Abstract
Human umbilical cord-derived mesenchymal stem cell (UC-MSC) transplantation has shown promise for the treatment of various diseases. For clinical applications, UC-MSCs have been stored in 0.9% saline, 5% dextrose, dextrose and sodium chloride injection, Plasma-Lyte A, 1% human serum albumin (1% HSA), or 5% HSA before administration, but the effect of storage conditions on the viability and biological function of the cells remains unknown. Freshly harvested UC-MSCs were resuspended and incubated in these solutions for 2, 4, or 6 h at 4°C or room temperature (24°C). Cell viability, apoptotic/necrotic fraction, poststorage growth potential, immunophenotype, immunosuppressive capacity, and differentiation capacity were analyzed. When stored in parenteral solutions, UC-MSCs showed progressive deterioration in survival viability and adhesion ability. After 6-h storage, the best viability and attachment rate of UC-MSCs decreased to 83.0 ± 1.6% and 71.8 ± 3.2%, respectively. Our results suggested that UC-MSCs in these conditions lose their viability in a short time. However, it seems that the other biological functions of the surviving UC-MSCs were little affected. Since UC-MSCs suspended in these mediums lose their survival viability in a short time to levels significantly below the permissible limits (70%) by FDA, precautions need to be taken on using these solutions as suspension medium and further studies on the optimal methods for preservation are urgent.
Introduction
Mesenchymal stem cells (MSCs) have an extensive capacity for self-renewal, proliferation, and differentiation into multiple lineages, as well as a role in immunoregulation. At present, MSCs are increasingly employed in cell therapy for a wide variety of immune disorders (18), degenerative diseases (41), and tissue injuries (12, 24, 26). MSCs can be isolated from different tissues (5, 16, 40), but the primary source is bone marrow (BM). However, alternative sources could be advantageous due to the invasive procedures required for isolating BM-derived MSCs and the significant age-related decrease (37) in their numbers and differentiation potential. Umbilical cord-derived mesenchymal stem cells (UC-MSCs), which are easily isolated from the umbilical cord normally discarded at birth, are a promising candidate source of MSCs. In addition to abundant supply, painless collection, and faster self-renewal, UC-MSCs also display similar biological characteristics to BM-MSCs. Together with the relative lack of ethical and safety issues associated with their use, UC-MSCs are considered to be an extremely promising alternative to BM-MSCs (6, 39) and have been extensively studied, including its biology characteristic (15, 43) and allogeneic transplantation for autoimmune diseases (19) and neurodegenerative disease (22, 44).
MSCs have been widely used in preclinical and clinical applications, with varying degrees of effectiveness (25). Many factors may affect the prognosis, such as cell sources (19), culture conditions (29), cell collection processes (33), the number of cells used (34), transplantation time (31), implantation method (14), and the variation between individual patients (13).
In addition to these factors, cell quality prior to transplantation is also important. The effects of storage conditions, such as solutions, temperatures, and durations of cell preservation, are particularly influential on cell quality. Before clinical therapy, it is necessary to store MSCs in parenteral solutions approved by Food and Drug Administration (FDA).
To date, there is no guideline for storing freshly MSCs, and few studies have been performed to define the optimal conditions for maintaining the viability of MSCs during storage. Since 0.9% saline, 5% dextrose, 5% dextrose in 0.9% sodium chloride (dextrose and sodium chloride injection), Plasma-Lyte A, 1% human serum albumin (1% HSA), and 5% HSA have previously been used as storage solutions before administration, it is essential to investigate the effect of these solutions on UC-MSC survival and biological functions.
In this study, we investigated how different approved storage solutions, used at different temperatures and for different periods of time, affected the viability, rate of apoptosis/necrosis, poststorage proliferation potential, surface phenotype, immunosuppressive capacity, and differentiation capacity of UC-MSCs harvested from in vitro culture for transplantation.
