Abstract
Islet autotransplant patients represent excellent subjects to assess the posttransplant impact of islet precursors, as chronic pancreatitis (CP) causes an elevation of ductal cells, pancreatic precursors cells, and hormone-positive acinar cells. The relationship between these cell types and autograft outcomes should be more apparent than would be the case in the context of an allograft program with confounding immunological variables. To improve diabetic control following total pancreatectomy for CP, nonpurified islets were autotransplanted into the liver. Pancreas specimens were recovered from 23 patients and stained for antigens including: insulin, glucagon, cytokeratin 19, cytokeratin 7, and PDX-1. In line with previous reports, the prevalence of ductal cells, non-islet endocrine cells and non-islet PDX-1-expressing cells was significantly higher in CP glands compared with normal pancreata. When correlating follow-up data (i.e., fasting and stimulated C-peptide/glucose levels and HbA1c%) with pancreas immunoreactivity, high levels of ductal cells, non-islet PDX-1-positive cells, and non-islet glucagon-positive cells were associated with superior outcomes, detectable up to 2 years posttransplant. To conclude, the acinar parenchyma and ductal epithelium of the CP pancreas show an upregulation of both endocrine and pre-endocrine cell types, which appear to have a positive effect on islet graft outcomes in autotransplantation setting.
Keywords
Introduction
It is well accepted within the field of islet transplantation that the number of islet equivalents transplanted is positively linked with islet graft success, with a critical mass of 10,000 IEQ/kg bodyweight shown to alleviate insulin dependence post allotransplant (24) and approximately 2,500 IEQ/kg following autotransplant [reviewed in Blondet et al. (1)]. Analysis of data collected within the Leicester islet autotransplant series showed that, although higher IEQs were frequently associated with insulin independence, this trend did not hold true in all circumstances (35) and, indeed, the Leicester group have reported two cases of sustained graft function following transplant of less than 1,000 IEQ/kg (13, 36). These findings therefore suggest that other factors, rather than pure islet mass, affect the insulin-secreting potential of the islet graft posttransplantation. Feasibly, this could be due to a range of factors; however, a review of the literature suggests that this is likely to be due to: the inclusion of cells that provide biotrophic support to the islets (12, 23) or the inclusion of cells that give rise to/transdifferentiate into the β-cell phenotype posttransplantation (3).
To date, there are a plethora of animal models and in vitro studies evidencing β-cell genesis from non-islet sources (2, 3, 10, 17). Furthermore, numerous histological studies of both normal and pathological pancreata describe the occurrence of cells displaying β-cell characteristics located outside of the islets of Langerhans (18, 34), the occurrence of pluripotent cells that have the potential to convert into islet cells (22, 26), and disease states that cause the upregulation of ductal epithelial cells (33), which are accepted to be a source of islet neogenesis (3). Therefore, the present study set out to explore whether the frequency of potential islet precursors had an impact on the long-term islet graft outcomes of chronic pancreatitis (CP) patients that had undergone total pancreatectomy and subsequent islet autotransplantation of nonpurified islets.
Chronic pancreatitis patients represent excellent subjects to study principally due to the fact that chronic pancreatitis induces a marked increase in tissue regeneration and remodeling, causing a distinct increase in the prevalence of potential islet precursors including: ductal epithelial cells, non-islet pancreatic and duodenal homeobox 1 (PDX-1)-expressing cells, and intermediate cells (hormone-positive acinar cells) (30, 33, 34). Therefore, it can be assumed that correlations between the prevalence of potential islet precursors in source pancreata and clinical outcomes would be more apparent in the auto- compared with the allotransplantation setting. Furthermore, autotransplantation of islets compared with allotransplantation has fewer confounding factors to consider and can therefore provide an uncomplicated clinical model to assess islet function posttransplant. Issues of patient immunosuppressive regimens, particularly toxicity and discontinuation of therapy, are avoided (4, 11, 20, 38), cold ischaemia times are consistent and relatively short as organ procurement and processing is generally completed on one site, and islets are usually transplanted directly following isolation without a period of transportation or culture (29). In addition, tissue processing is completed within a shorter period (less than 3 h) (29), largely due to the excision of the pancreas free of fat and the fact that simple syringe-driven perfusion is primarily utilized (35).
