Abstract
This study aimed to determine the potential of purified (Stro-1+) human mesenchymal precursor cells (hMPCs) to repair the injured spinal cord (SC) after transplantation into T-cell-deficient athymic RNU nude rats following acute moderate contusive spinal cord injury (SCI). hMPCs were isolated from the bone marrow (BM) stroma of SCI patients and transplanted as a suspension graft in medium [with or without immunosuppression using cyclosporin A (CsA)]. Extensive anatomical analysis shows statistically significant improvement in functional recovery, tissue sparing, and cyst reduction. We provide quantitative assessment of supraspinal projections in combination with functional outcomes. hMPC-transplanted animals consistently achieved mean BBB scores of 15 at 8 weeks postinjury. Quantitative histological staining revealed that graft-recipient animals possessed more intact spinal tissue and reduced cyst formation than controls. Fluorogold (FG) retrograde tracing revealed sparing/regeneration of supraspinal and local propriospinal axonal pathways, but no statistical differences were observed compared to controls. Immunohistochemical analysis revealed increased serotonergic (5-HT) and sensory (CGRP) axonal growth within and surrounding transplanted donor hMPCs 2 weeks posttransplantation, but no evidence of hMPC transdifferentiation was seen. Although hMPCs initially survive at 2 weeks posttransplantation, their numbers were dramatically reduced and no cells were detected at 8 weeks posttransplantation using retroviral/lentiviral GFP labeling and a human nuclear antigen (HNA) antibody. Additional immunosuppression with CsA did not improve hMPC survival or their ability to promote tissue sparing or functional recovery. We propose Stro-1+-selected hMPCs provide (i) a reproducible source for stem cell transplantation for SC therapy and (ii) a positive host microenvironment resulting in the promotion of tissue sparing/repair that subsequently improves behavioral outcomes after SCI. Our results provide a new candidate for consideration as a stem cell therapy for the repair of traumatic CNS injury.
Introduction
Spinal cord injury (SCI) results in disconnection of descending motor and ascending sensory pathways, leading to sensorimotor dysfunction and loss below the lesion. To date, a number of cellular transplantation approaches have been used in attempts to address these issues, reviewed in (55,80). Adult bone marrow stromal cells (BMSCs) have been transplanted into the mammalian nervous system including brain (43), peripheral nerve (32), and spinal cord (49) and are reported to (a) promote regeneration of lesioned axons into the graft (31,74), (b) differentiate into neurons (31), (c) remyelinate damaged myelin sheaths around CNS axons (2), and (d) improve the functional outcome after SCI. (31,46,53,73,74).
Most preclinical studies involving the application of adult BMSCs in the context of SCI treatment use heterogeneous populations of rat, human, or mouse cells (3). This can complicate the interpretation of results, making it uncertain whether the observed effects are due to the whole population or a specific subset of donor BMSCs. CD49c+/CD90+ sorted BMSCs (from different, normal human donors) have been reported to improve recovery in immunosuppressed rats after mild–severe SCI (29); modest results were also obtained with unpurified heterogeneous populations from four different normal human donors used to treat C3–C4 hemisection SCI in rats (53). Moreover, major differences in specific BMSC capabilities such as growth promotion, proliferation, differentiation, and cytokine profiles between different human donors (53,56) and even within the same donor (57,68) are likely to contribute to the outcome of hBMSC transplantation.
There is increasing interest in the application of stem cells from adult tissues as a basis for regenerative therapies, as these self-renewing cells have the capacity for multilineage differentiation and may act as promoters of tissue regeneration and repair under normal physiological conditions. Of particular interest in this regard are mesenchymal precursor cells (MPCs) (21,24,62). Originally described as precursor cells for the stromal tissue of the bone marrow (22), MPCs are defined by their capacity for differentiation into a range of tissues of mesodermal origin including bone, cartilage, and adipose tissue (16). The interest in potential therapeutic application of MPCs (52,77) is also due to the relative ease of isolation from bone marrow aspirates and in vitro cultivation. There is also data indicating the unique immunosuppressive properties of MPCs, which has led to the suggested application of these cells in both autologous and allogeneic transplant settings. With their reported multipotent differentiation potential, human MPCs (hMPCs) represent an attractive alternative source of stem cells for cellular therapies.
To date purified hMPCs, via a single immunoselection, from SCI patients have not been directly applied to SCI as a therapy; specifically highly purified Stro-1+ hMPCs that represent a restricted minor subpopulation of the stromal fraction from which all adult lineages derive. We are using the term hMPCs to define our Stro-1+ precursor population of cells that produce an adult BMSC population.
Putative stromal cell precursors have been identified in a wide variety of species, including human, by the ability to form colonies of fibroblasts as single cell preparations. The clonogenic progenitor responsible for fibroblast colony-forming units (CFU-Fs) is a small minor population that generates the full spectrum of stromal cell types, unlike other colonies that form only restricted tissues such as bone and soft connective tissue. Donor hMPCs can be purified to a minor subpopulation of adult cells that are characterized as CFU-Fs using stringent adherence techniques and/or fluorescence-activated cell sorting (FACS) using the monoclonal antibody Stro-1 (70). Stro-1 reacts with a trypsin-resistant cell surface antigen that is non-reactive with hematopoietic progenitors and is expressed by essentially all detectable clonogenic CFU-Fs (70).
The development of hMPCs are necessary for use in experimental studies aimed toward potential clinical applications; ideally, donor populations of hMPCs should be purified according to guidelines for the conduct of clinical trials for SCI [such as those developed by the International Campaign for Cures of Spinal Cord Injury Paralysis (ICCP)] (17). The present study uses Stro-1-selected hMPCs from SCI patients, since the use of autologous hMPCs from injured patients (73) would be a preferred protocol in transplantation therapy. Currently, populations of hMPCs are being utilized in clinical studies for diseases such as congestive heart failure, spinal fusion, and bone marrow regeneration.
In this study, purified multipotent hMPCs isolated from SCI patients transduced to express enhanced green fluorescent protein (GFP) (hMPCsGFP) by viral transduction were transplanted into an athymic RNU nude rat model following a moderate contusion injury; this was also performed with cyclosporin A (CsA) immunosuppression in a cohort of animals to ascertain potential beneficial outcomes from these hMPC therapies in combination with immune suppression. hMPCGFP therapy resulted in a marked improvement in functional behavioral recovery and promoted tissue sparing after acute injury, which highlights this selected cell population as a potential cellular therapy for SCI.
Materials and Methods
Animals
Adult female CBH-rnu/Arc (Athymic Nude) rats (120–150 g; Animal Resource Centre, Western Australia) were used in this study. All experimental procedures conformed to National Health and Medical Research Council Guidelines (Australia) and were approved by the University of Western Australia Animal Ethics Committee.
Isolation of hMPCs
Donor hMPCs were isolated and propagated using methods described previously (22,23). In brief, BM was aspirated from the iliac crest of two male volunteer donors with SCI after informed consent and ethical approval. The BM samples were separated over a Ficoll-Paque density gradient to yield light density (<;1.077 g/dl; Lymphoprep, Nycomed, Oslo, Norway) mononuclear cells. Following washing to reduce platelet contamination, the cells were resuspended in α-minimum essential medium (α-MEM; Gibco, Gaithersburg, MD, USA) supplemented with 20% (v/v) fetal calf serum (FCS; Invitrogen, Carlsbad, CA, USA) and transferred into tissue culture flasks precoated with human fibronectin (Sigma-Aldrich, St. Louis, MO, USA). Cultured cells were incubated at 37°C in low oxygen tension (5%, v/v), and after 2 days the nonadherent cells were removed. Adherent cells were maintained in α-MEM/20% FCS and then transduced with a retroviral vector encoding-enhanced green fluorescence protein (GFP) (67,82) (Fig. 1). These cells were denoted as hMPCsGFP and stored for transplantation experiments (see below); a subsample was also confirmed for Stro-1 expression using FACS (Developmental Studies Hybridoma Bank, University of Iowa, Iowa City, IA, USA) at passage number 1 (P1) in vitro.
