Abstract
Emerging evidence suggests that cell therapy with mesenchymal stem cells (MSCs) has beneficial effects on the injured heart. However, the decreased survival and/or adhesion of MSCs under ischemic conditions limits the application of cell transplantation as a therapeutic modality. We investigated a potential method of increasing the adhesion ability of MSCs to improve their efficacy in the ischemic heart. Treatment of MSCs with PKC activator, phorbol 12-myristate 13-acetate (PMA), increased cell adhesion and spreading in a dose-dependent method and significantly decreased detachment. When MSCs were treated with PKC inhibitor, that is, rottlerin, adhesion of MSCs was slightly diminished, and detachment was also decreased compared to the treatment with PMA. MSCs treated with both PMA and rottlerin behaved similarly to normal controls. In 3D matrix cardiogel, treatment with PMA increased the number of MSCs compared to the control group and MSCs treated with rottlerin. Expressions of focal adhesion kinase, cytoskeleton-associated proteins, and integrin subunits were clearly demonstrated in PMA-treated MSCs by immunoblotting and/or immunocytochemistry. The effect of PKC activator treatment on MSCs was validated in vivo. Following injection into rat hearts, the PMA-treated MSCs exhibited significantly higher retention in infarcted myocardium compared to the MSC group. Infarct size, fibrosis area, and apoptotic cells were reduced, and cardiac function was improved in rat hearts injected with PMA-treated MSCs compared to sham and/or MSC-implanted group. These results indicate that PKC activator is a potential target for niche manipulation to enhance adhesion of MSCs for cardiac regeneration.
Introduction
Mesenchymal stem cells (MSCs), also known as stromal cells, are a promising source of stem cells for regenerative therapy (8,24). Stem cell therapy provides a potential treatment for myocardial infarction (MI) through myocardial regeneration caused by transplantation of MSCs (16,17). However, the effects of clinical applications of MSCs have been confined to limited conditions and have been restricted by the poor survival of MSCs in vivo. To increase survival rates, several studies have focused on methods of strengthening MSCs (18).
One such method involves pretreatment of the stem cells to modify their cellular function (10). Several kinds of pretreatment can influence cellular mechanisms (34); for example, regulation of the protein kinase C (PKC) pathway is a recognized method of physiologically changing cells (36). PKC is a family of kinases consisting of at least 12 different isoforms (4,38). The conjunctiva contains at least eight of these isoforms and closely related PKCs (12,30). The serine/threonine kinase PKC induces phosphorylation of focal adhesion kinase, which is then transported to the cell surface (15). That is used to an actin-binding protein localized within cortical actin structures at the cell membrane (27). Adhesion to the matrix results in PKC-dependent phosphorylation from the actin structure (6). The PKC activator phorbol 12-myristate 13-acetate (PMA) has significant effects on the cells and plays a role in signaling related to cell adhesion (3). PMA is a phorbol ester, which is a well-known tumor promoter (31). However, previous studies have shown that cells treated with PMA survive better than nontreated cells (16). Furthermore, we investigated that PMA can induce specific differentiation of MSCs into cardiogenic cells without tumorigenesis (29). Since adhesion signaling mediates cell survival and apoptosis (37), it is likely that cells treated with PMA associate through adhesion involving several kinds of focal adhesion molecules and stress fibers. An increase in adhesion molecules such as focal adhesion kinase (FAK) and Src is associated with concomitant increases in tyrosine phosphorylation of cytoskeleton-associated proteins such as paxillin, vinculin, and talin at the same sites. FAK localizes to focal adhesion sites in cells (20), and phosphorylation of FAK is important for integrin-mediated cell attachment and spreading (32). Integrins are a family of αβ heterodimetric transmembrane adhesion receptors that participate in cell–cell or cell–matrix interactions within the immune system (7,21,26). Integrins are cell surface receptors for extracellular matrix molecules (ECMs) and play critical roles in a variety of biological process (10); for example, integrins activate the cellular focal adhesion-related kinases and bind to the ECM molecules and also regulate stress fibers. Several focal adhesion molecules have been identified, including FAK, Src, paxillin, talin, vinculin, α-actin, and tensin. An activated FAK/Src complex binds to potential substrates in the actin cytoskeleton, such as paxillin, talin, and vinculin. The relationship between adhesion signaling and the cytoskeleton has been investigated in several studies (23). However, there are no studies on the adhesion of implanted MSCs that were pretreated with PKC activator in the heart infarct zone.
We used an adhesion assay, detachment assay, and spreading assay to demonstrate that treatment with PKC activator increased the MSC adhesion rate. Immunoblotting and immunocytochemistry allowed the observation of FAK and integrin-related signaling that increased adhesion rate. To show enhanced adhesion of MSCs in vivo, the number of DAPI-labeled cells and histological analysis of rat hearts were observed after myocardial infarction (MI).