Materials and Methods
Isolation and Culture of Umbilical Cord Mesenchymal Stem Cells
The research protocol was approved by the Ethics Committee of the Institute of Medical Science, Shanxi Medical University. Informed consent was obtained from mothers who planned to deliver their healthy full-term babies. After delivery, umbilical cords were immersed in phosphate-buffered saline (PBS, pH 7.4) supplemented with antibiotics (penicillin, 200 μg/ml; streptomycin, 200 μg/ml; Hyclone, Thermoscientific, Logan, UT, USA), and processed within 4 h after collection. To isolate stem cells, the cord was washed twice in PBS and cut into small pieces (about 2 cm in length). Vessels were removed from the matrix to avoid endothelial cell contamination, and the rest of the cords were finely minced into 1- to 2-mm fragments, which were plated in 75-cm2 flasks (Corning, Corning, NY, USA). The cord sections were carefully covered with 10 ml of medium comprising low-glucose Dulbecco's modified Eagle's medium (LG-DMEM) supplemented with 10% fetal bovine serum (FBS), penicillin (100 μg/ml), and streptomycin (100 μg/ml) solution (all from Hyclone). Cultures were incubated at 37°C in a humidified CO2 incubator with a change of 10% FBS/LG-DMEM every 3–4 days. Approximately 2 weeks after expansion, colonies of fibroblast-like cells appeared. When cells reached 70–80% confluence, they were rinsed with PBS, harvested with 0.25% trypsin-EDTA (Gibco, Grand Island, NY, USA), split at a concentration of 1 × 104 cells/cm2, and transferred into another 75-cm2 flask for further expansion. Cells of passage 3 were used for subsequent studies. All experiments were replicated three times.
Characterization of MSCs
Expression of surface antigens was probed using anti-CD29-phycoerythrin (PE), CD44-PE, CD105-PE, CD34-electron coupled dye (ECD; PE-Texas Red), CD45-PE-Cyanin 5.1 (PC5) (Beckman Coulter, Brea, CA, USA) and determined by flow cytometry (Beckman Coulter FC500). Nonspecific IgG-PE, IgG-ECD, and IgG-P5 were used as isotype controls (Beckman Coulter).
pH and Osmolarity Adjustment
To evaluate the viability of UC-MSCs before transplantation, we also decided to modify these solutions under the physiological environment in order to figure out whether pH/osmolarity of solutions refined our results. It is known that the physiological pH is 7.2–7.4 and the osmolarity is 280–320 mOsm/L. Thus, we adjusted pH value of 0.9% saline, 5% dextrose and dextrose, and sodium chloride injection to 7.2 with 0.1 M NaOH. The level of osmolarity of dextrose and sodium chloride injection was modified with deionized water to 310 mOsm/L as osmolarity of the other solution fell within the appropriate range. Solution osmotic pressure was determined by measurement of the depression of freezing point using an osmometer (Model FM-8P, Shanghai Medical University Instrument Factory, Shanghai, China), and pH was measured immediately by a pH meter (Model PHS-3C, Shanghai Precision & Scientific Instrument Co., LTD, Shanghai, China).
Cell Viability Assay
The UC-MSCs at passage 3 were harvested using 0.25% trypsin (Gibco), washed three times, and resuspended at a concentration of 2 × 106 cells/ml. For storage studies, UC-MSCs were maintained in 0.9% saline (Baxter International, Deerfield, IL, USA), 5% dextrose (Baxter), dextrose and sodium chloride injection (Baxter), Plasma-Lyte A (Baxter), 1% HSA, and 5% HSA (Octapharma, Oberlaaerstra, Austria). These cells were assayed after 2, 4, and 6 h at 4°C or room temperature. At the end of each time point, a cell viability assay was performed using the trypan blue (Gibco) staining method. After removing some of the cell pellets and adding 0.4% trypan blue, cells were plated onto a slide, and unstained cells were counted as live cells. Unstored cells are fresh cells that did not undergo storage at time 0 h.
Cell Apoptosis and Necrotic Fraction
To assess apoptosis and necrosis, an Annexin V/propidium iodide (PI; Biovision, Milpitas, CA, USA) double staining assay was performed following the manufacturer's instruction. Briefly, following storage, the cells were resuspended in binding buffer at 5 × 105/ml, fluorescein isothiocyanate-conjugated Annexin V and PI were added to the cell suspension, and the resulting solution was incubated at room temperature in the dark for 5 min. Finally, the fraction of early apoptotic (Annexin V) and necrotic (or late apoptotic; PI) cells was calculated using flow cytometry (Beckman Coulter FC500).
