Abstract
Kidney disease (KD) is a life-threatening disease characterized by high morbidity and mortality in clinical settings, which can be caused by many reasons, and the incidence increases with age. However, supportive therapy and kidney transplantation still have limitations in alleviating KD progression. Recently, mesenchymal stem cells (MSCs) have shown great potential in repairing injury through their multidirectional differentiation and self-renewal ability. Of note, MSCs serve as a safe and effective therapeutic strategy for treating KD in preclinical and clinical trials. Functionally, MSCs ameliorate KD progression by regulating the immune response, renal tubular cell apoptosis, tubular epithelial–mesenchymal transition, oxidative stress, angiogenesis, and so on. In addition, MSCs exhibit remarkable efficacy in both acute kidney injury (AKI) and chronic kidney disease (CKD) through paracrine mechanisms. In this review, we outline the biological characteristics of MSCs, discuss the efficacy and mechanisms of MSCs-based therapy for KD, summarize the completed and ongoing clinical trials, as well as analyze limitations and new strategies, aiming to provide new ideas and approaches for the preclinical experiments and clinical trials of MSCs transplantation for KD.
Introduction
Kidney disease (KD) is a global public health problem that affects more than 750 million of the global population and causes 5 to 10 million deaths each year1,2, with the main common diseases being acute kidney injury (AKI) and chronic kidney disease (CKD). Previous studies have shown that the development and progression of KD were associated with obesity 3 , diabetes 4 , hepatitis B virus infection 5 , and so on. Meanwhile, KD progression is a risk factor for cardiovascular diseases 6 . In addition, clinical studies have found that the high incidence and poor prognosis of KD were relevant to clinical care and huge economic costs 7 , with approximately $10 billion spent annually on treating AKI 8 and more than $80 billion spent on caring for CKD without kidney replacement therapy 9 in the United States. Currently, the common therapeutic methods for KD include drugs, hemodialysis and peritoneal dialysis, and renal transplantation 10 . However, the expected efficacy was still not achieved due to the irreversibility of kidney injury, the toxic effects of drug therapy, the inconvenience of dialysis, and the shortage and high cost of kidney transplant donors 11 . Therefore, there is an urgent need to explore new therapeutic strategies to slow down the progression of KD.
In recent years, stem cells have been used as a new regenerative therapy for a variety of diseases, including KD 12 . Mesenchymal stem cells (MSCs), as one of the important members of the stem cell family, can be obtained from a variety of tissues such as bone marrow, adipose, umbilical cord, and peripheral blood and have powerful biological properties of immunomodulation, anti-inflammation, and tissue repair 13 . Preclinical and clinical trials have shown that MSCs possess reparative and protective effects on kidney injury14,15. Functionally, MSCs exert anti-apoptotic, antioxidant, anti-inflammatory, anti-fibrotic, and immunomodulatory activities through secreting trophic factors and delivering extracellular vesicles (EVs)16,17. Overall, MSCs are considered to be the most promising stem cell population for the treatment of KD.
In this review, we will focus on the current research advances and challenges in the use of MSCs for the treatment of KD. Preliminarily, this review provides an overview of the origin and biological properties of MSCs. Subsequently, we summarize the underlying mechanisms and clinical translational applications of MSCs in the treatment of KD. Finally, we analyzed the obstacles encountered in the use of MSCs for the treatment of KD and proposed corresponding strategies to cope with the limitations of MSCs in KD. Of note, this review aims to provide new ideas and directions for the treatment of KD with MSCs in preclinical experiments and clinical trials.
Biological Characteristics of MSCs
Mesenchymal stem cells are a class of adult stem cells with self-renewal and differentiation potential 18 . They have become a new means of treating KD because of their multidirectional differentiation potential, high proliferative capacity, immune regulation, and self-replication 19 . They can be obtained from a variety of tissues, including bone marrow, adipose tissue, umbilical cord, placenta, amniotic fluid, and dental pulp 20 (Fig. 1). Although MSCs are derived from different sources [eg, bone marrow mesenchymal stem cells (BMMSCs), adipose-derived mesenchymal stem cells (ADMSCs), and umbilical cord mesenchymal stem cells (UCMSCs)], they have similar differentiation and biological functions 21 . Numerous studies have confirmed that MSCs can differentiate a variety of cells (eg, osteoblasts, myoblasts, cardiomyocytes, and renal parenchymal cells) with different functional characteristics under different induction conditions 22 . Previous studies have shown that MSCs regulated immune activity and enhanced the expansion and differentiation potential of host cells through direct cell–cell contact or paracrine mechanisms, thus promoting the recovery of injured tissues 23 . Importantly, preclinical experiments and clinical trials have confirmed that treatment with MSCs significantly improved the progression of KD24,25. Functionally, the main regulatory roles of MSCs are as follows: (1) The recruited MSCs differentiate into functional cells to replace damaged cells, and (2) as a response to inflammatory cytokines, MSCs produce large amounts of cytokines, chemokines, growth factors, and exosomes, which stimulate angiogenesis, prevent cell apoptosis and epithelial–mesenchymal transformation (EMT) process, block oxidative stress, promote extracellular matrix (ECM) remodeling, and induce differentiation of tissue stem cells 26 . In recent years, several studies have proved that MSCs-secreted exosomes (MSCs-Exo) via paracrine mechanisms not only have the same effects as MSCs but also have the advantages of low transplantation risk, easy storage management, high controllability, low immunogenicity, high safety, high repairability, and so on 27 . The above studies suggest that MSCs exhibit great therapeutic effects on injury repair and immune-characterized diseases, and serve as shining stars of stem cells in the field of cell therapy and regenerative medicine.