Materials and Methods
Isolation and Culture of MSCs
MSCs were purified as previously described (28). Bone marrow from the femoral and tibial bones of 4-week-old male Sprague–Dawley rats (approximately 100 g) was aspirated with 10 ml Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin solution. Mononuclear cells were recovered after centrifugation in Ficoll-Paque™ PLUS (GE Healthcare, Uppsala, Sweden) and washed twice, resuspended in DMEM with 10% FBS, and plated in flasks at 1 × 106 cells per 100 cm2. Cultures were maintained at 37°C in a humidified atmosphere containing 5% CO2. After 48 h, nonadherent cells were discarded, and adherent cells were thoroughly washed twice with phosphate-buffered saline (PBS). Fresh complete medium was added and replaced every 3–4 days for approximately 10 days. The characteristics of MSCs were demonstrated by immunophenotyping. To verify the natures of the cultured MSCs, cells were analyzed for various surface and intracellular markers. Cells were harvested, washed with PBS, and labeled with the following antibodies conjugated with fluorescein isothiocyanate (FITC): cluster of differentiation 14 (CD14), CD34, CD71, CD90, CD105, CD106, and intercellular adhesion molecule-1 (ICAM-1). FITC-conjugated goat anti-mouse IgG (Jackson ImmunoResearch Laboratories, West Grove, PA) was used as secondary antibody and labeled cells. Labeled cells were assayed by flow cytometry and analyzed with CellQuest Pro Software (Becton Dickinson, San Jose, CA). MSCs were used for in vitro and in vivo experiments between passages 2 and 3.
Drug Treatment
To examine the ability of MSC adhesion, we used PMA (Sigma-Aldrich, St. Louis, MO) and Rottlerin (Merck Millipore, Billerica, MA). These chemicals were dissolved in dimethyl sulfoxide (DMSO) and were diluted at a final concentration of 100 nM and 3 μM with DMEM, respectively. PMA was treated for 4 h after rottlerin treatment for 10 min. DMSO with 0.1% (v/v) was used as a negative control.
Assays for Cell Adhesion and Spreading
Suspensions of 2 × 104 MSCs were added to each well of a six-well plate and allowed to attach for 1 h at 37°C and 5% CO2. Cells were treated with PKC activator (PMA) and inhibitor (Rottlerin) as indicated above. To determine MSC adhesion, cells were carefully washed three times with PBS, and then four separate fields were photographed with a phase contrast microscope, and the cells were counted. Each experiment was performed in triplicate wells and repeated at least three times. For spreading assays, MSCs were plated for 4 h on four-well plates under the conditions of the adhesion experiments described above. To determine the extent of MSC spreading, plates were washed three times with PBS, fixed with 3% formaldehyde, stained with Coomassie blue, and then destained. Finally, the four separate fields were photographed using a phase contrast microscope.
Trypsinization Assay
Cells were grown to 90% confluency on culture plates prior to trypsinization for 3 min at 37°C. The cells were then washed, and the remaining adherent cells were incubated for 1 h at 37°C. Lastly, the cells were counted in nine different regions per one plate.
Preparation of Cardiac Fibroblast-Derived Three-Dimensional Matrix (Cardiogel)
Viable cardiac fibroblasts (2 × 1 0 5 cells) were added to each well of a four-well plate and allowed to attach for 1 h at 37°C and 5% CO2. The medium was changed every 48 h until the matrix was denuded of cells. The matrix was carefully and gently rinsed with PBS. Next, 1 ml of prewarmed extraction buffer (0.5% Triton X-100, 20 mM NH4OH in PBS) was added to the matrix, and we observed cell extraction using an inverted microscope until no intact cells were observed. The plates were incubated at 37°C for 30 min with DNase (10 units of DNase per ml of PBS) to minimize the DNA debris. The matrix-coated plates were covered with a minimum of 3 ml PBS containing 100 U/ml of penicillin, 100 μg/ml of streptomycin, and 0.25 μg/ml of Fungizone for later use.
