Abstract
Mesenchymal stem cells (MSCs) are one of a few stem cell types to be applied in clinical practice as therapeutic agents for immunomodulation and ischemic tissue repair. In addition to their multipotent differentiation potential, a strong paracrine capacity has been proposed as the principal mechanism that contributes to tissue repair. Apart from cytokine/chemokine secretion, MSCs also display a strong capacity for mitochondrial transfer and microvesicle (exosomes) secretion in response to injury with subsequent promotion of tissue regeneration. These unique properties of MSCs make them an invaluable cell type to repair damaged tissues/organs. Although MSCs offer great promise in the treatment of degenerative diseases and inflammatory disorders, there are still many challenges to overcome prior to their widespread clinical application. Particularly, their in-depth paracrine mechanisms remain a matter for debate and exploration. This review will highlight the discovery of the paracrine mechanism of MSCs, regulation of the paracrine biology of MSCs, important paracrine factors of MSCs in modulation of tissue repair, exosome and mitochondrial transfer for tissue repair, and the future perspective for MSC-based therapy.
Introduction
The initiation of stem cell research can be traced back to 1963 when Becker, McCulloch, and Till first identified stem cells in mouse bone marrow (12). A subsequent growing body of evidence that confirms the existence and function of stem cells now makes them the optimal source for tissue engineering and regenerative medicine. According to statistical data registered on www.ClinicalTrials.gov to date, stem cell-based therapeutic approaches now total 4,230 globally; the transition from laboratory bench to bedside has begun. Based on the technical feasibility, promising curative effects, reduced economic cost, and circumvention of ethical issues, mesenchymal stem cells (MSCs), which account for up to 300 cases of the 4,230 (7.0%, 300/4,230), have become the most common and effective cell source in cell-based treatment. The fascinating therapeutic effects of MSCs in various life-threatening human diseases, including cerebral spinal cord injury, hematological disorders, cardiovascular diseases, diabetes, immune diseases, graft versus host diseases (GvHDs), and cancer, are well documented. Nonetheless, the in-depth mechanisms of how MSCs act remain a matter for debate and exploration. The generally putative concepts cover transdifferentiation, cell fusion, paracrine effects, microvesicles carrying messenger RNA (mRNA) or microRNA (miRNA) and mitochondrial transfer (Fig. 1) (8,9,16,31,34,35,43,97,101,136). This review will focus on the paracrine effects of MSCs, the most comprehensive and enduring mode of action that ascribes to functional recovery in both acute and chronic responses.
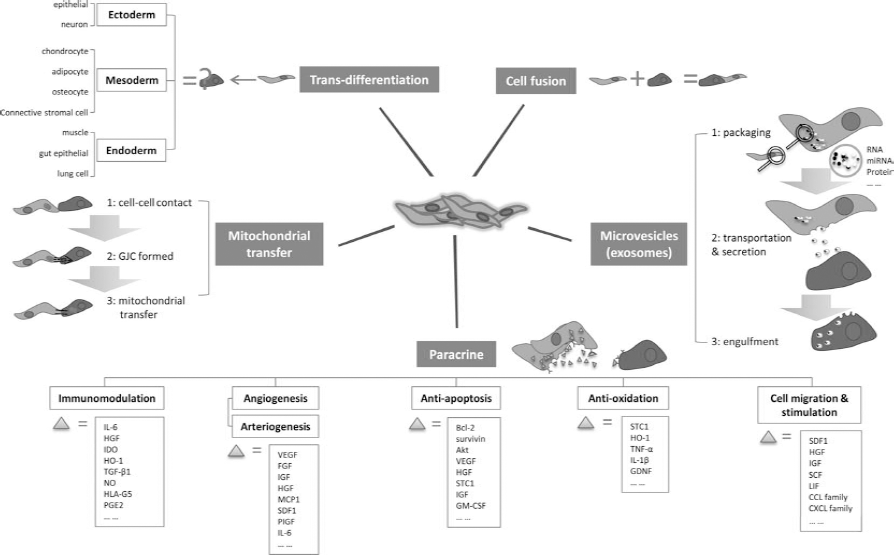
Action modes of MSCs. Transdifferentiation: Mesenchymal stem cells (MSCs) possess the ability to differentiate into another cell type, including ectoderm, mesoderm, and endoderm. Cell fusion: The MSC fuses with another cell to form a multinuclear cell known as syncytium. Mitochondrial transfer: The MSC makes contact with the adjacent cell, and a gap junctional channel (GJC) is built. The MSC transfers its mitochondria to the impaired cell through this GJC. Microvesicles: The MSC releases microvesicles containing RNA, microRNA (miRNA), and/or protein to the microenvironment. The cell nearby engulfs these microvesicles through the endocytosis process. Paracrine: The MSC secretes bioactive cytokines and chemokines that act on immunomodulation, angiogenesis/arteriogenesis, antiapoptosis, antioxidation, and cell migration/stimulation. Abbreviations: IL-6, interleukin-6; HGF, hepatocyte growth factor; IDO, indoleamine 2,3-dioxygenase; HO-1, heme oxygenase 1; TGF, transforming growth factor; NO, nitric oxide; HLA-G5, human leukocyte antigen class I molecule G5; PGE2, prostaglandin E2; VEGF, vascular endothelial growth factor; FGF, fibroblast growth factor; IGF, insulin-like growth factor; MCP1, monocyte chemotactic protein 1; SDF1, stromal cell-derived factor 1; PIGF, placental growth factor; IL-6, interleukin 6; Bcl-2, B-cell lymphoma 2; Akt, v-akt murine thymoma viral oncogene homolog 1; STC1, stanniocalcin 1; GM-CSF, granulocyte-macrophage colony-stimulating factor; TNF, tumor necrosis factor; GDNF, glial-derived neurotrophic factor; SCF, stem cell factor; LIF, leukemia inhibitory factor; CCL, chemokine C-C motif ligand; CXCL, chemokine C-X-C motif ligand.