Assay for Cell Adhesion
UC-MSCs at passage 3 were isolated by trypsinization. After storage in above solutions, suspensions of 1 × 104 UC-MSCs were added to each well of a 24-well plate (Corning) and allowed to attach for 24 h at 37°C and 5% CO2. To determine UC-MSC adhesion, plates were washed three times with PBS, fixed by 4% paraformaldehyde (Sigma-Aldrich, St. Louis, MO, USA), and stained by crystal violet (Sigma). To quantify UC-MSC adhesion, the number of attached cells was counted. Each experiment was performed in triplicate wells and repeated three times.
Population Doubling Time
To assess the population doubling time (PDT), the poststorage UC-MSCs were replated in a 24-well plate (Corning) at a concentration of 5 × 103 cells/well and detached daily at the same time for 3 successive days with cell counts calculated each day using a hemocytometer (Hausser Scientific, Horsham, PA, USA). Three replicates were carried out for each sample. The PDT was calculated using an online formula (www.doubling-time.com).
Additionally, when cells were 80% confluent after 3 days, they were trypsinized, counted, and reseeded at the same concentration of 5 × 103 cells/well in a new 24-well plate. The number of cells and the duplication time were calculated for three passages after storage.
Immunomodulation Capacity
To assess the immunosuppressive properties of poststorage UC-MSCs, lymphocyte proliferation assay was performed. Human peripheral blood lymphocytes/mononuclear cells (PBMCs) were isolated from healthy donors after informed consent and ethical approval by the Second Hospital of Shanxi Medical University, using Ficoll-Paque (Amersham Pharmacia Biotech, Uppsala, Sweden; 1.077 g/ml) density gradient centrifugation, and stimulated by phytohemagglutinin (PHA; Sigma, 50 μg/ml) for 96 h. The viable UC-MSCs after storage were replated into a 96-well (Corning) at a density of 1 × 104/well. At 70% confluence, UC-MSCs were inactivated by treatment with 50 μg/ml mitomycin-C (Sigma) for 1 h at 37°C. After the medium was removed, the inactivated UC-MSCs were washed twice with PBS and cocultured with 1 × 105/well PHA-induced PBMCs for 72 h at 37°C and 5% CO2. A cell counting kit-8 (CCK-8; Dojindo, Kumamoto, Japan) was used to assess the immunomodulatory impact, and the procedure was carried out according to the manufacturer's protocol. The inhibitory effect of UC-MSCs on lymphocyte proliferation was evaluated by comparing the optical density (OD) in wells cocultured on inactivated UC-MSCs with the OD of lymphocytes grown alone.
Differentiation
At the end of the last time point, the UC-MSCs were checked for their ability to differentiate into osteocytes and adipocytes.
For osteogenic differentiation, poststorage UC-MSCs were replated and incubated in LG-DMEM containing 10% FBS in six-well tissue culture plates, and then the medium was changed to osteogenic differentiation medium (Cyagen Biosciences, Sunnyvale, CA, USA) after 24 h of adherence. The medium was replaced every 2 days for 21 days. After differentiation, the cells were washed twice with PBS, fixed in a solution of 4% formaldehyde solution for 30 min, and stained for 5 min with Alizarin red (Cyagen Biosciences).
For adipogenic differentiation, poststorage UC-MSCs were incubated in LG-DMEM containing 10% FBS for 24 h of adherence, and then the medium was changed to adipogenic differentiation medium (Cyagen Biosciences). The medium was replaced every 2 days for 14 days. After differentiation, the cells were washed twice with PBS, fixed in a solution of 4% formaldehyde solution for 30 min, and stained for 30 min with Oil Red O (Cyagen Biosciences).
Statistical Analysis
The results were expressed as mean ± standard deviation (SD) of the mean. The results were analyzed using repeated-measures analysis of variance (ANOVA) and one-way ANOVA, post hoc multiple comparisons were made using Tukey–Kramer test. Statistical significance was accepted as p < 0.05.
Results
Characterization of Cultured UC-MSCs
Characterization of UC-MSCs was assessed by immunophenotyping and differentiation. The results indicated that UC-MSCs were positive for CD29, CD44, and CD105 and negative for CD34 and CD45 (Fig. 1A). In addition, UC-MSCs were shown to possess the ability to differentiate into osteocytes and adipocytes (Fig. 1B).
Immunophenotyping and differentiation of cultured UC-MSCs. (A) Immunophenotype of umbilical cord-derived mesenchymal stem cells (UC-MSCs) by flow cytometric analysis. (B) Osteogenic differentiation was examined by Alizarin red staining (left); adipogenic differentiation of UC-MSCs was assessed by Oil Red O staining (right).