Immunoblot Analysis
Cells were washed once in PBS and lysed in buffer (Cell Signaling Technology, Beverly, MA) containing 20 mM Tris (pH 7.5), 150 mM NaCl, 1 mM Na2-EDTA, 1 mM EGTA, 1% Triton, 2.5 mM sodium pyrophosphate, 1 mM β-glycerophosphate, 1 mM Na3VO4, 1 mg/ml leupeptin, and 1 mM phenylmethylsulfonyl fluoride. Protein concentrations were determined using the Bradford Protein Assay Kit (Bio-Rad Laboratories, Richmond, CA). Proteins were separated in a 12% sodium dodecyl sulfate-polyacrylamide gel and transferred to a polyvinylidene difluoride membrane. After blocking the membrane with Tris-buffered saline–Tween 20 (TBS-T, 0.1% Tween 20) containing 5% nonfat dried milk for 1 h at room temperature, the membrane was washed twice with TBS-T and incubated with primary antibody for 1 h at room temperature or overnight at 4°C. The membrane was washed three times with TBS-T for 10 min and incubated for 1 h at room temperature with horseradish peroxidase-conjugated secondary antibodies. After extensive washing, bands were detected by enhanced chemiluminescence reagent (Santa Cruz Biotechnology, Inc., Santa Cruz, CA). Band intensities were quantified using the Photo-Image System (Molecular Dynamics, Sunnyvale, CA). Immunoblot was performed with the following antibodies: phosphorylated FAK (p-FAK), FAK, and vinculin (1:1,000 dilution, Santa Cruz Biotechnology, Inc.), paxillin (1:500 dilution, Millipore), talin (1:500 dilution, Abcam, Cambridge, MA), Ras-related C3 botulinum toxin substrate 1 (Rac-1; 1:500 dilution, Cell Signaling), and β-actin (1:10,000 dilution, Santa Cruz Biotechnology, Inc.). The secondary antibodies were as follows: mouse anti-goat-horse radish peroxidase (HRP; 1:5,000 dilution), goat anti-mouse-HRP (1:4,000 dilution), and goat anti-rabbit-HRP (1:2,000 dilution, Enzo Life Sciences, Farmingdale, NY).
Immunocytochemistry
Cells were grown on four-well plastic dishes, washed twice with PBS, and then fixed with 4% paraformaldehyde in 0.5-ml PBS for 30 min at room temperature. The cells were washed again with PBS and permeabilized for 30 min in PBS containing 0.2% Triton X-100. Next, the cells were blocked with 10% goat serum in PBS and incubated for 1 h with primary antibodies. The MSCs were washed three times for 10 min with PBS and incubated with secondary antibodies for 1 h. All images were produced using an excitation filter under reflected light fluorescence microscopy (LSM 700, Carl Zeiss, Thornwood, NY). Immunocytochemistry was performed with the following antibodies: p-FAK, vinculin, integrin αvβ3, and integrin αV (1:100 dilution, Santa Cruz Biotechnology, Inc.), and paxillin and integrin α5 (1:200 dilution, Millipore). The secondary antibodies were as follows: Texas red-conjugated rabbit anti-goat, rhodamine red- conjugated donkey anti-mouse, rhodamine red-conjugated donkey anti-rabbit, FITC-conjugated donkey anti-mouse, and FITC-conjugated donkey anti-rabbit (1:500 dilution, Jackson ImmunoResearch Laboratories).
Induction of Myocardial Infarction
Experiments were conducted in accordance with the International Guide for the Care and Use of Laboratory Animals. The protocol was approved by the Animal Research Committee of the Yonsei University College of Medicine. Eight-week-old Sprague–Dawley male rats (approximately 250 g) were intubated under general anesthesia, and positive pressure ventilation (180 ml/min) was maintained with room air supplemented with oxygen (2 L/min) using a Harvard ventilator (Harvard apparatus, Holliston, MA). The heart was exposed through a 2-cm left lateral thoracotomy. The pericardium was incised and a 6–0 silk suture was placed around the proximal portion of the left coronary artery, beneath the left atria appendage. Ligature ends were passed through a small length of plastic tube to form a snare. For coronary artery occlusion, the snare was pressed onto the surface of the heart directly above the coronary artery, and a hemostat was applied to the snare. Ischemia was confirmed by the blanching of the myocardium and dyskinesis of the ischemic region. After 60 min of occlusion, the hemostat was removed, and the snare was released for reperfusion, with the ligature left loose on the surface of the heart. Restoration of normal rubor indicated successful reperfusion. The wounds were sutured, and the thorax was closed under negative pressure. Rats were weaned from mechanical ventilation and were returned to the cages to recover.
Labeling and Transplantation of MSCs
Cells were labeled with DAPI to measure cell viability by adding sterile DAPI solution to the culture medium for 30 min on the day of implantation, at a final concentration of 50 μg/ml. Cells were washed six times in PBS to remove unbound DAPI. DAPI-labeled cells were detached with 0.25% (w/v) trypsin and suspended in serum-free medium for grafting. For instant transplantation after induction of myocardial infarction, MSCs (1 × 106 cells) were suspended in 100 μl of serum-free medium with a 30-gauge needle on a Hamilton syringe and injected into anterior and lateral aspects of the viable myocardium bordering the infarction by dividing into three. There are four groups used in this study as follows: Normal, nonligated rat; Sham, ligated but not implanted rat; MSCs, ligated and MSCs implanted rat; PMA-MSCs, ligated and PKC-activated MSCs implanted rat. These animal groups were divided into three experimental groups. First group was injected with DAPI-stained MSCs for determining the survival rate of MSCs for 3 days after transplantation (n = 7, each). Second group was used for morphological analysis at 1 week (n = 7, each), and other group was used for determination of cardiac function (as described in the left ventricular catheterization section) at 3 weeks (n = 7, each).