Therefore, for the present study, the frequency of potential endocrine precursors was assessed in the pancreata of chronic pancreatitis patients that had undergone total pancreatectomy with concomitant (nonpurified) islet autotransplant (n = 23). Subsequently, the prevalence of each potential islet precursor within the source pancreas was correlated with islet graft performance over 5 years posttransplant to ascertain whether their inclusion had any impact on long-term islet graft outcomes.
Materials and Methods
Exclusions
Intact samples of CP pancreata were collected as a routine part of the pancreatectomy procedure. Of the 60 patients that have been transplanted as part of the Leicester islet autotransplantation series, only 44 [those collected prior to the Human Tissue (Scotland) Act 2006, UK] were eligible for use due to ethical constraints. Of these, 13 patients were excluded because they had received purified islets, a further four partial pancreatectomy patients (n = 4) were excluded due to the influence of the residual gland, and finally, samples could not be recovered for four patients; therefore, samples from n = 23 CP patients were available for use. Additionally, eight control pancreatic samples were obtained from non-heart-beating donors with no reported pancreatic disease. Ethical approval was obtained for all tissues used (UK Research Ethics Committee Reference numbers: 07/H0304/65 and 04/MRE04/77).
Immunohistochemistry
Samples of intact CP pancreata were collected, fixed and paraffin embedded as a routine part of the pancreatectomy clinical protocol. Sections were stained for insulin (1:10,000, ascetic fluid derived from cell line HB124 of the Hybridoma Bank, Baltimore, MD, US) glucagon (1:5,000), cytokeratin 7 (1:500), cytokeratin 19 (1:50), PDX-1 directed to the N-terminal (1:5,000, Abcam, Cambridge, UK), and PDX-1 C-terminal (1:500, Chemicon International, Temecula, CA, US). Before the application of the primary antibody, microwave antigen unmasking (850 W, 20 min) was used for all antibodies with the exception of insulin. The primary antibody was applied to each section, incubated at 37°C for 1 h, and then washed with Tris-buffered saline (TBS) + 0.1% bovine serum albumin (BSA) (3 × 5 min). The ChemMate Envision™ detection complex (mouse and rabbit secondary antibodies conjugated to a dextran polymer densely labeled with horseradish peroxidase) was then applied and incubated at room temperature for 30 min. Finally, after three further washes (× 5 min), the diaminobenzadine substrate (DAB) was used as the agent of antigen detection with hematoxylin used to counterstain. Sections were then dehydrated through graded alcohols to xylene, and coverslips were mounted using XAM (BDH, UK); all products were purchased from Dako (Glostrup, Denmark) unless otherwise stated.
Stained histological sections were analyzed using Axiovision imaging software with the exception of glucagon. The Axiovision data presented here represents the percentage of positive pixels per field; however, it is recognized that the actual percentage of positive cells are likely to exceed the Axiovision figures quoted due to the fact antibody staining was often restricted to either the nucleus or cytoplasm. Glucagon staining was carried out manually due to fact that positive cells associated with the ductal epithelium needed to be counted separately. Nine CP patients and one control patient had multiple samples (between two and seven tissue blocks); in these cases, all specimens were analyzed and median values were used for statistical analysis. The evaluation of all antigens excluded staining within the islets of Langerhans.
