Abstract
Current studies suggest that mesenchymal stromal cells (MSCs) improve acute kidney injury (AKI) via paracrine/ endocrine effects. We established human adipose tissue-derived stromal cells (hASCs) cultured in low (2%) serum (hLASCs), which have great potential of tissue regeneration. The present study was performed to investigate the therapeutic effects of hLASCs on AKI and to clarify the mechanisms involved. In low serum, hASCs proliferated well, while human bone marrow-derived stromal cells (hBMSCs) did not. hLASCs secreted higher levels of hepatocyte growth factor (HGF) and vascular endothelial growth factor (VEGF) than did hASCs cultured in high (20%) serum (hHASCs) or hBMSCs cultured in high serum (hHBMSCs). AKI was induced in nude rats by folic acid, and hLASCs, hHASCs or control medium were administered into the renal subcapsules. hLASCs significantly attenuated acute renal damage, while hHASCs showed far less effect. Furthermore, interstitial fibrosis observed on day 14 was less pronounced in the hLASCs group. Cell tracking experiment showed no evidence of transdifferentiation. Intravenous injection of hLASCs or hHBMSCs or subcapsular injection of hHBMSCs did not ameliorate AKI. Concerning the mechanisms, our in vivo experiments showed that HGF knockdown by siRNA impaired the ability of hLASCs to protect the kidney from acute injury whereas VEGF knockdown did not. In conclusion, hLASCs, but not hHASCs or hHBMSCs, ameliorated AKI via paracrine effects, and HGF is one of the key mediators.
Introduction
The importance of acute kidney injury (AKI) has been well recognized because AKI is associated with significantly increased mortality and the patients' outcomes are related directly to the severity of AKI. Prevention and effective treatment of AKI are a high priority (7). However, kidney targeted interventions beyond supportive therapy are currently not available.
Previous studies have demonstrated that administration of mesenchymal stromal cells (MSCs) accelerates the recovery of tissue injury in several organs including heart, liver, neuron, and pancreas (12, 23, 29, 30). Administration of bone marrow-derived stromal cells (BMSCs) has also been shown to protect the kidney from AKI induced by cisplatin, glycerol, and ischemia-reperfusion injury (4, 13, 14, 21, 26, 35).
Recently, it has been demonstrated that MSCs can be obtained from adipose tissue (41). Like BMSCs, adipose tissue-derived stromal cells (ASCs) have the potential to differentiate into various types of cells and tissues (10, 33). Previous studies suggest that ASCs may have an advantage over BMSCs. Firstly, adipose tissue is abundant and can be obtained repeatedly with minimal invasive procedure. Secondly, the number of stem cells in the fat is greater than that in the bone marrow (18). Lastly, in general ASCs grow faster than BMSCs (18, 22, 39).
In a previous study, we reported a novel culture system for human ASCs (hASCs) using low serum medium (17). hASCs cultured in low (2%) serum (hLASCs) secreted higher levels of hepatocyte growth factor (HGF) and vascular endothelial growth factor (VEGF) than in high (20%) serum (hHASCs). hLASCs also showed significantly greater therapeutic effects on rat hind limb ischemia than hHASCs. We have also demonstrated that ASCs cultured in low serum (LASCs) have great potential for transdifferentiation and immunosuppression (31, 40).
The precise mechanism that attenuates kidney injury after MSCs injection remains to be clarified (3). The process might involve recruitment of MSCs to the site of injury, transfer of microvesicles, or most likely paracrine/ endocrine effect (15). Several reports suggest that the renal protective properties of MSCs may be attributed to the action of insulin-like growth factor-1(IGF-1), HGF, and VEGF (16, 36, 37). In the present study, we hypothesized that hLASCs may have a great potential as a novel resource for cell therapy of AKI. Investigation of renoprotective factors of hLASCs would possibly provide new insight for the management of AKI.
The aim of the present study was to examine whether there are therapeutic advantages to using hLASCs rather than hHASCs or human BMSCs (hBMSCs) in an AKI model induced by folic acid injection in rats. In addition, we aimed to investigate the mechanisms involved in the treatment of AKI, focusing on HGF and VEGF.
