Abstract
ZNF281 is one of the core transcription factors in embryonic stem cells (ESCs) and has activation and repression roles in the transcription of ESC genes. A known target molecule of Zfp281 (the mouse homologue of ZNF281) is Nanog. However, NANOG is not expressed in most human multipotent stem cells (hMSCs). Here, we investigated the roles of ZNF281 with a gain- and loss-of-function study. The knockdown of ZNF281 in vivo and in vitro resulted in spontaneous osteochondrogenic differentiation and reduced the proliferation of hMSCs, as determined by cell morphology and molecular markers. When ZNF281-knockdown hMSCs were subcutaneously implanted into mice along with β-tricalcium phosphate (β-TCP), many cells were converted into osteoblasts within 4 weeks. In contrast, the overexpression of ZNF281 in hMSCs resulted in accelerated proliferation. The expression pattern of ZNF281 correlated well with the expression of β-CATENIN during differentiation and in the gain/loss-of-function study in hMSCs. The binding of ZNF281 to the promoter region of β-CATENIN was observed using a chromatin immunoprecipitation (ChIP) assay. In conclusion, we propose that ZNF281 plays an important role in the maintenance and osteogenic differentiation of stem cells via the transcriptional regulation of genes including β-CATENIN.
Keywords
Introduction
Stem cell therapy, especially with the use of human multipotent stem cells (hMSCs), is regarded as one of the most promising therapeutic tools (15, 16, 33, 50). A primary characteristic of hMSCs is their ability to differentiate into adipocytes, osteocytes, and chondrocytes (11). Some of the molecular regulatory mechanisms of hMSCs have been researched by us and others (1, 2, 22). However, the mechanisms by which self-renewal and differentiation is regulated on a molecular level are not well known.
Several core transcription factors play critical roles in embryonic stem cells (ESCs) (23), and some of these factors are expressed in hMSCs that largely conserve their functions. Octamer-binding transcription factor 4 type A (OCT4A) is expressed in human umbilical cord blood-derived MSCs (hUCB-MSCs), as in ESCs (39), and reduced expression gene 1 (REX1) expression in hMSCs is critical in the cell signaling of p38 mitogen-activated protein kinase (p38MAPK) via mitogen-activated protein kinase kinase 3 (MKK3) activation (2). Zinc finger protein 281 (ZNF281), initially named zinc finger DNA-binding protein 99 (ZBP-99), is one of the core transcription factors in ESCs (45). ZNF281 contains four Krüppel-type zinc fingers that collectively share 91% amino acid sequence similarity and 79% sequence identity with those present in ZBP-89. The predicted open reading frame of the ZNF281 cDNA encodes a 99-kDa protein. Electrophoretic mobility shift assays have shown that the ZBP-99 protein specifically binds to the GC-rich promoter elements in the gastrin and ornithine decarboxylase genes (26, 30). ZNF281 has also been identified as a myelocytomatosis oncogene (c-MYC)-associated protein using combined tandem affinity purification with mass spectral multidimensional protein identification technology (24). Zfp281, the mouse homolog of ZNF281, has two domains (an activation domain and a repressor domain) and has bifunctional roles in regulating the gene expression of mouse ESCs during differentiation. A putative direct target of Zfp281 regulation in mouse ESCs is the Nanog homeobox transcription factor (Nanog) (48). However, NANOG is not expressed or is barely expressed in hMSCs (39). The role of ZNF281 in human ESCs, as well as in adult stem cells, has not been studied thoroughly.
Activation of the canonical Wnt pathway is sufficient to maintain the self-renewal of both human and mouse ESCs (37). The canonical Wnt pathway is also important for the differentiation and self-renewal of MSCs. Periodic activation of the Wnt/β-catenin pathway provides a specific threshold that leads to the reprogramming of somatic cells after fusion (32). Wnt3a signaling is involved in the proliferation of rat bone marrow-derived MSCs and hMSCs and promotes the osteogenic differentiation of hMSCs (4, 31, 40). The activation of the canonical Wnt signaling is an early event in adipogenesis during MSC differentiation (5), and the activation of β-catenin induces myogenesis and inhibits adipogenesis in rat MSCs (40). Endogenous Wnt signaling promotes proliferation and suppresses osteogenic differentiation in human adipose tissue-derived MSCs (hAD-MSCs) (8). Endogenous canonical Wnt signaling is considered an autocrine process in primitive MSCs (12).
In this study, the hypothesis that ZNF281 expression in human adult stem cells has essential role in cell proliferation and differentiation was tested. Knockdown of ZNF281 in vivo and in vitro in adult stem cells resulted in spontaneous osteochondrogenic differentiation and reduced the proliferation of hMSCs.