Assessment of Glucose Homeostasis and Islet Function Posttransplant
Assessment of islet function was undertaken by C-peptide assay (ELISA DRG Diagnostics, Nottingham, UK) as a part of normal patient care. The lowest limit of sensitivity was 0.04 ng/ml. Follow-up assessments were made at 6 and 12 months posttransplant and annually thereafter, using the 75 g oral glucose tolerance test (OGTT). Prior to the assessment, patients were fasted overnight and had their insulin stopped for at least 12 h. Blood samples were taken and processed for the evaluation of plasma glucose and serum C-peptide levels at 0, 30, and 120 min (GTT 0, 30, and 120 and C-peptide 0, 30, and 120, respectively) additionally, blood samples were taken for assessment of glycated hemoglobin (HbA1c). Posttransplantation, stimulation indices (SI) were calculated as stimulated C-peptide levels (at either 30 or 120 min) divided by basal C-peptide levels, giving SI30 and SI120. Since many patients were recruited from centers at some distance from Leicester, many patients failed to attend an assessment every year.
Statistics
Median values and ranges or mean values ± SEM were used as descriptive statistics. The levels of antigen detected in chronic pancreatitic samples versus control pancreata were measured using the Mann–Whitney U test. Correlations between each antigen and metabolic markers of graft function were made using Pearson and Spearman rank correlations. All statistics were calculated on SPSS version 16, and graphs were created on both SPSS version 16 and Prism graph pad. Values of p < 0.05 were considered statistically significant.
Results
CP Pancreata Versus Control Tissue
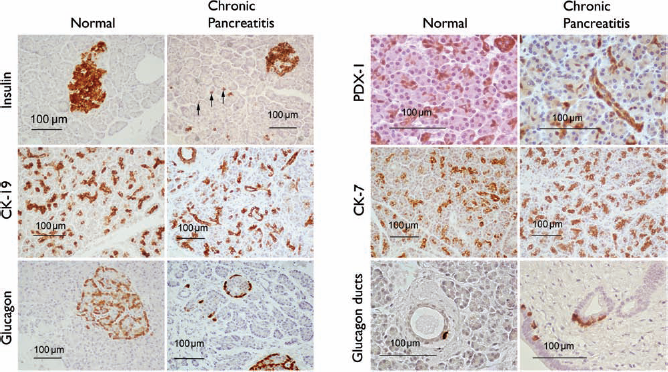
As anticipated, levels of potential islet precursors were largely higher in the CP group compared with the control group, indeed, insulin-positive non-islet endocrine cells (NIECs), PDX-1-positive cells, and cytokeratin 7- and 19-positive cells were found at significantly higher levels in the CP tissue (Figs. 1–3). Conversely, glucagon-positive NIECs and the subset of the glucagon-positive ductal epithelial cells were actually shown to be marginally lower in CP tissue; however, this finding did not reach significance (Fig. 1e, f).
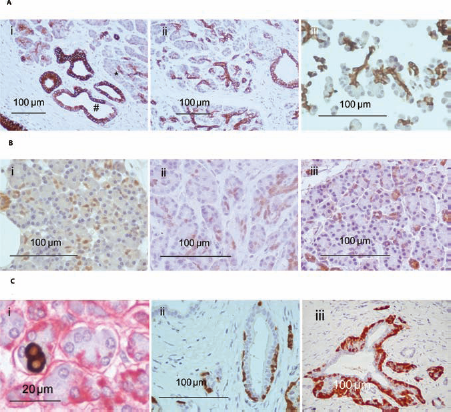
The prevelance of potential precursor cells within the normal and chronic pancreatitic (CP) pancreas. Expression of insulin-positive non-islet endrocrine cells (NIECs) and pancreatic and duodenal homeobox 1 (PDX-1) was significantly higher in CP patients compared with normal pancreata, 0.85 (0.15–4.46) versus 0.34 (0.18–0.50), p = 0.002 and 19.40 (7.07–34.00) versus 8.99 (4.57–17.10), p = 0.001, respectively (a, b). Additionally, both cytokeratin 7 (CK7) and CK19 expression were significantly higher in CP tissue, 23.40 (12.90–41.00) versus 17.60 (8.66–38.70), p = 0.044 and 34.20 (8.35–75.40) versus 17.70 (13.10–20.30), p = 0.001, respectively (c, d). No significant difference was found between CP and normal tissue with regards to glucagon-positive NIECs, 1.081 (0–3.889) versus 1.40 (0.50–3.10), p = 0.475 or glucagon-positive ducts, 0.10 (0–0.15) versus 0.08 (0–0.6), p = 0.688 (e, f), although levels were seen to be marginally higher in normal tissue. Typical examples of endocrine and ductal markers within the normal and chronic pancreatitic pancreas. Expression of insulin-positive