Histology of Cell-Transplanted Heart
At 7 days after implantation, the animals were sacrificed, and their hearts were excised. 2,3,5-Triphenyltetrazolium chloride (TTC) staining was used to assess myocardial tissue viability and to determine the size of the MI. Hearts were sectioned from apex to base into six 2 mm. Tissue slices were incubated in a 1% TTC solution, pH 7.4, at 37°C for 20 min. The tissues were fixed in 10% PBS-buffered formalin overnight at 4°C. The hearts were sectioned at horizontal axis, and the size of the MI was evaluated as a percentage of the sectional area of the infarcted tissue of the left ventricle (LV) to the sectional area of the whole LV. Both sides of each TTC-stained tissue slice were photographed with a digital camera.
To examine fibrosis area and apoptotic cells on histological analysis, each heart was perfusion-fixed with 10% (v/v) neutral-buffered formaldehyde for 24 h, transversely sectioned into four comparably thick sections, and embedded in paraffin using standard methods (28). Five-micrometer-thick sections were mounted on gelatin-coated glass slides to ensure that different stains could be used on successive tissue sections cut through the implantation area.
Fibrosis was analyzed by Masson's trichrome staining. The interstitial fibrosis areas in the MI following injection with PMA-treated MSCs were measured using metamophor and expressed as a percentage of the total LV.
The terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) assay was performed according to the manufacturer's instructions (Chemicon International, Temecula, CA). In brief, 5-μm-thick tissue sections were deparaffinized, rehydrated, and rinsed with PBS. The sections were pretreated with 3.0% H2O2, subjected to terminal deoxynucleotidyl transferase enzyme for 37°C for 1 h, and incubated in digoxigenin-conjugated nucleotide substrate at 37°C for 30 min. Nuclei exhibiting DNA fragmentation were visualized by adding 3,3- diamino benzidine (DAB; Vector Laboratories, Burlingame, CA) for 5 min. Lastly, sections were counterstained with methyl green. A coverslip was place on top of each section, and the sections were observed by light microscopy. For each group, five slices were prepared and five different regions were observed in each slice.
Left Ventricular Catheterization
Left ventricular catheterization was performed after infarction. While the animal was under anesthesia, a Millar Mikro-tip 2F pressure transducer (model SPR-838, Millar Instruments, Houston, TX) was introduced into the left ventricle via the right carotid artery. Real-time pressure–volume loops were recorded by a blinded investigator, and all data were analyzed offline with PVAN 3.5 software. The average was calculated of three measurements consisting of hemodynamic results of at least 10 pressure–volume loops. Relative volume units were normalized with heparinized blood in calibrated cuvettes.
Statistical Analysis
Data are presented as means ± SEM. The significance of differences between two groups was assessed by Student's t test. Comparisons between more than two groups were performed by one-way ANOVA using Bonferroni's correction. A value of p < 0.05 was considered significant.
Results
Effects of PKC Activation on MSC Adhesion
Adherence of the PMA-treated MSCs was observed from 2 to 8 h. At 4 h, there was a difference of the adhesion between DMSO-treated and PMA-treated MSCs (data not shown). MSC adhesion was induced by PMA at a concentration of 100 nM and gradually increased in a dose-dependent manner (Fig. 1A).
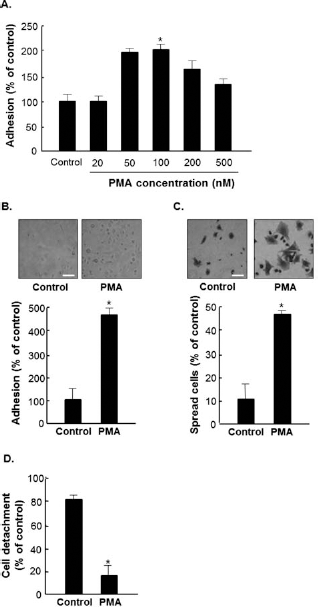
Changes in mesenchymal stem cell (MSC) adhesion induced by treatment with phorbol 12-myristae 13-acetate (PMA). (A) The adhesive properties of cultured MSCs treated with protein kinase C PKC activator (PMA). PKC activator was added when the cells were seeded, and the plates were incubated for 4 h under growth conditions at 37°C and 5% CO2. The plates were washed, and cell viability of adherent cells was assessed by Coomassie blue staining. (B) Representative microphotographs of MSCs treated for 4 h with PMA (100 nM, right) or with dimethyl sulfoxide (DMSO) (left). Scale bars: 100 μm. (C) Cell spreading morphology of MSCs treated with DMSO (left) or PMA (right) for 4 h. Scale bars: 100 μm. (D) The same number of MSCs was plated in each plate. The next day, PKC activators were added to each plate and a trypsinization assay was performed to measure the cell detachment rate. *p < 0.05 versus control.