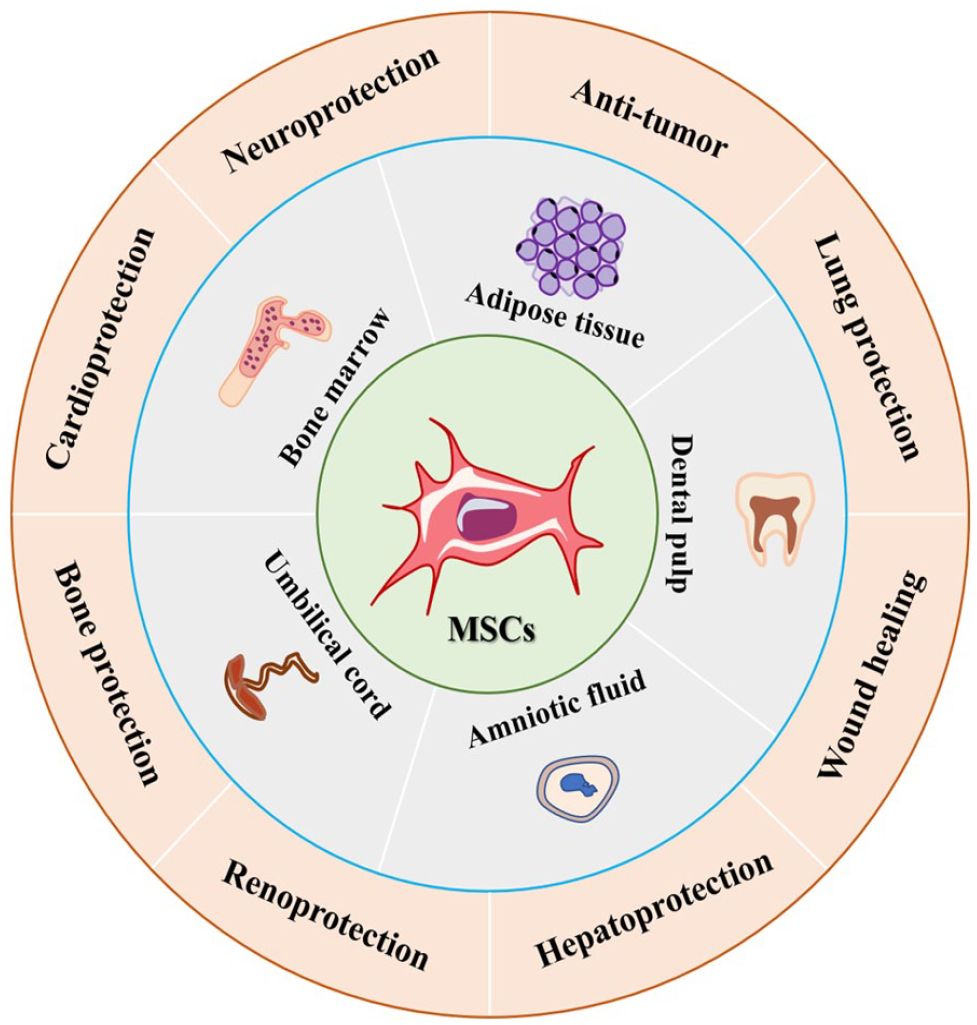
Sources and application of MSCs. MSCs: mesenchymal stem cells.
Efficacy and Mechanisms of MSCs Therapy in KD
Currently, MSCs are considered as new therapeutic tools for the treatment of KD because of their multidirectional differentiation, migration and homing, and paracrine effects17,28. Previous studies have shown that MSCs are effective and safe when used to treat organ injury29,30. Importantly, MSCs therapy promoted the recovery of renal function after renal pathogenesis through various mechanisms 31 (Fig. 2) such as anti-inflammation, anti-apoptosis, angiogenesis, anti-oxidative stress, anti-fibrosis, regulating autophagy, and senescence (Table 1).

Mechanisms of MSCs in the treatment of kidney diseases. ACN: aristolochic acid nephropathy; AKI: acute kidney injury; BTBRob/ob: Black and Tan Brachyury (BTBR) leptin deficiency; CKD: chronic kidney disease; CLP: cecal ligation and puncture; Col I: type I collagen; ECM: extracellular matrix; EMT: epithelial–mesenchymal transformation; FGF: fibroblast growth factor; FN: fibronectin; GLUT1: glucose transporter type 1; I/R: ischemia–reperfusion; HGF: hepatocyte growth factor; IL-1β: interleukin-1β; MAPK: mitogen-activated protein kinase; MDA: malondialdehyde; MI: macrophage infiltration; mtDNA: mitochondrial DNA; NF-κB: nuclear factor–kappa B; Nrf2/ARE: nuclear factor erythronid 2-related factor 2/antioxidant response elements; OXPHOS: oxidative phosphorylation; SDT: Spontaneously Diabetic Torii; α-SMA: alpha-smooth muscle actin; SOD: superoxide dismutase; SOX9: sex-determining region Y-box 9; STZ: streptozotocin; TNF-α: tumor necrosis factor–alpha; UUO: unilateral ureteral obstruction; VEGF: vascular endothelial growth factor.
Mechanisms of MSCs and Extracellular Vesicles in the Treatment of KD According to Published Studies From 2017 to 2022.