Materials and Methods
Culture Conditions
The basal culture medium was prepared as previously described (17). Cells were cultured under the following two conditions: a low serum culture medium containing 2% fetal bovine serum (FBS) (HyClone, Logan, UT, USA) and 10 ng/ml human fibroblast growth factor-2 (FGF-2) (Peprotech, Inc., Rocky Hill, NJ, USA) and a high serum culture medium containing 20% FBS and 10 ng/ml human FGF-2.
Preparation of hASCs and hBMSCs
Human abdominal subcutaneous adipose tissue was obtained with the written informed consent from patients undergoing surgery. All tissue samples were used with approval and according to the guidelines of the ethical committee at the Nagoya University Medical School (approval No. 505-2), and the study was performed according to the guidelines of the Declaration of Helsinki. hASCs were prepared as previously described (17). hBMSCs were purchased from LONZA (Basel, Switzerland).
Growth Speeds of hASCs and hBMSCs
hASCs from five patients or hBMSCs from three patients (2 × 105) were cultured for 30 days. Growth kinetics was calculated at each time point and shown as mean ± SD as described (17).
Measurement of HGF and VEGF Secreted From hASCs and hBMSCs
Secretion of HGF and VEGF was studied as described (17). Briefly, hLASCs and hHASCs obtained from five patients, and hBMSCs cultured in high serum (hHBMSCs) from three patients were expanded, and cells at passages 4 through 7 were used. The 24-h conditioned media were analyzed using HGF Otsuka enzyme-linked immunosorbent assay (ELISA) kit (Otsuka Pharmaceutical Co., Tokyo, Japan) or Quantikine Human VEGF Immunoassay (R&D Systems, Minneapolis, MN, USA). Data are expressed as nanograms of the secreted factor per 106 cells at the time of harvest.
Animals
Male F344/NJcl-rnu/rnu rats were purchased from CLER, Inc. (Tokyo, Japan).
Animals were housed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. The experimental protocols were approved by the Animal Experimentation Guidelines of Nagoya University Graduate School of Medicine.
Folic Acid-Induced Acute Kidney Injury
Eight-week-old male F344/NJcl-rnu/rnu rats weighing approximately 160–180 g were subjected to right heminephrectomy. Seven days later (day 0), AKI was induced in immunodeficient rats by the intravenous injection of folic acid (Sigma-Aldrich, St. Louis, MO, USA) at the dose of 200 mg/kg.
In Vivo Experimental Protocol 1: Subcapsular Administration of hASCs
Subcapsular injection of 6 × 106 of hLASCs, hHASCs, or control medium (Dulbecco's modified Eagle's medium, DMEM; Sigma-Aldrich) (each group n = 8) was given to the left kidney of AKI rats, immediately after folic acid injection. Blood samples were collected on days 0, 1, 2, 6, and 14, and blood urea nitrogen (BUN) and serum creatinine levels were measured by Mitsubishi Chemical Medience Co. Ltd (Tokyo, Japan). Kidney samples were taken on day 14. AKI was induced in another set of rats, and hLASCs, hHASCs, or control medium (each group n = 8) were injected in the same way. Rats were euthanized on day 2, renal cortical microcirculation was assessed using CCD video microscope (28), and kidney samples were taken for the study.
Histology and Immunohistochemistry
Kidney sample was processed for routine histology and immunohistology as described previously (32). Terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) assay was performed using in situ Apoptosis Detection Kit (Takara Bio, Otsu, Japan) as described (34). Immunostaining for monocytes/ macrophages (ED-1), α-smooth muscle actin (α-SMA), and type III collagen was performed on buffered formalin-fixed tissues. Antibodies used were mouse anti-rat monocyte/macrophage antibody (ED-1, BMA Biomedicals, Augst, Switzerland), mouse anti-α-SMA antibody (Dako, Glostrup, Denmark), rabbit anti-type III collagen antibody (Cosmo Bio, Tokyo, Japan), a conjugate of polyclonal goat anti-mouse IgG antibody or anti-rabbit IgG antibody, and horseradish peroxidase-labeled polymer (Histofine Simple Stain, Nichirei, Tokyo, Japan).