NIECs, PDX-1, and CK7 and CK19 was statistically higher in CP tissue. No significant difference was found when assessing glucagon-positive NIECs or glucagon-positive ducts. All photographs were taken at an original magnification of 200 × with the exception of PDX-1 staining and glucagon ducts that were taken at 400 ×. (A) Ductal cell staining within CP tissue. CK7 showed a gradient of staining with centro-acinar (*) cells exhibiting weak staining compared with larger ductal epithelia (#) (i). CK19 staining was consistent throughout the ductal epithelia (ii). (iii) Non-islet tissue following pancreas digestion (in this case in normal tissue) stained with CK19. Following digestion, non-islet tissue typically consists of small units of cells. (B) PDX-1 staining within CP tissue. The intensity of PDX-1 staining varied between patient samples (i–iii) and PDX-1 staining was typically nuclear staining. Sections stained using PDX-1 antibodies targeted toward either the N-terminus (ii) or C-terminus (iii) of the PDX-1 molecule produced a near-identical staining patterns. (C) Staining of non-islet endocrine cells (NIECs). The location insulin and glucagon-positive NIECs within the acinar parenchyma was confirmed by double-staining each hormone (brown) with laminin (red). (i) Insulin-positive NIECs. The majority of glucagon-positive NIECs were closely associated with midsize (intra and interlobular) ducts (ii, iii), although is of note that (ii) and (iii) show particularly high levels of glucagon staining.
Ductal Epithelial Cells
CK7 and CK19 were used to stain for ductal epithelial cells. CK7 staining was either absent or weak at the centroacinar position of each acini and showed an increased gradient of expression in the larger ducts (Fig. 3A, i) while CK19 stained all ductal and centroacinar cells evenly (Fig. 3A, ii). The frequency of CK19 positivity within CP tissue was approximately double that observed in control tissue [34.20 (8.35–75.40) vs. 17.70 (13.1–20.3), p ≤ 0.001], while CK7 positivity was more modestly upregulated [23.40 (12.90–41.00) vs. 17.60 (8.66–38.70), p = 0.044 in CP and normal tissue, respectively]. Due to the fact CK19 is a pan ductal marker while CK7 is largely restricted to the larger ducts; these data suggested that chronic pancreatitis caused a significant increase in the proportion of small ductal epithelial cells within the pancreas.
Correlation studies showed that a high prevalence of CK7-positive cells was associated with significantly lower HbA1c levels at 1 year posttransplant (r2 = −0.315, p = 0.025, n = 16) (Fig. 4a). In line with the CK7 results, high levels of CK19 were also associated with significantly lower HbA1c at 1 year posttransplant (r2 = −0.294, p = 0.029, n = 16) (Fig. 4b); additionally, fasting C-peptide secretion, perhaps the most pertinent marker of background islet function, was significantly higher with increased numbers of CK19-positive cells at 1 year posttransplant (r2 = 0.403, p = 0.015, n = 14) (Fig. 4c).
Graft function versus CK7 and CK19 antigen expression, significant correlations. CK19 (a pan-ductal epithelial marker) was observed to be correlated with significantly low glycated hemoglobin (HbA1c) (a) with high fasting C-peptide values at 1 year posttransplant (b). While CK7 (a marker of the medium to large duct) prevalence correlated with significantly low HbA1c at 1 year posttransplant (c).