Discovery of Paracrine Mechanism of MSCs
The secretion of cytoprotective factors by MSCs was first reported by Gnecchi and colleagues (34,35,57). The novel observation that modified MSCs overexpressing v-akt murine thymoma viral oncogene homolog 1/protein kinase B (Akt-MSCs) could prevent ventricular remodeling and reestablish heart function in less than 72 h following surgical myocardial infarction (MI) and cell transplantation raised the possibility of an action other than a myogenic pathway that would not be evident in such an extremely brief time period. Previous studies also pointed out that the limited frequency of the transplanted stem cell-derived cardiomyocytes (CMCs) was unlikely to be the main contributor to the marvelous amelioration of the ischemic organs (5,48,59,82). Thus, a new mechanism was proposed in which the injected MSCs might release trophic factors that contribute to myocardial protection following an ischemic insult. This hypothesis was then confirmed by evident improvement in cardiac performance following injection of conditioned medium (CM) collected from hypoxic Akt—MSCs (Akt—MSCs—CM) into an induced MI model. In vitro experiments also demonstrated that the hypoxic Akt—MSCs—CM could protect ventricular CMCs against apoptosis when subjected to a hypoxic challenge. Other valuable factors were also identified, including vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), hepatocyte growth factor (HGF), and thymosin β 4 (TB4), that were upregulated in the Akt—MSCs compared with the vector—MSCs. The paracrine effects of MSCs not only emerged as an original mechanism of action, but also inspired the future biological and clinical application of purified cytokines in ischemic injury. Kinnaird et al. (55) reported that growth of endothelial cells (ECs) and smooth muscle cells (SMCs) could be stimulated by the conditioned medium of MSCs (MSCs—CM) in a dosedependent manner. This phenomenon could be partly explained by the high level of VEGF and bFGF detected in the MSCs—CM. Nonetheless, neutralization using anti-VEGF and anti-bFGF antibodies could only partly impair this effect, suggesting other potential beneficial chemokines or cytokines needed to be determined (55). Subsequent studies located more salutary factors essential for remission of injury, which could be classified into five categories as follows.
Immunomodulation Factors
Bartholomew et al. first observed the immunomodulatory function of MSCs as evidenced by their dosedependent inhibitory effect on the cell proliferative response of allogeneic mitogen-stimulated lymphocytes in mixed lymphocyte culture (MLC) (11). The skin transplantation model also verified prolonged skin graft survival following intravenous (IV) administration of MSCs compared with a saline injection group. Contrary to this, Di Nicola's data demonstrated that this immunosuppressive feature was in part due to soluble factors as shown by the continued suppressive effect on the proliferation of T-lymphocytes in a Transwell system and excluded the possibility of cell—cell communication (21). The proliferation of T-cells could be sectionally restored with the addition of monoclonal antibody transforming growth factor-β1 (TGF-β1) or HGF, suggesting both cytokines were involved in the process. Inducible indoleamine 2,3-dioxygenase (IDO), an enzyme that catalyzes conversion from tryptophan to kynurenine and subdues the T-cell response to autoantigens and fetal alloantigens, was detected in MSCs stimulated by interferon-γ (IFN-γ) (77). With respect to IFN-γ-primed MSCs, another article proposed that IFN-γ played a crucial rule in the MSC—T lymphocyte interplay by upregulating the expression of B7 homolog 1 [B7-H1; also known as programmed death ligand 1 (PD-L1) or cluster of differentiation 274 (CD274)] on MSCs, a known coinhibitor molecule of the immune response (109). Selmani et al. discovered that human leukocyte antigen class I molecule G5 (HLA-G5) secreted by MSCs, possessing the ability to contact with allo-stimulated T-cells, was responsible for the immunosuppressive functions of MSCs on T-lymphocyte and NK (natural killer) cells, as well as the expansion of the inducible CD4-positive CD25 high-expressing forkhead box P3-positive (CD4+CD25highFOXP3+) regulatory T-cells (107). Apart from the influence on lymphocytes, the bioactive components of MSCs were shown to facilitate the transition of macrophages from the proinflammation phenotype M1 (classical-activated macrophage) to the anti-inflammation phenotype M2 (alternative-activated macrophage). This was proposed as one of the mechanisms that occur in the early stage of tissue deterioration (2,18). There are three major stages of the immune response: 1) antigen recognition and presentation; 2) T-cell activation, proliferation, and differentiation; 3) effective stage. The immunosuppressive privilege of MSCs not only exists during the second stage on T-cells but also modulates the very first step by interacting with antigen-presenting cells (APCs). Jiang et al. presented evidence that MSCs inhibited the differentiation, maturation, and function of dendritic cells (DCs) derived from CD14+ monocytes (46). The mature DCs that underwent MSC coculture showed reduced expression of CD83 and CD1a, both of which are markers of DC maturity. The costimulative molecules such as CD80 and CD86, as well as secreted IFN-γ, and interleukin 12 (IL-12) were also downregulated by MSC treatment, thus rendering T-cells anergic downward. To distinguish cell—cell contact from cytokine secretion, they set up a Transwell system with different MSC/monocyte ratios. The MSCs plated in the lower compartment were able to fully prevent monocytes from differentiation and maturation at a high MSC/monocyte ratio (1:10). It can thus be concluded that MSCs play the part of regulator throughout the immune response, including different phases, various cell types, and diverse modes of action.