ADMSCs: adipose-derived mesenchymal stem cells; AKI: acute kidney injury; ALT: alanine aminotransferase; AST: aspartate aminotransferase; BMMSCs: bone marrow mesenchymal stem cells; BUN: blood urea nitrogen; CR: creatinine; DN: diabetic nephropathy; ECM: extracellular matrix; EGF: epidermal growth factor; EMT: epithelial–mesenchymal transformation; ER: endoplasmic reticulum; FGF: fibroblast growth factor; FN: fibronectin; HGF: hepatocyte growth factor; H/R: hypoxia/reoxygenation; I/R: ischemia–reperfusion; IFN: interferon; IL: interleukin; KD: kidney disease; LPS: lipopolysaccharide; MAPK: mitogen-activated protein kinase; MCP-1: monocyte chemoattractant protein-1; MDA: malondialdehyde; MSCs: mesenchymal stem cells; NF-κB: nuclear factor–kappa B; PGE2: prostaglandin E2; ROS: reactive oxygen species; Scr: serum creatinine; SDT: Spontaneously Diabetic Torii; α-SMA: alpha-smooth muscle actin; SOD: superoxide dismutase; STZ: streptozotocin; TGF: transforming growth factor; TNF-α: tumor necrosis factor–alpha; UCMSCs: umbilical cord mesenchymal stem cells; UUO: unilateral ureteral obstruction; VEGF: vascular endothelial growth factor.
Anti-Inflammation
Previous studies have proved that activation of inflammation was an important part of the pathogenic process of KD, and macrophage infiltration and aggregation contribute to the acceleration of KD progression 83 . For example, a study by Heerspink et al. 84 showed that high plasma levels of tumor necrosis factor (TNF) receptor 1 and IL-6 were associated with the progression of diabetic KD. Martos-Rus et al. 85 found that the serum levels of inflammatory cytokines/chemokines were upregulated in patients with end-stage KD and activated the nuclear factor–kappa B (NF-κB) pathway, as well as increased peripheral monocytes and inflammatory polarization of macrophages were detected in the kidney tissue of mice with uremia model. In addition, macrophages play an important role in immune surveillance and maintaining the stability of the renal internal environment 86 , and macrophages derived from bone marrow can directly transform into myofibroblasts in the damaged kidney, accelerating the progression of pathogenic fibrosis87,88. Of note, MSCs can ameliorate kidney injury by inhibiting inflammation and promoting kidney repair 89 . For instance, treatment with UCMSCs significantly prevented the progression of diabetic nephropathy (DN) by reducing pro-inflammatory cytokines and secreting abundant epidermal growth factor and vascular endothelial growth factor (VEGF) 52 .
Recently, several studies have confirmed that MSCs-derived EVs play a major role in treating KD 15 . For example, MSCs-Exo treatment slowed the progression of ischemic–reperfusion injury (IRI) by inhibiting expressions of inflammatory factors [eg, IL-6, TNF-α, NF-κB, and interferon (IFN)-γ] 51 . Gao et al. 41 showed that ADMSCs-Exo inhibited inflammation of sepsis-related AKI by blocking the NF-κB pathway. Another study found that BMMSCs-Exo was a promising therapeutic approach for preserving CKD progression via reducing inflammation and degeneration 69 . Similarly, Song et al. 90 stated that MSCs-derived EVs serve as effective therapeutic strategies for CKD via upregulating anti-inflammatory M2 macrophages and regulatory T-cell numbers. In addition, it is well recognized that EV activity mainly involves the horizontal transfer of genetic materials91,92. For example, MSCs-EVs secrete insulin-like growth factor (IGF-1) receptor mRNA directly to renal tubular epithelial cells, as well as directly secreting IGF-1 and carrying IGF-1 receptors to promote kidney repair in AKI 93 . Several studies have confirmed that MSCs-derived exosomes enriched with miRNAs (eg, miR-15a, miR-15b, and miR-16 90 ) and/or chemokine receptors (eg, CCR2 94 and CXCR4 95 ) could ameliorate inflammation and kidney injury by reducing chemokines (eg, CX3CL1 96 and CCL2 97 ).
Anti-Apoptosis
Cell apoptosis is closely related to kidney injury and KD progression. Previous studies have shown that renal tubular epithelial cell apoptosis was detected in both animal AKI models and human kidney tissues of AKI 98 , and the expression of pro-apoptotic genes (Bax and caspase-3) was increased, while the expression of anti-apoptotic gene Bcl-2 was reduced. Of note, numerous studies have confirmed that MSCs implantation can inhibit apoptosis of renal tubular epithelial cells and thus restore renal function99,100. Guo et al. 33 confirmed that BMMSCs alleviated sepsis-induced AKI by inhibiting apoptosis and promoting mitophagy of renal tubular epithelial cells. Another study by Tseng et al. 101 found that hypoxic MSCs significantly reduced cell apoptosis in renal tubular NRK-52E cells exposed to hypoxia-reoxygenation as well as promoted renal tubular autophagy in acute renal IRI rats.
Of note, MSCs play a therapeutic role in KD through paracrine mechanisms. For example, HuMSC-Exo inhibited apoptosis of NRK-52E cells induced by cisplatin via activation of the ERK1/2 pathway 102 . Alasmari et al. 77 illustrated that exosomes derived from BMMSCs impeded the progression of CKD by interfering with fibrosis and apoptosis. Moreover, MSCs-Exo exhibited anti-apoptotic effect on KD progression by transferring miRNAs (eg, miR-199a-3p 44 and miR-424-5p 81 ). What’s more, exosomes released from MSCs preconditioned with melatonin blocked apoptosis by decreasing the levels of caspase-3 67 , which had a protective effect against CKD.
Pro-Angiogenesis
It has been reported that sparse peritubular capillaries, accompanied by reduced blood perfusion, limit the supply of interstitial oxygen to the kidney, ultimately leading to adverse consequences such as renal fibrosis and tubular atrophy, which accelerate KD progression 103 . Previous studies have shown that MSCs can survive for a long time after implantation into the injured kidney, promote renal interstitial capillary neovascularization, improve renal microcirculation, and inhibit renal fibrosis progression 104 . Meanwhile, MSCs derived from the kidney facilitated angiogenesis, vasculogenesis, and endothelial repair 105 . Numerous studies have demonstrated that MSCs transplantation increased the expression of VEGF mRNA in kidney tissues along with endothelial cell proliferation, reduced the loss of peritubular capillaries, and improved kidney function 106 .