Materials and Methods
Isolation of hMSCs and Cell Culture
Umbilical cord blood samples were obtained from the umbilical vein immediately after delivery. The cells were grown in D-media (formula no. 78–5470EF, Gibco BRL, Grand Island, NY) containing endothelial cell growth medium-2 (EGM-2) single quot and 10% fetal bovine serum (FBS) (Gibco BRL) as in our previous studies (1, 2, 39). The Tera-1 cell line was purchased from ATCC (HTB-105, Manassas, VA) and was maintained in low-glucose Dulbecco's modified Eagle medium (DMEM, Gibco BRL) containing 10% FBS (Gibco BRL). hAD-MSCs were isolated and characterized as previously reported (17). The hAD-MSCs were maintained in keratinocyte serum-free medium (K-SFM), which was supplemented with 2 mM N-acetyl-l-cysteine (Sigma-Aldrich, St. Louis, MO) and 0.2 mM l-ascorbic acid (Sigma-Aldrich). The cell growth rates were measured using the cumulative population doubling level (CPDL) and a cell counting kit-8 (CCK-8) (Dojindo Molecular Technologies, Inc., San Diego, CA) according to the manufacturer's instructions. The isolation and research use of hUCB-MSCs and hAD-MSCs were approved by the Boramae Hospital Institutional Review Board (IRB) with written consent. All procedures were approved by the IRB of Seoul National University (hUCB-MSC, #0603/001-002; hAD-MSC, #0600/001-001).
Flow Cytometry Analysis and Western Blots
A ZNF281 antibody (ab53574, Abcam) was conjugated to fluorescein isothiocyanate (FITC) (Abcam, Cambridge, UK), and a fluorescence-activated cell sorter (FACS) Aria system (Becton & Dickinson, Franklin Lakes, NJ) was used for flow cytometry analysis. For Western blot analysis, the cells were lysed with lysis buffer (20 mM Tris HCl, 150 mM NaCl, 1 mM EDTA, 1 μg/ml aprotinin, 1 μM leupeptin, 1 mM PMSF, 1 mM Na3VO4, and 1% Triton X-100, Sigma). The proteins were detected with the corresponding primary antibodies: ZNF281 (ab53574, Abcam), β-CATENIN (#9562, Cell Signaling Technology, Inc., Danvers, MA), phospho-β-CATENIN (p-β-CATENIN) (#9561, Cell Signaling Technology, Inc.), glycogen synthase kinase 3-β (GSK3-β) (#9315, Cell Signaling Technology, Inc.) and phospho-GSK3-β (p-GSK3-β) (#9336, Cell Signaling Technology, Inc.). After primary antibody incubation, the appropriate horseradish peroxidase-linked secondary antibodies (Zymed Laboratories, Inc., South San Francisco, CA) were used.
Vector Constructs and Transfection
A lentivirus vector was generated using the ViraPower™ lentiviral packaging mix (Invitrogen, Carlsbad, CA). Lipofectamine 2,000 (Invitrogen) was used to transfect SHGLYC-TRCN0000019684 (Z1), SHGLYC-TRCN 0000019685 (Z2), and SHC002 (random sequences inserted) (Sigma) into 293FT cells (Invitrogen). Z1 and Z2 targeted different sites in the ZNF281 coding region. The supernatant was harvested at 48 h after transfection. The hMSCs were infected with virus at a multiplicity of infection (MOI) of 10 in the presence of 6 μg/ml polybrene (Sigma) in the culture medium, and infected cells were selected by the addition of 3 μg/ml puromycin. For overexpression of ZNF281, the full coding region of ZNF281 was inserted into a pLenti6/V5 directional Topo vector (Invitrogen). Stable overexpression was selected using 5 μg/ml blasticidin (Sigma) in the culture medium.
Induction of Differentiation
To induce osteogenic differentiation, hMSCs were seeded and maintained at 70–80% confluency and incubated with low-glucose DMEM (Gibco-Invitrogen), 10% FBS (Gibco-Invitrogen), 0.1 μM dexamethasone (Sigma), 10 mM β-glycerophosphate (Sigma), and 50 μM ascorbate (Sigma) for 2–3 weeks (34). For adipogenic differentiation, the cells were seeded and maintained at 80–90% confluency and incubated with low-glucose DMEM (Gibco BRL), 10% FBS (Gibco BRL), 1 μM dexamethasone (Sigma), 10 μg/ml insulin (Sigma), 0.5 mM 3-isobutyl-1-methylxanthine (Sigma), and 0.2 mM indomethacin (Sigma) (34).
RT-PCR, PCR Array, and Chromatin Immunoprecipitation (ChIP) Assay
Total RNA was extracted with an Easy-spin™ Total RNA Extraction Kit (Intronbio, Sungnam, Korea) according to the manufacturer's instructions. The cDNA was synthesized using a SuperScript® III first-strand synthesis kit (Invitrogen) from 1 μg of total RNA. The primer sequences are shown in Table 1. Gene expression was also analyzed using real-time PCR with SYBR Green Master Mix reagents (Applied Biosystems, Foster City, CA). For the PCR array, the RT2 Profiler stem cell PCR array (catalog no. PAHS-405, SuperArray) was used according to the manufacturer's protocol. Semiquantitative RT-PCR was performed with a LightCycler 489 real-time PCR system (Roche, Indianapolis, IN). A ChIP assay was performed according to the manufacturer's protocol (catalog no. 17–295, Upstate Biotechnology, Lake Placid, NY).