Most recently, MSCs were found to act as immune modulators, rather like a double-edged sword. Li et al. proposed that the degree of nitric oxide (NO) production elicited by proinflammatory cytokines in the surrounding environment could influence MSCs to be either potently immunosuppressive or highly immune enhancing (66). While inducible nitric oxide synthase (iNOS) production was blocked, the immunosuppressive properties of MSCs diminished and reverted to promote T-cell proliferation, as well as the delayed-type hypersensitivity response by their chemotactic effect on immunocytes. This immunoenhancing effect of MSCs might be attributed to chemokines such as chemokine C-X-C motif receptor 3 (CXCR3) and chemokine C-C motif receptor 5 (CCR5) derived from themselves. To some extent, this gained support from the evidence that iNOS-/- MSCs failed to promote proliferation of CXCR3-/-CCR5-/- splenocytes. These results highlight the importance of evaluating inflammation status and intervention with regard to iNOS/IDO levels before MSCs can be broadly applied for immunological disease in both rodent models and human clinical trials. Other than suboptimal timing and dose of administered MSCs, it might partially explain the reasons why the manipulation of MSCs in GvHD showed mixed results, with some indicating an immunosuppressive potential and others not (22,113).
Angiogenic and Arteriogenic Factors
It has been proven that angiogenic and arteriogenic support accounts for amelioration of coronary artery disease (CAD) following bone marrow cell transplantation (89,90). Angiogenesis is evidenced by formation of a new blood network from the preexisting capillaries by sprouting and proliferation, while arteriogenesis is demonstrated by the collateral enlargement and muscularization of small arterioles to form larger arteries (28). Angiogenesis is tightly regulated by a competitive balance of the angiopoietins and inhibitors, known as “angiogenic switch” (88). Only when the increasing cytokines and cell adhesion receptors that impel neovascularization reach a certain level and concentration in the locoregional environment can they incrementally initialize the process of angiogenesis. It has been reported that the hypoxia-inducible factor 1 (HIF-1) signaling pathway can switch on this angiogenesis process in ischemic disease (123). HIF-1 is a nucleoprotein with transcription properties that regulates the expression of a variety of target genes with the ability to acclimatize and promote cell survival in an oxygen-deficient environment, including glycolytic enzymes that provide adenosine triphosphate (ATP) for cell metabolism, and proangiogenetic factors such as VEGF, FGF, NO, and insulin-like growth factor (IGF) (29,44,76,100). It is only under hypoxic conditions that HIF-1 can be stabilized and activate expression of the downstream proteins. The transcriptional activation mediated by HIF-1 results in an escalating concentration of the biological cytokines that trigger the vascular endothelial cells to proliferate, sprout, migrate, and infiltrate to develop new vasoganglion. Accompanied by pericytes, as well as optimal differentiation and apoptosis of the endothelial cells, the newborn vessels begin to form lumens. It is this physiological mechanism that led to the initiative to utilize MSCs for the treatment of ischemia: they express and produce VEGF, HGF, monocyte chemotactic protein 1 (MCP1; chemokine C-C motif ligand 2), and stromal cell-derived factor 1 (SDF1) that are critical for vascular network remodeling (54,55). Several studies attempting to explore the potential of MSCs for vascular regeneration have consistently reported increasing capillary density and better collateral perfusion following MSC manipulation, although whether the secretion of cytokines represents the paramount mechanism of action remains under dispute (41,50,62,79,83,108,110). In particular, a recent study by Dong et al. concerning the SDF1/CXCR4 axis revealed that CXCR4 expression in CMCs was a necessity for the trophic effects generated by MSCs following left anterior descending artery (LAD) ligation (23). The researchers injected MSCs into wild-type mice or conditional CMC—CXCR4 knockout mice post-MI induction. They observed an increased number of CMCs undergoing apoptosis and decreased cardiac progenitor cell (CPC) recruitment in the absence of CMC—CXCR4 expression, leading to depletion of MSC-mediated functional restoration. This was despite equivalent levels of implanted MSC infusion and increases in capillary density. It is reasonable to conclude from these results that the protective and restorative benefits mediated by MSCs are partially due to angiogenesis factors that lead to increasing vascular density and recovery of blood supply in the ischemic area. Nonetheless, the importance of coordination of the hibernating or susceptible cells in the vicinity cannot be underestimated.
