Abstract
We developed a clinical-scale cord blood (CB) cell ex vivo procedure to enable an extensive expansion of committed progenitors—colony-forming cells (CFCs) without impairing very primitive hematopoietic stem cells (HSCs). CD34++ cells, selected from previously cryopreserved and thawed CB units, were cultured in two steps (diluted 1:4 after 6 days) in the presence of stem cell factor (SCF), fms-related tyrosine kinase 3 ligand (Flt-3L), megakaryocyte growth and development factor (MGDF) (100 ng/ml each), granulocyte-colony stimulating factor (G-CSF) (10 ng/ml) in HP01 serum-free medium. HSC activity was evaluated in a serial transplantation assay, by detection of human cells (CD45, CD33, CD19 and CFC of human origin) in bone marrow (BM) of primary and secondary recipient NOD/SCID mice 6–8 weeks after transplantation. A wide amplification of total cells (~350-fold), CD34+ cells (~100-fold), and CFC (~130-fold) without impairing the HSC activity was obtained. The activity of a particular HSC subpopulation (SRCCFC) was even enhanced. Thus, an extensive ex vivo expansion of CFCs is feasible without impairing the activity of HSCs. This result was enabled by associating antioxidant power of medium with an appropriate cytokine cocktail (i.e., mimicking physiologic effects of a weak oxygenation in hematopoietic environment).
Introduction
Although frequently assimilated to “stem cells” (18), the human cord blood (CB) CD34+ cell population is extremely heterogeneous from a functional viewpoint. As an acceptable approximation (8,31), it could be considered to be composed of i) committed progenitors [or colony-forming cells (CFCs)], representing a relative majority; ii) a low number of short-term repopulating stem cells [usually revealed by the functional in vitro and in vivo assays as long-term culture initiating cells (LTC-ICs) or cells generating the committed progenitors in the secondary liquid cultures (pre-CFCs)]; iii) the primitive stem cells that exhibit the capacity of in vivo engraftment and could be evidenced by transplantation to NOD/SCID (or another immunodeficient strain) mice [Scid repopulating cells (SRCs)]; iv) the most primitive and rare population of stem cells that could be demonstrated on the basis of their capacity to maintain the human stem cell potential after being transplanted to the first generation of recipient mice (i.e., on the basis of their capacity to engraft the secondary recipient mice). Any culture system aimed to expand the CD34+ cells results in the production of precursors and mature cells and, in most cases, in the simultaneous amplification of committed progenitors. Whereas production of precursors and mature cells results from enhanced differentiation of committed progenitors in ex vivo cultures, the amplification of committed progenitors results mainly in their production from the stem cells differentiating rapidly in culture and, hence, exhausting themselves.
Most probably, these facts could explain the positive effect of transplanted expanded cells on the shortening of posttransplantation neutropenia. This is the reason why the mature cells, precursors, and committed progenitors should be amplified as much as possible. There is, however, another opposite demand to an ex vivo expansion procedure: to maintain the long-term engraftment capacity, the activity of very primitive stem cells in the expansion product should be preserved or, even better, amplified. In order to reach this goal, enabling the expansion of the whole CD34+ content from one CB unit for transplantation without taking its substantial part as “unmanipulated CB fraction,” many groups studied the different culture conditions. We present here a clinical-grade procedure for the ex vivo expansion of cord blood CD34+ cells isolated from previously frozen cord blood units (7) with or without volume reduction, allowing the amplification of total cells by factor ~350 and of committed progenitors by factor ~130 (mean values) without significantly impairing the activity of primitive stem cells. Our two-step clinical-grade serum-free culture system [stem cell factor (SCF), fms-related tyrosine kinase 3 ligand (Flt-3L), megakaryocyte growth and development factor (MGDF) (100 ng/ml), granulocyte-colony stimulating factor (G-CSF) (10 ng/ml)], initially upgraded (7) on the basis of the experimental data of Douay's group (17) in one-step cultures with the medium IRWIN (no longer commercialized) (7), was subsequently improved with serum-free medium Macopharma HP01 (13).