In addition, MSCs-derived EVs ameliorated AKI progression by promoting angiogenesis (enhancing renal VEGF levels) in vivo and in vitro 107 . Eirin et al. 108 proved that the autologous ADMSCs-EVs improve the renal microvascular system in pigs with metabolic renal vascular diseases. Another study found that melatonin-stimulated MSCs-Exo isolated from patients with CKD promoted angiogenesis in ischemic diseases through the upregulation of miR-4516 109 . Mechanistically, MSCs promote angiogenesis through paracrine secretion of some bioactive substances related to angiogenesis [such as VEGF, hypoxia-inducible factor 1-alpha (HIF-1α), platelet-derived growth factor-BB (PDGF-BB), stromal cell-derived factor 1 (SDF-1), and angiogenin]34,52,110, as well as differentiation to vascular endothelial cells 111 and smooth muscle cells 112 . For example, ADMSCs transplantation significantly increased peritubular vascular density and the number of CD31- and vWF (von Willebrand factor)-positive cells in renal interstitium and peritubular area of mice with IRI injury, as well as improved blood perfusion in the kidney of mice 113 . The above studies suggest that MSCs have beneficial effects against KD progression by promoting renal angiogenesis and preventing peritubular capillary loss.
Anti-Fibrosis
Renal interstitial fibrosis is the common pathological hallmark of CKD progression, and eventually inevitably develops into end-stage KD 114 , causing a huge socioeconomic burden. Increasing evidence has confirmed that EMT of renal tubular cells is a key event in renal interstitial fibrosis, characterized by fibroblast proliferation and an imbalance between ECM production and degradation115,116, and inhibition of renal tubular EMT may be a potential therapeutic strategy for the treatment of CKD 117 . Numerous studies have shown that MSCs, as a protective mediator of renal interstitial fibrosis, can play an important regulatory role in the process of EMT through their anti-fibrotic activity and paracrine mechanisms, delaying tubular EMT and improving renal fibrosis 118 . For example, Tang et al. 119 showed that BMMSCs treatment prevents renal interstitial fibrosis by blocking the Akt/GSK3β/Snail signaling pathway in adenine-induced CKD. Another study proved that glial cell line–derived neurotrophic factor–modified ADMSCs suppressed EMT and renal fibrosis via inhibition of the PI3K/Akt pathway in CKD 120 .
As the research progresses, subsequent studies have shown that MSCs-Exo exerts anti-fibrotic and EMT-suppressive effects by delivery of genetic information to target cells, thereby alleviating renal fibrosis in CKD. For instance, Grange et al. 121 found that EVs of MSCs can inhibit and reverse the progression of glomerular and tubule-interstitial fibrosis in the DN mouse models by downregulating fibrosis-related genes (eg, Serpia1a, TIMP1, MMP3, collagen I, and Snail). The MSCs-Exo inhibited the EMT process of transforming growth factor (TGF)-β1–treated renal tubular epithelial cells and renal fibrosis in a unilateral ureteric obstruction (UUO)–induced renal fibrosis mouse model via delivery of miRNA-122a 73 and miR-186-5p 82 . Liu et al. 79 found that UCMSCs-Exo exhibits anti-fibrotic effects in CKD through the inactivation of the reactive oxygen species (ROS)–mediated p38 mitogen-activated protein kinases/extracellular signal-regulated kinase (MAPK/ERK) pathway.
Anti-Oxidative Stress
Oxidative stress is involved in the development and progression of KD, including AKI 122 and CKD 123 . Several studies have confirmed that oxidative stress induces renal tubular inflammation, fibrosis, and renal tubular epithelial cell apoptosis, and resulted in promoting the progression of KD124,125. Meanwhile, the kidney acts as an essential organ for the production of reactive oxygen species (ROS), and oxidative stress is a mediator of CKD progression 126 . In recent years, numerous studies have proved that MSCs were reported to be used as an antioxidant therapeutic drug in the treatment of KD98,127,128. Therefore, the regulation of oxidative stress is the essential mechanism of MSCs-based treatment in KD. Recently, Song et al. 129 showed that MSCs alleviated adriamycin-induced nephropathy by inhibiting oxidative stress and NF-κB-mediated inflammation. Another study showed that valsartan- and melatonin-modified MSCs improved renal architecture and function in CKD by diminishing oxidative stress 130 .
In addition, several preclinical studies demonstrate that MSCs-derived EVs promote tissue repair and reduce oxidative stress in KD 131 . For instance, Zhang et al. 132 revealed that human Wharton’s jelly MSCs-EVs could protect the kidney against IRI by mitigating oxidative stress. Another study confirmed that exosomes derived from UCMSCs prevented AKI progression via suppressing renal oxidative stress and inflammation as well as improving kidney function of kidney failure 48 . Cao et al. 133 showed that human placenta MSCs-Exo reduced oxidative stress and mitochondrial fragmentation in a renal IRI model through activation of the Nrf2/keap1 pathway.