Antiapoptotic Factors
To prevent programmed cell death, MSCs not only restore the microhemodynamics but also synthesize and secrete proteins that are classic inhibitors of apoptosis, such as B-cell lymphoma 2 (Bcl-2), survivin, and Akt (86,125). The ratio of Bcl-2 to Bcl-2-associated X protein (Bax) determines the sensitivity of the cells to a pathological stimulus (87). The predominantly expressed Bcl-2 will prevent the release of caspase activators; thus, cells are less likely to respond to the apoptotic signaling and vice versa (36). Tang et al. detected downregulated Bax expression in the ischemic myocardium following autologous MSC transplantation (117). Pan et al. showed lower expression of Bax, TNF receptor superfamily, member 6 (FAS), and caspase 3 (CASP3) at both a transcriptional and translational level in MSC—CM-treated LO2s (a human normal liver cell line) subjected to H2O2 challenge, a laboratory setting intended to mimic ischemic—reperfusion (IR) injury in the liver, resulting in protection of hepatocytes against apoptosis (93). It is also worth noting that as well as the ability of MSCs to synthesize proteins that directly repress apoptosis, they are also able to secrete cytokines that either neutralize the apoptotic pathway or enhance survival. Gerber et al. established that VEGF could prevent serum starvation-induced apoptosis by upregulating Bcl-2 expression in vascular endothelial cells (33). VEGF also participated in an antiapoptotic process by phosphorylated activation of focal adhesion kinase (FAK), a critical prosurvival signal that acts by suppressing p53-mediated apoptosis (42,72,73). The bioactive molecules against apoptosis secreted by MSCs not only affect nearby cells but also fulfill their own mission. Studies by Wang and colleagues showed that hypoxic preconditioning repressed the apoptotic index of MSCs by stabilizing mitochondrial membrane potential and elevating the secretion of VEGF and Bcl-2 (124).
Antioxidative Factors
The concept of oxidative stress (OS) was derived from the free radical theory of Sohal et al. in their research on aging and life span (91,111). OS is initiated by the disequilibrium of oxidation and antioxidation in response to physiochemical or physiological stimuli. When an oxidative reaction prevails, the following process should occur: infiltration of inflammatory cells, release of protease, and accumulation of oxidative by-products referred to as ROS (reactive oxygen species), including oxygen ions, oxygen free radicals, and peroxides. Insufficient or surplus ROS may give rise to pathogenesis known as ROS-related diseases, such as carcinogenesis, immune disorders, inflammation, neurodegeneration, or angiocardiopathy (27,47,49,74). Studies by Ohkouchi and others highlighted the protective function of MSC-derived stanniocalcin 1 (STC1) by reducing ROS-related apoptosis (85). STC1 also featured in another article with regard to its antioxidative activity in CMCs (69). This indicates that the successful harvest of MSCs in heart disease was mediated partially by releasing STC1. Another study described xenogenic MSC engraftment that ameliorated the redox environment in a lipopolysaccharide (LPS)-induced acute lung injury (ALI) model by enhancing expression of antioxidative enzyme heme oxygenase-1 (HO-1) and reducing expression of malondialdehyde (MDA), an indicator of lipid peroxidation (64). The enriched concentration of proinflammatory cytokines tumor necrosis factor-α (TNF-α), IL-1β, and IL-6, but not IL-10, existed in the MSC-treated group and may play a role in this modulatory activity. Studies by Zarjou et al. supported the importance of HO-1 by the use of HO-1-/- MSCs, in which less effective production of SDF1, VEGF, and HGF was detected (134). With respect to IR injury, MSCs were beneficial; they encouraged expression of HO-1 in renal IR induced by cisplatin or surgery (71,135). Another group who studied neurodegenerative dysfunction reported a similar “shield/barrier” effect conferred by MSCs on monoaminergic perikarya and monoamine neurotransmitter transporter function exposed to nitric oxide (NO)-induced OS. They identified another trophic factor, glial-derived neurotrophic factor (GDNF) (127). The operative molecules secreted by MSCs vary in different experimental settings, probably due to nonuniform ROS inductors leading to different levels of ROS production. Nonetheless, it is evident that MSC therapy has an extensive influence on the redox context due to these antioxidative factors.
