myeloid leukemia (CML) relapse-like mouse model. On day 0, 105 Lineage negative (Lin–) syngenic cells, retrovirally transduced with the p210 breakpoint cluster region/V-abl Abelson murine leukemia viral oncogene homolog 1 (
All recipients but one (30/31), developed the clinical features of a CML-like myeloproliferative syndrome. Despite the variations in transduction efficacies, mice from the different experiments displayed very similar disease latencies and clinical characteristics. The animals died or were euthanized at days 28–60 after transplantation (Fig. 1B), when severe clinical signs developed (altered fur, reduced movements and hunched back, and one ataxia). The weekly peripheral blood (PB) analysis (Fig. 2) showed an increased percentage of granulocytes and circulating NGFR+ cells (Fig. 2A), mostly mature granulocytes and metamyelocytes, and scarce blasts. At death day PB displayed 7 ± 6% NGFR+ leukocytes (Fig. 2B). Necropsies showed large livers, marked splenomegalies (Fig. 2D), and consistent increased extramedullar hematopoiesis in various organs, in which the histology was analyzed (Fig. 3). The normal structures of the spleen (Fig. 3A) were replaced by leukemic hematopoiesis (Fig. 3E), consisting predominantly of immature but also mature white blood cells. This hematopoiesis was mainly leukemic with extensive NGFR expression, shown by immunohistochemical staining (Fig. 3F) and detected by flow cytometry analysis (75 ± 8%). Bone marrows were hypercellular (Fig. 3G), had a scarce representation of the red lineage, and showed mainly neoplasic myeloid cells at different stages of maturation, of which 24 ± 6% were NGFR+ as detected by cytometry (Fig. 2C) and by NGFR+ expression (Fig. 3H). Livers presented invasive hematopoietic leukemic infiltration of sinusoids and portal spaces (not shown). In contrast to human CML, mice displayed leukemic infiltration of the lungs, with frequent leukemic thrombus and infarcted zones, as well as extensive intraparenchymal hemorrhage (not shown). Engraftment studies showed mixed chimerism of CD45.1- and CD45.2-expressing cells detected by flow cytometry in the weekly PB analysis and the BM analysis at death day (data not shown). No NGFR expressing CD3+ or B220+ lymphocytes were detected, neither in PB nor in BM (not shown). In short, recipients developed a myeloproliferative syndrome that recapitulated the main features of CML.

Peripheral blood, bone marrow, and spleen characteristics of the CML relapse group mice. (A) Representation of the weekly monitoring of the peripheral blood (PB) granulocyte and leukemic NGFR+ cell evolution, starting on day 21 until the death day of each mouse compared to the HSCT control group mice. Gray area: cell range of HSCT control group engraftment. (B) Peripheral blood and (C) BM NGFR+ expressing cells at death day of the CML relapse group. The horizontal line represents the mean value. (D) Splenomegaly of the same group of animals compared to a HSCT control group spleen (Ctrl.spl.).
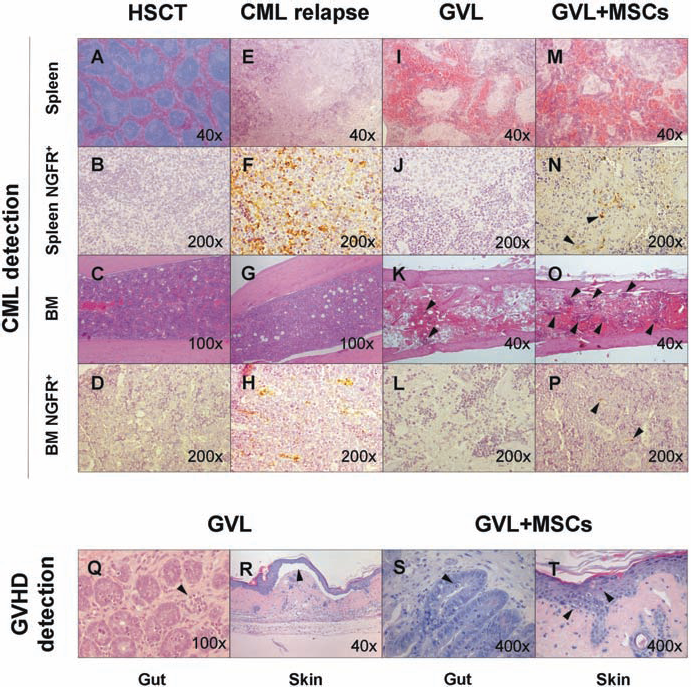
Histological characteristics of the organs of the CML relapse, GVL, and GVL + MSCs study groups compared to HSCT control group. CML detection: (A) HSCT normal spleen section (H&E, original magnification: 40×), (B) with no NGFR+ cell detection (IHC, original magnification: 200×). (C) HSCT bone marrow (H&E, original magnification: 100×), (D) with no NGFR+ detection (IHC, original magnification: 200×). (E) Spleen section of CML relapse group (H&E, original magnification: 40×) showing normal spleen structure replacement by hematopoietic cell infiltration. (F) CML relapse group spleen showing NGFR+ infiltration of leukemic cells (IHC, original magnification: 200×). (G) CML relapse group hypercellular leukemic bone marrow with scarce representation of the red lineage (H&E, original magnification: 100×). (H) Bone marrow NGFR+ leukemic infiltration (IHC, original magnification: 200×) in CML relapse group. (I) Disruption of spleen normal structure by fibrosis with hematopoietic infiltration (H&E, original magnification: 40×) in GVL group. (J) GVL group spleen representative image showing no NGFR+ leukemic cell detection (IHC, original magnification: 200×). (K) Aplastic bone marrow (H&E, original magnification: 40×) showing fibrosis, scarce hematopoietic focus (arrows) with (L) no NGFR+ leukemic cell detection (IHC, original magnification: 200×). (M) Disruption of normal spleen structure with fibrosis, white pulp depletion, and hematopoietic foci (H&E, original magnification: 40×), showing (N) leukemic NGFR+ expressing cells (arrows) (IHC, original magnification: 200×) in GVL + MSCs group spleen. (O) GVL + MSCs group hypoplastic bone marrow combining aplastic and cellular (arrows) areas (H&E, original magnification: 40x), showing (P) isolated leukemic NGFR+ cells (arrows) (IHC, original magnification: 200x). GVHD detection: (Q) Apoptotic cells and exploding crypt (arrow) with apoptotic bodies at the basis of the gut crypts (Grade II GVHD) in GVL group (H&E, original magnification: 100x). (R) Apoptosis of epidermal basal keratinocytes and subepidermal blister by separation of dermo-epidermal junction (arrow) in a Grade III GVHD GVL group mouse (H&E, original magnification: 40×). (S) Apoptotic cells at the basis of gut crypts (arrow) in Grade I GVHD in a GVL + MSCs mouse (H&E, original magnification: 400×). (T) Vacuolar degeneration of epidermal basal cells and apoptotic keratynocytes (arrows) in Grade II GVHD in a GVL + MSCs mouse (H&E, original magnification: 400x). GVL, graft-versus-leukemia, GVHD, graft-versus-host disease; H&E, hematoxylin-eosin staining; IHC, immunohistochemistry staining; MSCs, mesenchymal stromal cells.
Some of the mice developed a slower course of the disease with a milder clinical profile and low detection of NGFR+ cells in PB leukocytes (1.6 ± 1%). However, an increase in granulocyte count progressively developed, and mice died between days 66 and 72 after transplants, with the sole long-term survival of one mouse (1/31) that did not develop leukemia (Fig. 1B). Leukemic cell areas in spleen and in BM (5.4 ± 2% of NGFR+ cells) were detected. Although we could not determine the cause of death in these mice, their consistent death within a limited time period, compared to the prolonged survival of the HSCT control group, points to a leukemic-related event. The only surviving mouse of the group was euthanized on day +120. Necropsy showed that it was a healthy mouse, with no evidence of leukemic disease.
As a control group, to verify the transduced HSC capacity to generate CML, lethally irradiated B6D2F1 recipients were infused with 105 Lin-
Infusion of Haploidentical Splenocytes Prevents the Development of Leukemia Relapse in a GVL Effect Mouse Model
Next, we studied if haploidentical T lymphocytes infused with the haploidentical BM cells could control CML development in this CML relapse-like model by a GVL effect (Fig. 4A and Table 1, GVL). In the course of 2 weeks, mice showed PB leukopenia, ruffled fur displaying bald patches, hunched back, reduced movements and body weight loss, a clinical state similar to that of GVHD control group. Around days 13-36 after transplant, all the mice died or had to be sacrificed (Fig. 4B). Weekly PB analysis showed progressive leukopenia (Fig. 4C) that reached 1.7 ± 0.2×109/L leukocyte counts at death day. Similar observations were detected in the mice of the GVHD control group (Fig. 4B, C). In the GVL group, only a very small proportion of NGFR+ cells could be detected in PB at death day (mean 0.3 ± 0.1%) (Fig. 4C, D). Necropsies confirmed the absence of splenomegaly, hepatomegaly, or lung detectable leukemic infiltrations, with no significant NGFR+ cell detection. Bone marrows were hypoplastic, with varying grades of fibrosis (Fig. 3K), and no significant NGFR+ cell detection (mean 0.6 ± 0.3% NGFR+ cells) (Figs. 3L and 4D). Spleens showed disruption of the normal structure by fibrosis (Fig. 3I), sinusoidal dilatation, and white pulp depletion, the cellularity mainly consisting of maturative hematopoiesis with only isolated NGFR+ cells detection (mean 0.5 ± 0.2% NGFR+ cells) (Fig. 3J). The hematopoietic cells in weekly analysis of PB and the BM at death day were of donor origin (data not shown). Thus, the splenocyte infusion essentially prevented the development of CML by a GVL effect. GVHD as the death cause was determined by the clinical signs and confirmed by histological analysis that detected grade II–III gut GVHD in 100% of the analyzed animals (17/17) (Fig. 3Q); grade I–II liver GVHD in 88.2% (15/17); and grade I–III skin GVHD in 88.2% (15/17) (Fig. 3R).