In this study, MSCs were treated with a PKC activator, and adhesion was measured by an adhesion assay and a spreading assay since cell spreading is involved in cell adhesion and migration. The adhesion rate of MSCs increased about 4.4-fold with treatment of 100 nM PMA for 4 h (Fig. 1B). To determine whether PMA modulates spreading, the cells were stained with Coomassie brilliant blue. Spreading cells showed that MSCs treated with PKC activator were increased about 4.5-fold compared to control group (Fig. 1C). To examine the effect of PKC activator on adhesion rate, MSCs were plated for 1 day and treated with PMA for 4 h. Each plate was subjected to a trypsinization assay. As shown in Figure 1D, MSCs treated with PMA had an adhesion rate of 4.85-fold higher than that of normal controls and showed a reduced detachment ratio. The data confirmed that an increased adhesion rate led to increased cell survival.
Effect of PKC Activation and Inhibition on MSC Adhesion
To confirm the effect of PKC activation and inhibition on adhesion rate, a cell-spreading assay was performed. Cells were treated with PMA and rottlerin. MSCs treated with PKC activator PMA showed increased adhesion compared to the control group, whereas MSCs treated with rottlerin showed decreased adhesion. MSCs treated with both activator and inhibitor exhibited adhesion rates similar to those of the control group (Fig. 2A). The detachment assay showed the same trend as in Figure 1D. MSCs treated with PMA had detachment ratios that were 25% of the ratios in the control group, whereas MSCs treated with both activator and inhibitor had detachment ratios similar to that of the control group (Fig. 2B).

Changes in MSC adhesion induced by PKC activator and inhibitor. (A, B) Representative microphotographs of MSCs treated with PMA (100 nM) and rottlerin (3 μM). (A) Cells were treated with DMSO or PMA for 4 h after rottlerin treatment for 10 min. *p < 0.001 versus control and **p < 0.001 versus PMA only. Scale bars: 100 μm. (B) When MSCs were treated with both PKC activator and inhibitor, the cells were treated in each plate with the PKC inhibitor for 10 min, then with the activator. Each plate was subjected to a trypsinization assay. Scale bars: 100 μm. *p < 0.05 versus control, and **p<0.05 versus PMA only.
Changes in Cell Adhesion to the Cardiogel Following Treatment with PKC Activator and Inhibitor
Cardiogel mimics the environment of myocardial cells using cardiac fibroblasts (1,5). The distinctive 3D matrix adhesions differ from classically described 2D adhesions in structure, localization, and function. As shown in Figure 3, cell adhesion to the cardiogel 3D matrix significantly increased in PMA-treated MSCs compared to MSCs treated with DMSO. Cells grown for 2 h were compared to cells grown for 16 h. The difference in adhesion was over twice greater on cardiogel than on the fibronectin-coated plates (Fig. 3A, B).
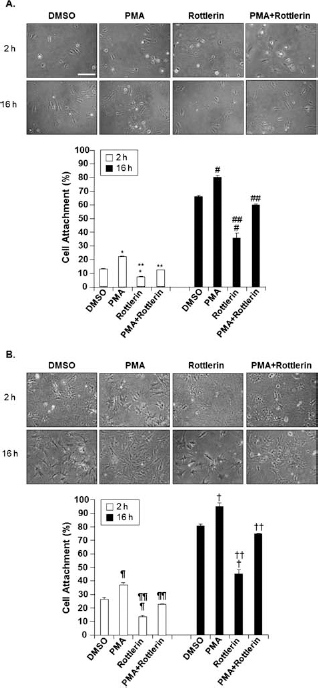
Adhesion rate of MSCs treated with PKC activator or inhibitor on 2D and 3D matrices. (A) Adherence of MSCs in a 2D fibronectin (Fn) matrix. MSCs were plated on a Fn-coated plate and incubated for 2 h (upper panel) and 16 h (lower panel). *p < 0.001 versus DMSO, **p < 0.001 versus PMA, #p < 0.005 versus DMSO and ##p < 0.005 versus PMA. (B) Adherence of MSCs in a 3D cardiogel matrix. Cells were grown on cardiogel for 2 h (top) and 16 h (bottom). ¶p < 0.01 versus DMSO, ¶¶p < 0.01 versus PMA, †p < 0.05 versus DMSO, and ††p < 0.005 versus PMA. Cells were treated with PKC activator and inhibitor at the time of plating. Several separate fields were photographed using a phase contrast microscope. Scale bars: 50 μm.