Cell Migration, Homing/Targeting, and Stimulation
Several studies using different cell tracking approaches have revealed that following IV administration, MSCs are widely distributed to nonhematopoietic tissues, including the gastrointestinal tract, kidney, skin, lung, thymus, and liver. This suggests that MSCs patrol the body until triggered by what might be metaphorically compared to “criminal behavior” (19,20). Indirect evidence supporting this hypothetical notion has been well documented in various disease models, in which MSCs showed faster mobilization and better retention at sites of injury following systemic or local intratissue infusion (10,45,52,95). The inherent tumor-trophic migratory properties of MSCs have been employed as gene/drug carriers to deliver therapeutic, effective, targeted therapy to carcinomas and metastatic diseases (129). Thus, investigations oriented toward the motivation of this migration behavior proliferated. The intention was to maximize the therapeutic potential of MSCs by establishing a more efficient platform of stem cell homing and/or targeting. Several pathways or attractants have been discovered. Kitaori et al. demonstrated that inducible SDF1 expression in the periosteum of a live bone graft was required to recruit MSCs for endochondral bone repair (56). By neutralizing SDF1 with an anti-SDF1 antibody or antagonizing CXCR4 with TF14016, new bone formation was significantly reduced following IV injection of MSCs. This suggests the involvement of the SDF1/CXCR4 axis in MSC-mediated tissue repair and regeneration. Under hypoxic conditions, MSCs exhibited enhanced mobility toward SDF1 in a concentration-dependent manner, accompanied by elevated cellular CXCR4 expression (134). In accordance with the in vitro experiments, the in vivo study showed increased MSC retention in the infarct region 5 days after MI, when ischemic-related SDF1 reached the fastigium. When cells were pretreated with LY294002 [a reagent that selectively inhibits phosphoinositide 3-kinase (PI3K)/Akt], a smaller number of implanted cells targeted the injured area with enlargement of the infarcted fibrotic area compared with nontreated cells. This indicated that the PI3K/Akt pathway was involved in the chemotactic response of MSCs to the SDF1/CXCR4 axis. In a separate but similar experiment, the LAD occlusion-induced SDF1 expression in the infarcted myocardium resulted in accumulation of bone marrow-derived cells delivered by IV injection (1). This time, the author employed AMD3100, an antagonist of CXCR4, to confirm the instrumental role of the SDF1/CXCR4 signaling pathway in regard to stem cell homing. Another noteworthy finding in this study was that SDF1 alone could not trigger the grafted cells to migrate, indicating the requirement for another concomitant secretion or pathway stimulation. In addition, SDF1 serves as a chemoattractant signal to the endogenous CXCR4+ stem cells. Otsuru's paper stated that the expression of SDF1 in vascular endothelial cells promoted migration of CXCR4+ bone marrow-derived osteoblast progenitor cells from circulating blood to the region of osteogenesis (92). In another study, Tang et al. reported that CM of genetically modified MSCs with VEGF expression that contained more SDF1 achieved better left ventricular performance than unmodified MSCs by massive mobilization and homing of bone marrow stem cells and cardiac stem cells (116). While mounting evidence has demonstrated that upregulation of SDF1 is a required, though not unique, signal for better localization of stem cell targeting, other studies focused on the other side of this axis, that is, CXCR4. Contrary to expectations, forced expression of CXCR4 into the myocardium by direct gene transfer prior to MI surgery resulted in increased infiltration of inflammatory cells and apoptosis of CMCs (14). If the manipulation of CMC—CXCR4 is not as beneficial as predicted, it is likely that CMC—CXCR4 expression serves as an indication of CMCs that are in a self-protective state with reduced energy consumption, or even hibernating as a means of surviving in the ischemic episode. The necessity of myocardial CXCR4 expression was analyzed recently using a conditional CMC—CXCR4 null mouse model (23). In the CMC—CXCR4 null mice, increased numbers of CMCs underwent programmed cell death in the border zone following MI plus stem cell therapy. This phenomenon diminished in the absence of stem cell infusion. In addition, far more CPCs accumulated and localized in response to MSCs in the wild-type mice compared with CMC—CXCR4 null mice. Consequent to this, the concept was proposed that CMC—CXCR4 expression is required in MSC-mediated cytoprotection and CPC recruitment. Based on these data, it is possible to decipher a reciprocal relationship between the cells, either endogenous or exogenous, and surrounding milieu regarding SDF1/CXCR4 binding in the presence of MI: the increasing MI-induced SDF1 in the infarct region helps to attract endogenous CXCR4+ stem cells, in addition to migration of exogenous MSCs to the lesion as a result of the increasing concentration of SDF1; the localized and stabilized distribution of stem cells then augments the SDF1 signal by synthesizing and releasing more SDF1; this process works as an amplifying hierarchy, within which CMC—CXCR4 expression is required for the implanted MSCs to recruit cardiac stem cells.
In addition to chemotactic-like properties, MSCs also help revitalize endogenous stem cells to accumulate and proliferate. Using the in situ chromosome Y FISH technique, Lin et al. distinguished male donor bone marrow cells from female host cells and established that bone marrow cells accounted for only 11% of the proliferating epithelial cells in a kidney IR model. This suggests a regenerative capacity generated by the inherent hibernating stem via revitalization or rejuvenation (68). In the pig MI model, c-kit (v-kit Hardy—Zuckerman 4 feline sarcoma viral oncogene homolog) and Ki-67 (antigen identified by monoclonal antibody Ki-67) were detected at the peri-infarct region in the MSC-treated group, implying endogenous stem cell recruitment and reentry of CMCs to the cell cycle and duplicate status (6). MSCs—CM were as able as MSCs to mobilize cardiac stem cells in the first 3 days, suggesting that the paracrine signaling accounted for part of the stem cell homing effects in the acute phase, though not comparable with what could be achieved from direct MSC delivery in long-term results (37). In addition to SDF1, other members of the MSC secretion pool, such as HGF and IGF, were also shown to enhance mobilization and proliferation of cardiac stem cells (121).