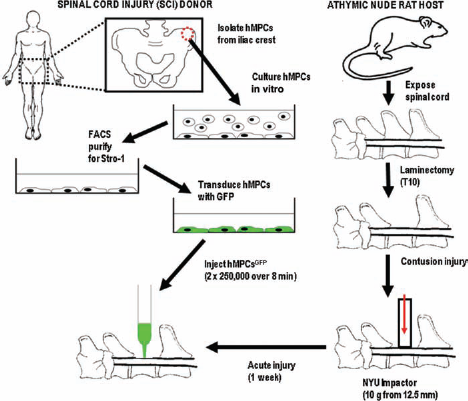
Schematic for spinal cord therapy (SCI) therapy using donor human mesenchymal precursor cells (hMPCs) isolated from SCI patients. Isolated donor hMPCs are grown in culture, and transduced with green fluorescent protein (GFP) using a retrovirus. Frozen hMPCsGFP are thawed, washed, and then injected into the contused spinal cord of athymic nude rats 1 week after a moderate contusion injury using an NYU impactor device. Donor Stro-1-selected hMPCs were also tested for Stro-1 reactivity after 24–48 h in culture using fluorescence-activated cell sorting (FACS). As a control, human dermal fibroblasts transduced with GFP (hDFsGFP) were also transplanted.
In Vitro Culture of hMPCs
Thawed hMPCsGFP that were not used for transplantation were grown in α MEM/10% FCS in order to determine any neuronal phenotype/marker expression (83). The cells were stained using immunocytochemistry techniques after 24–48 h in culture using the panel of antibodies described in the immunohistochemistry section below.
Human Dermal Fibroblast (hDFs)
The normal human dermal fibroblast (CC-2511, Lonza, Inc., Williamsport, PA, USA) was used as a control cell type for injections after SCI in cohorts of animals (n = 8) and also for some in vitro culture experiments as a direct comparison for donor hMPCsGFP. All procedures used for hMPCs transplantation were reproduced when using donor hDFs. The hDFs were routinely maintained in Dulbecco's modified Eagle's medium (DMEM; Gibco) plus 10% (v/v) FCS. Donor hDFs were transfected with lentiviral GFP (82) (flap-Ub promoter-GFP-WRE; Addgene, Cambridge, MA, USA) at a multiplicity of infection (MOI) of 400 for 16 h prior to use; the resulting hDFsGFP were then transplanted using the same procedure for hMPC transplantation. hDF phenotype/marker expression was also determined after 24–48 h in culture using the panel of antibodies described in the immunohistochemistry section below.
Protocol for Spinal Cord Surgery
Athymic nude rats were anesthetized with 1.5% (v/v) halothane (Rhone-Poulenc Chemicals Pty. Ltd., Baulkham Hills, NSW, Australia) combined with nitrous oxide (60%) and oxygen (38.5%). Amacin ophthalmic eye ointment (Provet, Queensland, Australia) was applied before the rats were placed on a heating pad (37°C). Partial laminectomy at vertebral level T9–T10 exposed the SC underneath without disrupting the dura. Using an NYU impactor (Wise Young; Rutgers University, New Brunswick, NJ, USA) (59), a moderate contusion injury was induced by dropping a 10-g weight from 12.5 mm above the exposed SC. After surgery, the muscles were closed in layers and the incision closed with wound clips. Rehydrating saline (2 ml, IP) was administered immediately following surgery. Rats were housed in filter top cages with ample autoclaved bedding, chow and water. The cages were positioned so as to be half-on/half-off their heating pads (37°C) for 1 day following surgery to provide a cooler environment if needed and prevent hyperthermia and dehydration. To prevent wound and bladder infections, Benacillin [0.02 ml/100 g body weight, 300 U/ml, IM (150 mg/ml procaine penicillin, 150 mg/ml benzathine penicillin, 20 mg/ml procaine hydrochloride; Troy Laboratories Pty. Ltd., Glendenning, NSW, Australia)] was administered at 2, 4, and 6 days after surgery and again after hMPCGFP transplantation (at day 7). Buprenorphine (Temgesic, Provet; 0.01 ml/100 g body weight, 300 U/ml, IP) was also administered daily for 4 days after surgery.
Preparation of hMPCs Suspensions for Implantation
Donor hMPCsGFP isolated from individual SCI patients were used as vials of frozen cells. Prior to transplantation, these cells were thawed, washed in phosphate-buffered saline (PBS), and adjusted to a cell density of 5 × 105 cells in 4 μl of medium (α-MEM only). The viability of hMPCGFP suspensions was determined by trypan blue staining (Sigma; showing at least 95% viability).
Experimental Design
At 1 week following SCI, rats were subjected to the following procedures in regime (A): (i) SCI alone (noted as “no further treatment” or “injury only”), (ii) SCI plus α-MEM medium injection (“injury-plus medium”), and (iii) SCI plus donor cell transplantation (“injury-plus hMPCs” or “injury-plus hDFs”). In regime (B), rats subjected to (i) “injury only” or (ii) “injury-plus medium” and (iii) “injury-plus hMPCs” were also administered the immunosuppressant Cyclosporin A (CsA; LC Laboratories, Woburn, MA, USA; 20 mg/kg, every second day, IM) until sacrifice. For regimes (A) and (B), rats were monitored for functional behavioral recovery and analyzed at 1, 2, 4, and 8 weeks (n = 8 at each time point). All animals subjected to surgery were included for both morphological (immunocytochemical staining, tissue sparing, and Fluorogold tracing) and behavioral analyses described below (i.e., n = 8 for all analyses).
Transplantation of hMPCs, hDFsGFP, and Postoperative Care
Two injections of 2.5 × 105 donor hMPCsGFP and hDFsGFP (total of 5 × 105 cells) in 4 ml medium were delivered into the contused SC (an injection at each side of the center of the lesion) at a 1-mm depth using a small glass capillary (tip diameter, 60 μm) attached to a 10-μl Hamilton syringe. Injections were performed at 1 week (acute) after SCI using a Harvard microinjection apparatus (0.5 μl/min over 8 min). The micropipette was left in place for 1 min to minimize efflux of the inoculum after pipette withdrawal. Muscle layers were sutured and skin wounds closed. Buprenorphine (analgesic; 0.01 ml/100 g body weight, 300 U/ml, IP) and Benacillin (antibiotic; 0.02ml/100 g body weight, 300 U/ml, IM) were administered as described above, in addition to saline injected subcutaneously to prevent dehydration and normalize blood pressure. Control rats received either SCI, SCI with α-MEM injection (no hMPCsGFP), or SCI with hDF injection with no further therapeutic intervention.
Functional Behavior
Performance of the impaired hind limb to assess spontaneous movements of control injured and hDFGFP- and hMPCGFP-transplanted rats was investigated via the open field Basso–Beattie–Bresnahan (BBB) locomotor rating scale (6). The BBB rating score is composed of 22 nonlinear operational definitions (0–21 scale) studying several aspects involved in the locomotion of quadrupedal animals such as weight support, plantar stepping, and forelimb–hindlimb coordination. Rats from all treatment groups were observed walking at different time points following SCI. There were always two observers present who scored each animal independently. BBB scores for each regime were averaged and rounded up from 0.5 to 1.0 or down from 0 to 0.4 to achieve whole numbers. Table 1 represents the BBB scores and descriptive pattern of locomotion for the reported results in this study.
Summary of Key Observed Basso–Beattie–Bresnahan (BBB) Locomotor Rating Scores in This Study (6)

Perfusion and Tissue Processing
At sampling times, rats were euthanized by lethal injection of sodium pentobarbitone (Provet; 50 mg/100 g) and transcardially perfused with 100 ml of heparinized Dulbecco's PBS wash (Gibco), followed by 100 ml of 4% (w/v) paraformaldehyde (Sigma) in PBS pH 7.4. The head and vertebral column were dissected from each animal and postfixed for 24 h. The brain and spinal cord were extracted from the skull and vertebra and then stored intact in 0.1 M PBS (pH 7.4). The position of the injury in the SC was measured from the caudal edge of the cerebellum to confirm that all animals were lesioned at the same level. A 2-cm segment was cut from the SC, with the lesion at the midpoint of this segment, and embedded in 1% (w/v) gelatin (Sigma). Using a CO2-freezing microtome (Polycut, Reichert-Jung, Australia), proximal and distal SC close to the grafts (1 cm) was cut sagitally in 40-μm frozen sections, while the brain and brainstem were cut transversely in 50-μm frozen sections. Consecutive series of sections were transferred to 24-well plates containing 0.1 M PB with 0.01% (w/v) sodium azide (Sigma) and stored at 4°C until processed for immunohistochemistry.