The PCR Primers Used in Experiment
Osteogenesis In Vivo and Animal Care
The hUCB-MSCs were sequentially infected with a β-galactosidase-expressing lentivirus (pLenti6/V5-GW/lacZ, Invitrogen) followed by ZNF281 knockdown (Z1) or vehicle control lentivirus and subsequently underwent selection. β-Tricalcium phosphate (β-TCP) has been proven to be an effective bone formation additive for hMSCs in vivo (6). β-TCP powder was a generous gift from the Biomaterials Center, National Institute for Materials Science, Japan. β-TCP (700 mg) was mixed with 1 × 106 hUCB-MSCs and diluted with 0.5 ml saline. The hUCB-MSCs and the β-TCP mixture were implanted into nude mice subcutaneously. Nude mice were sacrificed at 2, 4, 8, and 12 weeks after implantation. Each experimental group contained three animals. Alizarin red S staining and LacZ staining were performed using established protocols (7). Decalcification of samples was performed using 4% formic acid/hydrochloric acid solution (Sigma), and the samples were neutralized with ammonia solution (Sigma). All animal experiments followed the guidelines for Animal Experiments of Seoul National University and were approved by the Seoul National University IRB (SNU-100111-4).
Statistical Analysis
The change in the CT of the target genes was calculated as ACT = (CT of target genes) - (CT of GAPDH). The ratio of the target gene to the housekeeping gene (GAPDH; glyceraldehyde-3-phosphate dehydrogenase) was calculated and expressed as 2-ΔCT. This ratio was then used to evaluate the expression level within each target gene in ZNF281-inhibited hUCB-MSCs and vehicle control-infected hUCB-MSCs. To determine the changes in gene expression, the normalized gene expression of the target genes in ZNF281-inhibited hUCB-MSCs was divided by the normalized expression of the same target gene in the vehicle-control samples. The data from the quantitative RT-PCR are presented as the mean values ± standard deviation from three independent clones of hUCB-MSCs. Student's t test (Microsoft Excel) was used for the analysis and calculation of p values between control and treated cells. For comparison between the two experimental groups and controls, multiple statistical comparisons between groups were performed by one-way ANOVA, with post hoc Bonferroni correction.
Results
ZNF281 Was Expressed in hMSCs and Knockdown of ZNF281 Resulted in Reduced Proliferation of hMSCs
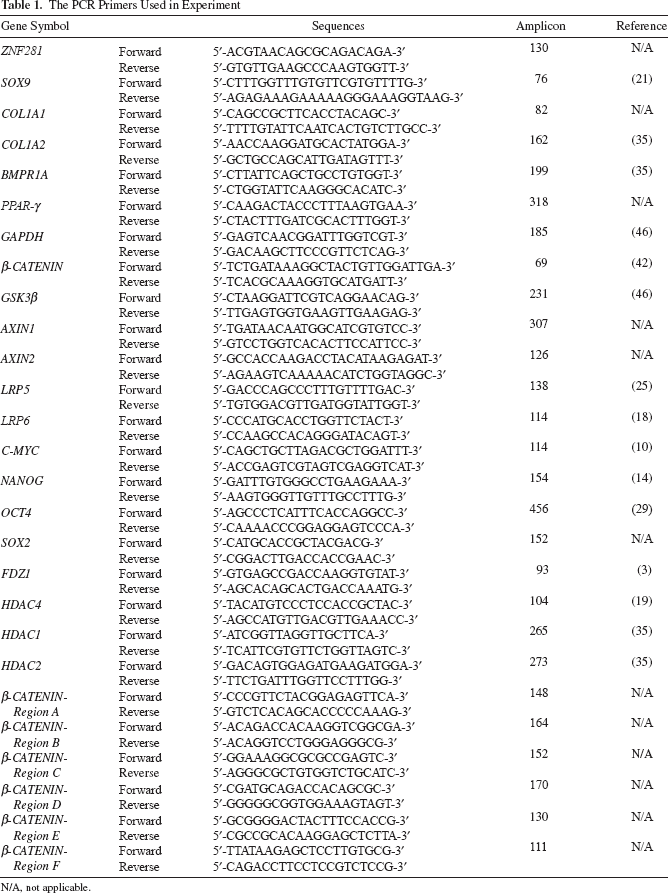
ZNF281 was found to be expressed in hUCB-MSCs and hAD-MSCs. The expression levels of ZNF281 in hUCB-MSCs and hAD-MSCs were similar to those in the Tera-1 cell line, which was derived from an embryonic carcinoma and has multidifferentiation abilities, as do ESCs (Fig. 1a). ZNF281 expression was observed in hUCB-MSCs from passage (P) 3 through P9. More than 98% of the hUCB-MSCs were ZNF281 positive, as assessed by flow cytometry analysis (Fig. 1b). Two different sites within ZNF281 were targeted (Z1 and Z2), and a random sequence was used as a vehicle control (VC), using a lentiviral delivery system during the stable inhibition study. After selection, the passage number was reset to zero after inhibition (PI0). After antibiotic selection, hUCB-MSCs were cultured for 3 or 4 days until lentivirus-infected cells reached approximately 70% confluence. The inhibition of ZNF281 expression in hUCB-MSCs was confirmed by Western blotting and semiquantitative RT-PCR analysis at PI1. The expression levels of ZNF281 in Z1- and Z2-infected hUCB-MSCs were reduced to approximately 30% and 50% of those of the vehicle control-infected hUCB-MSCs, respectively (Fig. 1c). Cell numbers were counted after trypsin treatment at PI1, and the cells were seeded in new dishes at the same density for each experimental group. Cell counts were performed with three independent clones isolated from three individuals until PI3. According to the cumulative proliferation data, Z2-infected hUCB-MSCs showed a significant decrease in proliferation rates (Fig. 1d).