Regulation of Paracrine Biology of MSCs
Gnecchi's group reported approximately 650 transcripts that expressed differentially between Akt—MSCs and vector—MSCs (70). This suggests that genetic modification of MSCs by overexpression of one pivotal gene that acts as a “switch” might alter the profile of the released factors and thus maximize the therapeutic potential. Intense efforts have been made to hone MSCs and make them more effective and efficient. The paracrine effects should hold most appeal because of the wish to have an extensive, stable, and lasting mode of action.
Preconditioning of MSCs
Gnecchi et al. demonstrated that hypoxic treatment of the Akt—MSCs facilitated release of trophic factors such as VEGF, bFGF, HGF, IGF, and TB4 (35). The hypoxic Akt—MSCs—CM provided cytoprotection and induced spontaneous contraction of the adult rat ventricular cardiomyocytes (ARVCs) when exposed to prolonged hypoxia. Other groups compared the secretome in normal and hypoxic conditions: most suggested that the contents of the concentrated medium of MSCs cultured in a hypoxic environment were superior. Chang et al. reported that both HGF and VEGF were elevated in the MSCs maintained in 0.5% oxygen for 24 h (13). They then concentrated the medium and injected it into the rat traumatic brain injury (TBI) model every 12 h consecutively for 3 days, with consequent findings of increasing neurogenesis as well as alleviation of motor and cognitive dysfunction. Another group highlighted the fact that under normal physiological conditions, MSCs originate in areas of low oxygen concentration such as bone marrow. They thus need to accommodate themselves to the higher oxygenation that is present when they are isolated and expanded ex vivo (21% oxygen) (104). They then need to immediately reacclimatize to a hypoxic state when applied as a cell source for ischemic disease models. Subjecting MSCs to a premature hypoxic period may allow them a “buffering period” and thus improve their tissue regenerative potential. In this project, it was shown that preculturing MSCs in a low-oxygen environment activated the Akt and met proto-oncogene (cMET, receptor of HGF) signaling pathway, contributing to enhanced neovascularization and stem cell homing in a surgical hindlimb ischemia model. Another study by Leroux et al. also went some way to confirm that hypoxic preconditioning of MSCs promoted their survival capacity, and vascular and tissue reconstruction, albeit via a wingless-related mouse mammary tumor virus (MMTV) integration family, member 4 (Wnt4)-dependent pathway (63).
In addition to physiological preconditioning, another feasible method may be cytokines/chemicals. Application of cytokine/chemicals by three different ways improved the therapeutic efficiency of MSCs: administration of cytokines/chemicals to MSCs prior to transplantation, aimed at optimizing the secretome with better migration toward the injured tissue; pretreatment at the site of injury, with the objective of attracting more stem cells for tissue repair; or simultaneous injection of cytokines/chemicals and MSCs. As an example, Luo et al. pretreated MSCs with IL-1β and TGF-β, and showed synergistic advantages of this combination on VEGF production, as well as functional restoration post-MI (75). Pasha et al. showed SDF-1-pretreated MSCs suppressed cell apoptosis, enhanced engraftment increased vascular density, and improved myocardial function via SDF/CXCR4 signaling (94). Cui et al. indirectly upregulated SDF1/CXCR4 expression with a nitric oxide donor, diethylenetriamine (DETA)-NONOate (17). They demonstrated that preconditioning of the animal with DETA-NONOate 24 h following middle cerebral artery occlusion promoted MSC engraftment. In Pons's study, VEGF and MSCs were coinjected to MI hearts, which then showed the expected improvement in stem cell mobilization and cardiac function (99).
Genetic Modification of MSCs
Their ability to migrate toward damaged tissue makes MSCs the optimal vector for therapeutic agents. Gnecchi et al. not only defined the paracrine mechanism but also demonstrated that genetically engineered MSCs with Akt expression were superior to vector—MSCs in many aspects (34,35). Since then, many attempts have been made to equip MSCs with curative genes to aid functional recovery. Li et al. engineered MSCs with Bcl-2, and the modified cells presented better apoptotic tolerance, cell survival, and more VEGF secretion than control MSCs (65). Other genetically engineered MSCs, including those for survivin, SDF1, CXCR4, HGF, PI3K, and IGF, showed similar results of improving angiogenesis, left ventricular ejection fraction (LVEF), endogenous stem cell recruitment, contractile function, and reduction of LV remodeling effects (24,25,58,112,115,128). Most of these selected genes are considered to be trophic factors secreted by physiological MSCs or possess the ability to activate Akt signaling directly or indirectly, based on maintaining the fundamental characteristics of MSCs and safety concern. Genetic manipulation could improve cell survival and control the MSC secretome by engineering the gene of interest; nonetheless, overexpression could bring unexpected effects, either from the delivering approach or the targeting genes. For example, overexpressing bFGF or platelet-derived growth factor-β (PDGF-β) led to highly proliferating MSCs and increases in osteogenesis, while upregulation of TGF-β1 hindered both osteogenic and adipogenic differentiation (26). It should nonetheless be noted that while the genetically modified MSCs showed enhanced therapeutic efficacy, a very stringent evaluation of their physiological characteristics with regard to safety and undesired effects is required prior to their application in clinical trials.