Changes in Adhesion-Related Signaling by Regulation of PKC Activation
Previous results showed that PMA-induced upregulation of PKC affected the adhesion ability of MSCs. The expression of adhesion-related molecules in MSCs treated with PKC activator was evaluated using immunoblot analysis to determine whether adhesion of MSCs causes an increased upregulation of adhesion-related signaling.
In particular, there was an increase in phosphorylation of FAK (p-FAK) in MSCs treated with only PKC activators relative to control MSCs (Fig. 4A); in MSCs treated with PMA, p-FAK increased by 28.5% compared to the nontreated group. There was also an increase in expression levels of paxillin, talin, and vinculin following PMA treatment; paxillin expression increased by 46.2% (Fig. 4B), talin expression increased by 33% (Fig. 4C), and vinculin expression increased by 20% (Fig. 4D). However, expression of these proteins was decreased in MSCs treated with PKC inhibitor and was recovered in cells treated with both PKC activator and inhibitor compared to PKC inhibitor-treated group; FAK phosphorylation increased by 50%, paxillin expression increased by 50% (Fig. 4B), talin expression increased by 45% (Fig. 4C), and vinculin expression increased by 47.5% (Fig. 4D).

Expression of adhesion-related proteins and small-GTPase proteins in MSCs treated with PKC activator. Each panel shows Western blot data for the expression of adhesion-related proteins. (A) MSCs treated with PMA expressed phosphorylated focal adhesion kinase (FAK). Densitometric analysis was performed, and the ratio of the expression level was calculated relative to that of the nonphosphorylated form. MSCs treated with PMA showed higher expression of paxillin (B), talin (C), and vinculin (D) than the other groups. (E) Activity of the Rho GTPase Rac-1 was examined by Western blot analysis. Densitometric analysis was performed, and the ratio of the band intensity of each protein was calculated relative to that of β-actin. *p < 0.01 versus nontreated group and **p<0.05 versus PMA-treated group.
Altered Expression of Small GTPase-Proteins in MSCs Treated with PKC Regulators
The expression of Rac-1 was measured to investigate the activity of the ras homolog gene family guanosine triphosphatase (Rho GTPases). At cell adhesion sites, PKC can impact the actin cytoskeleton through its influence on Rho GTPases, and Rac-1 is downstream of phosphoinositide 3 (PI3)-kinase and PKC in a pathway involving integrin β cytoplasmic domain function in cell spreading. The expression level of Rac-1 was increased twofold in MSCs treated with PMA and decreased in cells treated with the inhibitor (Fig. 4E). When MSCs were treated with both activator and inhibitor, Rac-1 expression was similar to that of control cells.
Expression and Colocalization of Adhesion-Related Molecules and Stress Fibers in MSCs Treated with PKC Regulators
Next, immunocytochemistry was used to test whether or not adhesion molecules colocalized within the cell. Double immunostaining showed colocalization of vinculin and p-FAK (Fig. 5A). Colocalization of integrin α5 (α5β1) and paxillin (Fig. 5B) was also observed, as well as colocalization of integrin αvβ3 and paxillin (Fig. 5C) and integrin αV and vinculin (Fig. 5D). MSCs treated with PMA exhibited more intense colocalization than the MSCs in the control group. The intensity of colocalization in MSCs treated with the PKC inhibitor rottlerin was lower than that in the control group, whereas MSCs treated with both PKC activator and inhibitor showed similar signal intensity to the control. Specific staining of several focal adhesion molecules was observed in the MSCs, and there was a distinct difference between control MSCs and MSCs treated with PMA.

Intracellular localization of adhesion-related proteins in MSCs treated with PKC activator and inhibitor. (A) Immunofluorescence staining of vinculin and phosphorylated FAK. (B) Immunofluorescence staining of paxillin and integrin α5 showed their colocalization in the cytosol. (C) Immunofluorescence staining of paxillin and integrin αvβ3. (D) Immunofluorescence staining of vinculin and integrin αV. MSCs were stimulated with 100 nM PMA and 3 μM rottlerin, fixed at each time point, and stained with secondary antifluorescein isothiocyanate (FITC), rhodamine, and DAPI. The localization of adhesion-related molecules and nuclei was observed using a confocal laser scanning microscope. Scale bars: 20 μm.
MSCs Treated with PKC Activator Improved Myocardial Infarction In Vivo
To address the effect of PMA-treated MSCs in vivo, DAPI-labeled MSCs were transplanted into the border region between the infarcted area and the normal area. Most of the cells treated with PKC activator were attached to the infarct border zone of the LV (Fig. 6A). To further evaluate the adhesive function of MSCs by PKC activation in vivo, we observed a histological analysis in cell-transplanted rats. We determined the infarct sizes in the LV using TTC staining. PMA-MSC induced a 4.1-fold decrease in infarct size compared to sham (Fig. 6B). Using Masson's trichrome stain, we investigated the degree of fibrosis in the infarct zone. Fibrotic tissue was substantially lower in PMA-MSC group than in sham group by about 15% (Fig. 6C). The incidence of TUNEL-positive myocardial cells was significantly reduced by 20% in the ligated hearts transplanted with PMA-MSCs compared to sham (Fig. 6D).