Tissue Sparing
Assessment of spinal tissue sparing was carried out using cresyl violet staining on every fourth sagittal section to determine the volume of spared spinal tissue (55). In each section, the total number of pixels in a 2.5-mm-long SC segment was determined, with the lesion epicenter in the middle, as well as the area of damaged spinal tissue around it. The border of the damaged tissue was defined by the absence of healthy cells and an obvious discontinuity in density. Measurements of each section were summed per rat and multiplied by 4 (four sections were analyzed across the lesion) to give the total number of pixels in the analyzed SC segment. Spared tissue was calculated as the difference between the numbers of pixels in the area of damaged tissue versus the whole segment.
Quantitative Analysis of Retrograde Axonal Tracing, Fluorogold Cell Labeling, and Tissue Sparing
Axon sparing and regrowth was evaluated using mapping and counting of Fluorogold-labeled neurons in spinally projecting brain areas using one in six brain sections for (i) “injury only,” (ii) “injury-plus medium,” and (iii) “injury-plus hMPCsGFP” groups. hDFsGFP-treated animals were not included in this analysis. A total of 0.5 ml of 4% (w/v) Fluorogold (FG, Fluorochrome LLC, Denver, CO, USA) in 0.9% saline was injected via a small glass capillary (tip diameter, 60 μm) using a Drummond Nanoject II device (World Precision Instruments, Sarasota, FL, USA) into both left and right sides of the spinal cord at 0.75- and 1.25-mm depths distal to the injury (8 mm from the distal part of the contusion site) (59) at 2 weeks before sacrifice. The 8-mm distal injections were utilized to avoid leakage of the FG into the lesion site as well as the proximal spinal cord; any leakage would prevent correct labeling of projecting supraspinal projections to the distal spinal cord. Analysis of 50-μm transverse brain sections was performed after image acquisition using a fluorescence microscope (DMRBE, Leica, Wetzlar, Germany) with a motorized XYZ stage, a Nikon 1200F digital camera, Image ProPlus 4.5.1 software, and a Scope Pro image tiling plug in (Media Cybernetics, Rockville, MD, USA). Excitation of FG was performed at 400–450 nM and FG-positive cells in each brain section were analyzed using its emission band of 500–600 nM. Total numbers of FG-labeled cells were counted in vestibular, reticular, red nucleus, cortical layer V, raphe, and trigeminal regions throughout the whole brain.
Analysis of SC Sections Using Immunohistochemistry
The anatomical relationship between grafted cells and regrowing axons for each regime, and whether donor cells differentiate under influence of the surrounding host SC environment was assessed. This was performed via analysis on frozen longitudinal sections across the contused area of the SC using a panel of antibodies directed against: GFP (#AB3080, Millipore, Billerica, MA, USA), human nuclear antigen (HNA, #MAB1281, Millipore, USA), β-III tubulin (#PRB-435P, Covance, Princeton, NJ, USA), PAN neurofilament (#18-0171, Zymed, San Francisco, CA, USA; data not shown), anti-neurofilament RT97 (monoclonal antibody supernatant), glial fibrillary acidic protein (GFAP, #Z0334, Dako, Glostrup, Denmark), p75 (#G323A, Promega, Madison, WI, USA), S100 (#Z0311, Dako), axonal markers 5-hydroxytryptamine (5-HT; serotonergic, #20080, Immunostar, Inc., Hudson, WI, USA), calcitonin gene-related peptide (CGRP; sensory) (#24112, Diasorin, Stillwater, MN, USA), and microtubule-associated proteins MAP5 (early) (#M4528, Sigma; data not shown) and MAP2A (late) (#M1406, Sigma; data not shown). Fibronectin (FN) (#F3648, Sigma), laminin (LAM) (#L9393, Sigma) and proliferating cells (proliferating cell nuclear antigen [PCNA, #MAB424, Millipore] and Ki67 [#MAB4190, Millipore]) were analyzed, as well as infiltrating CD68+ve macrophages (ED1, #MCA341R, Serotec, Raleigh, NC, USA). Immunostaining patterns were compared between hMPCGFP-transplanted rats and unimplanted lesioned control animals. Dilutions of primary antibodies were at 1/400 to 1/500 in PBS/1% normal goat serum [NGS]/0.02% Triton X-100, except for ED1 (1/200), PCNA (1/200), HNA (1/40), MAP5 (1/250) and RT97 (1/10). Primary antibodies were detected using cyanine 3 (CY3)-conjugated secondary antibodies (Invitrogen) appropriate for the host species at 1:500 dilution. Semiquantitative analysis of GFP-positive donor cell survival was performed by visual assessment of at least three sections (as centrally located within the spinal cord as possible) of each animal in each treatment group for both GFP and HNA staining. The amount of donor cells observed was graded according to the scale shown in Table 2.
Semiquantitative Assessment of Donor Cell Survival After Acute Spinal Cord Injury (SCI)

Donor human green fluorescent protein expressing mesenchymal precursor cells (hMPCsGFP) or human green fluorescent expressing human dermal fibroblasts (hDFsGFP) were transplanted on day 7 after SCI and remaining numbers assessed visually. No donor hMPCsGFP remained at 1 month after transplantation, whereas a few remaining hDFsGFP were observed 1 month after transplantation. +++, >51 cells per section analyzed; ++, between 11 and 50 cells per section; +, 1–10 cells per section; -, no cells in section analyzed. CsA, cyclosporin A.
Electron Microscopy Analysis of SC Sections
Spinal cords from additional injury-only control (n = 2) and injury-plus hBMSC (n = 2) rats were removed and fixed in 2% (v/v) glutaraldehyde containing 100 mM sucrose and 50 mM phosphate buffer. The graft site was cut transversely into rostral, lesion, and caudal (distal) regions, and sections further fixed in buffered osmium tetroxide were dehydrated and embedded in Epon Araldite. Sagittal sections (1 μm) were stained with toluidine blue. Ultrathin sections of interest were prepared and stained for electron microscopy.
Statistical Analysis
Using GraphPad Prism v4.03 and InStat v3.06 for Windows (GraphPad Software, San Diego, CA, USA), statistical analyses were performed using either one- or two-way analysis of variance (ANOVA), except for BBB scoring, which used a Kruskal–Wallis analysis (nonparametric ANOVA) plus Dunn's multiple comparisons testing.
Results
In Vitro Expression Profile of Donor hMPCsGFP and hDFsGFP
Phenotypic analysis of hMPCsGFP cultured up to 5 days from frozen in α MEM/10% FCS (Fig. 2) revealed no reactivity for GFAP, S100, p75, or RT97 (data not shown). Large amounts of fibronectin (Fig. 2A) and laminin (Fig. 2B) were present. Small amounts of β-III tubulin expressed in the cytoplasm of hMPCsGFP close to the nucleus (Fig. 2C). Control hDFsGFP did not stain positive for any of the cell phenotype markers GFAP, S100, p75, or RT97 described above in vitro (data not shown) but did stain positively for fibronectin (Fig. 2E), laminin (Fig. 2F), and β-III tubulin (Fig. 2G). The human nuclear antigen (HNA) antibody positively identified viable donor hMPCsGFP and hDFsGFP in vitro, since it is constitutively expressed in all human cells (Fig. 2D, H).