Materials and Methods
Isolation and Immunophenotypical Analysis of CD34+ CB Cells
CB units, previously banked in the CB bank of Bordeaux Transfusion Center (Etablissement Français du Sang Aquitaine-Limousin) as volume-reduced (4) or nonreduced samples, were thawed in the presence of DNase and CD34+ selection made [Isolex 300i (mean purity 59.2 ± 16%) or Variomacs (mean purity 94.85 ± 5%) (Miltenyi Biotech, Paris, France)] as described previously (7). Only CB samples rejected in the course of the securization from the technical, logistical, and organizational reasons were used. Informed consent to use the CB for experimental purposes (in the case it was not suitable for banking) was systematically obtained from the mothers before sampling. The experiments were performed on discarded CB units in compliance with French regulation (article R1243-49 of “Code de Santé Publique”) and with a related authorization granted to the Laboratory.
The CD34+ cell concentrations/purities in CB samples before freezing and after selection, as well as the CD34+ cell number in cultures, were detected following the recommendation of the International Society of Hematotherapy and Graft Engeneering (29) as previously described (16). Briefly, three-color fluorescence was used to detect viable CD45+/CD34+ cells that were counted directly by using tricount microbeads (Beckton Dickinson, San Jose, CA) by means of a flow cytometer (FACS Calibur, Becton Dickinson, San Jose, CA). For phenotypic characterization of fresh and cultured cells, the following fluorescein-coupled monoclonal antibodies were used: anti-CD13 (PE), anti-CD14 (FITC), anti-CD33 (PE), anti-CD61 (FITC) (all from Beckton Dickinson) and anti-CD41 (PE) (Pharmingen, San Diego, CA). The cells were counted by automatic cell counter (Cell Dyn 3000, Abbott, Rungis, France).
Expansion Cultures
CD34+ cells from one CB sample were divided in two parts used for two culture conditions (with and without IL-3; 0.5 ng/ml, R&D Systems, Minneapolis, MN). The cultures were initiated with 20 × 103 CD34+ cells per milliliter in HP01 medium (Macopharma, Tourcoing, France) (13) supplemented by SCF (100 ng/ml; Amgen SAS Neuilly sur Seine), MGDF (100 ng/ml; Amgen SAS Neuilly sur Seine), Flt-3L (100 ng/ml;, R&D Systems), G-CSF (10 ng/ml; Neupogen, Amgen SAS Neuilly sur Seine). The volume of culture varied depending on the CD34+ number obtained from each CB sample. This cell suspension was injected in culture cell bags (VueLife 25 or 118, AFC, Gaithersburg, MD) and cultured at 5% CO2 in water-saturated air (Incubators Stericult 200, Thermo Scientific or IGO 150 Cell-life Jouan) for 6 days, when the cultures were fed with the fresh cytokine-supplemented medium (4 volumes of fresh medium to 1 volume of culture) and continued until day 14. The analysis of cellular content and phenotype was performed at days 6, 10, and 14 of culture.
The serum-free medium (13), all cytokines (SCF, MGDF, G-CSF, and Flt-3L), bags, CD34+ selection devices, etc., used in this study were of clinical grade.
Detection of Committed Progenitors (CFCs)
The committed progenitors [CFC = (CFU-GM + CFU-Mix + BFU-E)] were detected using the cytokine-supplemented methylcellulose ready-to-use kit ID (Stem Alpha, Saint Clement les Places, France) (10). CD34+ cells were seeded in methylcellulose cultures in concentration of 500 cells/ml in 35-mm petri dishes (NUNC, Roskilde, Denmark). The cultures were incubated for 14 days and the colonies (>50 cells) enumerated on an inverse microscope (Leica, Wetzlar, Germany).