Morphological Analysis
To evaluate tubulointerstitial injury, periodic acid Schiff (PAS)-stained kidney sections were analyzed using a quantitative grading as described previously (24). The numbers of TUNEL-positive cells and ED-1-positive cells were counted in 10 nonoverlapping fields (200x magnification) of the cortex and the numbers were expressed per field. Renal fibrosis was assessed by quantification of Masson's trichrome (MT), α-SMA or type III collagen-positive area using the MetaMorph 6.3 image analysis program (Universal Imaging, West Chester, PA, USA) as described (20).
Measurement of HGF and VEGF Concentrations in the Renal Cortex
Renal cortex samples taken on day 2 were homogenized with RIPA Buffer (Pierce, Rockford, IL, USA) and centrifuged, and the supernatants were collected. The levels of growth factors were measured using Human HGF Otsuka ELISA kit (Otsuka), Quantikine Human VEGF Immunoassay (R&D Systems), Rat HGF EIA kit (Institute of Immunology, Tokyo, Japan), and Rat VEGF ELISA kit (Invitrogen, Carlsbad, CA, USA).
In Vivo Experimental Protocol 2: hLASCs Tracking Study
Subcapsular injection of 6 × 106 hLASCs was given to rats with folic acid-induced AKI and kidney samples were removed on day 2 or day 14 (each group n = 6). Frozen sections were stained with mouse monoclonal anti-Lamin A/C antibody (Novocastra/Vision BioSystems, Norwell, MA, USA) followed by goat anti-mouse IgG antibody (Histofine).
In Vivo Experimental Protocol 3: Intravenous Injection of hLASCs
Tail vein injection of 6 × 106 of hLASCs or PBS (each group n = 6) was given to rats with AKI immediately after folic acid injection. Blood samples were taken on days 0, 1, and 2 for the measurement of BUN and serum creatinine.
In Vivo Experimental Protocol 4: Subcapsular or Intravenous Injection of hHBMSCs
Subcapsular administration of hHBMSCs (6 × 106) or control medium (each group n = 6), or intravenous injection of hHBMSCs (6 × 106) or PBS (each group n = 6) was performed in rats with AKI immediately after folic acid injection. Blood samples were taken on days 0, 1, and 2 for the measurement of BUN and serum creatinine.
Transfection of siRNA Into hLASCs
ON-TARGET plus SMART pool siRNA targeted to human HGF or human VEGF was purchased (Dharmacon, Lafayette, CO, USA) and tested at concentrations of 5, 10, and 50 nM. Forward transfection procedure was used into hLASCs in a 24-well format. hLASCs (2 × 104) were seeded onto 24-well plates (BD Falcon, Franklin Lakes, NJ, USA) in 500 ml of low serum medium without antibiotics. The next day, diluted siRNA duplex with the diluted Lipofectamine RNAiMAX (Invitrogen) in Opti-MEM I Reduced Serum Medium (Gibco, Carlsbad, CA, USA) was transfected into hLASCs. After 6 h of incubation, medium change was performed. Knockdown efficiency was detected on the protein level in the cultured medium measured by ELISA kits; HGF Otsuka ELISA kit (Otsuka), Quantikine Human VEGF Immunoassay (R&D Systems) at 48 and 96 h after transfection. Controls consisted of hLASCs treated with ON-TARGET plus Non-targeting pool (negative control siRNA) (Dharmacon), Lipofectamine RNAiMAX transfection agent only, and untreated cells.
In Vivo Experimental Protocol 5: Subcapsular Administration of HGF or VEGF Knockdown hLASCs
In the folic acid-induced rat AKI model, 2 × 106 of hLASCs transduced with HGF siRNA (hLASCs-siHGF), VEGF siRNA (hLASCs-siVEGF), negative control siRNA (hLASCs-siCtrl) or control medium (each group n = 9) were injected into the subcapsular space of the kidney at 2 days after transfection. Blood samples were collected on days 0, 1, 2, and BUN and creatinine levels were measured.
Statistical Analysis
Statistical analysis was performed using the software program, Stat View 5.0 (SAS Institute, Cary, NC, USA). Student's t test was used to determine the significant differences between two groups. Two-way analysis of variance (ANOVA) was employed to determine the significant difference among three or four groups. When a statistical difference was indicated by ANOVA, further analysis was performed using Scheffe to determine the difference between any pair of groups. A value of p < 0.05 was considered statistically significant. All values are expressed as mean ± SD.