ZNF281 expression and growth retardation after ZNF281 inhibition. (a) Zinc finger protein 281 (ZNF281) expression in tera-1, human adipose tissue-derived mesenchymal stem cells (hAD-MSCs), and human umbilical cord blood-derived (hUCB)-MSCs. ZNF281 expression was constantly continued until P9 of hUCB-MSCs in semiquantitative RT-PCR. (b) Flow cytometry analysis of hUCB-MSCs for ZNF281 at P5. Over 98% of hUCB-MSCs are positive for ZNF281. FITC, fluorescein isothiocyanate. (c) The expression of ZNF281 after shRNA producing lentivirus infection with Western blot and semiquantitative RT-PCR. The expression of ZNF281 was reduced in Z1- and Z2-infected hUCB-MSCs. (d) Cell count was performed at the end of passage and the cumulative proliferation rates were measured until passage 3 after ZNF281 inhibition (PI3). VC, vehicle control (SHC002); Z1, ZNF281 knockdown 1 (SHGLYC-TRCN0000019684); Z2, ZNF281 knockdown 2 (SHGLYC-TRCN0000019685); P, passage; PI, passage after ZNF281 inhibition. GAPDH, glyceraldehyde 3-phosphate dehydrogenase. Error bars represent standard deviation. *p < 0.05; **p < 0.01.
Adipogenic Potentiality Was Decreased but Osteogenic Potential Was Increased in ZNF281-Knockdown hMSCs
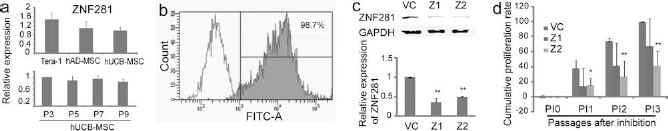
The adipogenic differentiation ability of ZNF281-knockdown hUCB-MSCs was consistently lower than that of the vehicle control-infected hUCB-MSCs in three independent hUCB-MSC clones (Fig. 2a). The number of Oil Red O stained cells was severely reduced in ZNF281-knockdown hUCB-MSCs after adipogenic induction. When the expression of adipogenic marker genes was compared, the expression levels of peroxisome proliferator-activated receptor gamma (PPAR-γ) and adipocyte protein 2 (aP2) were severely decreased in ZNF281-knockdown hUCB-MSCs, compared to the vehicle control-treated hUCB-MSCs in both Z1- and Z2-infected cells after adipogenic induction (Fig. 2b). Osteogenic differentiation studies were initiated at P5 and P8 for the hUCB-MSCs, and after antibiotic selection, osteogenesis was induced in P6 and P9. The osteogenic differentiation ability of hUCB-MSCs following induction was also altered in ZNF281-knockdown hUCB-MSCs. However, unlike the inhibitory effect observed in adipogenesis, ZNF281-knockdown hUCB-MSCs showed accelerated osteogenesis compared to the vehicle control-infected hUCB-MSCs, a finding confirmed with Alizarin red S staining (Fig. 2c). The expression levels of the osteogenic marker genes bone morphogenetic protein receptor type 1a (BMPR1A), runt-related transcription factor 2 (RUNX2), sex-determining region Y box containing gene 9 (SOX9), and collagen type 1 a 2 (COL1A2) were increased in ZNF281-knockdown hUCB-MSCs at 7 days after osteogenic induction compared to the vehicle control-infected hUCB-MSCs (Fig. 2d). For extended cultures of hUCB-MSCs (i.e., P9 or P10), hUCB-MSCs occasionally lost their normal osteogenic potential. However, ZNF281-knockdown hUCB-MSCs were able to differentiate into osteogenic lineage cells after osteogenic induction, even though the vehicle control-infected hUCB-MSCs could not properly differentiate (Fig. 2c).
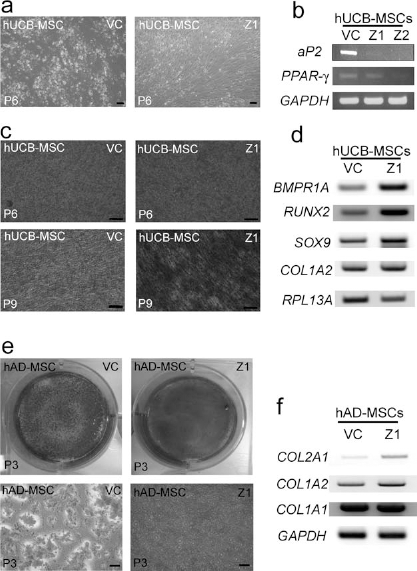
Differentiation study after ZNF281 inhibition. (a) Adipogenic differentiation after ZNF281 inhibition at passage 6 (P6) of hUCB-MSCs. The number of Oil Red O stained cells decreased in ZNF281-knockdown hUCB-MSCs (Z1) compared with that of the vehicle control-infected hUCB-MSCs (VC). (b) RT-PCR of molecular marker genes after adipogenic induction at P6. The expressions of adipocyte protein 2 (aP2) and peroxisome proliferator-activated receptor gamma (PPAR-γ) were decreased in ZNF281 knocked-down hUCB-MSCs after adipogenic induction. (c) Osteogenic differentiation after ZNF281 knockdown at P6 and P9. ZNF281-knockdown hUCB-MSCs showed accelerated osteogenic potential compared to the vector control-infected hUCB-MSCs, especially in P9. (d) RT-PCR of molecular marker genes after osteogenic induction at P6. The expressions of bone morphogenetic protein receptor type 1a (BMPR1A), runt-related transcription factor 2 (RUNX2), sex-determining region Y box containing gene 9 (SOX9), and collagen type 1 a 2 (COL1A2) were increased in ZNF281 knocked-down hUCB-MSCs after osteogenic induction. Ribosomal protein L13A (RPL13A) was used as a loading control. (e) Osteogenesis of hAD-MSCs. Osteogenesis was increased after ZNF281 knockdown (Z1), which was confirmed with Alizarin red S staining. (f) The expressions of osteogenic marker genes were increased in ZNF281 knocked-down (Z1) hAD-MSCs after osteogenic induction compared to the vehicle control-infected (VC) hAD-MSCs in RT-PCR. Scale bar: 50 μm.