Detection of Stem Cells by Their In Vivo Repopulating Capacity (SRC)
The animal experiments were performed in compliance with the French regulation (License No: 3306002). A certain number (20, 40, and 80 × 103) of CD34+ cells at day 0, as well as their whole day 14 progeny (14,20), were injected to irradiated (3.5 Gy; 60Co source; Gamatron, Siemens, France) 8–10-week-old NOD/SCID mice (Central animal-keeping facility of Bordeaux 2 University), two to nine animals per condition. After 6–8 weeks, the animals were sacrificed and their femoral mononuclear bone marrow cells isolated and analyzed by flow cytometry (FACSCalibur; Becton Dickinson) for human CD45 (PC5-coupled anti-human antibody, Immunotech, Marseille, France). Femora were isolated and the bone marrow flushed with 1 ml of RPMI-1640 complemented with 20% fetal calf serum (FCS). After separation on Ficoll, mononuclear cells were incubated with rat serum (Stem Cell Technologies, Meylan, France) at 4°C (5% of final volume) in order to block Fc receptors. Cells were washed (PBS, EDTA 5 mM, human albumin 0.4%) and incubated with anti-human CD45, CD19, and CD33 antibodies. Cells were then analyzed on a flow cytometer FACSCalibur. To avoid false-positive results due to control isotype, we used the noninjected mice as controls (14), showing constantly a “background” less than 0.5%.
Serial Engraftment of NOD/SCID Mice: The Detection of Several Functional Subsets of Stem Cells
The recipient mice (9–15 per condition) were treated as explained and injected with the anti-CD122 [produced in our laboratory thanks to the TM-β1 hybridoma originating from Dr. Tanaka laboratory (30)] (100 μg/mice) 24 h prior to the transplantation of cells (80 × 103 cells at day 0 or their progeny at day 14). They were sacrificed 6–7 weeks later. The bone marrow from both femurs was flushed out in 1 ml of medium; a negligible volume of this suspension (65 μl) was used for cell counting, flow cytometry detection of human antigens [in addition to CD45 (see above), CD19 (PE), and CD33 (FITC) (Beckton Dickinson) were analyzed]. For human clonogenic progenitor detection, 10–20 × 103 cells per dish were plated in methylcellulose supplemented with human cytokines [SCF, IL-3, GM-CSF, and erythropoietin (Epo)] (kit Stem Alpha I), and “AB” human plasma. Thus, the human chimerism in femoral bone marrow of these anti-CD-122-treated mice was analyzed as well as the repopulation of their bone marrow with functional human progenitors, to get insight on two subpopulations of SRCs before and after expansion: a) one relatively less primitive, based on the total human cell repopulation (human CD45) and the other, relatively more primitive, based on the generation of human clonogenic progenitors in primary recipient's bone marrow.
Total cellular content from both femurs (suspension in 1 ml) of each primary recipient was centrifuged. The cells were resuspended in 30 μl of RPMI and injected intrafemorally (27) to the secondary recipient NOD/SCID mice, conditioned for transplantation as were the primary recipients. They were sacrificed 6–7 weeks later and the same flow cytometry analysis and functional colony-forming assay were performed as explained for primary recipients. The injected and noninjected (contralateral) femurs were analyzed separately. As explained previously, the cells of noninjected control mice (14) constantly showed a fluorescence “background” less than 0.5%.
Statistical Analysis
Student's t-test was used to assess the significance of difference between the experimental conditions. For multiple comparisons of mean values Kruskall-Wallis test was used; if a significant difference was obtained, the values were compared by Wilcoxon test with the Bonferroni correction.
Results
Expansion Efficiency
The fold expansion of total cells, CD34+ cells, and committed progenitors (CFCs) increased during the culture as evidenced at intermediary time points (day 6 and day 10) and at culture endpoint (day 14) (Fig. 1A). Mean day 14 expansion was ~350-fold, ~100-fold, and ~130-fold for total cells, CD34+ cells, and CFCs, respectively. Analyzing the mean values, the addition of IL-3 to the cytokine cocktail increased fold expansion 20–25%, but due to the important individual variations, these differences were not statistically significant. In fact, more detailed analysis of data revealed that only 5 of 10 samples increased the expansion of total cells at day 14 upon IL-3 addition at a dose of 0.5 ng/ml to the expansion cocktail. So, if only these samples “responsive” to this low IL-3 dose at the “borderline” of cell responsiveness (~50% of response) (“IL-3-responsive samples” in the further text) were taken into consideration, nearly three times better expansion was evidenced for total cells and CFCs (Fig. 1B), compared with the same samples without IL-3. It is interesting, however, that IL-3-responsive samples had five to six times lower fold expansion without IL-3 than corresponding samples that did not respond to IL-3 stimulation (Fig. 1, not shown). The absolute number analysis of CD34+ cells before and after expansion is shown in Table 1.