Results
Growth Kinetics of hASCs and hBMSCs
The cell proliferation speeds of hASCs and hBMSCs were compared under low serum (2%) and high serum (20%) culture conditions. hHASCs grew the fastest, followed by hLASCs. Of note, hLASCs grew faster than hHBMSCs. Low serum cultured hBMSCs (hLBMSCs) proliferate too slowly to be used in the following experiments (Fig. 1A).

(A) Growth speeds of human adipose tissue-derived stromal cells (hASCs) and human bone marrow-derived stromal cells (hBMSCs) under low serum (2%) or high serum (20%) culture conditions. High serum cultured human adipose tissue-derived stromal cells (hHASCs) grew the fastest, followed by low serum cultured human adipose tissue-derived stromal cells (hLASCs), and human bone marrow-derived stromal cells cultured in high serum (hHBMSCs). Low serum cultured human bone marrow-derived stromal cells (hLBMSCs) did not proliferate well. (B) Secretion of growth factors by hLASCs, hHASCs, and hHBMSCs. The levels of hepatocyte growth factor (HGF) and vascular endothelial growth factor (VEGF) were significantly higher in the hLASCs-conditioned medium than those in the hHASCs or hHBMSCs-conditioned medium. ∗p < 0.05 versus hHASCs or hHBMSCs, ∗∗p < 0.01 versus hHASCs or hHBMSCs.
Growth Factor Secretion by hASCs and hBMSCs
Since HGF and VEGF are known to promote recovery from ischemic tissue injury (17, 25, 27), secretion of these molecules was analyzed. hLASCs secreted higher levels of HGF and VEGF than did the hHASCs or hHBMSCs at passages 4 through 7 while no difference was observed between the levels of HGF or VEGF secreted by hHASCs and hHBMSCs (Fig. 1B).
Effects of Subcapsular Injection of hLASCs and hHASCs
Renal Function
An experimental model of AKI was induced in immunodeficient rats, and hLASCs, hHASCs, or control medium were injected into the subcapsular space of the kidney immediately upon folic acid injection. Rats treated with control medium demonstrated a marked rise in BUN and serum creatinine on days 1 and 2, whereas rats given hHASCs showed preserved renal function on day 2. Interestingly, hLASCs further suppressed the increase of BUN and serum creatinine on days 1 and 2 (Fig. 2).

Renoprotection by subcapsular administration of low serum cultured human adipose tissue-derived stromal cells (hLASCs). Immunodeficient rats were given intravenous injection of folic acid on day 0, followed by subcapsular administration of hLASCs, high serum cultured human adipose tissue-derived stromal cells (hHASCs), or control medium. Blood urea nitrogen (BUN) and serum creatinine were measured on days 0, 1, 2, 6, and 14. hLASCs markedly attenuated renal injury, as reflected by the significantly lower BUN and serum creatinine levels on days 1 and 2, whereas hHASCs showed less renoprotection. ∗p < 0.05 versus control, ∗∗p < 0.01 versus control, #p < 0.05 versus hHASCs, ##p < 0.01 versus hHASCs. Hpf, high power field.
Tubular Injury
Examination of PAS-stained kidney sections taken from AKI rats treated with control medium showed severe tubular cell degenerative changes with necrosis and luminal casts on day 2 (Fig. 3A). hLASCs greatly attenuated the tubular injury, while hHASCs partially attenuated the tubular damage (Fig. 3B, C). The severity of the tubular damage, including tubular dilatation, degeneration, and cast formation was scored (24). Treatment with hLASCs resulted in significantly better scores than the control or hHASCs group. In contrast, the hHASCs-treated group failed to show significantly better scores than the control group (Fig. 3D).