Regulating Autophagy
Autophagy is a type II programmed cell death 134 that can be activated to promote cell survival 135 or resulted in cell death 136 by stimulating various physiological and pathological factors. A basal level of autophagy occurs as a self-eating cellular process to degrade cytosolic proteins and subcellular organelles in lysosomes, recycle the cytoplasmic components, and regenerate cellular building blocks and energy, thus maintaining cellular and tissue homeostasis in all eukaryotic cells137,138. Recently, transplantation of MSCs has emerged as an effective strategy in regenerative medicine to repair injured organ function via regulating autophagy 139 . For instance, hypoxic MSCs alleviate AKI progression by promoting renal tubular autophagy 32 . Feng et al. 140 found that transplantation of sirtuin3-overexpression amniotic fluid stem cells serves as promoting therapeutic strategies for DN through activation of mitophagy and inhibition of apoptosis. Other studies confirmed that UCMSCs enhanced autophagy in advanced oxidation protein products–treated HK-2 cells through inactivation of the PI3K/Akt/mTOR pathway141,142. Intriguingly, upregulation of autophagy remarkably increased the secretion of TGF-β1 from MSCs and suppressed the proliferation of CD4+ T lymphocytes 143 , whereas inhibition of autophagy reduced the responsiveness of T cells to mitogen IL-2 and increased the production of immunosuppressive prostaglandin E2 144 . For example, Yuan et al. 25 showed that MSCs ameliorate kidney injury in DN via eliciting macrophages into anti-inflammatory phenotype and elevating PGC-1α (peroxisome-proliferator-activated receptor-γ coactivator-1alpha)/TFEB (transcription factor EB)-mediated lysosome-autophagy. In addition, autophagy is active in the physiological state or can be activated by cellular stresses such as oxidative stress 145 . Gergin et al. 146 demonstrated that transplanted MSCs inhibited oxidative stress in colistin-induced nephrotoxicity by modulating autophagy. Autophagy and oxidative stress are correlated, and the underlying mechanisms of MSCs-based treatment have not been fully explored.
At the same time, some researchers demonstrated that MSCs-Exo has become a research focus for targeted therapy of KD 147 . Wang et al. 50 discovered that UCMSCs-Exo preprocessing can prevent cisplatin-induced AKI in vivo and in vitro by activating autophagy. Jia et al. 148 identified that UCMSCs-Exo can prevent cisplatin-induced AKI by activating autophagy. Ebrahim et al. 71 confirmed that MSCs-Exo enhances autophagy and then slows the progression of DN via activating the mTOR pathway.
Senescence
Cellular senescence is a specialized cell state of permanent cell cycle arrest caused by the accumulation of cellular damage due to a variety of stressors such as telomere shortening, DNA damage, oxidative stress, and activation of oncoproteins80,149. Senescent cells are known to be present at increased levels in KD, and accumulation of senescent cells is thought to facilitate renal fibrosis, DN, severe AKI, and decay in renal function150,151. Several studies have shown that the removal of senescent tubular cells in the kidney by transgenic or pharmaceutical approaches reduced features of tissue aging and efficiently ameliorated glomerulosclerosis, inflammation, and renal function152–154. Of note, ADMSCs transplantation can alleviate ischemia–reperfusion (I/R)-induced kidney injury through reducing renal senescence 155 . Rodrigues et al. 156 found that UCMSCs can prevent IRI-induced renal senescence in AKI.
In addition, several studies have demonstrated that MSCs-derived exosomes exhibit therapeutic effects on KD by regulating cell senescence157,158. For example, Wang et al. 159 showed that MSCs-derived exosomal let-7b-5p ameliorates cisplatin-induced AKI by reducing renal senescence and cell apoptosis. Another study confirmed that treatment with exosomes derived from MSCs efficiently reduced senescence in renal tubular epithelial cells by diminishing the transcription of senescence markers and senescence-associated secretory phenotype factors 80 . In addition, the paracrine effects of MSCs were enhanced after pretreated with metformin and inhibited MSCs senescence by suppressing SA-β-gal activity, p16Ink4a expression, and p53 and NF-κB activation, thus effectively reducing CKD inflammation and fibrosis 160 . Taken together, the above studies have proven that MSC-EVs are effective in treating KD.
Clinical Trials of MSCs Therapy in KD
In the last decade, the beneficial efficacy of MSCs in the treatment of KD has been confirmed in multiple cellular and animal experimental models. For example, MSCs-base therapy was first shown to promote renal tubular regeneration and improve renal function in cisplatin-induced AKI mice models in 2004 161 . Subsequent studies have also confirmed that MSCs alleviate other animal models of AKI induced by ischemia–reperfusion, glycerol, sepsis, cecal ligation and puncture162,163, and so on. In addition, MSCs transplantation significantly alleviated CKD progression by inhibiting renal tubular epithelial cell apoptosis, EMT process, and inducing cell autophagy15,164. For example, Liu et al. 165 showed that treatment with BMMSCs restricted inflammation and renal damage in the IRI model. Of note, several clinical studies are completed or ongoing to evaluate the safety and efficacy of MSCs for the treatment of KD according to ClinicalTrials.gov (Table 2). For example, a phase I/II clinical trial by Swaminathan et al. 166 showed that BMMSCs alleviated inflammatory response in patients with AKI by secreting anti-inflammatory factors. Two other clinical trials (NCT00698191 and NCT01741857) confirmed that UCMSCs transplantation for refractory systemic lupus erythematosus improved disease activity and renal function, and reduced proteinuria, as well as no adverse events occurred. Currently, a total of seven and nine clinical trials are ongoing to evaluate the safety and efficacy of MSCs in patients with DN and CKD. However, MSC-based therapy is limited by the low survival rate of MSCs when used to treat severe KD 167 . Several factors such as poor control of the disease, cellular microenvironment, anoikis, ischemia, inflammation, and ROS production reduce the efficacy of MSC-based therapies168,169. Some preclinical studies have suggested that the preconditioning or cotreatment of MSCs protects them from the harmful environment at the site of damage and improves their function 23 , including cytokines or natural/chemical compounds. In addition, as several clinical trials are in recruiting status, it is worthwhile to further consider and explore whether there are safety issues and insignificant efficacy of MSCs for the treatment of KD. Therefore, we will further explore multiple treatment strategies based on MSCs for KD after obtaining the results of the existing clinical trials to prolong the survival of patients and delay the progression of KD.
The Ongoing Clinical Trials of MSCs Therapy in KD.