Representative photomicrographs of hMPCsGFP and hDFsGFP in vitro: An immunocytochemical analysis of phenotype. Freshly thawed donor hMPCsGFP (A–D) show extensive deposition of fibronectin (FN) (A), laminin (LAM) (B), and moderate cytoplasmic expression of βIII tubulin (BIIITUB) (C). Cultures of hMPCs showed strong nuclear staining of human nuclear marker (HNA) (D). Green fluorescent protein expressing human dorsal fibroblasts (hDFsGFP) (E–H) showed strong staining for fibronectin (E), but lower staining of laminin with a speckled appearance particularly around the cell nucleus (F). Fibrous βIII tubulin expression was seen on the human dermal fibroblasts and showed long spindly cell protractions (G). Immunostaining for HNA also showed strong nuclear expression in the hDFs as with the hMPCs. Fluorescent staining for cyanine 3 (Cy3) (red), GFP (green), and Hoechst 33342 (blue) is shown in all figures. Scale bars: 100 μm.
Functional Behavioral Recovery
Acute (1 week postinjury) hMPCGFP-transplanted animals demonstrated marked improvement of walking patterns (weight support and coordinated gait) compared to the injury-only and medium-injected controls using the open field scoring method BBB (Fig. 3A, B; also refer to Table 1). Similarly, hDFGFP-transplanted animals also showed improved functional recovery compared to injury-only and medium-injected controls, but not to the extent of hMPCGFP-treated animals. The dotted line on each graph indicates donor cell transplantation therapy at day 7 after injury. Rats subjected to contusive thoracic SCI alone without any further therapeutic intervention normally recover to achieve mean BBB scores of 10 (59). Here, control rats recovered to achieve mean BBB scores of 11 from 2 weeks postinjury onwards and eventually leveled out to scores of 12–13 from 4 weeks postinjury until the end of analysis at 8 weeks. In marked contrast, rats transplanted with hMPCsGFP consistently achieved an average BBB score of 15 (Fig. 3A) from 4 weeks posttransplantation (5 weeks postinjury until the end of analysis at 8 weeks). hDFGFP-transplanted animals achieved an average BBB score of 14 from 5 weeks posttransplantation (Fig. 3A).

Open field functional recovery after acute SCI. Average Basso–Beattie–Bresnahan (BBB) scores for athymic nude rats after moderate SCI alone (black square), SCI plus medium injection (red square), SCI with hMPCGFP transplantation (blue triangle), or SCI with hDFGFP transplantation (green triangle) after acute SCI only (A) and acute SCI with additional cyclosporin A (CsA) immunosuppression (B). Standard deviation is shown (n = 8). (A) ∗Significant differences between the injury-only and injury-plus hMPCs (p < 0.05); #significant differences between injury-plus hMPCs and injury-plus hDFs (p < 0.05). (B) Immunosuppression with CsA did not further improve upon hMPC treatment alone (∗p < 0.05, Kruskal–Wallis ANOVA), although significant differences to injury-only were achieved after 42 days posttransplantation with hMPCs rather than 28 days seen in the non-CsA-treated rats. Overall, the functional improvement following hMPC therapy represents a significant switch from noncoordinated locomotor function after injury to a marked, coordinated locomotor recovery.
Significant differences (p < 0.05) between control, injury, and medium and hMPCGFP-transplanted animals (Fig. 3A) began to emerge 3 weeks (day 28 postinjury) after hMPCGFP injection. There was no statistically significant difference in average BBB scores between hDFGFP-transplanted and injury-only and medium-injected controls prior to this time period. At 4 weeks posttransplantation (5 weeks postinjury), functional recovery between hMPCGFP-and hDFGFP-transplanted animals became statistically significant (p < 0.05) (Fig. 3A), but not at 6, 7, or 8 weeks posttransplant.
In CsA-treated rats, hMPCGFP-transplanted animals again showed improvement of walking patterns (weight support and coordinated gait), with average BBB scores of 15 at 4 weeks posttransplantation (5 weeks postinjury), compared to injury-only and medium-injected controls. Interestingly, the averaged BBB scores of both the control injury and medium-injected groups more consistently reached 13 (Fig. 3B), which represented a slightly higher trend than those same groups not treated with CsA.
Transplantation of hMPCs Promote Tissue Sparing
Spinal tissue sparing was calculated from the damaged area (including cysts, intact tissue, degenerate tissue, and transplanted hDFsGFP and hMPCsGFP) from the field of view within a 2.5-mm-long SC segment 1 week and 2 months after transplant (Figs. 4 and 5). There was a significant (p < 0.01) marked reduction in cyst area percentage when comparing both injury-only and injury-plus medium injection control groups to hMPCGFP-treated groups, respectively (Figs. 4B and 5B). There were reductions from (1) 39–40% in injury-only and injury-plus medium to 15% in hMPCsGFP at 1 week posttreatment (Fig. 4B) and (2) from 39–42% in injury-only and injury-plus medium to about 6% in hMPCsGFP at 2 months posttreatment (Fig. 5B). There was also a significant (p < 0.05) marked reduction in cyst area percentage when comparing both injury-only and injury-plus medium injection groups to hDFGFP-treated groups, respectively (Figs. 4B and 5B), with reductions (i) from 39–40% (control) to 23% (hDFGFP-treated) at 1 week (Fig. 4B) and (ii) from 39–42% (controls) to 12% (hDFGFP-treated) at 2 months posttreatment (Fig. 5B).

Reduced cyst size and increased tissue sparing after hMPCGFP and hDFGFP therapy at 1 week posttransplantation. (A) The significant increases in intact tissue remaining (∗p < 0.05) between injury-only and injury-plus hDFs, injury-plus hMPCs and injury-plus hMPC/CsA treatment groups were observed. (B) There was a significant reduction (∗p < 0.05, ∗∗p < 0.01) in cyst volume observed between the injury-only and injury-plus hDFs, injury-plus hMPCs, and injury-plus hMPC/CsA treatment groups. (C) There was a significant reduction (∗p < 0.05) in the level of cyst/degenerate tissue observed between injury-only and injury-plus hDFs, injury-plus hMPCs, and injury-plus hMPC/CsA. Treatment with CsA did not improve tissue sparing outcomes, even when used in combination with hMPC therapy. ∗,∗∗Statistical significance of p < 0.05 and p < 0.01; n = 8.

Reduced cyst size and increased tissue sparing after hMPCGFP and hDFGFP therapy at 2 months posttransplantation. (A) There was a significant increase (∗∗p < 0.01 and ∗∗∗p < 0.001) in intact spinal cord tissue observed between injury-only and injury-plus hDF, hMPC, and hMPC/CsA treatment groups. (B) Treatment with hDFs, hMPCs, and hMPCs/CsA showed significant decreases (∗p < 0.05 and ∗∗p < 0.01) in cyst volume when compared to the injury-only group. (C) There was an observed decrease (∗∗∗p < 0.001) in cyst/degenerate tissue between injury-only and injury-plus hMPC treatment groups. In addition, there was a significant decrease (∗∗p < 0.01) between injury-plus medium and injury-plus hMPC treatment groups. Significant decreases between injury-only and injury-plus hDF and injury-plus hMPC/CsA (∗∗p < 0.01) treatment groups were also observed. Treatment with CsA did not improve tissue sparing outcomes, even when used in combination with hMPC therapy. ∗,∗∗,∗∗∗Statistical significance of p < 0.05, p < 0.01 and p < 0.001, respectively; n = 8.
There were also significant increases in the amount of intact spinal tissue (sparing) following hMPCGFP and hDFGFP transplantation at both 1 week and 2 months, when compared to injury-only or injury and injury-plus medium injection groups (Figs. 4A and 5D). At 1 week posttreatment (Fig. 4A), the amount of intact spinal tissue was significantly higher from 47–52% in injury-only and injury-plus medium injection groups to 68% in hMPCGFP-treated rats and 68% in hDFGFP-treated rats (p < 0.05). At 2 months posttreatment (Fig. 5A), there was a significant increase (p < 0.001) in the amount of intact spinal tissue from 43% injury-only and 51% in injury-plus medium injection groups compared to 89% in hMPCGFP-treated rats. This was in contrast to a significant increase (p < 0.05) in the amount of intact spinal tissue from 43% to 51% in injury-only and injury-plus medium compared to 80% in hDFGFP-treated animals.