Tubular damage and macrophage infiltration. (A–C) Representative micrographs show the kidney sections taken on day 2 from AKI rats treated with control medium (A), AKI rats treated with high serum cultured human adipose tissue-derived stromal cells (hHASCs) (B), and those treated with low serum cultured human adipose tissue-derived stromal cells (hLASCs) (C), and followed by periodic acid Schiff (PAS) staining. The hLASCs group showed significantly better tubular necrosis scores than the other groups (D). (E–G) Terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) staining performed on the kidney sections taken on day 2 from the control group (E), the hHASCs group (F), and the hLASCs group (G). hLASCs significantly decreased the number of apoptotic cells as compared to the other groups (H). (I–K) ED-1 staining performed on the kidney sections taken on day 2 from the control group (I), the hHASCs group (J), and the hLASCs group (K). The numbers of ED-1-positive cells were significantly lower in the hLASCs group than the other groups (L). Original magnification: 200x (in all micrographs). Scale bar: 100 mm (A–C, E–G, I–K). ∗∗p < 0.01 versus control, #p < 0.05 versus hHASCs, ##p < 0.01 versus hHASCs.
Tubular Cell Apoptosis
TUNEL assay was performed on the kidney sections obtained on day 2 (Fig. 3E–G). Transplantation of hLASCs significantly decreased the numbers of TUNEL-positive cells as compared to control or hHASCs, while hHASCs did not (Fig. 3H).
Macrophage Infiltration
Macrophage infiltration was evident in the interstitium of the AKI rats treated with control medium (Fig. 3I). hHASCs treatment slightly suppressed macrophage infiltration (Fig. 3J). Further suppression was observed in the hLASCs group (Fig. 3K). Quantification of ED-1-positive cells showed a significant reduction in the number of macrophages in the hLASCs group while no significant suppression was observed in the hHASCs group (Fig. 3L).
Growth Factors in the Kidney
Since hLASCs produced significantly higher levels of HGF and VEGF than hHASCs in vitro, the concentrations of these growth factors in the renal cortex were measured on day 2. In the hLASCs group, the human HGF and VEGF levels were significantly higher than in the hHASCs group (Fig. 4A). The rat HGF level was significantly higher in the hLASCs group compared with the control group, while the hHASCs group did not show a significant difference. In contrast, no differences were observed in the rat VEGF levels among the three groups (Fig. 4B).

(A, B) Levels of hepatocyte growth factor (HGF) and vascular endothelial growth factor (VEGF) in the renal cortex. HGF and VEGF concentrations were measured in the homogenates of the renal cortex samples taken on day 2 from AKI rats given subcapsular injection of low serum cultured human adipose tissue-derived stromal cells (hLASCs), high serum cultured human adipose tissue-derived stromal cells (hHASCs), or control medium. Levels of human HGF and VEGF were significantly higher in the hLASCs group as compared to the hHASCs group (A). In the hLASCs group, rat HGF concentration was significantly higher than in the control group. No differences were observed in the levels of rat VEGF among three groups (B). (C) Renal cortical microcirculation. Velocity of the capillary blood flow and the capillary blood flow volume were significantly higher in AKI rats given subcapsular injection of hLASCs than those given the control medium, while hHASCs did not show significant effects. ∗p < 0.05 versus control, ∗∗p < 0.01 versus control, ##p < 0.01 versus hHASCs.
Direct Visualization of the Renal Cortical Capillaries
The effects of hLASCs or hHASCs on renal cortical microcirculation were examined by analyzing the direct images obtained with a CCD video microscope system on day 2. The blood flow velocity was significantly faster and the blood flow volume was greater in the hLASCs group than in the control or the hHASCs groups (Fig. 4C).
Subsequent Interstitial Fibrosis
MT staining was performed on the kidney sections obtained on day 14 (Fig. 5A–C). Quantitative analysis with the use of the Meta Morph 6.3 image computer program demonstrated that the MT-positive area was significantly reduced in the hLASCs group but not in the control group (Fig. 5D). α-SMA staining was performed on the kidney sections obtained on day 14 (Fig. 5E–G). Administration of hLASCs significantly decreased the interstitial α-SMA-positive area as compared to the control medium or hHASCs (Fig. 5H). Type III collagen staining was also performed (Fig. 5I–K). Quantification of type III collagen-positive area on day 14 showed a significant reduction in the hLASCs group while no significant change was observed in the hHASCs group (Fig. 5L).