ZNF281 is also expressed in hAD-MSCs, which can differentiate into osteogenic cell lineages. Therefore, osteogenic differentiation was studied in hAD-MSCs. In ZNF281-knockdown hAD-MSCs, the number of Alizarin red S-stained cells was significantly increased 3 weeks after osteogenic induction compared to the vehicle control-infected hAD-MSCs (Fig. 2e). The expression levels of COL2A1 and COL1A2, which are chondrogenic and osteogenic markers, were increased in ZNF281-knockdown hAD-MSCs compared to the vehicle control-infected hAD-MSCs 1 week after osteogenic induction (Fig. 2f). The expression of COL1A1, an MSC marker, was not significantly changed after ZNF281 knockdown. The expression levels of GAPDH and RPL13A (ribosomal protein L13A) were used as loading controls.
ZNF281 Knockdown Accelerated the Osteogenesis of hUCB-MSCs In Vivo
To evaluate the role of ZNF281 in osteogenesis in vivo, ZNF281-knockdown hUCB-MSCs were mixed with β-TCP and implanted subcutaneously on the right side of the back of nude mice. The left side of the back was implanted with vehicle control-infected hUCB-MSCs that were also mixed with β-TCP. All hUCB-MSCs were prelabeled with LacZ, which was performed with β-galactosidase-expressing lentivirus, before ZNF281 inhibition. The conversion of normal hUCB-MSCs into osteoblasts takes 3 months after implantation with β-TCP (6). Calcium droplets among LacZ-positive cells were found using Alizarin red S staining of ZNF281-knockdown hUCB-MSCs not only near the β-TCP cluster but also in the interstitial area, which was not found in control hUCB-MSCs until 4 weeks after implantation (Fig. 3b, magnified).

Osteogenic differentiation of hUCB-MSCs in vivo. (a) LacZ and Alizarin red S staining in the decalcified tissue of 2 weeks after implantation. Most LacZ-positive cells were present in the interstitial area. (b) LacZ and Alizarin red S staining of 4 weeks after implantation. Calcium droplets were found in the LacZ-positive cells of ZNF281 knocked-down hUCB-MSCs not only near β-tricalcium phosphate (β-TCP) but also in the interstitial area (magnified). Scale bars: 50 μm.
PCR Array Results Indicate That the ZNF281 Knockdown Induced the Osteochondro Differentiation of hUCB-MSCs
Z2-infected hUCB-MSCs exhibited very low proliferation rates (Fig. 1c). Therefore, Z2-infected hUCB-MSCs were not used for long-term osteogenic differentiation experiments (Fig. 2c–f) but were used for analysis of gene expression pattern changes (Fig. 4a–c). The expression levels of 84 genes that are frequently cited in stem cell research were examined by real-time RT-PCR at the end of PI1 in three independent clones of hUCB-MSCs derived from three individuals. Several genes with irregular expression patterns among the hUCB-MSC clones were excluded. The expression levels of two genes in ZNF281-knockdown hUCB-MSCs increased more than fourfold compared to the levels in the vehicle control-infected hUCB-MSCs (Fig. 4a). The most significant increase in expression in ZNF281-knockdown hUCB-MSCs was seen for aggrecan (ACAN), which was expressed at a level 244-fold higher than in the vehicle control-infected hUCB-MSCs (p = 0.019). The expression of cyclin D2 (CCND2) in ZNF281-knockdown hUCB-MSCs was also increased compared to the vehicle control-infected hUCB-MSCs. In contrast, the expression of the following six genes decreased by more than 4-fold in the ZNF281-knockdown cells (compared to the vehicle control-treated hUCB-MSCs): fibroblast growth factor 1 (FGF1), chemokine CXC motif ligand 12 (CXCL12), S100 calcium-binding protein (S100B), ATP-binding cassette subfamily G member 2 (ABCG2), histone deacetylase 2 (HDAC2), and insulin-like growth factor 1 (IGF1).