A comparison of the percentage cyst plus degenerative tissue at the injury site also revealed a significant reduction (p < 0.01) at 1 week posttreatment (Fig. 4C) from 53% in injury-only and 48% injury-plus medium injection groups to 31% (hMPCGFP-treated) or 32% (hDFGFP-treated) animals. At 2 months posttransplantation treatment significant reductions of cyst degenerative tissue was seen from 57% in injury-only groups (p < 0.001) and from 49% in injury-plus medium injection groups (p < 0.01) were observed when compared to 11% in the hMPCGFP-treated group (Fig. 5C). Significant reductions (p < 0.01) from 57% (injury-only control groups) and from 49% (injury-plus medium injection groups) were observed when compared to 21% in the hDFsGFP. hMPC-transplanted groups treated with CsA showed less reduction of cyst plus degenerate tissue at 2 months posttreatment than hMPCs alone (Fig. 5C). The use of CsA did not affect any of the treatment groups in a number of respects: (1) animals in control (injury-only and injury-plus medium injection groups) with CsA treatment groups were not significantly different to control (no CsA treatment) groups and (2) hMPCGFP treatment plus CsA treatment groups were similar to hMPCGFP treatment only groups in cyst size and cyst and degenerative tissue reductions; (3) there were differences in tissue sparing between hMPCs alone (89%) and hMPCs plus CsA (79%) to control with CsA treatment groups.
Analysis of Transplanted hMPCsGFP: Phenotypic Expression Profile In Vivo
The integration and viability of hMPCsGFP and control hDFsGFP in vivo, using immunofluorescence expression of GFP by hMPCs in combination with anti-HNA immunostaining, allowed both their characterization and analysis of interaction with host cells and axons. If used in combination with ED1 staining, it can assist in assessing whether macrophage scavenging of HNA and GFP has occurred. Unless otherwise stated, there was no apparent difference in either hMPC survival or phenotype expression in vivo following transplantation into nonimmunosuppressed (A) or CsA-immunosuppressed (B) experimental regimes. Donor cell survival was assessed semiqualitatively, because full quantification using GFP and HNA double labeling would rely on the HNA antigen being stable at all postoperative times. Previous quantitative analysis using Hoechst 33342 and GFP was not used in this study because there is no convincing evidence that the technique is accurate, especially when trying to correlate between nuclei and cytoplasmic stains as reporter genes can be silenced or significantly downregulated.
Transplanted hMPCsGFP and hDFsGFP survived in contused SCs of athymic nude rats after 1 week, showing evidence of inducing axonal growth within and around the lesion site/transplantation area. Grafted cells were seen in close association with host glial cells (such as astrocytes and Schwann cells) within the lesion site (Fig. 6). Immunohistochemical staining of spinal cord sections containing donor hMPCsGFP in vivo revealed that 1 week after transplantation β-III tubulin positive axons were seen surrounding the groups of cells surviving within the lesion site (Fig. 6A). Transplanted hMPCs were surrounded by p75-positive profiles, which indicate the migration of Schwann cells into the transplant site (Fig. 6B, C). At no time were hMPCsGFP positive for p75. Adjacent sections of the hMPC-transplanted spinal cords showed the GFAP-positive astrocytic glial reaction to the transplanted hMPCs. GFP-positive hMPCs intermingled with host astrocytes within the spinal cord parenchyma (Fig. 6D). S100 immunostaining showed strong expression surrounding the hMPCs, without the cells themselves expressing the protein within the cytoplasm (Fig. 6E). Lastly, RT97-positive fibers were often expressed in close proximity to transplanted hMPCsGFP both surrounding and within areas of surviving transplanted cells (Fig. 6F). Similar in vivo glial and axonal profiles were observed in hDFGFP-treated animals except for the level of p75-positive Schwann cells present within the lesion site where Schwann cells were closely aligned with transplanted fibroblasts rather than randomly intermingle (data not shown). Rats treated with CsA did not alter phenotypic expression (using immunohistochemistry) on hMPCs over time.
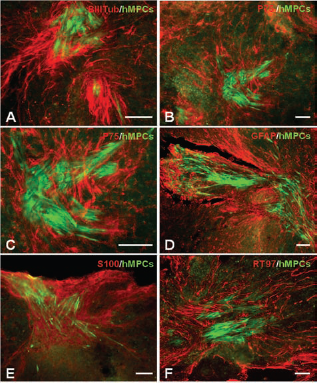
Immunostaining of nude rat spinal cord sections following transplantation of hMPCsGFP. Fluorescent photomicrographs show host tissue reaction and in vivo hMPC phenotype at the site of injection after 1 week (A–F) following transplantation. Antibodies specific for regrowing and spared axons (βIII tubulin) (A), Schwann cells (p75 and S100) (B, C, E), astrocytes (glial fibrillary acidic protein; GFAP) (D), and medium–high positive neurofilament positive axons (RT97) (F) were used (red) and are shown in close proximity to transplanted donor hMPCsGFP (green). hMPCs were seen within the lesion site and shown to be in small groups and incorporated well into the spinal cord parenchyma. Scale bars: 100 μm.
We used an antibody to HNA as an additional marker to retroviral and lentiviral GFP identified viable donor hMPCs and hDFs in vivo (Fig. 7). At 1 week posttransplantation, hMPCsGFP were seen within the transplantation zone in the contused spinal cord. The hMPCs were closely aligned, tightly packed together (Fig. 7A), and positive for HNA (Fig. 7B). Additional samples of hMPC-transplanted spinal cords, but with the additional treatment with CsA, showed transplants still surviving in groups (Fig. 7C) and coexpressing HNA (Fig. 7D). This marker was used to assess donor hMPC integration, dispersal/migration within the spinal cord, and approximation of cell survival in vivo over time. Some hMPCs were found to have lost GFP expression after 1 week (Fig. 7E), but the majority of cells were dual labeled. Overall there was no apparent difference in the GFP and HNA expression profiles between hMPC-transplanted animals (with or without CsA treatment).

Immunostaining of spinal cord sections with human nuclear marker (HNA) antibody following hMPCGFP transplantation. Fluorescent photomicrographs of transplantation site 1 week postinjection of hMPCs labeled with GFP (A), same field as previous showing colabeling with HNA (Cy3) and showing close association with GFP fluorescence (B). hMPCGFP-transplanted spinal cord in addition to CsA treatment indicating two groups of surviving cells, which have dual overlapping label for both GFP and HNA (C, D). Adjacent spinal cord section within the lesion site at higher power showing GFP- and HNA-positive hMPCs at 1 week posttransplantation. Interestingly, some cells have downregulated some of their native GFP fluorescence but are clearly labeled by the use of the HNA-specific antibody (see arrows) (E). Scale bars: 100 μm.
Two weeks after transplantation, numbers of donor hMPCs and hDFs (detected using GFP/HNA) were drastically reduced, indicating these cells were not surviving in the SC. Donor cell survival was semiquantitatively assessed, and the results are represented in Table 2. One month after transplantation, immunohistochemical staining of spinal cord sections revealed there were β-III tubulin-positive axons within the lesion site (Fig. 8A) as well as p75-positive profiles resembling migrating Schwann cells into the lesion site (Fig. 8B). GFAP (Fig. 8C) and S100 (Fig. 8D) immunohistochemistry revealed the presence of activated glial cells close to the lesion/transplantation site. RT97-positive axons were seen to enter the lesion site in (Fig. 8E). Similar glial and axonal-stained profiles were observed in animals at 1 month following the transplantation of hDFsGFP. At 1 month, there were no viable donor hMPCsGFP and very few donor hDFsGFP (Fig. 8F) when using HNA staining or GFP expression.