Development of graft-versus-leukemia effect mouse model. (A) Experimental design to generate a GVL effect study group. On day 0, 2.5×107 splenocytes from C57Bl/6 haploidentical donor were infused into lethally irradiated B6D2F1 mice, together with 105
Reduced Ad-MSC Capacity to Prevent GVHD in the CML Relapse Mouse Model
In a previous work, we showed that MSC infusion prevented GVHD and increased mouse survival to 60–80% in a haploidentical HSCT mouse model (41). To investigate if Ad-MSC immunosuppressive properties could affect the GVL effect, Ad-MSCs were injected in the group of mice with CML relapse in which haploidentical splenocytes prevented the development of the disease (GVL group) (Fig. 5). Three doses of 2–2.5×105 Ad-MSCs were weekly infused into the GVL recipients, starting on day 0 of HSCT (Fig. 5A and Table 1, GVL + MSCs). To avoid deaths by embolism, we chose to infuse MSCs intraperitoneally.

PB weekly analysis showed normal leukocyte counts with a very low proportion of NGFR+ cells (0.7 ± 0.2%) at death day (Fig. 5C) that occurred on days 13–55 after HSCT (Fig. 5B) in 25/26 mice. At that time, only moderate GVHD clinical signs appeared in the recipients, whose survival curve was essentially similar to that of the GVL or GVHD groups, although with a mildler slope. The only survivor, sacrificed on day +90, was a healthy mouse. Necropsies showed spleens, livers, and lungs of normal appearance. BM histology showed heterogeneous cellularity, with hypoplastic and maturative cellular areas in the same bone (Fig. 3O), and 2.5 ± 0.6% NGFR+ cells detected by cytometry analysis and by immunohistochemical staining (Fig. 3P). Spleens showed a disrupted structure, with fibrosis and sinusoidal dilatation, and areas of maturative hematopoiesis (Fig. 3M), with scarce NGFR+-expressing cells (Fig. 3N). Chimerism studies showed engraftment of donor hematopoietic CD45.1 cells in the weekly PB analysis and the BM analysis at death day. GVHD histological analysis detected grade I–II gut GVHD in 94.1% of the analyzed animals (16/17) (Fig. 3S), grade I–II liver GVHD in 70.5% (12/17), and grade I–II skin GVHD in 64.7% (11/17) (Fig. 3T). These results point to the death cause being mainly GVHD. Since we detected leukemic cells in these mice, our results suggest that mice succumbed to GVHD before the final outcome of leukemia could be studied.
In one experiment, we studied if additional Ad-MSC doses could control GVHD and favor leukemic relapse. Five mice of the GVL group that received the three planned Ad-MSCs doses were additionally infused with 2–2.5×105 Ad-MSCs on days 19, 22, 24, and 25 after HSCT. However, despite the Ad-MSC dose increase, only transient improvement was obtained, and all the mice died around day 27 (data not shown). This implied a week of delay in the death of the recipients compared to mice that received the three standard Ad-MSC infusions that died around day 20 in this particular experiment. Again, necropsies showed that the death cause was a consequence of GVHD, and no increase in leukemic relapse was detected.
The GVHD prevention control groups that received the haploidentical BM and splenocytes but no NGFR+ cells (Table 1; GVHD) after the three intraperitoneal Ad-MSC injections had an average of 60% (56–70%) survival (Fig. 5B). This confirmed that the chosen doses and administration schedule of Ad-MSCs were efficacious in rescuing a significant proportion of the mice from lethal GVHD in the absence of leukemic cells.