Fold expansion of total cells, CD34+ cells, and colony-forming cells (CFCs) in the course of the culture; effect of a limiting dose of IL-3. Fold expansion represents the ratio between the number of cells belonging to a category (total cells, CD34+ cells, CFCs) at the end of culture (day 14) and those at the beginning of culture (day 0). All cord blood units (CBUs) tested taken into consideration (N = 10) (A) or only those that responded by an increase in fold expansion to the low dose of IL-3 (0.5 ng/ml) added to the cytokine cocktail (N = 5) (B). (C) Expansion of clonogenic progenitors in the course of the culture. Insight to CFU-GM, BFU-E, and CFU-Mix compartment (N = 10). (D) Evolution of differentiation markers in expansion cultures. For each time point the white bars show the expression on fresh (day 0) cells (N = 7).
The Absolute Number of CD34+ Cells per Whole CB Unit (Extrapolated Theoretically on the Basis of Expansion of Half of the Samples)
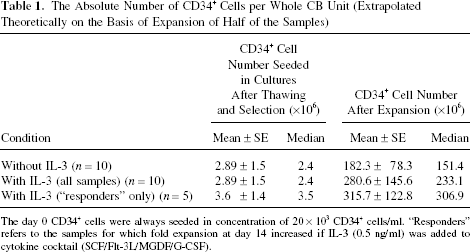
The day 0 CD34+ cells were always seeded in concentration of 20 × 103 CD34+ cells/ml. “Responders” refers to the samples for which fold expansion at day 14 increased if IL-3 (0.5 ng/ml) was added to cytokine cocktail (SCF/Flt-3L/MGDF/G-CSF).
Committed Progenitors in Expansion Product
Among the CFCs, CFU-GM progenitors were the dominant type (~90%) in the expansion product, irrespective of IL-3 treatment (Fig. 1C). In IL-3-supplemented cultures there was a trend of CFU-Mix to increase with time, but statistical evaluation did not confirm such an increase. Furthermore, the relative participation of the different class of progenitors in expansion product seems not to be influenced by the IL-3 presence whether all or only IL-3-responsive samples were analyzed (not shown).
Immunophenotypic Characterization of Cells in Expansion Product
In the course of expansion the percentage of CD34+ cells gradually decreased (Fig. 1D) to ~20% at day 14. Over 80% of cells constantly expressed CD13 and CD33 during the expansion culture but only <10% were CD14+ at day 14. On the other hand, the expression of megakaryocytic lineage markers progressively increased to reach ~20% at day 14 (Fig. 1D). Here again, the outcome was not affected by IL-3 addition to culture medium.
Maintenance of Stem Cell Subsets During Expansion
In order to get insight into the heterogeneous stem cell compartment, we employed first the classical SRC assay, injecting three different cell doses into nonlethal irradiation-conditioned NOD/SCID mice (9). While two cell doses (40 and 80 × 103 day 0 cells or their day 14 progeny) resulted in a high level of transplanted mice chimerism, the lowest dose (20 × 103 day 0 cells or their day 14 progeny) engraftment was relatively less efficient (Fig. 2), allowing to evaluate the percentage of positive versus negative mice and the human chimerism in murine bone marrow (each point represents one mouse). These experiments indicated that SRC activity was maintained during cell expansion in vitro, and that IL-3 addition did not determine any difference.

Maintenance of stem cells at the end of the expansion culture with respect to day 0 (cells of day 0 used for injection). Evaluation on the basis of engraftment of NOD/SCID mice. A limiting dilution analysis. *A cell dose (20, 40, and 80 × 103) of CD34+ cells at day 0, as well as the whole day 14 progeny (expansion product) of a cell dose was injected (IV) to non-lethally irradiated (3.5 Gy) 8–10-week-old NOD/SCID mice without any treatment destined to enhance the SRC seeding efficiency. After 6–7 weeks, the animals were sacrificed and their femoral mononuclear bone marrow cells isolated and analyzed by flow cytometry judged on the basis of human CD45 chimerism in their bone marrow. The “positivity” threshold was established to be 0.5% on the basis of the analysis of noninjected mice standard (dashed line). Each point represents one mouse.