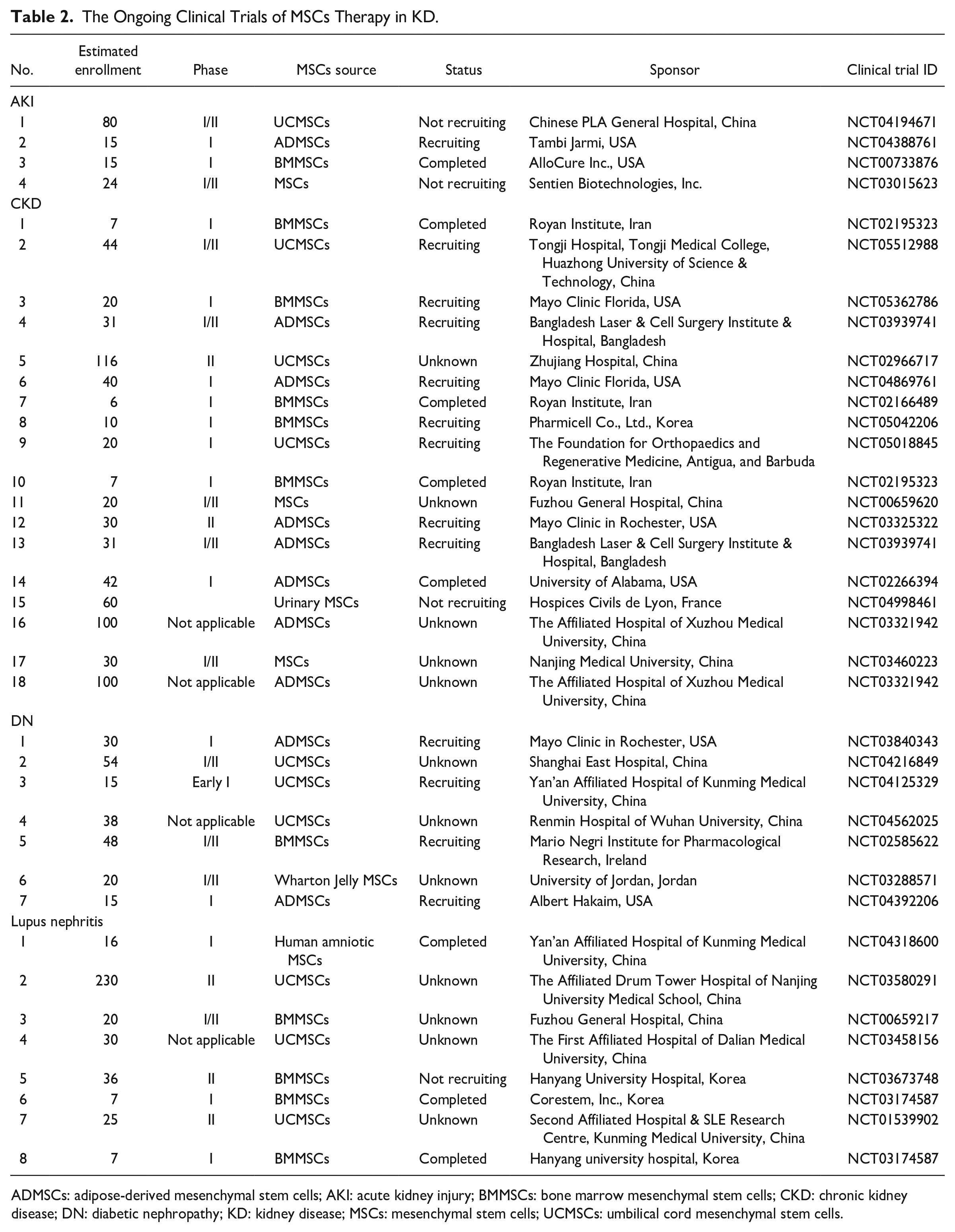
ADMSCs: adipose-derived mesenchymal stem cells; AKI: acute kidney injury; BMMSCs: bone marrow mesenchymal stem cells; CKD: chronic kidney disease; DN: diabetic nephropathy; KD: kidney disease; MSCs: mesenchymal stem cells; UCMSCs: umbilical cord mesenchymal stem cells.
Current Challenges of MSCs Therapy in KD
Selection of MSCs Source
Interestingly, the results of animal models and clinical trials have confirmed that MSCs have shown positive results for the treatment of various KD, and no adverse effects or serious adverse complications have been observed. Currently, MSCs are widely available in clinical trials, but the ultimate goal is to use MSCs to delay KD progression and avoid its progression to end-stage renal disease. Therefore, the choice of autologous or allogeneic MSCs for transplantation should be considered in clinical applications. Autologous MSCs have low immunogenicity and no risk of infection, but the longer time required for autologous cell preparation may limit their practical application in clinical treatment. Allogeneic MSCs can be selectively derived from young healthy donors and have the potential to be produced rapidly and in large quantities in vitro culture, significantly reducing costs 118 , while the use of allogeneic MSCs includes a higher risk of immunological reactions and shorter cell survival times following injection 170 . Although transplantation with autologous MSCs is safer and more ethical than allogeneic MSCs, there are still some problems in clinical applications. First, after autologous MSCs are extracted, the in vitro culture cycle is long, which may not fully meet the needs of the body. Second, there is a significant difference between the secretion and immune regulation of autologous MSCs 171 . However, a clinical study reported that injections of allogeneic or autologous BMMSCs were both associated with low rates of treatment-emergent serious adverse events (such as immunologic reactions) in patients with ischemic cardiomyopathy 172 . Further studies to overcome the immune rejection caused by allogeneic MSCs during the treatment process are necessary. Owing to the many sources of allogeneic MSCs and the high efficiency of in vitro culture, the treatment of immune rejection caused by allogeneic MSCs is still receiving widespread attention 173 . On the contrary, an obvious solution is to immediately use autologous MSCs as a ready-made product. In addition, new products such as acellular exosomes and MSCs derived from human pluripotent stem cells are exciting developments that are attracting significant attention 174 .