The gene expression changes after ZNF knockdown and during osteogenesis of hUCB-MSCs. (a) Log–log scatter plot of PCR array. Spots below the bottom line represent down-regulated genes and spots above the top line represent genes upregulated more than fourfold in ZNF281-knockdown hUCB-MSCs (Z2) when compared to the vehicle control (VC) infected hUCB-MSCs. (b) Semiquantitative RT-PCR results of canonical WNT/β-CATENIN signaling genes after ZNF281 inhibition. The expressions of β-CATENIN, GSK3-β and AXIN2 were significantly decreased in ZNF281-knockdown hUCB-MSCs (Z1 and Z2). (c) Western blot analysis of β-CATENIN and glycogen synthase kinase 3-β (GSK3-β) from total cell lysate. The expressions of β-CATENIN and GSK3-β were decreased in ZNF281-knockdown hUCB-MSCs. The expression of p-β-CATENIN increased, but the expressions of phosphorylated GSK3-β at serin-9 (p-GSK3-β) were varied in ZNF281-knockdown hUCB-MSCs. (d) The gene expression changes after osteogenic induction of normal hUCB-MSCs. The expression of ZNF281 was decreased at 8 h after osteogenic induction and continued its level until 7 days after osteogenic induction. β-CATENIN expression decreased at 24 h and rebound at 3 days after osteogenic induction. Axis inhibition protein 2 (AXIN2) expression increased steadily until 3 days after osteogenic induction. SOX9 expression increased at 24 h after osteogenic induction. Hours (h) and days (d) after osteogenic induction. Error bars represent standard deviation. **p < 0.01.
β-CATENIN Expression Was Significantly Decreased in ZNF281-Knockdown hUCB-MSCs
Canonical WNT/β-CATENIN signaling is important in osteogenic differentiation (13), and the ZNF281 knockdown enhanced the osteogenic differentiation of hUCB-MSCs. The expression levels of canonical WNT signaling genes, including β-CATENIN, were significantly affected as shown by the PCR array, although the changes were less than fourfold (Fig. 4a). Therefore, the expression patterns of the canonical WNT/β-CATENIN signaling-related genes were examined in ZNF281-knockdown hUCB-MSCs. The expression of β-CATENIN in ZNF281-knockdown hUCB-MSCs was significantly lower than that in the vehicle control-infected hUCB-MSCs in three independent clones of hUCB-MSCs isolated from three individuals. The expression levels of β-CATENIN in Z1- and Z2-infected cells were reduced to roughly 40% and 50%, respectively, of the expression level in vehicle control-infected hUCB-MSCs (Fig. 4b). The expression levels of GSK3-β and axis inhibition protein 2 (AXIN2) were also lower in ZNF281-knockdown hUCB-MSCs than in the vehicle control-infected hUCB-MSCs. The expression levels of AXIN1, frizzled-1 (FDZ1), dickkopf-related protein 1 (DKK1), low-density lipoprotein receptor-related protein (LRP5), and LRP6 were not consistent among the different ZNF281-knockdown hUCB-MSC clones (data not shown). The protein expression levels of β-CATENIN and GSK3-β in ZNF281-knockdown hUCB-MSCs were reduced compared to those in the vehicle control-infected hUCB-MSCs (Fig. 4c). The phosphorylation of β-CATENIN is one of the most important mechanisms affecting β-CATENIN stability in mammalian cells. The level of phosphorylated β-CATENIN was higher in ZNF281-knockdown hUCB-MSCs than in vehicle control-infected hUCB-MSCs (Fig. 4c).
The Expression Pattern of β-CATENIN Was Changed After Osteogenic Induction of Normal hUCB-MSCs
To investigate the roles that ZNF281 and other genes play in the osteogenic differentiation of hUCB-MSCs, in vitro osteogenic differentiation studies were performed with normal hUCB-MSCs (Fig. 4d). The osteogenic differentiation of hUCB-MSCs occurs in less than 2 weeks. Alizarin red S-positive cells, which are osteoblasts, appeared at the end of the first week after osteogenic induction. Therefore, the gene expression patterns in normal hUCB-MSCs after osteogenic induction were investigated until the 7th day after induction (Fig. 4d). The expression of ZNF281 in hUCB-MSCs after osteogenic induction was reduced to roughly half that of undifferentiated cells at 8 h, and expression continued to be suppressed for the entire course of the experiment (Fig. 4d). The expression of β-CATENIN did not change at 8 h but was downregulated at 24 h after osteogenic induction and then returned to initial levels at day 3 postinduction. At 7 days after osteogenic induction in hUCB-MSCs, β-CATENIN expression dramatically increased. The expression of AXIN2 gradually increased until 3 days after the osteogenic induction of the hUCB-MSCs. The expression of SOX9, a transcription factor that is known to play key roles in osteogenic cells (49), increased at 24 h and remained at an elevated level until 7 days after osteogenic induction of the hUCB-MSCs (Fig. 4d). Therefore, the cell fate of hUCB-MSCs is probably decided within 24 h after differentiation induction, and the change in ZNF281 expression precedes the change in β-CATENIN expression during the osteogenic differentiation of normal hMSCs.