In order to estimate more closely the activity of the different stem cell populations before and after expansion, we performed the transplantation of primary and secondary NOD/SCID recipient mice treated by anti-CD122. As presented in Figure 3, the first generation of mice (injected IV) showed comparable bone marrow chimerism for human cells expressing CD45, CD33, and CD19 antigens before and after expansion with or without IL-3 (Fig. 3). The analysis of human CFC in recipients' bone marrow (each point represents one mouse), however, showed twice more progenitors (statistically highly significant) after expansion without IL-3 than at day 0 or after expansion with IL-3 (Fig. 3). These data indicate the maintenance of SRCCFC subpopulation after expansion with IL-3, and its twofold amplification without IL-3.
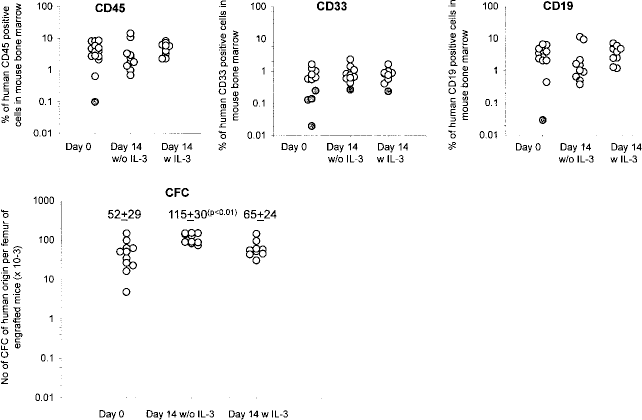
Maintenance of stem cells at the end of expansion cultures with respect to day 0: evaluation on the basis of engraftment of NOD/SCID mice. The day 0 CD34+ cells (80 × 103) or their whole day 14 progeny (expansion product) were injected (IV) to non-lethally irradiated (3.5 Gy) 8–10-week-old NOD/SCID mice pretreated with anti-CD122, in order to enhance the seeding efficiency of primitive stem cells. The animals were sacrificed after 6–8 weeks, and the presence of human cells endowed by immunophenotypical markers (CD45, CD33, CD19) or cells with the colony-forming ability (CFCs) in femoral bone marrow was analyzed. Each point represents one mouse. The mice negative for the human antigens (the background of fluorescence found in noninjected mice <0.5%) are represented by shaded points. p < 0.01 compared both to day 0 and day 14 with IL-3 conditions (Kruskal-Wallis test followed by Wilcoxon test with Bonferroni correction).
The cellular content of both primary recipients' femora was injected directly into the bone marrow (one femur per mouse only) of secondary recipients. Six weeks later, analysis demonstrated that the very primitive stem cells were not exhausted in the course of expansion, whether or not IL-3 was present (Fig. 4, each point represents a mouse). The analysis of human CFCs in injected femurs provides data concerning the most primitive populations of human HSCs that could be obtained using the NOD/SCID model. The results presented (Fig. 4) confirm the maintenance of this very primitive HSC subset in the course of expansion with or without IL-3.

Maintenance of very primitive stem cells at the end of expansion cultures with respect to day 0: evaluation on the basis of engraftment of secondary recipients—analysis of injected femur. The cellular content of both femora of primary recipients (the mice presented in Fig. 3) was injected directly in bone marrow of one femur of non-lethally irradiated (3.5 Gy) 8–10-week-old NOD/SCID mice pretreated by anti-CD122. The animals were sacrificed after 6–8 weeks, and the presence of human cells endowed by immunophenotypical markers (CD45, CD33, CD19) or cells with the colony-forming ability (CFCs) in femoral bone marrow was analyzed. Each point represents one mouse. The mice negative for human antigens (the background of fluorescence found in noninjected mice <0.5%) are represented by shaded points.
Finally, the contralateral femur (noninjected) was analyzed in secondary recipient mice in order to estimate the impact of expansion on the migration capacity of stem cells persisting in bone marrow of the primary recipients (Fig. 5). The CD45+ expression and the human CFC analysis showed that the expansion did not influence the migration capacity of very primitive stem cells. The relatively lower expression of human CD33 and CD19, however, indicates that the expansion affects the differentiation capacity of these very primitive stem cells.