Important Paracrine Factors of MSCs in Modulation of Tissue Repairs
Paracrine Factor-Activated MSCs in Immunomodulation
Mesenchymal stem cells have the ability to suppress the immune response and can even induce immune tolerance in certain conditions through cell—cell contact and soluble factors. An increasing number of studies have suggested that the soluble factors of MSCs are a key requirement of their immune regulatory properties (30,114,130). Liu et al. demonstrated that the immunogenicity of MSCs was heightened during the increasing passage number and could not suppress lymphocyte proliferation in vitro (70). Nonetheless, soluble factors collected from the coculture of MSCs and lymphocytes could suppress lymphocyte proliferation without the need for cell—cell contact. Ren et al. found that the proinflammatory cytokine-activated MSCs could secrete chemokines to recruit lymphocytes (102). The MSCs concentrated their action on the localized lymphocytes by secreting TGF-β and NO with two effects: inhibition of proliferation and promotion of apoptosis. Yang et al. reported that soluble factors from the culture supernatant of MSCs could suppress T-cell proliferation, in which IL-10 and IDO played important roles (132). TGF-β, HGF, prostaglandin E2 (PGE2), HLA-G5, IL-6, CCL2, CCL5, and other chemokines have also shown that the paracrine mechanism of MSCs can modulate regulatory T-cells (Tregs) and the immune response (39,84,105,118).
Polchert et al. found that IFN-γ-pretreated MSCs reduced GvHD more efficiently than the MSCs without IFN-γ pretreatment (98). It has been suggested that the immunomodulatory ability of MSCs is activated by inflammatory factors secreted by lymphocytes. In the acute host versus graft reaction (HvGR) and graft versus host reaction (GvHR), the activated Th1 cell releases cytokines such as IL-2 and IFN-γ to promote lymphocyte proliferation and amplify the immune response. Alternately, these proinflammatory cytokines also revitalize the immunosuppressive properties of MSCs, which release soluble factors in turn to suppress HvGR and GvHR.
MSCs have demonstrated their practical application in terms of HvGR and GvHR in vivo. Aksu et al. reported that autologous MSCs could limit the toxicity of allogeneic bone marrow transplantation and delay GvHD onset when coinfused four times with unmodified donor bone marrow (3). Ge et al. found that infusion of MSCs with rapamycin achieved long-term cardiac allograft survival by inducing Tregs that could suppress the acute rejection response and induce immune tolerance (32). Other studies have demonstrated that soluble factors such as TGF-β can promote the viability of regulatory cells, indicating an indirect pattern to modulate the immune response (118,131). Despite these advances, the use of the paracrine effects of activated MSCs to treat organ transplantation and GvHD remains in its infancy, and further animal and human studies are required.
Paracrine Factors and Cardiovascular Diseases
MSCs have emerged as a very promising cell type for the treatment of cardiovascular disease both in small animal studies and large animal models (15,53,67, 78, 96,106,120,133). The exact extent to which these cells form new cardiac myocytes and improve cardiac function nonetheless remains highly controversial (7,81). The disproportion between tremendous functional recovery and low rates of cell engraftment and persistence suggests an indirect primary mechanism other than structural integration of transplanted cells into ischemic myocardium. Thus, the ability of MSCs to produce a variety of trophic and immunomodulatory factors that can directly promote cell survival and reduce inflammation posttransplantation has attracted great attention (7,34,81,119). While Gnecchi and Kinnaird have already focused on the car-dioprotective properties of MSCs via a paracrine effect (34,35,54,55,57,70,79,80), the current focus is to locate the most efficient factors with high tissue specificity or optimal design with synergistic effects to maximize myocardial survival postinfarction. For instance, Gnecchi's research identified secreted frizzled-related protein 2 (Sfrp2) as the most dramatically upregulated protein (100-fold in transcriptional level) in the Akt—MSCs (70). As an extension of this research, He and others confirmed the tissue-regenerative capacity of Sfrp2 by simple administration to the infarcted areas (4,38,80). Zhang et al. proposed that combined treatment with Wnt11 and bone morphogenetic protein 2 (BMP-2) increased the cardiomyogenic potential of MSCs and raised the possibility that preconditioning MSCs with inducible factors might augment the transdifferentiation odds (137). Indeed, the paracrine mechanism of MSCs facilitates the discovery and discrimination of the most powerful soluble factors and, above all, makes possible the broader future application and conversion of these factors into novel therapeutic applications for clinical off-the-shelf therapy. Kanki et al. showed intracoronary injection of SDF1 could improve ventricular function in experimental ischemia/reperfusion injury even 3 h after the onset of ischemia (51). In a clinical trial of 178 patients, high-dose VEGF administration improved treadmill tests, angina class, and quality of life assessments at day 120 (40). Nonetheless, not all the beneficial cytokines identified from the MSCs—CM achieved satisfactory consequences as expected. For example, granulocyte-colony-stimulating factor (G-CSF) alone did not yield any encouraging alteration in MI patients, despite its definite safety and feasibility (103,122). The reasons for this contradiction might be varied: the dose, timing, and delivery method need to be optimized; the combination of cytokines at different concentrations and time points might heighten the synergistic effects. Though these issues still wait to be addressed, it is worth noting that non-cell-based alternative cytokine delivery does circumvent the major drawbacks such as quantification control, immunological rejection, concerns about infection and carcinogenesis, compared with direct stem cell injection. It also facilitates the establishment and optimization of curative standard.