PDX-1-Positive Cells
Within CP and normal tissue PDX-1 staining was largely observed within the acinar parenchyma (and sporadically within the ductal epithelia) as nuclei staining, although diffuse cytoplasmic staining was also evident (Fig. 3B). Within this study, normal tissue displayed a median level 8.99% (4.57–17.10), which is higher than previously suggested (31, 37). However, in line with the findings of Song and colleagues (30), the number of PDX-1-positive cells was higher in CP specimens over control tissue [17.70% (13.10–20.30) vs. 8.99% (4.57–17.10), p = 0.001]. High PDX-1 levels were shown to be significantly correlated with lower fasting glucose levels at both 6 months and 2 years posttransplant (r2 = −0.345, p = 0.031, n = 15 and r2 = −0.423, p = 0.022, n = 12, respectively) and with lower stimulated glucose (GTT120) at year 2 (r2 = −0.345, p = 0.045, n = 12) (Fig. 5).
Graft function versus PDX-1 antigen expression, significant correlations. Scatter plots displaying the relationship between the percentage of PDX-1-positive staining and islet graft outcomes posttransplant. PDX-1 was observed to be significantly correlated with lower fasting glucose (mmol/L) at both 6 months and 2 years (a, b) and stimulated glucose at 2 years posttransplant (c). GTT0, glucose tolerance test at 0 min.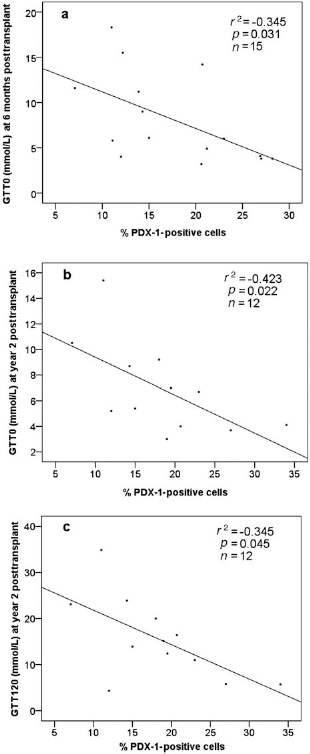
The Insulin-Positive Non-Islet Endocrine Cells (NIECs)
Within this study, insulin-positive NIECs were almost exclusively intermediate cells (i.e., hormone-positive cells located within the acinar parenchyma) (Fig. 3C, i) with few insulin-positive ductal cells. Unlike the aforementioned cell types, insulin NIECs were shown to occur at low frequency of 0.85% (0.15–4.46%), and their occurrence was not significantly associated with markers of graft function.
Glucagon-Positive NIECs and Glucagon-Positive Ductal Cells
The majority of glucagon-positive NIECs were found within the acinar parenchyma, but unlike insulin-positive NIECs, an appreciable proportion was observed closely associated with the ductal epithelium (Fig. 3C, ii, iii). The median percentage of glucagon-positive NIECs observed totaled 0.23% (0–5.71%), and despite their low frequency, a positive correlation was observed between their occurrence and high stimulation index levels (SI30) recorded at 6 months posttransplant (r2 = 0.420, p = 0.043, n = 10) (Fig. 6a).
Graft function versus glucagon-positive NIECs, significant correlations. Scatter plot displaying the relationship between glucagon-positive NIECs and islet graft outcomes posttransplantation. The percentage of glucagon-positive NIECs was shown to be significantly correlated with a higher SI30 at 6 months posttransplant (a). Glucagon-positive ducts were shown to significantly correlate with lower stimulated glucose levels (GTT30) at 6 months (b) and 2 years (c), lower HbA1c% at 2 years (d), and increased SI120 at 6 months posttransplant (e).
Investigations into the subset of glucagon NIECs restricted to the ductal epithelium, observed at a frequency of 0.019% (0–2.57%), revealed a similar magnitude of improved outcomes as CK19 and PDX-1 (Fig. 6b–e). Notably, high levels of glucagon-positive ductal cells were seen to be strongly correlated with reduced stimulated glucose levels (GTT30) at 6 months (r2 = −0.292, p = 0.046, n = 14), reduced GTT30 at year 2 (r2 = −0.507, p = 0.009, n = 12), low HbA1c at year 2 (r2 = −0.450, p = 0.009, n = 14), and a higher stimulation index (SI120) at 6 months posttransplant (r2 = 0.531, p = 0.026, n = 9) (refer to Fig. 6b–e).