Estimation of the migration capacity of repopulating cells after expansion with respect to day 0: evaluation on the basis of the repopulation of noninjected femur of secondary recipients. The results concern the same secondary recipients mice described in Figure 4 legend. The noninjected, contralateral femur was analyzed, as explained in Figure 4 legend, for human cells endowed by immunophenotypical markers (CD45, CD33, CD19) or for cells with the colony-forming ability (CFCs). Each point represents one mouse. The mice negative for human antigens (the background of fluorescence found in noninjected mice <0.5%) are represented by shaded points.
Discussion
In this article we presented a clinical-scale technique for the massive expansion of hematopoietic progenitors without impairing the activities of primitive and very primitive stem cells. The latter conclusion, based upon our data on the engraftment of secondary recipient NOD/SCID mice, is an additional argument suggesting that the very primitive stem cells responsible for the long-term engraftment are not exhausted in the course of our ex vivo expansion performed at clinical scale. This conclusion is similar to that obtained in cocultures of CB cells with mesenchymal stem cells (10) but substantially different from the other one derived from expansion experiments performed with the cytokine cocktail composed of SCF, MGDF, and G-CSF but without Flt-3L (21). The latter study established a requirement for two CB cell fractions to be transplanted: expanded cells to provide rapid short-term engraftment and unmanipulated cells to provide stem cells for long-term engraftment. The clinical-grade results presented here seem to authorize the expansion of the whole CB sample, and its transplantation alone, as sparing and transplantation of nonmanipulated cells is not required. This is important, since the mass of expanded cells could be doubled.
Four clinical trials concerning the transplantation of ex vivo-expanded cord blood CD34+ cells confirmed the feasibility of this strategy. However, they did not reveal a shortening of the posttransplantation neutropenia period after transplantation of the expansion product (6,15,24,28). In our opinion, the main reason for this is a still insufficient, despite its expansion ex vivo, total number of progenitors (CFCs), and CD34+ cells transplanted per kilo of donor weight: the median values in cited studies varied between 0.88 and 2.1 × 105 CD34+ cells/kg and between 0.27 and 1.02 × 105 CFC/kg (since only a fraction of sample was expanded). In addition, apart from two cohorts of patients [cohort “B” in one study (28) and all patients in another study (6)], the expanded fraction was transplanted 10 or 12 days after the nonmanipulated fraction. In comparison to these studies, our protocol, if the CD34+ cells from the whole CB unit were expanded, is expected to render between 10- and 100-fold more CD34+ cells and CFC/kg available for transplantation into the patients. This estimation is based on i) efficiency of expansion shown here and applied to the real content of CD34+ and CFU-GM in CB units in our bank [since data from this study (Table 1) mainly concern the samples with insufficient total and/or CD34+ cell number for banking], ii) the recovery of CD34+ cells after volume reduction (4) and selection (7). This way, the full contribution of expansion to the shortening of the posttransplantation neutropenia period could be evaluated. We already achieved a substantial shortening/abrogation of the posttransplantation neutropenia by transplanting to myeloma patients CD34+ cells mobilized into peripheral blood and expanded in our laboratory without unmanipulated cell support (1). Furthermore, a recent preliminary clinical trial (5), comparing the effects of transplantation of two nonmanipulated CB units to that of unmanipulated unit along with a second unit that had undergone Notch-mediated ex vivo expansion, confirms our estimations: the mean value of 6 × 106/kg (0.93–13 × 106/kg) of expanded CD34+ cells significantly reduced the period of posttransplantation neutropenia (>500 cells/μl) to 16 days (median) comparing to 24 days of two unmanipulated units (5).