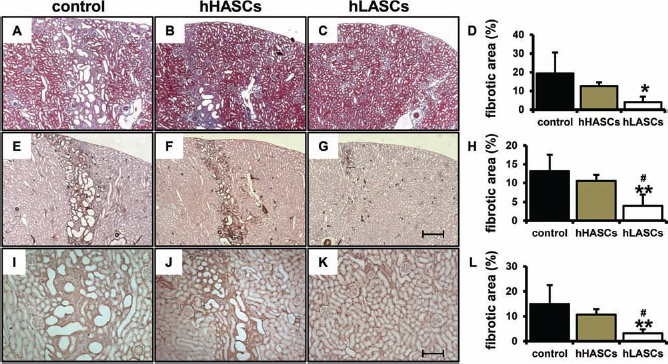
(A–L) Subcapsular administration of low serum cultured human adipose tissue-derived stromal cells (hLASCs) decrease subsequent renal fibrosis. Representative micrographs show the kidney sections taken on day 14 from rats given control medium (A, E, I), high serum cultured human adipose tissue-derived stromal cells (hHASCs) (B, F, J), or hLASCs (C, G, K). Masson's trichrome (MT) (A–C), α-smooth muscle actin (α-SMA) (E–G), and type III collagen (I–K) staining were performed. (D, H, L) Quantitative analysis of fibrotic area with the use of the Meta Morpho demonstrated that renal fibrosis detected by MT (D), α-SMA (H), and type III collagen (L) was reduced in the hLASCs group. Original magnifications: 50x (in A–G) and 100x (in I–K). Scale bars: 400 μm (A–C, E–G) and 200 μm (I–K). ∗p < 0.05 versus control, ∗∗p < 0.01 versus control, #p < 0.05 versus hHASCs.
Fate of Transplanted hLASCs
Human lamins A and C immunostaining was performed on the kidney sections from rats with AKI on days 2 and 14 after hLASCs transplantation into the subcapsular space. hLASCs were detected only in the subcapsule of the kidney on day 2 (Fig. 6A), and the transplanted cells remained alive in the subcapsule on day 14 (Fig. 6B).

Representative micrographs of low serum cultured human adipose tissue-derived stromal cells (hLASCs) transplanted into the subcapsule of the kidney of AKI rats. hLASCs stained by anti-human Lamins A and C were detected in the subcapsule of the kidney on days 2 (A) and 14 (B). Original magnification: 100x. Scale bar: 200 μm (A, B).
Therapeutic Effects of Intravenous Injection of hLASCs
In order to study whether or not intravenous injection of hLASCs is as effective, AKI was induced by folic acid injection followed by intravenous administration of hLASCs. The increase in the levels of BUN and serum creatinine was not statistically significant (Fig. 7A).

Renal function after the intravenous injection of low serum cultured human adipose tissue-derived stromal cells (hLASCs), subcapsular injection of high serum cultured human bone marrow-derived stromal cells (hHBMSCs), or intravenous injection of hHBMSCs. Immunodeficient rats were given folic acid on day 0, followed by intravenous injection of hLASCs (A), subcapsular injection of hHBMSCs (B), or intravenous injection of hHBMSCs (C). Blood urea nitrogen (BUN) and serum creatinine levels were measured on days 0, 1, and 2. No significant difference was observed in any set of experiments.
Therapeutic Effects of Subcapsular or Intravenous Administration of hHBMSCs
To investigate whether hHBMSCs are renoprotective in AKI, hHBMSCs were injected into the subcapsular space of the kidney or the tail vein, immediately after folic acid injection. Subcapsular administration of hHBMSCs showed a tendency to reduce BUN and serum creatinine elevation, which was not statistically significant (Fig. 7B). Intravenous injection of hHBMSCs did not decrease the elevation of BUN and serum creatinine (Fig. 7C).
Knockdown of HGF or VEGF in hLASCs by siRNA
Knockdown efficiency was determined on the protein levels in the culture media supernatants of hLASCs at 48 and 96 h after transfection. Treatment with either HGF or VEGF siRNA at a concentration of 10 nM proved to be the most effective. In the hLASCs-siHGF and hLASCs-siVEGF, greater than 80% knockdown level of HGF or VEGF expression was observed at 48 and 96 h after transfection compared to the hLASCs-siCtrl. HGF or VEGF concentrations in the supernatants of untreated hLASCs, hLASCs treated with transfection agent only, or hLASCs-siCtrl were similar (Fig. 8)

Knockdown of human hepatocyte growth factor (HGF) and vascular endothelial growth factor (VEGF) in low serum cultured human adipose tissue-derived stromal cells (hLASCs) in vitro. Human HGF and VEGF protein concentrations in the cell culture supernatants of hLASCs treated with specific siRNA at 48 and 96 h after transfection. Either hLASCs transduced with HGF siRNA (hLASCs-siHGF) or hLASCs transduced with VEGF siRNA (hLASCs-siVEGF) yielded a greater than 80% knockdown. hLASCs, hLASCs treated with transfection agent only, and hLASCs transduced with negative control siRNA (hLASCs-siCtrl) showed almost equivalent HGF and VEGF concentrations.