Transplantation Protocol of MSCs
Currently, MSCs are mostly transplanted in animal experiments by intravenous, arterial, intraperitoneal, and local injections for KD, whereas clinical transplantation of MSCs includes arterial and intravenous injections. Previous studies have shown that MSCs transplantation via arterial injection was more effective than intravenous injection in promoting renal regeneration 175 . Moreover, local injection of MSCs also plays a positive role in renal repair 176 , but this route was less commonly used in clinical practice. Importantly, different transplantation modalities have a significant impact on the survival and homing rate of MSCs, and the optimal implantation modality needs to be determined 177 . Furthermore, the timing of MSCs injection, the number of injections, the number of cells per injection, exploring the optimal strategy for MSCs migration to the damaged site, understanding the interactions between MSCs and other tissue cells, and the adverse effects of MSCs after transplantation (eg, low differentiation in vivo and tumorigenesis) 178 , all of which pose challenges for MSCs to move from basic experiments to clinical applications.
Migration and Survival of MSCs
A prerequisite for the efficacy of MSCs is the ability to migrate to damaged tissues. Previous studies have found that MSCs can localize to diseased sites 179 , but only a small fraction of MSCs 180 . Several studies found that the migration of MSCs in vivo was regulated by various surface adhesion molecules (eg, CD44, VLA-4/VCAM1, SDF-1/CXCR4, and CXCL5/CXCR2)181–183. Importantly, pretreatment with cytokines or active substances can improve the localization/migration ability of MSCs. For example, MSCs modified by CXC chemokine receptors (such as CXCR3 184 and CXCR4 185 ) exhibited better migration and localization abilities. In addition, enhanced migration and anti-inflammatory activities of MSCs mediated by the transient ectopic expression of CXCR4 and IL-10 or IL-35186,187. However, the source, culture, and amplification methods of MSCs may affect the expression of their localized surface molecules 188 , as well as the cell activity, therapeutic effects, and safety of modified MSCs were difficult to control. Meanwhile, there is a lack of effective strategies to precisely localize MSCs to damaged tissues.
Safety of MSCs Transplantation
With the gradual increase of studies on the application of MSCs in clinical practice, the safety of MSCs has received widespread attention. In the phase I clinical trial by Liu et al. 189 , no physical abnormalities were found in healthy volunteers after receiving BMMSCs infusion at a 2-month follow-up. Wang et al. 190 conducted a toxicity study of UCMSCs transplantation in 32 macaques and no adverse reactions were observed. Ra et al. 191 evaluated the safety of ADMSCs preparations using an animal model of ulcerative colitis and no toxicity or tumorigenicity was found in immunodeficient mice. Hu et al. 192 showed that no severe adverse reactions or tumorigenicity was observed in clinical trials with either autologous or allogeneic transplantations of MSCs. These results indicated that MSCs were relatively safe in the treatment of diseases. Currently, no US Food and Drug Administration (FDA)-approved MSCs on market for disease treatment, whereas some MSCs-approved products for human disease are in other countries (Table 3). Meanwhile, most clinical studies of MSCs are still in the early stage, as well as the source, isolated, purified methods, and injection route of MSCs are different. Therefore, the safety of MSCs needs to be summarized and improved with continuous clinical trials.
Current Approved in South Korea, Europe, Japan, and Other Countries With MSCs for Diseases.
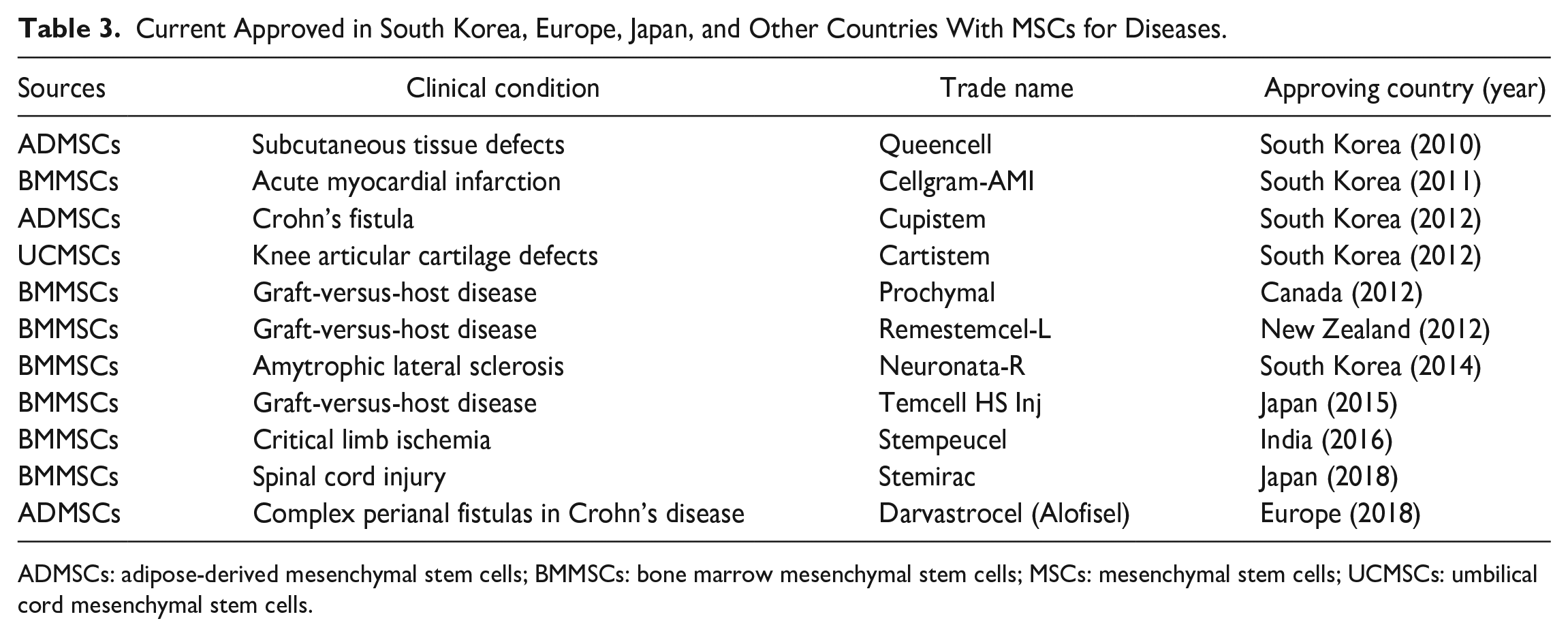
ADMSCs: adipose-derived mesenchymal stem cells; BMMSCs: bone marrow mesenchymal stem cells; MSCs: mesenchymal stem cells; UCMSCs: umbilical cord mesenchymal stem cells.