Exosome and Mitochondrial Transfer for Tissue Repair
Timmers and colleagues demonstrated that IV and intracoronary injection of MSC—CM significantly restored ventricular performance in a porcine model of IR injury (119). Of note, only the CM-containing products >1,000 kDa (100-220 nm) contributed to the cytoprotective effects, implying that the paracrine signaling might function as a large complex rather than a single small molecule. Coimmunoprecipitation by ultracentrifugation identified that these particles contain plasma membrane phospholipids such as cholesterol, sphingomyelin, and phosphatidylcholine, as well as exosome-associated proteins CD81, CD9, and apoptosis-linked gene 2-interacting protein X (Alix). Electron microscopic examination of the size and morphology revealed that this group of bioactive particles released by MSCs were exosomes (60). The purified exosomes at a dose of 0.4 μg were then delivered to an IR injury 5 min prior to reperfusion and showed comparable capacity in reducing infarct size to that of a 3.0-μg dose of MSC—CM. Proteomics, transcriptomics, miRNA array, or other high-throughput technologies are practical to exploit the content in the vesicles. With proteomic profiling using mass spectrometry and antibody array, Lai et al. reported 857 proteins in the exosome proteome, in which 20S proteasome was determined as the main contributor to cardioprotection by reducing the amount of misfolded proteins during acute MI (61). It appears that MSCs are working in a more intelligent and efficient way than we expect by encapsulating functional proteins or regulatory RNAs into exosomes, via whose phospholipid envelop a rapid intracellular delivery pathway is created, thus allowing MSCs to mount an early response to stimuli.
Contrary to this, the most recent work by Islam et al. provided in vivo evidence of mitochondrial transfer from MSCs to host cells in the ALI model induced by LPS instillation (43). With live imaging, they observed instilled MSCs attached to the alveoli and intercellular dye exchange began, suggesting the involvement of gap junctional channels (GJCs). Fluorescence recovery after photobleaching (FRAP) occurred in alveolus-attached MSCs, affirming the existence of GJCs. The FRAP could be blocked by a nonspecific GJC blocker or the specific connexin43 (Cx43) inhibitor, providing further reverse proof of the involvement of GJCs and Cx43 in cell—cell contact between MSCs and the alveolar epithelium. The expression of GJC-competent Cx43 was indispensable to mediate a successful mitochondrial transfer, supported by the evidence that either mutant Cx43 or ablation of Cx43 led to failure of GJC formation. The internalized mitochondria acted functionally by increasing the ATP concentration in the recipient pulmonary alveoli, leading to bioenergetic restoration and protection in the acute phase of lung injury. Nevertheless, this successful delivery also compromised the energy of MSCs with a resultant decrease in other cell behaviors that benefit the surrounding or remote areas, such as secretion capability, migration ability, and exosome packaging. It was clear that the MSCs played multiple roles, trying to repair or normalize the tissue during disease emergencies, though the combined or neutralized effects and the proportions of each substation among these different modes of action require further investigation.
Perspective
It is undisputable that MSC therapy contributes to restoration of structural integrity and functionality of damaged tissue, but their fundamental and detailed biological mechanisms require further elucidation. The many effects of MSCs, including transdifferentiation, cell fusion, paracrine, exosome secretion, and mitochondrial transfer, each have drawbacks that need to be addressed before maximal benefit is obtained: transdifferentiation and cell fusion seem to occur in too low a frequency to account for the meaningful improvement; exosome secretion and mitochondrial transfer faces the problem of finding a robust and scalable cell source with sufficient quantity and quality to generate exosome encapsulation and energy transportation. With regard to paracrine actions, the limitations must not be overlooked. For instance, some cytokines or chemokines released from MSCs may be harmful, such as TNF-α and IL-6. This may explain the modest benefit of MSC transplantation observed in clinical trials (126). Nonetheless, mounting evidence suggests that the secretion profile can be readily improved by preconditioning or genetic manipulation. Furthermore, the paracrine action provides the possibility to apply one trophic factor alone or combined as a cocktail therapy for disease-oriented treatment. The advantage of MSC-based therapy is that it can maintain a sustainable moderate release and concentration of the trophic factors that might be varied according to diverse microenvironments and situations. It seems that these cells are working smartly and systematically, to adapt themselves in harsh disease conditions, with the aim of restoring physiological status. Thus, the paracrine effects of MSCs hold great promise as a controllable, manageable, and feasible route, by which the transition from bench to bedside becomes more feasible in the near future.
Footnotes
Acknowledgments
This study was partly supported by grants from the GRF/RGC (HKU 722510M to Q. Lian), National Natural Science Grant of China (81170896), and small project funding from The University of Hong Kong (201111159183 to Q. Lian). The authors declare no conflicts of interest.