Implantation of MSCs treated with PKC activator into rat hearts. (A) Detection of viable MSCs. Stem cell engraftment after transplantation into infarcted myocardium was analyzed by counting the numbers of DAPI-stained MSCs. Scale bars: 100 μm. (B) Measurement of infarct size. Transplantation of MSCs reduced the left ventricle (LV) infarct size as assessed by 2,3,5-triphenyltetrazolium chloride (TTC) staining at 1 week post-MI. The outline is infarct zone. Scale bars: 2 mm. (C) Observation of fibrosis area. Fibrosis area/LV in heart was stained by Masson's trichrome. Original magnification (bottom): 200x. Scale bars: 2 mm (top) and 100 μm (bottom). (D) Quantitative analysis of apoptotic cell index. Apoptotic cells were stained by terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL). Arrows are TUNEL-positive cells. Original magnification (bottom): 200x. Scale bars: 100 μm. *p < 0.05 versus MSC.
To measure the heart function by cell therapy, we performed invasive hemodynamic measurements by closed-chest approach. The cardiac function of rats in the PMA-MSC group, which was calculated using pressure–volume loops, showed improvement after MI at 3 weeks. In parameter, end-systolic volume was decreased in PMA-MSC group compared to sham (79.70 ± 3.33 vs. 105.37 ± 16.98, p < 0.01) or MSC group (79.70 ± 3.33 vs. 93.99 ± 14.69, p < 0.01). Stroke volume of PMA-MSC was also improved. In systolic indices, hearts of PMA-MSC group had a dramatically higher ejection fraction 21 days post local cell implantation compared to the sham (62.27 ± 1.74 vs. 41.97 ± 4.38, p < 0.01) or MSC group (62.27 ± 1.74 vs. 49.37 ± 3.92, p < 0.01). Furthermore, stroke work was also significantly improved in the PMA-MSC group compared to the MSC-transplanted group (Table 1).
Effect of PMA-MSC on Cardiac Function in MI

PMA-MSC, phorbol 12-myristate 13-acetate-treated mesenchymal stem cell; ESV, end-systolic volume; EF, ejection fraction; SV, stroke volume; SW, stroke work.
p < 0.01 versus PMA-MSC.
p < 0.05 versus PMA-MSC.
Discussion
In this study, we investigated MSC modification by pretreatment with PKC activator. Previous research has highlighted the importance of PKC in integrin-mediated cell adhesion and spreading, as well as in cell migration, FAK phosphorylation, and focal adhesion formation. Different approaches have been used to study the specific role of PKC in integrin signaling. PKC also appears to be a key intermediate between integrins and FAK signaling in muscle cells and other cell types. Several studies have indicated that PKC activation is required for FAK phosphorylation in cells. Although PKC and FAK colocalize at focal adhesion sites, the precise functional relationship between these two kinases is not known (9,13,21–23,36).
This study provides evidence that key signaling small molecules can control factors downstream of the PKC pathway that may mediate cellular changes. These results add to the understanding of the cellular components involved in the integrin-signaling cascade and the regulation of cellular adhesion and spreading (Fig. 1).
To determine the importance of PKC activation in the adhesion process, MSCs were treated with PKC activator and inhibitor. The adhesion rate of the cells treated with PKC activator increased, whereas the adhesion rate of cells treated with PKC inhibitor decreased, when compared to MSCs in the control group. When MSCs were treated with both activator and inhibitor, the adhesion rate was similar to that of the control group, although there were minor differences experiments (Fig. 2).
Fibronectin and vitronectin, components of the extracellular matrix, are involved in cell growth and development (4). For this reason, a 3D matrix experiment was performed to demonstrate the functional effect of PKC activator on cells after in vivo culture. Previous studies have shown that, during in vivo cardiogenesis, myofibrils are initially distributed in sparse, irregular myofibillar arrays, which gradually mature into parallel arrays of myofibrils and ultimately align into densely packed sarcomeres (28,36). In general, the adhesion rate in cardiogel increased, and cells treated with PKC activator showed a greater adhesion rate increase than the other samples (Fig. 3).