Islet Equivalents
In line with the analysis of the preceding cell types studied, total IEQs transplanted (rather than IEQs/kg bodyweight) were utilized. Within this patient group, islet IEQ was seen to significantly correlate with high levels of C-peptide 30 at year 1 posttransplant (r2 = −0.283, p = 0.0412, n = 15) (Fig. 7), while at year 3, a near significant correlation was observed between IEQ and increased levels of C-peptide 30 (r2 = 0.346, p = 0.0806, n = 10).
Graft function versus IEQs, significant correlation. Scatter plot showing a positive correlation between total IEQs transplanted and stimulated C-peptide (C-peptide 30) at year 1 posttransplantation.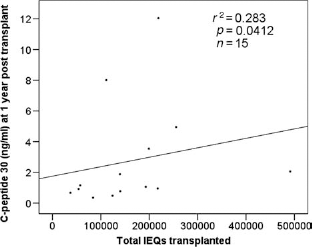
Cell Type Correlations
Further correlations between each antigen marker (and islet IEQ) showed that high levels of glucagon-positive cells associated with the ductal epithelium were positively correlated with CK19 expression (r2 = 0.267, p = 0.015, n = 22).
Discussion
The main aim of the present study was to assess the impact of potential islet precursor cells on islet graft outcomes, with the hypothesis being that such cells contribute to long-term insulin secretion posttransplantation. Although this study employs a much less rigorous approach than certain in vitro and lineage-based models that use either genetic or physical labeling of individual cell types to identify β-cell neogenesis, its importance lies in the fact that data are based on transplantation of human tissue, which has been under the influence of a natural (i.e., not experimentally induced) disease course (chronic pancreatitis) and takes into account the long-term function of the islet graft.
Primarily, this study, in line with earlier reports, demonstrates that ductal epithelial cells, PDX-1-positive cells and non-islet hormone-positive cells are upregulated in response to chronic pancreatitis (30, 33, 34). Secondly, perhaps due the distinct upregulation of these potential islet precursors, this study was able to successfully establish correlations between high levels of ductal cells, PDX-1-positive cells and glucagon-positive NIECs and clinical markers of islet graft function, this is in spite of the fact that the correlations should be heavily confounded by the differing levels of islet equivalents transplanted, patient factors [i.e., body mass index (BMI)] and transplant factors (i.e., instant blood mediated inflammatory reaction) (14, 15).
If indeed these potential islet precursors do go on to produce islets posttransplant, we believe that their potency is due to the sheer volume of cells transplanted (within the source tissue approximately 34% of cells were CK19 positive and 19% PDX-1 positive); additionally, postdigestion, ductal structures and acini (which harbor PDX-1-positive cells) represent small tissue units (Fig. 3A, iii), and therefore, the potential risk of central necrosis, which is a significant cause of ischemia in islets posttransplantation (6, 7), would be lessened.
Interestingly, this study suggests improved islet graft outcomes in association with high levels of PDX-1-positive cells transplanted. This finding is contentious, due to the fact that it is widely accepted that PDX-1-expressing cells are restricted to the islets of Langerhans in the adult pancreas (8). However, it is the opinion of some that the low-intensity staining of acini produced by PDX-1 antibodies represents specific staining due to the fact it is nonuniform and often nuclear (37). Additionally, improvements in standard immunohistochemistry techniques and available antibodies may have aided PDX-1 detection within this study. By employing Axiovision image analysis, this study was able to objectively show appreciable levels of non-islet PDX-1-positive cells within normal tissue (the lowest level recorded was 4.57% PDX-1 positivity), additionally significantly higher levels of PDX-1-positive cells were detected within CP patients and this finding concurs with small animal models showing increased PDX-1 expression in response to pancreatic injury (5). Moreover, the validity of staining is strengthened by the fact that PDX-1 antibodies directed to both the carboxyl terminal and N-terminal regions of the PDX-1 molecule produced near identical staining patterns, differing just in the levels of accompanying background stain (Fig. 3B, ii, iii).