Others
Except for the current challenges mentioned above, clinical applications of MSCs have other limitations. For example, tissue sources and isolation methods can influence MSC proliferation and differentiation potential193,194. In addition, microenvironment, donor age, and environmental factors affect the genetic stability of MSCs. No consensus on the standard properties (eg, phenotype, differentiation potential, physiological functions, and biological properties) of MSCs has been developed 195 . Of note, MSCs can only proliferate for a limited number of passages in vitro and will eventually enter a senescence state 196 . Progressively slow growth and lack of differentiation of high-passaged MSCs have been reported in several studies197,198. Other important challenges are the isolation and culturing of MSCs using xenofree conditions 199 as cells grown in media containing fetal bovine serum and other animal or bacterial products cannot be used for clinical purposes. Thus, a better understanding of the origin, biological properties, and function of MSCs derived from different tissues could provide insight into what truly is an “MSC.”
Improvement of MSCs’ Therapeutic Effect in KD
MSC-based therapy has been widely studied for KD therapy and has been shown to result in improved renal function and the recovery of damaged renal tissues in animal studies and clinical trials 200 . However, the limited effects of the current therapy for KD drive the need for the development of novel strategies such as preconditioning, genetic modifications, and strategies for scalability. For instance, several cytokines and natural/chemical compounds have been shown to have protective effects by enhancing cell survival and proliferation 201 . Docosahexaenoic acid (DHA) is a necessary omega-3 fatty acid found in the blood and the kidney. The 14S,21R-dihydroxy-doxosa 4Z,7Z19Z,12E,16Z,19Z-hexaenoic acid (14S,21R-dHDHA) has been identified as a new DHA-derived lipid mediator, and treatment with this compound has been shown to enhance the function of MSCs. In vitro and IRI mouse models, MSCs treated with 14S,21R-dHDHA show reduced apoptosis and inflammatory responses, and improved renal function 202 . Other studies have shown that the pharmacological agent, S-nitroso N-acetyl penicillamine (SNP), a nitric oxide donor associated with cytoprotective and tissue-protective effects, promoted MSCs functionality by increasing cell proliferation and survival in renal IR model 203 . Moreover, administration of SNP-treated MSCs resulted in a significant improvement in renal function and increased the expression of pro-survival and pro-angiogenic factors in ischemic renal tissue. Darbepoetin-α is an erythropoietic agent that shows similar protective and hematopoietic effects and reduces kidney damage in an animal model of renal IRI 204 . In a mice model of renal ischemia, the administration of melatonin-pretreated MSCs increased the secretion of angiogenic cytokines and the survival of engrafted MSCs in CKD-associated ischemic sites. Moreover, miRNAs (eg, miR-146a-5p 205 , miR-19a-3p 206 , miR-374a-5p 207 , and miR-34a 208 )-modified MSCs ameliorated KD progression via reducing inflammation, oxidative stress, renal fibrosis, cell apoptosis, and so on.
Conclusion
Numerous studies have confirmed the safety and tolerability of MSCs transplantation for the treatment of KD209–212. Given the increasing incidence of KD worldwide, MSCs-based therapy appears to be an innovative intervention approach with tremendous potential for the management of KD, but there is still much work to be done before MSCs can be used for clinical treatment on a large scale. First, the issues of donor heterogeneity, mass production, immunogenicity, and cryopreservation of MSCs need to be addressed. Second, how to make MSCs with more efficient targeting ability, more precise immunomodulatory function, and safer application effect by artificial means need to be studied. Existing studies have provided several strategies, including genetic engineering, microparticle engineering, and preculture, which theoretically improve the efficiency of MSCs application. Third, the detailed underlying mechanisms of MSCs for the treatment of KD and the functional role of targeting kidney-related injury need to be further explored. Fourth, the therapeutic safety of MSCs (eg, carcinogenic) remains controversial. With the advancement of novel biotechnology, there are many strategies to enhance the efficacy and safety of MSCs (such as drug conjugation, hypoxia condition, cytokine pretreatment, and genetic modification), but long-term efficacy has not been proven and standardized clinical trial protocols are still needed. Currently, the treatment of KD is limited to drug therapy, dialysis, and renal transplantation, whereas MSCs transplantation has emerged as a promising alternative therapy and has been supported by evidence from relevant clinical studies213,214. From the perspective of functional improvement and clinical parameters, the results of clinical trials are more favorable, and the development of new technologies is expected to overcome current barriers to the clinical application of MSCs therapy. In conclusion, with the continuous innovation of treatment protocols and more and larger scale clinical trials in the future, MSCs-based therapy is expected to become a major “tool” for the treatment of KD.
Footnotes
Author Contributions
F.C. and N.C. are responsible for the acquisition, analysis and interpretation of the data, and drafting of the manuscript. Others contributed to the critical revision of the manuscript.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the National Natural Science Foundation of China (Grant No. 81860144).