Effect of Cellular Storage Solutions on Viability of UC-MSCs
The harvested UC-MSCs were suspended in 0.9% saline, 5% dextrose, dextrose and sodium chloride injection, Plasma-Lyte A, 1% HSA, and 5% HSA stored at 4°C or room temperature. After 6 h of storage, the best cellular viability was only 83.02 ± 1.6% in 0.9% saline at 4°C, which is significantly decreased (p < 0.05) compared with control (Fig. 2A). Similarly, at room temperature, viability was significantly reduced in all solutions and the best viability of UC-MSCs in 0.9% saline was 76.9 ± 1.9% after 6 h of storage (Fig. 2B). These results indicated that the viability of UC-MSCs suspended in the above solutions decreased quickly over a short period of time (p < 0.05).
Viability of UC-MSCs suspended in different solutions at 4°C and room temperature. (A) Cell viability for UC-MSCs at 4°C at 2, 4, and 6 h. (B) Cell viability for UC-MSCs suspended in the same solutions and times as for (A) but at room temperature. *p < 0.05 versus unstored UC-MSCs. HSA, human serum albumin.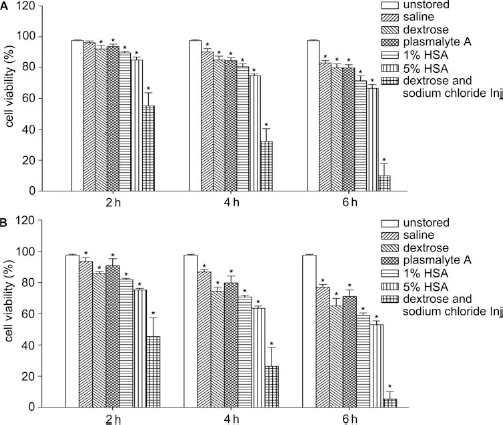
Considering that pH and osmolarity changes may affect viability in the above solutions, we adjusted the pH and osmolarity values of 0.9% saline, 5% dextrose, and dextrose and sodium chloride injection, reevaluated their effects on the preservation of cells, and found that these modifications did not change the viability of UC-MSCs significantly. After 6 h of storage at 4°C, cellular viability in adjusted saline and dextrose was 82.2 ± 1.3% and 78.1 ± 2.0% (data not shown), respectively, and the improvement did not reveal a statistically significant difference. By contrast, the viability of UC-MSCs suspended in dextrose and sodium chloride injection was increased after adjusting its osmotic pressure. After 6 h of storage at 4°C, cell viability in adjusted dextrose and sodium chloride injection was 77.4 ± 2.7% (data not shown). However, this value is still low for clinical applications compared with the value for 0.9% saline.
Effect of Storage Solutions on Cellular Apoptosis and Necrosis
Apoptosis and necrosis in UC-MSCs resuspended in 0.9% saline, 5% dextrose, dextrose and sodium chloride injection, Plasma-Lyte A, 1% HSA, or 5% HSA at 4°C or room temperature were evaluated at 2, 4, and 6 h poststorage (Fig. 3). Following storage in these solutions, the fraction of necrosed UC-MSCs gradually increased over time (p < 0.05) (Fig. 3B, D). The fraction of apoptotic cells showed no significant difference among the groups (data not shown), indicating that the storage solutions mostly affected the rate of necrosis (Fig. 3A, C).
UC-MSC apoptotic and necrotic cell fractions suspended in cellular storage solutions at 4°C and room temperature. (A) Representative flow cytometry data of UC-MSCs at 4°C for 6 h. (B) Fraction of necrotic cells (%) for UC-MSCs at 4°C for 2, 4, and 6 h. (C) Representative flow cytometry data of UC-MSCs at room temperature for 6 h. (D) Fraction of necrotic cells (%) for UC-MSCs suspended in the same solutions and durations as in (B) but at room temperature. *p < 0.05 versus unstored UC-MSCs.