PMA is a nonphysiological activator of PKC. In previous reports, PMA has been shown to influence cell adhesiveness. Other studies report that ligand stimulation of tyrosine kinase growth factor receptors can activate PKC in several cell types (2). Therefore, it is possible that PKC activator will not only affect cell adhesion but will also influence cell survival. Heidkamp et al. reported that increased cell survival is involved in downstream of activation of the mitogen-activated protein kinase (MAPK) cascades and demonstrated an increase in extracellular signal-related kinase 1/2 (ERK1/2) activation in neonatal rat ventricular myocytes after 8 h of PKC epsilion overexpression. Their studies examined ERK1/2 activation at 4, 8, 24, and 48 h after PKC induction (11,14) and indicate that PKC signaling is related to cell survival. Cell survival is in turn related to cell adhesion, and so increased cell survival is related to increased cell adhesion. PKC levels in the cell cytosol correlate with diacylglycerol (DAG)-induced formation of FAK/Src signaling complexes important for regulating the adhesion machinery (11,12,14). The integrin cytoplasmic domain plays a key role in focal contacts and is involved in the activation of FAK. Integrins also bind the proteins paxillin and vinculin (28). Integrins in the extracellular matrix associate with paxillin and vinculin and then crosslink actomyosin stress fibers and attach the fibers to focal contacts (14). Subsequently, integrin clustering leads to association of the protein tyrosine kinase Src and the adaptor protein p130Cas with the membrane also associated with the focal contacts (32).
We showed that modulation of the PKC signal in MSCs affected their adhesion. It was already known that PKC activation is significantly induced before FAK phosphorylation during adhesion (25,30,35). Here, we showed that activation of PKC by PMA induced increased adhesiveness of MSCs and increased protein synthesis of integrin (Figs. 4 and 5).
In downstream signaling, RhoA is induced by PMA, suggesting that PMA plays a role upstream of the RhoA GTPase Rac-1 in controlling cell adhesion (13). Rac-1 expression increased in the MSCs treated with PKC activator. In contrast, cells treated with the PKC inhibitor rottlerin exhibited decreased Rac-1 expression (33). RhoA GTPase is one of the critical regulators of cell motility and cytoskeleton function. Previous studies examined the role of RhoA GTPase in the regulation of monocyte adhesion to vascular endothelium using monocytes pretreated with a 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitor or a statin (33). Although a similar inhibitory action toward RhoA has been shown for statin, the mechanisms underlying the pyridines are not likely to inhibit intracellular cholesterol synthesis or the important intermediates required for the activation of small GTP proteins, including the RhoA GTPase Rac-1 (33). Instead of statin, PKC activator was used in this study. Statin and PKC activator show similar activation of MSCs; they both trigger downstream activation of the Ras/MAP kinase and Rho/Rac-1 pathways (37). It has been demonstrated that Rho family GTPases play important roles in the regulation of integrin clustering and the subsequent interaction of integrin with focal adhesion molecules (19,39). This explains the activation of the Rac-1 protein observed in PMA-treated MSCs (Fig. 4E). Colocalization of adhesion-related molecules was demonstrated using immunofluorescence methods (Fig. 5). Moreover, vinculin and p-FAK were colocalized, indicating that the stress fibers were affected by the phosphorylation of FAK (Fig. 5A). Colocalization of integrin and paxillin was also demonstrated (Fig. 5B, C). MSCs treated with PMA showed increased colocalization compared to the control group, compared to MSCs treated with inhibitor, and compared to MSCs treated with both activator and inhibitor. Colocalization of integrin-vinculin was also confirmed (Fig. 5D).
The in vivo portion of this study aimed to confirm the survival of modified MSCs that were treated with PKC activator in rat hearts. PMA-treated MSCs were shown to have enhanced survival in the myocardium, as assessed by counting the number of DAPI-labeled cells (Fig. 6A). According to our in vivo histological data, transplantation of PMA-MSCs resulted in a further decrease in infarct size, fibrosis area, and apoptotic cell index (Fig. 6B, C). In the functional test, the cardiac function of rats in the PMA-treated MSC-transplanted group improved significantly as assessed by various parameters (Table 1).
In conclusion, the activation of PKC induces cell adhesion, which in turn enhances PKC signaling and integrin signaling complexes, resulting in activation of FAK and its downstream pathways. These findings may explain the relationship between activation of PKC by small molecule stimulation and cell adhesion as follows: formation of a complex of activated PKC/integrin signal/FAK may enhance cell adhesion through phosphorylation and activation of FAK. In an in vivo study, when MSCs treated with PKC activator were injected into the MI rat heart, the cells showed effective adhesion in the border zone and association with myocytes. Targeted therapies that activate PKC in MSCs may improve the survival of injected cells in the treatment of cardiac MI.
Footnotes
Acknowledgments
This research was supported by a Korea Science and Engineering Foundation Grant funded by the Korea government (MEST) (2011-0019243, 2011-0019254), a grant from the Korea Health 21 R&D Project, Ministry of Health and Welfare, Republic of Korea (A085136 and A120478). The authors would like to thank Minji Seung from the Undergraduate Biological Science Studies at University of California at Santa Barbara and SoMin Jeon from the Daewon Foreign Language High School at Seoul, Republic of Korea, for technical assistance and research involvement. The authors declare no conflict of interest.