Immunostaining of spinal cord sections following transplantation of hMPCsGFP and hDFsGFP. Sections stained with specific antibodies to βIII tubulin (A), p75 (B), GFAP (C), S100 (D), and RT97 (E) show the host glial and neuronal response at the site of injection 1 month after transplantation of hMPCs. No surviving hMPCs were visible within the lesion site and adjoining spared tissue. Surviving GFP-positive hDFs were seen within the lesion site but in small numbers (F). Scale bars: 100 μm.
Extracellular matrix molecules (ECM) laminin and fibronectin (Fig. 9A–C) were expressed in close proximity to hMPCsGFP and hDFsGFP, indicating in vivo expression comparable to their in vitro expression profiles. These ECM molecules could also be derived from host cells including Schwann cells and endothelial cells (laminin) or fibroblasts (fibronectin) induced to migrate to the lesion by the injury and in conjunction transplanted cells. At 2 months post hMPCGFP transplantation, the distribution of β-III tubulin axonal profiles, p75-positive Schwann cell profiles, GFAP-and S100-positive astrocytes, and RT97-positive axons, as well as laminin- and fibronectin-positive structures, also revealed little change in spatial and temporal expression compared to earlier time points. There were significant numbers of ED1-positive cells in the hMPCGFP-transplanted cords at 1 month (Fig. 9D) and 2 months (data not shown). Immunocolocalization of intact hMPCsGFP (viable GFP-positive donor hMPCs) with macrophages using ED1 was problematic due to the autofluorescent properties of macrophages after double labeling with these antibodies. However, an influx of immunoregulatory (ED1+ve) macrophages in and around the lesion site coincided with the disappearance of intact donor hMPCsGFP. There were significant levels of ED1+ve cells in hMPCGFP and hDFGFP-treated spinal cords as seen in control injury-only or medium-injected cords. This indicates that, despite the T-cell deficiency of the athymic nude rat, there still appears to be a cellular immune reaction to the transplanted hMPCsGFP and that accumulation of ED1+ve cells continues up to 2 months after the disappearance of the hMPCsGFP. Similar decreasing hMPCGFP numbers with evidence of ED1+ve cells was also observed in CsA-treated animals up to 2 months after hMPCGFP transplantation. Sensory calcitonin gene-related peptide (CGRP)-positive and supraspinal serotonergic (5-HT)-positive fibers were identified both in and around lesion site at 2 months post hMPC transplantation (Fig. 9E–H respectively) as indicated by the arrows.

Immunostaining of spinal cord sections following transplantation of hMPCsGFP. Photomicrographs show laminin (LAM) deposition at 1 week close to and within the GFP-positive transplant (A) and at 2 months but with very few surviving hMPCsGFP (B) and fibronectin at 2 months posttransplantation (C). ED1+ macrophages have infiltrated the transplant/lesion site in large numbers and are still present 2 months after initial transplant (D). In addition, numerous sensory axons (arrows), which were Sensory calcitonin gene-related peptide (CGRP+; E, F) and regenerating serotonergic (5-HT+) fibers (G, H), were seen within the lesion site and in the distal spinal cord after transplantation of hMPCs. Scale bars: 100 μm.
Up to 2 months postinjury, immunostained spinal cord sections from injury-only and injury-plus medium-injected control animals showed fibronectin expression within and lining the lesion site; immunoreactivity for laminin showing new blood vessel growth was evident and small numbers of RT97-positive axons within the lesion. GFAP-positive processes within the lesion were evident, but only within 100–150 μm from the lesion edges. p75 Schwann cells were present when dorsal roots were close to the lesion site, and these were in close proximity to some β-III tubulin-positive fibers.
At no time point posttransplantation (1 week, 1 month, or 2 months) of hMPCs was there any colocalization of GFP/HNA-positive human cells with either PCNA or Ki67 immunostaining, indicating that there was no proliferation of transplanted donor hMPCsGFP in vivo.
Quantitative Analysis of (Fluorogold) Axonal Tracing
Mapping and counting of FG-labeled neurons in spinally projecting brain areas (55) was performed using Image Pro Plus analysis software. The numbers of FG-labeled neurons in raphe nuclei, reticular, vestibular, rubrospinal, trigeminal, and cortical regions of injury-only control, medium-injected control, and hMPCGFP-transplanted rats varied but were not statistically different (ANOVA) at 2 months after acute SCI, either with or without CsA treatment (Fig. 10). The mean total number of FG-labeled neurons, as counted in one of six sections throughout the whole of the brain in injury-only control, medium-injected control, and hMPCGFP-transplanted rats is shown in Table 3. Only in the raphe nuclei was there an apparent increase (approximately twofold) in the number of FG-labeled neurons between injured-only and hMPC-transplanted rats; however, this was not statistically significant using the test we employed.

Retrograde Fluorogold tracing of axonal pathways at 2 months after transplantation. The mean number of Fluorogold (FG)-labeled cells counted in cortical (A), reticular (B), rubrospinal (C), vestibular (D), trigeminal (E), and raphe (F) brain sections is shown for each treatment. One of six sections were analyzed for total FG counts in all animals at 2 months (n = 8). A low-power ultraviolet image of representative sections from hMPC-transplanted animals is shown top right for each region, with a higher power image inserted. White, injury-only; red, medium-injected; blue, hMPC-injected. Standard deviation is shown. Statistical analysis of the results showed no significance between groups.
Mean (±SEM) Total Number of Fluorogold (FG)-Labeled Neurons After Spinal Cord Injections 8 mm Distal From the Contusion Site Edge

As counted in one of every six sections throughout the whole of the brain [with or without cyclosporin A (CsA)] in injury-only, medium-injected, and hMPCsGFP-transplanted rats.
Electron Microscopy of SC Regions Caudal to Injury
A small number of additional spinal cord sections immediately caudal (distal) to the injury site were prepared for electron microscopy to compare injury-only (n = 2) and hMPC-treated animals (n = 2) (Fig. 11). In Figure 11A, ultrathin sections medial lateral to (areas 1 and 3) and lateral ventral to (areas 2 and 4) the injury site revealed large areas of cyst and degenerative tissue in injury-only animals compared to more intact (spared) tissue seen in the hMPC-treated group (Fig. 11B). In injury-only, the dorsal column and central gray matter area are filled with macrophages and large cysts (asterisk). The spinal cord segment also shows a very thin border of white matter surrounding the injury zone. This zone is thicker in the hMPC-treated spinal cords. Thinly myelinated axons are also present in the hMPC-treated animals (see areas 1–4) indicating potentially new myelinated axons or preservation of myelin. More unmyelinated axons (hash) were also present (see 2) in the hMPC-treated group but rarely in the injury-only group.

Photographs of spinal cord sections caudal to injury site after processing for electron microscopy. Semithin plastic 1-μm sections were taken from the control (A) and hMPC-transplanted (B) rats and stained with toluidine blue (see large pictures) to see the full extent of tissue changes at 2 months after injury. Additional ultrathin sections, lateral (1,4) and ventral (2,3) zones, within 1-mm distance caudal to the site of impact revealed less degeneration and regions of increased myelination in hMPC-treated (n = 2) versus injury-only (n = 2) animals. Scale bar: 5 μm. #Unmyelinated axon; ∗cysts.
Discussion
This study demonstrates purified (Stro-1+) hMPCs isolated from SCI patients and injected into the rat SC 7 days after SCI: (i) markedly improved functional recovery after moderate contusive SCI, (ii) increased the amount of spared (intact) tissue at 1 week and 2 months time points, and (iii) generated increases in neuronal and glial marker expression both in and around the injury site after treatment. These results were achieved despite the lack of transdifferentiation of donor hMPCs into neurons and glia and also despite transplanted hMPCs not surviving in the injured rat SC beyond 4–8 weeks. Even without long-term survival of hMPCs, there was a marked beneficial effect of this cellular treatment after acute SCI. Quantitative analysis of brain nuclei by retrograde tracing (FG) revealed the presence of preserved supraspinal axonal pathways such as raphespinal. However, the tracing showed high variability within treatment groups and indicated trends, rather than statistically significant improvement in supraspinal innervations 8-mm distal from the transplantation site after hMPC therapy. Further treatment of the rats using CsA did not improve functional recovery, tissue sparing, hMPC survival, or any other observation over and above those regimes without CsA immunosuppression.