The Overexpression of ZNF281 in hUCB-MSCs Resulted in the Upregulation of β-CATENIN
To elucidate the function of ZNF281, a gain-of-function study was performed in hUCB-MSCs. The full-length ZNF281 cDNA under the control of the cytomegalovirus (CMV) promoter was inserted into a lentiviral vector and transfected into hUCB-MSCs. The expression levels were compared with those in control-lentivirus-infected hUCB-MSCs. The expression levels of ZNF281 and β-CATENIN increased approximately threefold, as determined by semiquantitative RT-PCR after antibiotic selection (Fig. 5a). The expression levels of GSK3-β, LRP-5, AXIN2, and DKK1 were also higher in ZNF281 overexpressing hUCB-MSCs than in control-lentivirus-infected hUCB-MSCs. The overexpression of β-CATENIN and ZNF281 was confirmed at the protein level (Fig. 5b). Cell proliferation was measured using a CCK-8. After starting at the same cell densities, ZNF281 overexpressing hUCB-MSCs grew faster than control-lentivirus-infected hUCB-MSCs from three different cell lines (Fig. 5c). The expression levels of β-CATENIN always followed the expression pattern of ZNF281. Therefore, a mechanism for the direct regulation of β-CATENIN expression by ZNF281 in hUCB-MSCs was proposed. To confirm this mechanism, the binding of ZNF281 to the promoter region of β-CATENIN was examined using the ChIP assay. Six possible regions within the evolutionarily conserved area around the promoter of β-CATENIN were targeted using PCR. A strong positive signal (region C) was found near the 5′ end, upstream of the first exon of β-CATENIN of hUCB-MSCs (Fig. 5d).

The gain of function study for ZNF281 in hUCB-MSCs and ChIP assay. (a) The expressions for ZNF281 and β-CATENIN were upregulated by approximately threefold in semiquantitative RT-PCR. (b) Western blot of ZNF281 and β-CATENIN after ZNF281 overexpression with using a lentiviral vector. GAPDH used as a loading control. (c) Cell growth was measured with a cell counting kit-8 (CCK-8). Cell growth was significantly increased in ZNF281 overexpressing hUCB-MSCs (ZNF-over) compared to the vehicle control-infected hUCB-MSCs (VC). (d) ZNF281 binding was found in the β-CATENIN promoter region (region C) with ChIP assay. Semiquantitative PCR results. Error bars represent standard deviation. **p < 0.01.
Tera-1 Cells and the Expressions of HDACs Were Altered After ZNF281 Expression Changes
The expression level of β-CATENIN in Tera-1 cells was lower after ZNF281 knockdown, and the expression level of ACAN was higher in ZNF281-knockdown Tera-1 cells compared to the vehicle control-infected Tera-1 cells (Fig. 6a), a result similar to that observed in hUCB-MSCs.

The gene expression changes of tera-1 and histone deacetylases (HDACs). (a) Semiquantitative RT-PCR after ZNF281 knockdown in tera-1 cells. The expression of β-CATENIN was decreased, but the expression of aggrecan (ACAN) was increased after ZNF281 knockdown (Z1) compared to the vehicle control-infected (VC) tera-1. (b) The expressions of HDAC2 and HDAC4 decreased in ZNF281 knocked-down hUCB-MSCs compared to the vehicle control-infected hUCB-MSCs with semiquantitative RT-PCR. The expression of HDAC4 significantly increased in constantly ZNF281 overexpressing hUCB-MSCs (ZNF-over) compared to the vehicle control-infected hUCB-MSCs (VC). (c) The model of stemness and differentiation in hMSCs. ZNF281 possibly has a role for the proliferation and cell-fate decision of hMSCs via the β-CATENIN and octamer-binding transcription factor 4A (OCT4A) regulation. Error bars represent standard deviation. VC, vehicle control; Z1, ZNF281 knockdown 1; Z2, ZNF281 knockdown 2. **p < 0.01.
In the PCR array, the expression of histone deacetylase 2 (HDAC2) was dramatically changed (Fig. 4a). The activity of HDAC is essential for maintaining the self-renewal, multipotency, and cellular senescence of MSCs (20, 27, 28). Therefore, further study focused on the expression of HDAC gene family members. Among the class I and class II HDAC family members, the expression levels of HDAC1, HDAC2, and HDAC4 were examined using semiquantitative RT-PCR. The expression level of HDAC1 was too varied among the clones to show any significant changes between ZNF281-knockdown and vehicle control-infected hUCB-MSCs (Fig. 6b). For cells in which ZNF281 had been overexpressed using the lentivirus vector, the expression of HDAC2 was not significantly changed. However, the expression of HDAC4, a class II HDAC, was significantly reduced in Z2-infected hUCB-MSCs compared to the vehicle control-infected hUCB-MSCs from three independent hUCB-MSCs clones. The expression of HDAC4 also significantly increased when ZNF281 was overexpressed (Fig. 6b).
The expression levels of c-MYC, SOX2, and REX-1 in ZNF281-knockdown hUCB-MSCs did not show any consistent results when compared with the vehicle control-infected hUCB-MSCs because their expressions varied with each hUCB-MSC clone. The expression of OCT4A, which is responsible for the pluripotency of ESCs (29) in ZNF281-knockdown hUCB-MSCs, was consistently reduced as demonstrated by RT-PCR analysis (data not shown). The expression of NANOG in hUCB-MSCs was not recognized regardless of whether ZNF281 was inhibited or overexpressed in hUCB-MSCs.