Although it has proved difficult to study the conversion of adult non-islet PDX-1-positive cells into β-cells in vitro or posttransplantation, transfection experiments carried out in vitro have shown that PDX-1 uptake by ductal cells is accompanied with an acquisition of β-cell characteristics including transcription of genes involved in insulin production (25). Through mechanisms that are yet to be elucidated, it is conceivable that pancreas digestion and subsequent transplantation promotes β-cell genesis from PDX-1-positive islet precursors, although the exact mode of β-cell genesis is in doubt. Work carried out by Miyatsuka and colleagues (19) used a transgenic mouse model to show that sustained PDX-1 expression within the acinar parenchyma cells induced an acinar-ductal transition through signal transducer and activator of transcription 3 (STAT3) in a cell-autonomous manner. The Miyatsuka study provides an attractive mode by which PDX-1-positive cells may convert into the β-cell phenotype, that is, via the ductal phenotype, and indeed within this patient cohort a number of patient samples exhibited PDX-1 positivity, which was largely reminiscent of CK19 staining (Fig. 3B, ii, iii), supporting the supposition that PDX-1-positive cells may, in part, contribute to the ductal cell mass posttransplant. However, when analyzing the entire CP cohort (n = 23), no significant concordance between PDX-1 expression and CK19/CK7 expression was observed.
The role of ductal cells following islet transplantation is currently gaining momentum. Work carried out by Bogdani and colleagues in 2003 was able to show that pediatric ductal cells transplanted into a nude mouse model were able to give rise to a small population of insulin-positive cells at 1 month posttransplant (2) while a later clinical study by Street et al. was able to show that allotransplantation of high levels of ductal cells (within an islet graft) was positively correlated with a superior response to an oral glucose tolerance at 2 years posttransplant (32). Although there is a growing body of experimental data showing that ductal cells provide islets with biotrophic support (12, 21, 23), precursor– progeny relationships between ductal cells and islet cells have also been confirmed using lineage-based experiments (3). The data presented within this study suggest superior graft function up to 2 years posttransplant while islet equivalents transplanted could not be associated with improved graft outcomes past 1 year posttransplant, it is therefore possible that the presence of the ductal cells in islet autograft goes beyond providing islets with biotrophic support.
While the small ductal cells of the pancreas represent a recognized source of endocrine precursors (9, 10, 16), glucagon-positive cells associated with the duct potentially represent cells committed to islet neogenesis. This supposition is based on early studies by Pictet and colleagues and Rall et al., which suggest that glucagon is the first hormone detected during pancreas development and that the first secretory cells within the pancreas display granule characteristics consistent with the α-cell (27, 28). However, whether the α-cells associated with the duct give rise to islets is still in question. Correlation analysis revealed a significant positive relationship between glucagon-positive duct cells and CK19-positive cells, possibly suggesting that glucagon-positive cells are produced at a constant rate throughout the ductal epithelium and are therefore a surrogate marker for ductal cells. However, this supposition does not hold fast in all circumstances. One patient within the series had exhibited extensive weight loss principally due to the pain associated with eating and had undergone a previous partial pancreas resection in an attempt to remove the source of pain. Upon completion of pancreatectomy (and islet auto transplant), the patient who now had a BMI of <15 showed extensive and aberrant glucagon expression particularly associated with the ductal epithelia; indeed, certain ducts were enveloped in glucagon-positive cells (Fig. 3C, ii, iii). This finding potentially suggests that in extreme circumstances most, if not all, ductal epithelial cells have the ability to give rise to endocrine cells. Additionally, if the emergence of such cells does suggest a commitment to islet neogenesis, one could hypothesize that this phenomenon is an evolutionary attempt to maximize insulin output at times of severe starvation.