Purified Donor hMPCs: In Vitro and In Vivo Phenotype
In this study we chose a specific Stro-1 selection from a population of human mesenchymal cells isolated from BM of SCI patients. This was a prospective immunoselection aimed to initiate mesenchymal culture at higher purity by specifically isolating the clonogenic mesenchymal precurors from BM with a higher proportion of immature Stro-1+ precursor cells to provide an increased multipotent stem cell source (62). hMPCs provided a selected population known to be enriched for CFU-F and after replating from frozen stocks had retained the Stro-1 antigen in approximately 55% of cells (Plant, unpublished observations). New published reports in the rat have argued other cell populations express the Stro-1 antigen such as endothelial cells and perivascular cells (41), but the distribution and identity of these cells has not been confirmed as being present in human. This is further complicated by evidence from Crisan and colleagues (14), who suggest perivascular (pericytes) cells sorted from diverse fetal and adult tissues have the properties and ancestry of MSCs, and can be developed into clones that can yield osteocytes, chondrocytes, and adipocytes. Therefore, the potential exists that Stro-1 cells identified as endothelial and perivascular as reported by Lin and colleagues (41) are in fact MSCs (14) and potentially have Stro-1 immunoreactivity. Further stringent in situ analyses combined with isolation techniques using the Stro-1 antibody in human tissue sections of bone marrow would provide great insight into the role of these selected cells.
We believe the Stro-1-selected hMPC population sourced from the bone marrow represents a significant step forward in understanding the potential of hMPCs as a treatment for spinal cord repair and the mechanisms involved, which otherwise would be more difficult to decipher with heterogeneous adherent cell populations. Interestingly, recent evidence has been reported on the role of pericytes after injury to the spinal cord (19). Class A pericytes lining the blood vessel wall were found to migrate in response to injury and filled the lesion site. This may be a first response to injury and provide a stabilization of the injury zone, instead of solely a scar forming function. Further analysis in the role of endogenous pericytes with mesenchymal transplants would be worthy of further investigation.
Our results provide no direct evidence that donor hMPCs from SCI patients transdifferentiate into neuronal (39), glial or astrocyte phenotypes in vivo. This is in contrast to the controversy that exists in neuronal differentiation of heterogeneous adherent populations of donor adult BMSCs in vivo after transplantation and their contribution toward morphological and functional improvements after SCI therapy (52,57). hMPCs isolated from SCI patients expressed low levels of the marker β-III tubulin (often considered a neuronal marker) and no other neuronal, astrocytic (GFAP), or other glial markers such as p75 and S100 in vitro when cultured alone. Our hMPC results are supported by evidence from other studies characterizing gene expression patterns of adult mesenchymal cells before and after neural differentiation. These studies revealed the expression of several neural genes and glial cell transcripts prior to any differentiation treatment (10,27,28,75).
In our laboratory, undifferentiated donor hMPCs show no evidence of differentiation into myelinating glial lineages and did not myelinate embryonic (E16) dorsal root ganglion (DRG) cultures in vitro, even though they are shown to express glial markers such as S100 and GFAP (Hodgetts and Plant, unpublished data). Many potential cell transplantation candidates have shown remyelination in the adult CNS following injury including Schwann cells (18), olfactory ensheathing glial cells (OEGs) (65), oligodendroglial progenitor cells (7), adult neural progenitors (37), embryonic stem cells (13), and BMSCs (52); each differ in their myelination capability (47). Although our data suggest that donor Stro-1+ hMPCs show no direct myelination, this does not preclude them from promoting host myelination in vivo by facilitating a more permissive environment for such endogenous mechanisms. This could be mediated either directly or indirectly by Schwann cells, oligodendroglia, or host stem/progenitor cell activation. Myelination is also determined by lesion type, age, and genetic background of the recipient so Stro-1+ cells could be shown to provide compact myelin when provided with the right conditions.
Extensive and continued deposition of fibronectin and laminin was seen in the lesion site from GFP and human nuclear marker (HNA)-positive hMPCs with both extracellular matrix molecules present as early as 7 days posttransplantation and remained for the entire time frame of analysis (2 months). These depositions could provide axons with a permissive matrix through which they can grow, be ensheathed, or allow for more efficient host remyelination of intact axons.
Donor hMPCs Survival After Transplantation
To date, Stro-1-purified hMPCs isolated from SCI patients have not been directly examined in the context of SCI and carefully analyzed in a reproducible injury model that cell numbers controlled for. Transplanted hMPCs initially survived well in the injured athymic RNU nude rat spinal cord but were not detected at 2 months via the use of viral transduction with GFP and/or staining with the HNA antibody. At this time, ED1 macrophage infiltration remained high. In comparison, control donor hDFs survived up to 2 months after transplantation, although with dramatically reduced numbers. Immunosuppression with CsA did not improve donor hMPC survival, compared to hMPCs only-treated athymic RNU nude rats. Different studies have reported prolonged survival of transplanted rodent BMSCs following SCI, ranging from 4 weeks to 3 months, while others reported rapid loss of donor cells within 1–5 weeks (52). Two studies using adult hBMSCs, rather than hMPCs, into rat SCI models reported similar losses of donor cells with numbers decreasing from 2 weeks and continuing to 11 weeks (no cell survival) (29,53). Due to the unknown selection criteria of cells in these studies, one cannot rule out differential survival of mesenchymal subpopulations.
The highly abnormal thymus rudiment in this rat model generates a significant T-cell immunodeficiency, which has led to their use in xenotransplantation; however, immune function that is independent of the thymus appears to be normal in these animals (12). Together with our data showing that CsA immunosuppression does not alter any hMPC survival outcome, this would suggest that a specific T-cell-mediated immune response in the athymic RNU nude rat was not responsible for the eventual loss of donor hMPCs. It is possible that donor hMPCs succumbed to another functional component of the innate immune response, such as a host natural killer (NK) cell attack, complement or dendritic cells (64). Recent published studies suggest that adult BMSCs are target cells for activated NK cell attack (71). Accordingly, the efficacy of transient immunodepletion of NK cells as part of a therapy to promote the survival of donor Stro-1+ hMPCs using blocking and/or depleting antibodies (30) remains to be determined as a potential additional strategy. In some studies, it may be an assumption that prolonged cell survival can be attributed to the total heterogeneous population of donor adult BMSCs; rather, we feel it may be due to specific subpopulations within the graft. Indeed, donor Stro-1-selected hMPCs from SCI patients may have altered expression patterns compared to normal donors, which may affect their ability to survive following transplantation.
The immunosuppressive properties of donor adult hBMSCs have been reported, but we did not investigate such a role of Stro-1+ hMPCs in our model. Properties include a wide range of cytokine expression, chemokine expression, and immune cell suppression mechanisms (1), which may in turn function as survival and/or differentiation factors for neural progenitor cells, with subsequent effects on neurogenesis and CNS plasticity. However, secretion of pro-inflammatory cytokines from adult Stro-1+ hMPCs may also have a negative effect on recovery by increasing neutrophil and macrophage infiltration and inhibiting axonal growth (40). The acute inflammation following SCI results in additional tissue damage and this proinflammatory environment may also be detrimental to the protective growth and repair-promoting properties of donor hMPCs. However, other studies have also shown a positive, regenerative potential of certain immune effector cells (66). The balance, timing, and nature of protective and destructive factors produced by the immune response after SCI are not yet fully understood (60); any mechanism through which hMPC therapy might improve functional recovery after SCI may be altered in response to encountering the microenvironment following transplantation. Further differences may also depend on the variation from different hMPC donors (53,77) such as age, gender, health status, and genetic makeup.