Discussion
This study addressed the roles of ZNF281 in the growth and differentiation of adult stem cells. The cell growth decreased after ZNF281 knockdown and increased after ZNF281 overexpression in hMSCs. The multipotency of hUCB-MSCs was also altered in ZNF281-knockdown hUCB-MSCs. The adipogenic differentiation ability was reduced, but the osteogenic differentiation ability was increased in ZNF281-knockdown hUCB-MSCs. The activation of Wnt/β-catenin signaling is important for early events in the adipogenesis of MSCs (5). Therefore, the depressed adipogenic differentiation of ZNF281-knockdown hUCB-MSCs was suggested to be caused by a decrease in β-CATENIN expression followed by ZNF281 knockdown. In contrast, the osteogenic differentiation in ZNF281-knockdown hUCB-MSCs was accelerated. During the osteogenic induction of normal hUCB-MSCs, the expression of ZNF281 decreased for 8 h after osteogenic induction. The expression of SOX9 increased in tandem with the downregulation of β-CATENIN at 24 h after the osteogenic induction of the hUCB-MSCs (Fig. 4d). During the initial step of osteogenic differentiation in hUCB-MSCs, β-CATENIN expression followed the ZNF281 expression pattern, which implied that ZNF281 could regulate β-CATENIN expression at this initial step.
The results of the osteochondrogenic differentiation of hUCB-MSCs after ZNF281 knockdown were supported by the PCR array data. The most highly upregulated gene in ZNF281-knockdown hUCB-MSCs was ACAN, a marker for osteochondro progenitor cells (43). After ZNF281 knockdown in hUCB-MSCs, CCND2 was upregulated and IGF1 was downregulated, which is a gene expression pattern similar to that seen in the osteogenic transdifferentiation of MSCs (38).
Wnt/β-catenin (i.e., canonical) signaling in MSCs is one of the critical signals for maintaining stemness and multipotency (4, 12). The expression of WNT/β-CATENIN signaling-related genes was observed in ZNF281-knockdown hUCB-MSCs and compared to that in the vehicle control-infected hUCB-MSCs. The expression level of β-CATENIN correlated well with that of ZNF281 expression in hUCB-MSCs. The promoter region of β-CATENIN contains a GC-rich domain, which is a supposed target sequence for ZNF281 in genomic DNA (30, 48). The binding of ZNF281 to the promoter region of β-CATENIN was found in the ChIP assay, implying the direct regulation of β-CATENIN expression by ZNF281 in stem cells. However, it was suggested that this direct regulation of β-CATENIN by ZNF281 would be limited in undifferentiated stem cells because the β-CATENIN expression levels were not correlated with those of ZNF281 after stem cell differentiation (Fig. 4d).
Transcriptional regulation is not the only way to control the canonical WNT/β-CATENIN signaling pathway. The amount of phosphorylated β-CATENIN (Ser33/37/Thr41), a degraded form of the protein, increased even though the levels of total β-CATENIN decreased in ZNF281-knockdown hUCB-MSCs. The phosphorylation of the tyrosine at position 216 in GSK3-β activates the phosphorylation and degradation of β-CATENIN (47). The function of GSK3-β is inhibited by phosphorylation at serine-9 (p-GSK3-β) (41). The amount of p-GSK3-β in ZNF281-knockdown hUCB-MSCs varied and did not increase consistently in the repeated experiments. Therefore, the degradation of β-CATENIN accelerated concurrently with the downregulation of β-CATENIN transcription. Loss-of-function mutations for β-catenin have been introduced into osteochondrogenic progenitor cells of osteoblast precursors in mice, where, as in our study, the progenitor cells differentiated into chondrocytes alone (36).
HDACs are essential catalytic components of the transcription silencing machinery (9). Hdac4 regulates chondrocyte hypertrophy and endochondral bone formation in mice. Hdac4-null mice displayed premature ossification of developing bones as a result of ectopic and early-onset chondrocyte hypertrophy. Conversely, in vivo overexpression of Hdac4 in proliferating chondrocytes inhibited chondrocyte hypertrophy and differentiation (44). The stable overexpression of ZNF281, using lentivirus vectors, resulted in the upregulation of HDAC4 in hUCB-MSCs. However, the transient overexpression of ZNF281 using plasmid vectors resulted in no significant changes to HDAC4 expression until 72 h after transfection (unpublished data). Therefore, the expression changes of HDAC4 in hUCB-MSCs might be a secondary effect of osteogenic differentiation induced by ZNF281 expression changes.
In conclusion, β-CATENIN activation in hMSCs might lead to adipogenic differentiation (5), but β-CATENIN inactivation leads to osteochondrogenic differentiation (4) during the early fate decision step of hMSCs. Stable expression of β-CATENIN in hMSCs might be guaranteed not only by the autocrine process (12) but also by the stable expression of ZNF281 (Fig. 6c). ZNF281 knockdown can induce the osteochondrogenic differentiation of hMSCs. These phenomena occur not only in hUCB-MSCs but also in hAD-MSCs and in vivo.
Footnotes
Acknowledgments
This work was supported by the National Research Foundation (NRF) of Korea grant funded by the Korea government (MEST, 2010-0020265) and by Basic Science Research Program through the NRF funded by the Ministry of Education, Science and Technology (550-20100030). The authors declare no conflict of interest.