Effect of Storage Solutions on Cell Proliferation
Adhesion is the first step for the survival of attached cells and so poststorage proliferation capacity was investigated by cell adhesion assay and PDT. As shown in Figure 4, storage solutions altered cell adhesion significantly. The best adhesion ability of UC-MSCs after replanting was markedly reduced to around 70% after 6 h (p < 0.05) of storage at 4°C in 0.9% saline (Fig. 4A), which is very close to the minimum viability (70%) for cell transplantation acceptable by the FDA. Similar results were obtained in the other solutions. Fortunately, the proliferation potential of the remaining attached cells was maintained. Although the PDT of poststorage UC-MSCs was increased, there was no significant difference to control after one passage (Fig. 4B, D).
Poststorage growth potential for UC-MSCs suspended in different solutions at 4°C and room temperature for 6 h. (A) Cell adhesion assay of UC-MSCs suspended in different media for 6 h at 4°C. (B) Population doubling time (PDT) of poststorage UC-MSCs at 4°C for three successive passages. (C) Cell adhesion assay for UC-MSCs suspended in the same solutions and durations as in (A) but at room temperature. (D) PDT of poststorage UC-MSCs at room temperature for three successive passages. *p < 0.05 versus unstored UC-MSCs.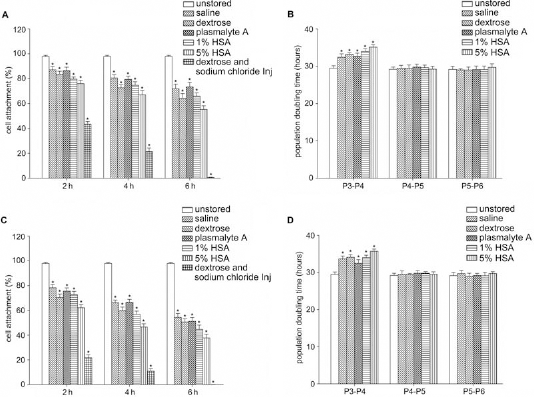
Effect of Storage Solutions on Surface Phenotype
We next investigated whether suspending in various parenteral solutions for 6 h at 4°C or room temperature altered the surface phenotype of UC-MSCs. To this end, we performed flow cytometry experiments designed to detect their marker surface antigens, CD29, CD44, and CD105. There were no statistical differences in expression levels between stored and unstored UC-MSCs (Fig. 5).
Immunophenotype of UC-MSCs in parenteral solutions for 6 h at 4°C and room temperature.
Immunomodulatory Effect of Poststorage UC-MSCs
To investigate the immunosuppressive capacity of poststorage UC-MSCs, we cocultured induced PBMCs and viable UC-MSCs suspended in different solutions for 6 h at 4°C or room temperature. As shown in Figure 6, the proliferation of PBMCs induced by PHA was significantly suppressed by unstored UC-MSCs (p < 0.05, n = 3). The inhibitory effects of viable UC-MSCs after storage were only slightly less than the unstored (p > 0.05, n = 3).
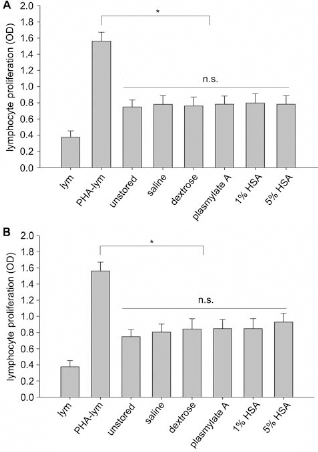
Immune suppression by coculture of UC-MSCs with PHA-induced lymphocytes. (A) Immune suppression of UC-MSCs after being stored in parenteral solutions for 6 h at 4°C. (B) Immune suppression of UC-MSCs after being stored in parenteral solutions for 6 h at room temperature. *p < 0.05 versus phytohemagglutinin (PHA)-induced lymphocytes; n.s., not significant. Abbreviations: lym, lymphocytes; PHA-lym, PHA-induced lymphocytes.
Effect of Storage Solutions on Differentiation
The UC-MSCs that attached to the culture dish after retrieval from storage were further subjected to in vitro differentiation into osteocytes and adipocytes. After induction, UC-MSCs from all groups exhibited multidifferentiation ability (Fig. 7).

Poststorage differentiation for UC-MSCs suspended in different solutions. (A) Differentiation capacity of UC-MSCs after being stored in parenteral solutions for 6 h at 4°C. (B) Differentiation capacity of UC-MSCs after being stored in parenteral solutions for 6 h at room temperature. Osteogenic differentiation was examined by Alizarin red staining; adipogenic differentiation of UC-MSCs was assessed by Oil Red O staining.