The maintenance and even expansion of primitive stem cells (LTC-IC) and simultaneous expansion of committed progenitors, in the presence of Flt-3L and Tpo in long-term liquid cultures (25 weeks) (25), has been achieved. However, these cultures were serum supplemented and, although a 10 × 106-fold expansion of progenitors was obtained after 25 weeks, their expansion after 2 weeks (the time still acceptable for clinical setting of cord blood cell expansion) was only fivefold. The short-term cultures (<14 days) developed later, usually adopting Flt-3L, Tpo, and SCF as basic cytokine cocktail, achieved an expansion comparable with that of our study (19,26,33). Our approach, aimed to develop a clinically applicable procedure of expansion, took the advantage of the Flt-3L/Tpo(MGDF)/SCF association, completing it with G-CSF, in combination with a medium of new generation showing a better efficiency than previously used clinical-grade medias and being used in our laboratory for the clinical expansion/autologous transplantation trial with myeloma patients (22). The association of a medium with a powerful system of anti-oxidants with MGDF (Tpo) [stabilizing HIF1α transcripts (34)] mimics the physiologically low O2 environment of hematopoiesis, whereas the other cytokines (SCF, G-CSF, Flt-3L) in relatively high doses provoke a “regenerating bone marrow-like” effect [reviewed in Ivanovic & Boiron (12)].
A recent study performed with enriched CD133+ CB cells suggested, on the basis of the analysis of bone marrow repopulation after intrafemoral injection of expanded cells (SCF, Flt-3, Tpo), that expansion alters significantly the migration properties of SRCs (32). Our data concerning a more primitive population of stem cells (Fig. 5) would be in favor of the alteration in myeloid and lymphoid differentiation potential of expanded primitive stem cells rather than their migration capacity. However, due to numerous reasons, the xenogenic NOD/SCID transplantation model could not be considered as appropriate to study the migration of human cells, so these results should be taken with caution.
By analyzing our own and literature data (11), we concluded that serum-free cultures supplemented with at least Flt-3L and Tpo could enable IL-3 to act predominantly by stimulating the proliferation of primitive stem cells without acting in favor of their differentiation. To achieve this stem cell-enhancing effect of IL-3, it was suggested that the lowest dose still active should be used to decrease the probability of its prodifferentiation action (11,12,14). We tested this hypothesis by using a very low dose of IL-3 (0.5 ng/ml). This limiting efficiency dose ensured ~50% response in terms of ex vivo amplification of progenitors, so the responsive samples could be considered as stimulated by the lowest IL-3 dose still acting on stem and progenitor cells under the tested conditions. The results obtained confirmed our hypothesis as well as the fundamental phenomenon shown (2) since the production of committed progenitors in the cultures of IL-3-responsive samples increased significantly, compromising neither the SRC activity nor that of the very primitive stem cells engrafting the secondary recipient mice. This potential advantage of IL-3 is not, however, directly transposable to the clinical expansion cultures of CB CD34+ cells because: i) a clinical-grade IL-3 molecule accepted by health authorities is not available at the moment, ii) an IL-3 dose-escalation study should be carried out in order to establish the lowest 100% efficient dose (100% responsive samples), and iii) it should be evaluated if it still enables a beneficial effect on the expansion of progenitors, without provoking the exhaustion of stem cells. These concerns (including the evaluation of very primitive stem cell activity by a serial transplantation of NOD/SCID mice) will be the object of our future studies.
Since the clinical-grade MGDF used in this study is no longer available, a clinical-grade Tpo molecule is replacing MGDF in our ex vivo expansion protocol. The major points of this study carried out with MGDF were confirmed with Tpo (manuscript in preparation), and MGDF was replaced by Tpo to perform clinical expansions/autologous transplantations using mobilized CD34+ cells of myeloma patients (22). The clinical trial (allogeneic adult transplantations) based on the CB expansion protocol without IL-3 presented here (slightly modified: Tpo to replace MGDF and 12 instead of 14 days of culture) is ongoing under the responsibility of Professor N. Milpied (http://clinicaltrials.gov/ct2/show/NCT01034449).
Of course, all these considerations concern the hematopoietic transplantation context. The other transplantation contexts (3,23) could impose other objectives of ex vivo expansion and demand some other approaches and means.
Footnotes
Acknowledgments
The authors would like to thank Dr. Tanaka (Osaka, Japan) for the gift of the TM-β1 hybridoma cell line, as well as to Mrs. Elisabeth Volkmann (language corrections) and Mrs. Pascale Potié (graph design and manuscript formatting). This work was supported by R&D funds of Aquitaine-Limousin Branch of French Blood Institute (Bordeaux).