Effects of HGF or VEGF Knockdown hLASCs on Renal Function
The in vivo effects of hLASCs-siHGF or hLASCs-siVEGF on renal function were studied in the rat AKI model. In the hLASCs-siCtrl goup, renal injury assessed by BUN and serum creatinine levels on days 1 and 2 was significantly attenuated compared to the control. hLASCs-siHGF significantly reduced the renoprotection exerted by hLASCs-siCtrl. hLASCs-siVEGF also reduced renoprotective effects, which however was not significant (Fig. 9).

Hepatocyte growth factor (HGF) knockdown reduced renoprotective effects of low serum cultured human adipose tissue-derived stromal cells (hLASCs) in vivo. Immunodeficient rats were given intravenous injection of folic acid on day 0, followed by subcapsular administration of hLASCs transduced with HGF siRNA (hLASCs-siHGF), hLASCs transduced with vascular endothelial growth factor (VEGF) siRNA (hLASCs-siVEGF), hLASCs transduced with negative control siRNA (hLASCs-siCtrl), or a control medium. Blood urea nitrogen (BUN) and serum creatinine values were higher in the hLASCs-siHGF-treated group than in the hLASCs-siCtrl-treated groups on days 1 and 2. ∗∗p < 0.01 versus control, #p < 0.05 versus hLASCs-siHGF.
Discussion
The present study provides clear evidence that administration of hLASCs into the renal subcapsule protects the kidney from acute injury induced by folic acid as well as subsequent fibrosis more efficiently than does administration of hHASCs. In contrast, hHBMSCs failed to offer significant renoprotection.
hBMSCs have been shown to secrete a variety of growth factors, which promote tissue regeneration (1, 6, 11, 35). In a previous study, we have shown that hLASCs secrete higher levels of HGF and VEGF and promote ischemic tissue recovery more efficiently than hHASCs (17). In the present study, we further compared hASCs and hBMSCs regarding the proliferation and growth factor secretion and found that hASCs showed better growth rate than did hBMSCs and that a high serum medium resulted in a greater proliferation rate than a low serum medium. Of note, hLASCs proliferated better than hHBMSCs. Concerning the growth factors, hLASCs secreted higher levels of HFG and VEGF than did hHASCs or hHBMSCs. These results indicate that hLASCs are a useful and powerful tool in regenerative cell therapy.
In order to deliver MSCs to the target organ efficiently, we tried direct subcapsular injection (8, 19). When we administered hLASCs at 5 h after induction of AKI, a significant therapeutic effect was not obtained (data not shown). The tubular damage in this model may have already been established by this time. We then administered hLASCs immediately after folic acid injection. And we found that administration of hLASCs showed significant renoprotection while hHASCs showed only moderate effects. Our result—that a subcapsular delivery of hHBMSCs failed to show significant therapeutic effects—suggests a clear advantage for hLASCs over hHBMSCs. Since hLASCs secreted high levels of HGF and VEGF, the levels of these molecules in the kidney samples were measured. We found that the concentrations of HGF and VEGF were significantly higher in the renal cortex of the hLASCs group than the hHASCs group, suggesting that hLASCs produced and secreted high levels of HGF and VEGF in vivo. Moreover, we demonstrated that rat HGF concentration in the renal cortex was significantly higher in the hLASCs group. It was previously reported that HGF promotes the expression of HGF itself (38). Therefore, we assume that the human HGF produced by hLASCs promoted further rat HGF expression, thus attenuating tubular injury. Of interest, only a single delivery of hLASCs at day 0 reduced the subsequent fibrosis evaluated on day 14.