Discussion
UC-MSCs display the characteristics of MSCs as defined by the International Society for Cellular Therapy (ISCT) (7). The accumulated evidence strongly suggests that UC-MSCs are excellent candidates for regenerative medicine (11, 19, 28, 45).
For transplantation, UC-MSCs must be detached from their culture expansion vessel and stored in a parenteral solution that is approved by FDA immediately prior to infusion into the patient. In fact, immediate transplantation of UC-MSC is not always possible under clinical conditions. Many surgical and cell transplantation processes (e.g., burr hole surgeries for Parkinson's disease or intra-coronary injections for myocardial infarction), as well as long-distance transportation of UC-MSCs for transplantation, may increase the waiting time prior to transfusion. Cryopreservation is a well-established alternative for long-term (months to years) MSC storage. However, its low recovery rate, low vitality rate, and requirement for toxic cryoprotectants make it impractical for short-term (i.e., 24–48 h) storage of MSCs(8).
There are, however, no guidelines for the storage of freshly prepared MSCs prior to transplantation. Furthermore, the effect of storage conditions on cell viability has not drawn sufficient attention. Regarding BM-MSC transplantation, different groups have used different transplantation methods and holding solutions (1, 2, 4, 9, 27, 38,42). Venkataramana et al. (41) stored harvested cells in 0.9% saline, Wang et al. (42) resuspended cells in PBS, Mohamadnejad et al. (26) suspended cells in medium 199 (M199), and Mohyeddin-Bonab et al. (27) preserved cells in 1% HSA. None of these studies provided information regarding cell function prior to transfusion into patients, and it is probable that clinical outcomes were affected by the storage conditions used. Additionally, PBS, M199, and other culture medium (e.g., DMEM, etc.) are only suitable for in vitro use and not for injection. To our knowledge, no previous investigations into the effects of parenteral solutions on the viability and biological function of UC-MSCs have been performed. Investigating the influence of the storage conditions under which the UC-MSCs are stored before transplantation is therefore highly essential.
We designed the present study to evaluate the suitability of 0.9% saline, 5% dextrose, dextrose and sodium chloride injection, Plasma-Lyte A, 1% HSA, or 5% HSA for the preservation of UC-MSCs for different storage times. The results surprisingly indicated all conditions tested caused a rapid deterioration in the rate of survival of UC-MSCs. However, the proliferation rate, the surface marker expression profile, and the differentiation capacity of the surviving cells were unaffected.
The solutions we tested (0.9% saline, 5% dextrose, dextrose and sodium chloride injection, Plasma-Lyte A, 1% HSA, or 5% HSA) are FDA approved for direct administration into patients. The first three are commonly used for both intravenous infusion and the dilution of various drugs, while the latter three are typically used to store MSCs for transplantation. Cells for transplantation are often preserved at 4°C to minimize deterioration, while transplantation itself is conducted at room temperature, and so the combined effect of both storage solution and temperature on cellular biological function should be investigated.
In the present study, the viability of UC-MSCs was first evaluated by counting trypan blue-stained cells on a hemocytometer. This simple method is widely used to estimate the number of viable cells. It is surprising that viability of UC-MSCs decreased rapidly during short-term liquid storage. UC-MSCs could be stored in either 0.9% saline or 5% dextrose at 4°C for 6 h with maximum viability of 83% and 80%, respectively, or at room temperature for 6 h with maximum viability of 77% for 0.9% saline and 65% for 5% dextrose. When stored in dextrose and sodium chloride injection at either 4°C or room temperature, viability of UC-MSCs decreased to less than 60% after 2-h storage. Plasma-Lyte A, 1% HSA, and 5% HSA are typically used as storage media for MSCs prior to transplantation. How ever, none of them supported the survival of UC-MSCs. Additionally, the viability of UC-MSCs was significantly reduced in HSA in a concentration-dependent manner: the higher the HSA concentration, the lower the viability of UC-MSCs. This phenomenon was validated by determining the apoptotic and necrotic cell fractions in poststorage UC-MSCs. The results indicated that the necrosis rate, rather than the apoptosis rate, increased in all solutions.