The present study demonstrates a number of significant correlations between increased levels of potential islet precursors transplanted and improved islet graft function, despite this, the results are subject to certain flaws. The major limitation is related to the small number of tissue samples available for study (n = 23) and incomplete patient follow-up; as such, many of the correlations presented are based on the results of between 9 and 16 patients. Furthermore, the correlation coefficients presented in Figures 4–7 are arguably low despite the significant p values, and therefore, the possibility of type I errors is of real concern. However, as already outlined, the strength of the correlations presented would have inevitably been affected by various patient and islet confounding factors, particularly the number of IEQs transplanted.
A further potential weak point of this study is that despite the high number of correlation analyses carried out (nine metabolic endpoints, six time points, and six predictor variables equating to 324 results), only 12 were seen to be statistically significant. All of the significant results were apparent within the first 2 years posttransplant, and this is likely to be due to reduced cohort sizes when analyzing data at years 3, 4, and 5 posttransplant. Furthermore, there is a likelihood that patient factors, that is, weight gain and lifestyle become increasingly important in the longer term, confounding any initial predictors of graft failure or success. Interestingly, a review of the correlations carried out between the islet precursors (PDX-1-positive cells, ductal cells, and glucagon- positive NIECs) and patient outcomes within the first 2 years posttransplant showed that, although many correlations did not reach significance, in the majority of cases (85%), a negative correlation was observed between increased precursors and increased glucose values (glucose 0, 30, and 120 and HbA1c) and positive correlation between increased islet precursors and increased C-peptide values (C-peptide 0, 30, 120 and SI30,120). These trends support the theory that transplantation of islet precursors is associated with higher C-peptide secretion and lower blood glucose values and also add credence to the significant results presented within Figures 4–7. However in the future, a larger patient cohort would be necessary to definitively determine whether islet precursors do contribute to islet function posttransplant.
Assuming that the data presented here are reflective of the fate of the nonpurified islet posttransplant, it therefore follows that purified islet grafts should be associated with inferior graft function compared with nonpurified preparations. In support of this theory, unpublished regression analysis of 31 consecutive patients within the Leicester series has shown that both fasting glucose and HbA1c are statistically lower over a 5-year period, while stimulated glucose and C-peptide values were equivalent in both groups. Although these data are encouraging, it must also be highlighted that patients receiving purified islets were predominantly transplanted during the early years of the Leicester program and therefore these data may reflect increased experience of personnel and improvements in islet isolation reagents.
Conclusions
The data presented here provide a preliminary study into the impact of potential islet precursors on long-term islet graft function. In line with previous findings, this study suggests that transplantation of non-islet precursor cells may significantly improve islet graft outcomes. Although the data presented here relate to the chronic pancreatitic pancreas, our analysis also shows appreciable levels of PDX-1-positive cells, glucagon-positive ductal cells, and most obviously ductal epithelial cells within the normal tissue. With islet transplantation groups increasingly seeking methods of predicting the success/failure of islet graft, this study and others suggest that it may prove useful to correlate both islet IEQs and the prevalence of islet precursor cells with long-term graft outcomes.
Footnotes
Acknowledgments
The authors would like to thank Professor Michael Nicholson for the use of his research facilities and Dr. Gareth Bicknell for the management of the islet isolation facilities within the University Hospitals of Leicester. Funding source: The University Hospitals of Leicester NHS Trust. Authorship: M. A. Webb participated in experimental design, laboratory work, data analysis, and manuscript preparation. J. J. Chen participated laboratory work and data analysis. Severine C. Illouz participated in the islet transplantation procedures and patient follow-up. Cristina A. Pollard participated in the islet transplantation procedures and patient follow-up. B. Dennison participated in laboratory work. K. P. West participated in experimental design and data analysis. R. F. L. James participated in experimental design and manuscript editing. A. R. Dennison participated in experimental design and manuscript editing. The authors declare no conflicts of interest.