To date, the way MSCs promote tissue regeneration remains controversial. Previous studies have suggested that the renoprotective effects are attributable to the replacement of damaged tubular cells by transdifferentiation of MSCs (9, 26). However, recent studies have suggested that almost no MSCs are transdifferentiated into renal cells when applied to the AKI model (5). In our study, hLASCs remained alive in the renal subcapsule at days 2 and 14 after injection, showing no evidence of migration and transdifferentiation. A recent study has demonstrated that intravenous injection of either BMSCs or ASCs protected against cisplatin-induced AKI via an endocrine effect since even administration of conditioned culture medium alone ameliorated renal injury (4). Based on these findings, we then tried intravenous administration of hLASCs and hHBMSCs and found that neither of them showed significant therapeutic effects in our model. These results suggest that hLASCs exert renoprotection mainly via a paracrine effect.
In order to specifically evaluate the contribution of HGF or VEGF toward hLASCs mediated renoprotection, we conducted a HGF or VEGF knockdown experiment using siRNA. It has already been reported that knockdown of VEGF by siRNA lessened the therapeutic effect of MSCs in the ischemia-reperfusion AKI model (37). However, there are no reports regarding the effects of HGF knockdown. In our study, VEGF knockdown decreased the effectiveness of hLASCs, which however was not statistically significant. In contrast, we showed for the first time that knocking down HGF in hLASCs significantly reduced their therapeutic activity, suggesting that HGF is a predominant mediator of renoprotection in the folic acid-induced AKI model.
There are several possible explanations concerning the difference between the VEGF and HGF knockdown effects. hLASCs secreted 6–10 times more HGF than did hHASCs, whereas the level of VEGF secretion by hLASCs was only 2–4 times higher. The effects of VEGF knockdown may have been offset by a high level of HGF. Another explanation is that HGF was particularly effective in the model employed in our study. VEGF has been shown to protect endothelial cells from injury, while HGF is known to protect not only endothelial cells but also renal tubular cells. The contribution of VEGF may be greater in the ischemic renal injury model where endothelial cells are severely damaged. In folic acid-induced AKI, where renal tubular cells, not endothelial cells, are the primary target of tissue injury, the contribution of HGF may be greater. Furthermore, the superior anti-inflammatory effect shown by hLASCs may also be attributed to the action of HGF.
Of note, HGF knockdown did not fully cancel the renoprotection of hLASCs, indicating that all of the paracrine effects of hLASCs cannot be attributed to a single growth factor. Cell therapy, as a new strategy, exerts the combined actions of several factors to simultaneously reduce tissue damage and is therefore potentially more effective than any single growth factor or drug therapy.
What is interesting is the fact that a clear difference exists in the potency of renoprotection between hLASCs and hHASCs even derived from the same donor patient. We propose that these ASCs are of great use as tools in future research. Studying the molecular differences between hLASCs and hHASCs and examining the function, for instance, by using a siRNA system as employed in this study, will further reveal molecular mechanisms, leading to the development of novel therapies.
That hHBMSCs did not show significant renoprotection in the present study may be attributable to the model employed. In order to evaluate chronic fibrosis, we used folic acid-induced AKI (2), while previous studies used AKI models induced by cisplatin, glycerol, or ischemia-reperfusion injury (4, 13, 14, 21, 26, 35). Moreover, we used nude rats whose immune reaction is compromised. Therefore, it was difficult to evaluate the immunosuppressive effects of ASCs or BMSCs in this study.
In conclusion, we demonstrate that subcapsular administration of hLASCs protects the kidney from acute tubular injury. Our data suggest that HGF is an important mediator in the renoprotective effects of hLASCs and provide evidence that hLASCs will be considered a new source for cell therapy with providing a mixture of several humoral factors. Moreover, hLASCs and hHASCs are useful for a deeper investigation of the molecular mechanisms involved in organ protection.
Footnotes
Acknowledgments
The authors thank N. Asano, Y. Sawa, and N. Suzuki for excellent technical assistance. This study was supported in part by a Grant-in-Aid for Progressive Renal Diseases Research, Research on Intractable Disease, from the Ministry of Health, Labor, and Welfare of Japan. The authors declare no conflict of interest.