A high proliferation rate is one of the key properties of UC-MSCs. Cell adhesion is a prerequisite for cell proliferation: following attachment, UC-MSCs spread and then divide. Previous studies have shown that the adhesion rate of MSCs was more than 95% 4 h after plating (35). To achieve the maximum adhesion, UC-MSCs were allowed to adhere for 24 h following retrieval from storage due to their initial lag phase prior to cell growth (32). After 24 h of replanting, the highest rate of adhesion was only 72% in 0.9% saline at 4°C for 6 h. At room temperature, cell attachment was even lower. For cell therapy, the minimum acceptable cell viability specification provided by the FDA is 70% (10). Thus, 6 h of storage was set as the end point of our observation.
Cell survival requires a very strictly controlled environment that provides the appropriate physiological pH, osmolarity, suspension medium electrolytes, and nutrients. Therefore, the rapid deterioration of survival of UC-MSCs during suspension in solutions containing 0.9% saline, 5% dextrose, and dextrose and sodium chloride injection should not be surprising because of their low-pH levels (e.g., 0.9% saline: 4.5–7.0; 5% dextrose: 3.2–5.5; dextrose and sodium chloride injection: 3.5–5.5). It is highly possible that the poor ability of dextrose and sodium chloride injection to preserve UC-MSCs is associated with its high osmotic pressure. In order to confirm the effects of pH and osmolarity, we made simple modifications to the storage media by increasing the pH values of 0.9% saline, 5% dextrose, and dextrose and sodium chloride injection to the physiologically relevant value of 7.2 and adjusting the osmotic pressure of dextrose and sodium chloride injection to 310 mOsm. However, the viability of the UC-MSCs was not noticeably increased by these modifications (data not shown). The detrimental storage effects of 5% dextrose can probably be attributed to the huge difference of glucose concentrations between 5% dextrose (50 g/L) and the culture medium (1 g/L). Although Pal et al. (30) reported that the best short-term storage solution for BM-MSCs was 5% dextrose, numerous studies substantiated that a high glucose concentration (lower than 50 g/L) affects the regenerative potential of MSCs (3) and induces the replicative senescence (20, 36). These data potentially explain our results with 5% dextrose.
Plasma-Lyte A, 1% HSA, and 5% HSA are typically used to prepare suspensions of MSCs, but their storage effects were also not favorable in our study. The poor viability of UC-MSCs in Plasma-Lyte A might be associated with a lack of ample nourishment for cell survival. The similar results between 0.9% saline and Plasma-Lyte A suggested that the reasons for the poor effects of 0.9% saline, 5% dextrose, and dextrose and sodium chloride injection may lie outside of their pH and osmolarity because the pH of Plasma-Lyte A is 7.4 and its osmolarity is in the physiological range (294 mOsm/L). For HSA, the presence of some vestigial substances during its preparation might contribute to its weak effectiveness (23). Similar results have been described by Lane (17).
Our results suggest that the survival of UC-MSCs stored in 0.9% saline, 5% dextrose, dextrose and sodium chloride injection, Plasma-Lyte A, 1% HSA, or 5% HSA is poor. In order to evaluate whether the biological functions of the viable UC-MSCs was affected by storage, we investigated their proliferation potential, immunosuppressive capacity, and differentiation capacity. The PDT of the stored UC-MSCs was slightly longer than unstored cells, and the proliferation potential of stored UM-MSCs recovered following one passage. The expression of surface markers in stored UC-MSCs and differentiation capacity in viable UC-MSCs were obviously not affected.
As previously shown, the viability of UC-MSCs was poorly maintained during 6 h of storage in our test solutions. Therefore, these solutions are not suitable for the preservation of UC-MSCs because the resulting viability is lower than that required by the FDA for clinical application. Although the proliferation, immunophenotype, immunosuppressive capacity, and multiple lineage differentiation properties of attached viable UC-MSCs were largely unaffected by storage, transplanted UC-MSCs may undergo significant cell death, which could significantly reduce the potential benefits provided by their transplantation. We propose that, until optimal storage conditions are identified, harvested UC-MSCs should be preserved at 4°C and be transplanted within 6 h. The identification of suitable storage solutions for the preservation of UC-MSCs is thus of great importance for cell transplantation.
Footnotes
Acknowledgments
We especially thank Cuiping Zhao for providing grammar aid on this manuscript. The authors declare no conflicts of interest.