In summary, the mice of the GVL group that were infused with Ad-MSCs died of GVHD relatively early after transplants. Although very few leukemic cells were found, we cannot assert that these leukemic cells would have been capable of causing an increase in leukemic relapse. Ad-MSC infusions seem to have slightly delayed GVHD development and death day as compared to the GVHD group, but our results show that, in the presence of
BCR/ABL Transduced Lin– Cells Enhance T Lymphocytes' In Vitro Proliferative Response
To evaluate if the reduced capacity of MSCs to prevent GVHD was related to the presence of leukemic cells, the T lymphocytes' response to
Discussion
One of the major curative effects of allogeneic HSCT is mediated by the GVL effect, in which donor's T lymphocytes recognize allogeneic antigens present in leukemic cells as nonself and destroy them (17,19,33). However, this beneficial effect of donor T-cells is closely associated with the potentially severe complication GVHD. One of the possible drawbacks of immunosuppression used to prevent GVHD in allogeneic graft recipients is the impairment of the GVL effect. Therefore, the development of animal models in which the balance between positive and negative consequences of preventing GVHD can be investigated are of the greatest importance.
In the present work, we have generated a GVL effect in a mouse model of CML-like relapse. We chose CML because this is the disease in which GVL effect is more efficacious with delayed lymphocyte infusions to treat relapses after allo-HSCT (17). Aiming to mimic a leukemic relapse generated by leukemic residual cells observed in transplanted patients, in our model the leukemic cells represented only a small proportion of the infused cells in the transplant after myeloablative conditioning regimen. To avoid possible strain-related problems, we chose to generate a CML-like myeloproliferative syndrome by transducing syngenic hematopoietic progenitors, instead of infusing leukemic cell lines (29,38,39). The
In a previous work, we have described in a mouse model that three 5×104 Ad-MSCs intravenous infusions administered weekly early after HSCT prevents GVHD development (41). In this study, we wanted to test if the Ad-MSCs' immunosuppressive properties had any impact on the GVL effect in our newly developed CML relapse mouse model. As mentioned, MSCs have been used to treat life-threatening GVHD, with significant increases in an important proportion of patients' survival and no evident adverse consequences (12,22). However, a special consideration has to be taken with the use of MSCs to prevent GVHD, particularly in standard risk HSCT, for this strategy could contribute to weaken the GVL effect and favor an increase in leukemic relapses. In this sense, some experimental works have demonstrated permissiveness to the development of solid tumors after MSC injections in immunocompetent mice as well as increased metastatic potential (8,9,15).
MSC immunosuppressive effects act in a dose dependent manner (1,21,24,30). In order to intensify the possible development of a leukemic relapse, we decided to increase significantly the number of Ad-MSCs injected to prevent GVHD in order to avoid any dose-dependent masking of possible relapses.
The results obtained when three 2–2.5×105 Ad-MSCs/week doses were infused into the GVL mice group were consistent throughout the six experiments performed but totally unexpected. As described, lymphocyte infusion prevented the development of leukemia of the GVL study group, and mice died of GVHD in a similar time period as the GVHD group. When Ad-MSC infusions were added to this group of mice, we had anticipated a moderation of the GVL effect by lowering T lymphocyte reactivity, and a possible appearance of leukemic relapses in at least a proportion of mice. However, although the clinical course was milder and deaths were delayed, recipients died of GVHD and not of leukemia, regardless of the very high dose of Ad-MSCs infused, that represented about 30–35×106 MSCs/kg of the recipient mice. Contrary to what we detected in the GVL group in which practically no NGFR+ could be detected, in the Ad-MSC recipients a small proportion of leukemic cells survived T lymphocyte infusion, demonstrated by NGFR+ cells detected in BM and spleen in the necropsies.
We did not foresee what seemed to be Ad-MSCs' lack of efficacy in preventing GVHD, although as expected, a 60% survival of the mice of the GVHD prevention control group was observed. We investigated if additional MSC injections could control GVHD and eventually favor leukemic relapse. Five mice of the GVL group that received the three planned MSC doses were infused four Ad-MSCs additional doses. Again, lethal GVHD developed and no increase in leukemic relapse was detected. These results suggest that the reduced efficacy of Ad-MSCs in preventing GVHD in a CML setting is probably a leukemic-related subject rather than MSC dose-related. Its combination with other immunosuppressive treatments has to be evaluated.
An exacerbation of the immune response of the donor's T lymphocytes to leukemic
In our in vivo model, the immune response to
Taken together, our results show in this CML relapse/GVL mouse model that Ad-MSCs have a modest efficacy in preventing GVHD, in contrast to what was detected when
Footnotes
Acknowledgments