Transplantation of Purified hMPCs: Tissue Sparing
hMPCs significantly reduced cyst size within the injured SC up to 2 months posttreatment (p < 0.05), as well as a significant increase in the amount of intact spinal tissue (tissue sparing, p < 0.001) observed. Transplantation of hDFs used as a control donor cell also resulted in significant reduction in the cyst size within the injured SC up to 2 months posttreatment (p < 0.05), as well as significant increase in the amount of intact spinal tissue (tissue sparing, p < 0.001). Previous studies incorporating fibroblasts as a cell transplantation strategy in rats and primate models reported small but significant improved functional recovery, if assessed. Improvement in tissue sparing and axonal regrowth outcomes are seen when transduced fibroblasts were transplanted to express growth factors (8,9,20,26,35,36,42,43,45,49,51,69,72,78,79,81) or other modifying factors (58), but not in unmodified or control fibroblasts. Our results indicated increased tissue sparing at 2 months in the hMPC group compared to hDF-treated groups, highlighting that hMPCs have a significant role in reducing tissue loss and secrete different and perhaps more potent bioactive molecules to those in hDFs.
It is our opinion that the increase in tissue sparing and reduction of tissue loss could not be solely attributed to the effects of the donor material simply filling the cyst volume in the injured spinal cord, particularly when no hMPCs were visible after 5 weeks. Despite large amounts of ECM molecules produced from donor hMPCs themselves, the amount of degenerative tissue at the injury site was significantly reduced. This may be due to the immunomodulatory properties of hMPCs reducing the amount of secondary damage after SCI or stimulation of mitosis of tissue intrinsic progenitors whose progeny reform the tissue (11). Importantly, the amount of intact SC tissue continued to increase up to 2 months after injection, even though donor hMPCs had disappeared. This suggests that hMPC therapy may result in a prolonged endogenous response (50) to promote tissue sparing or repair by remyelination of damaged (but still intact) axons. Our electron microscopy results of spinal cord sections also suggested less axonal degeneration and higher levels of myelinated tissue caudal to the injury site in hMPC-treated animals. Additional treatment of SCI rats with CsA immunosuppression did not further improve tissue sparing outcomes in hMPC-treated animals. This is consistent with results reported by Rabchevsky et al. (63) but does not support the reported neuroprotective effects observed by others (33,34,44,48). The further reduction of T-cells using CsA in the athymic RNU nude rat indicated that T-cells were not the primary mechanism of hMPC loss in this study and that CsA had no increased efficacy on the repair properties of the hMPCs in this model.
Improved Functional Recovery After hMPCs Transplantation
Increased BBB scores indicating a positive change in functional recovery emerged 3–4 weeks following transplantation of hMPCs. Rats subjected to hMPC therapy consistently achieved a mean BBB score of 15 (p < 0.05). Interestingly, at 5 weeks, the hMPC-treated rats achieved significant increased BBB scores to that of the hDF group (p < 0.05). However, both hMPC and hDF transplants were shown to be statistically (p < 0.05) significant from injury and injury-plus medium groups 6–8 weeks following transplantation. Differences in scores were evident between the hMPCs and hDFs at these time points, but due to internal group variation were not significant. hMPC-treated rats achieved BBB scores of 16, which was not seen in the hDFs at any time points. This indicates a significant toe clearance and parallel stride. BBB scores reached at 4 weeks postinjury are comparable to previously published studies using SCI with rodent BMSCs (54) in Wistar rats or human bone marrow stromal cell transplantation (29,77) into Sprague–Dawley rats. Animals treated with CsA alone in injury-only and injury-plus medium groups showed increases in BBB scores of up to 13 even without hMPC or hDF treatments, but when CsA was combined with hMPCs, it did not provide improved BBB outcomes to hMPCs alone. This could indicate that the general immunosuppressive actions of CsA may improve functional recovery after acute SCI (with or without the need for hMPC therapy) and that these effects may be strain-specific (15). It is important to note that, even though the applied Kruskal–Wallis test is an analysis of variance for nonparametric data, the BBB scale is neither linear nor exponential; higher BBB values represent a score comprised of increasing numbers of variables that are often unrelated to previous (lower) scores. We feel that careful interpretation should always be exercised when equating biological significance with statistical significance in such cases. With this caveat in mind, the improvement in functional recovery was more pronounced in hMPC-treated animals compared to injury and medium-injected control animals. Our results suggest that hMPCs therapy results in a significant transition from noncoordinated locomotor function after injury toward a marked, coordinated locomotor recovery. Transdifferentiation into neural cells (38) as a potential mechanism for recovery of function remains controversial (52,57).
Axonal Sparing and Regeneration
Axonal regeneration may contribute to some of the functional recovery observed in this study. This may involve collateral sprouting of noninjured axons (25), although functional improvements do not necessarily reflect the extent of anatomical regeneration and reconnection of descending nerve tracts within the injured spinal cord. While there was no statistically significant evidence of sparing or regeneration of both serotonergic (5-HT) and sensory (CGRP) axons, they were observed in large numbers within the lesion sites and innervated into the distal stump close to the lesion of hMPC-transplanted animals.
Serotonergic (5-HT)-positive axons traversed through the lesion site but may not have contributed to the FG raphe numbers in the brain because they did not reach the injection of the FG at approximately 8 mm distal from the injury site. This does not preclude local circuitry innervations 1–2 mm distal from the injury site which potentially could stimulate downstream central pattern generator activation within the spinal cord (80).
An increase of spared axonal pathways such as reticular and vestibular spinal projections could facilitate spontaneous plasticity in the form of axonal sprouting or modified use (compensatory behavioral strategies) of spared projections, leading to an improvement in functional outcomes (4,5). Our results showed no statistically significant differences between treatment groups or CsA immunosuppression regimes in FG-labeled neurons of raphe nuclei, reticular, vestibular, red nucleus, trigeminal, or cortical regions at 2 months after acute SCI. As such, we cannot correlate any trends of increasing numbers of FG-labeled supraspinal pathways to any axonal regenerative/ sparing event or to the functional recovery observed in hMPC-treated rats.
Summary
Our results show hMPC therapy using a purified selected sub population of hMPCs (Stro-1+) from SCI patients markedly improves functional recovery, promotes tissue sparing, and induces local axonal growth without evidence of transdifferentiation of the transplanted hMPCs into either neuronal or glial phenotypes. The hMPCs provide a supportive environment for the activation of these repair mechanisms before the donor hMPCs finally succumb to an immune response. We believe ECM deposition and or neurotrophin secretion are involved. In addition several putative growth factors such as brain-derived neurotrophic factor (BDNF) or related molecules could be released by the hMPCs, which are known to affect functional recovery in CNS injury (8,35,61,76). hMPCs may secrete immunomodulatory cytokines and thereby reduce secondary damage and enhance neuroprotection. Or they may activate an endogenous host response that could result in decreased demyelination and/or increase remyelination by recruited Schwann cells or oligodendrocytes. We believe these beneficial effects can be improved by enhancing the survival of donor hMPCs in vivo; this could be via immunomodulation of the innate host immune response (84) rather than general (CsA) immunosuppression.
Experiments using purified, selected subpopulations of hMPCs isolated from SCI patients will facilitate a better understanding of the effects that specific parameters have on recovery of function. These may include prolonged survival of hMPCs in the graft, costimulation with granulocyte macrophage colony-stimulating factor (GM-CSF) (85), or repeated hMPC transplantations via intraspinal or vascular routes and may therefore result in further improvement in both anatomical and behavioral recovery.
Footnotes
Acknowledgments
We wish to express sincere thanks to Seok Voon Lee, Sarah Lovett, Alysia Buckley, Anne Kramer, and Dr. Ajanthy Arulpragasam for their technical contribution. We thank Christine Plant for critical writing and editing of the manuscript and Guy Ben Ary (CellCentral, ANHB, University of Western Australia) for his assistance in image acquisition and analysis. We gratefully acknowledge Dr. David Haylock and Genevieve Whitty (both at ASCC) for their help in hMPC cultures and Dr. Hu Ying (ANHB, UWA) for his help in statistical analyses of results. GWP's laboratory was supported by the National Health and Medical Research Council (RD Wright Fellowship No. 303265), Spinal Cord Society of Australia, Eileen Bond, The Neurotrauma Research Program of Western Australia, and The Australasian Spinal Research Trust. The authors declare no conflicts of interest.
